Venus flytrap on:
[Wikipedia]
[Google]
[Amazon]
The Venus flytrap (''Dionaea muscipula'') is a
 The leaf blade is divided into two regions: a flat, heart-shaped
The leaf blade is divided into two regions: a flat, heart-shaped
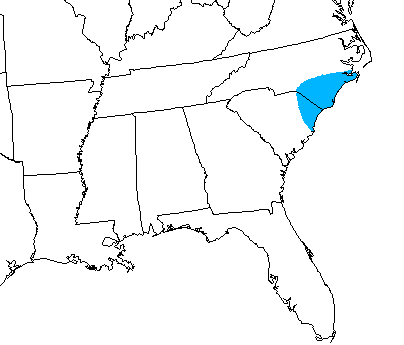 The Venus flytrap is found in
The Venus flytrap is found in
''Dionaea muscipula''.
Burke Museum of Natural History and Culture. The nutritional poverty of the soil is the reason it relies on such elaborate traps: insect prey provide the nitrogen for

 The Venus flytrap is one of a very small group of plants capable of rapid movement, such as ''
The Venus flytrap is one of a very small group of plants capable of rapid movement, such as ''
Comparative physiology of the Droseraceae ''sensu stricto'' – How do tentacles bend and traps close?
Proceedings of the 4th International Carnivorous Plant Society Conference. Tokyo, Japan. pp. 77–81. Flytraps show an example of memory in plants; the plant knows if one of its trigger hairs have been touched, and remembers this for a few seconds. If a second touch occurs during that time frame, the flytrap closes. After closing, the flytrap

 Carnivory in plants is a very specialized form of
Carnivory in plants is a very specialized form of
 Plants can be propagated by seed, taking around four to five years to reach maturity. More commonly, they are propagated by clonal division in spring or summer. Venus flytraps can also be propagated ''in vitro'' using
Plants can be propagated by seed, taking around four to five years to reach maturity. More commonly, they are propagated by clonal division in spring or summer. Venus flytraps can also be propagated ''in vitro'' using
Images and movies of the Venus flytrap ''(Dionaea muscipula)''
at
Venus Flytrap Growing Guide and Distribution Map
Venus flytrap origins uncovered – BBC
Criminal Podcast Episode Five: Dropping Like Flies
{{Authority control Articles containing video clips Carnivorous plants of North America Dionaea (plant) Endemic flora of the United States Flora of North Carolina Flora of South Carolina Plant cognition Plants described in 1768 Symbols of North Carolina Vulnerable plants
carnivorous plant
Carnivorous plants are plants that derive some or most of their nutrients from trapping and consuming animals or protozoans
Protozoa (singular: protozoan or protozoon; alternative plural: protozoans) are a group of single-celled eukaryot ...
native to subtropical
The subtropical zones or subtropics are geographical zone, geographical and Köppen climate classification, climate zones to the Northern Hemisphere, north and Southern Hemisphere, south of the tropics. Geographically part of the Geographical z ...
wetland
A wetland is a distinct ecosystem that is flooded or saturated by water, either permanently (for years or decades) or seasonally (for weeks or months). Flooding results in oxygen-free (anoxic) processes prevailing, especially in the soils. The ...
s on the East Coast of the United States
The East Coast of the United States, also known as the Eastern Seaboard, the Atlantic Coast, and the Atlantic Seaboard, is the coastline along which the Eastern United States meets the North Atlantic Ocean. The eastern seaboard contains the coa ...
in North Carolina
North Carolina () is a state in the Southeastern region of the United States. The state is the 28th largest and 9th-most populous of the United States. It is bordered by Virginia to the north, the Atlantic Ocean to the east, Georgia and So ...
and South Carolina
)''Animis opibusque parati'' ( for, , Latin, Prepared in mind and resources, links=no)
, anthem = " Carolina";" South Carolina On My Mind"
, Former = Province of South Carolina
, seat = Columbia
, LargestCity = Charleston
, LargestMetro = ...
. It catches its prey—chiefly insect
Insects (from Latin ') are pancrustacean hexapod invertebrates of the class Insecta. They are the largest group within the arthropod phylum. Insects have a chitinous exoskeleton, a three-part body ( head, thorax and abdomen), three pairs ...
s and arachnid
Arachnida () is a class of joint-legged invertebrate animals (arthropods), in the subphylum Chelicerata. Arachnida includes, among others, spiders, scorpions, ticks, mites, pseudoscorpions, harvestmen, camel spiders, whip spiders and vinegaroon ...
s—with a trapping structure formed by the terminal portion of each of the plant's leaves, which is triggered by tiny hairs (called "trigger hairs" or "sensitive hairs") on their inner surfaces.
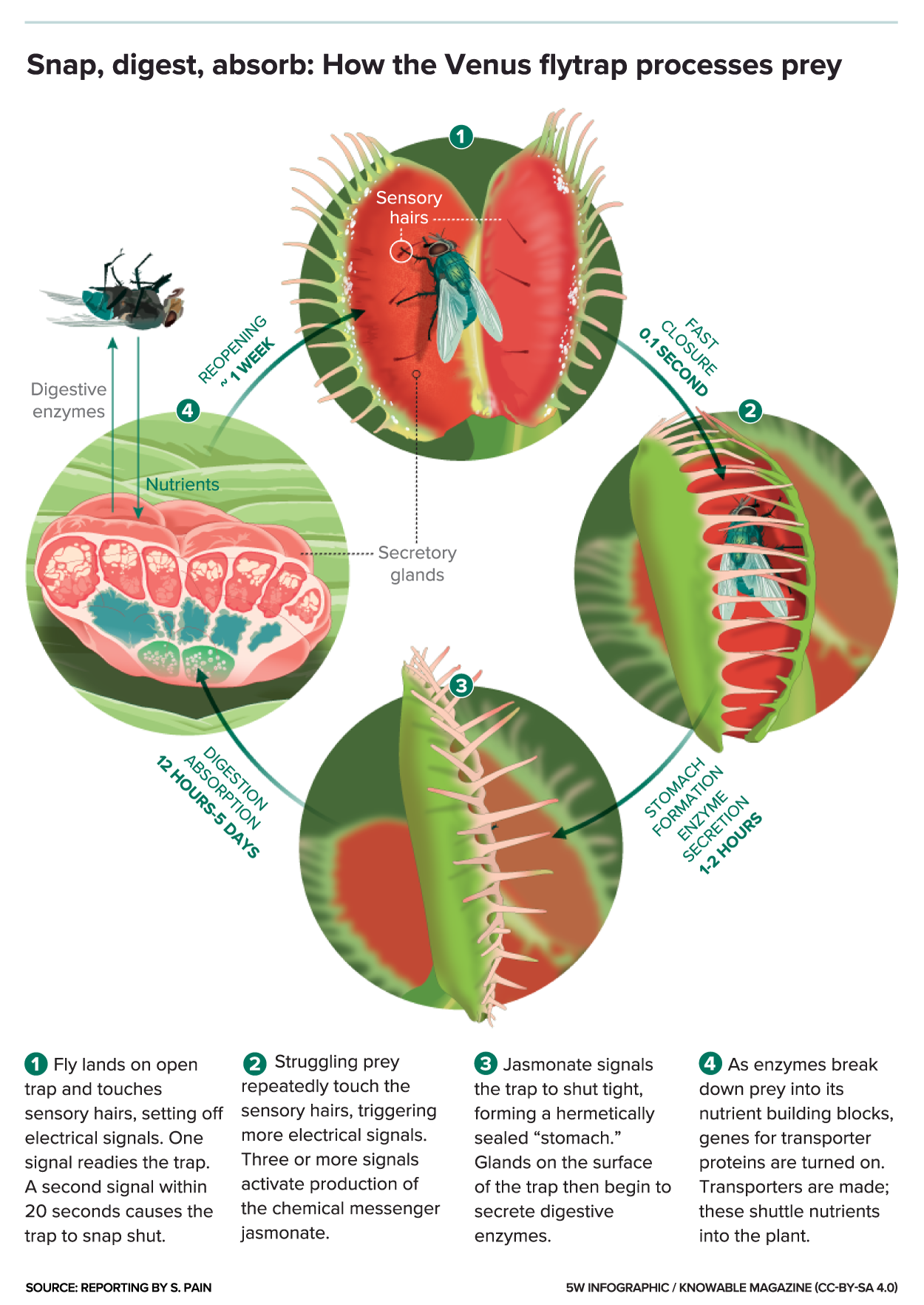
When an insect or spider crawling along the leaves contacts a hair, the trap prepares to close, snapping shut only if another contact occurs within approximately twenty seconds of the first strike. Triggers may occur with a tenth of a second of contact. The requirement of redundant triggering in this mechanism serves as a safeguard against wasting energy by trapping objects with no nutritional value, and the plant will only begin digestion after five more stimuli to ensure it has caught a live bug worthy of consumption.
''Dionaea'' is a monotypic
In biology, a monotypic taxon is a taxonomic group (taxon) that contains only one immediately subordinate taxon. A monotypic species is one that does not include subspecies or smaller, infraspecific taxa. In the case of genera, the term "unispec ...
genus
Genus ( plural genera ) is a taxonomic rank used in the biological classification of extant taxon, living and fossil organisms as well as Virus classification#ICTV classification, viruses. In the hierarchy of biological classification, genus com ...
closely related to the waterwheel plant ('' Aldrovanda vesiculosa'') and sundews (''Drosera
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
''), all of which belong to the family Droseraceae
Droseraceae is a family of carnivorous flowering plants, also known as the sundew family. It consists of approximately 180 species in three extant genera. Representatives of the Droseraceae are found on all continents except Antarctica.
Descr ...
.
Although widely cultivated for sale, the population of the Venus flytrap has been rapidly declining in its native range. The species is currently under Endangered Species Act
The Endangered Species Act of 1973 (ESA or "The Act"; 16 U.S.C. § 1531 et seq.) is the primary law in the United States for protecting imperiled species. Designed to protect critically imperiled species from extinction as a "consequence of ec ...
review by the U.S. Fish & Wildlife Service.
Etymology
The plant's common name (originally "Venus's flytrap") refers toVenus
Venus is the second planet from the Sun. It is sometimes called Earth's "sister" or "twin" planet as it is almost as large and has a similar composition. As an interior planet to Earth, Venus (like Mercury) appears in Earth's sky never fa ...
, the Roman goddess of love. The genus name, ''Dionaea'' ("daughter of Dione"), refers to the Greek goddess Aphrodite
Aphrodite ( ; grc-gre, Ἀφροδίτη, Aphrodítē; , , ) is an ancient Greek goddess associated with love, lust, beauty, pleasure, passion, and procreation. She was syncretized with the Roman goddess . Aphrodite's major symbols include ...
, while the species name, ''muscipula'', is Latin for both "mousetrap" and "flytrap". The Latin word ("mousetrap") is derived from ''mus'' ("mouse") and ("trap"), while the homonym
In linguistics, homonyms are words which are homographs (words that share the same spelling, regardless of pronunciation), or homophones ( equivocal words, that share the same pronunciation, regardless of spelling), or both. Using this definiti ...
word ("flytrap") is derived from ("fly") and ("trap").
Historically, the plant was also known by the slang term "tipitiwitchet" or "tippity twitchet", possibly an oblique reference to the plant's resemblance to human female genitalia. The term is similar to the term tippet-de-witchet which derives from tippet
A tippet is a piece of clothing worn over the shoulders in the shape of a scarf or cape. Tippets evolved in the fourteenth century from long sleeves and typically had one end hanging down to the knees. A tippet (or tappit) could also be the long ...
and witchet (archaic term for vagina
In mammals, the vagina is the elastic, muscular part of the female genital tract. In humans, it extends from the vestibule to the cervix. The outer vaginal opening is normally partly covered by a thin layer of mucosal tissue called the hymen ...
). In contrast, the English botanist John Ellis, who gave the plant its scientific name in 1768, wrote that the plant name tippitywichit was an indigenous word from either Cherokee
The Cherokee (; chr, ᎠᏂᏴᏫᏯᎢ, translit=Aniyvwiyaʔi or Anigiduwagi, or chr, ᏣᎳᎩ, links=no, translit=Tsalagi) are one of the indigenous peoples of the Southeastern Woodlands of the United States. Prior to the 18th century, t ...
or Catawba Catawba may refer to:
*Catawba people, a Native American tribe in the Carolinas
*Catawba language, a language in the Catawban languages family
*Catawban languages
Botany
*Catalpa, a genus of trees, based on the name used by the Catawba and other N ...
. The plant name according to the ''Handbook of American Indians'' derives from the Renape word ''titipiwitshik'' ("they (leaves) which wind around (or involve)").
Discovery
On 2 April 1759, the North Carolina colonial governor,Arthur Dobbs
Arthur Dobbs (2 April 1689 – 28 March 1765) was a British colonial official who served as the seventh governor of North Carolina from 1754 until 1764.
Early life and career
Dobbs was born in Ayrshire, Scotland, where his mother had been sen ...
, penned the first written description of the plant in a letter to English botanist Peter Collinson. In the letter he wrote: "We have a kind of Catch Fly Sensitive which closes upon anything that touches it. It grows in Latitude 34 but not in 35. I will try to save the seed here." A year later, Dobbs went into greater detail about the plant in a letter to Collinson dated Brunswick, 24 January 1760.
This was the first detailed recorded notice of the plant by Europeans. The description was before John Ellis' letter to ''The London Magazine
''The London Magazine'' is the title of six different publications that have appeared in succession since 1732. All six have focused on the arts, literature and miscellaneous topics.
1732–1785
''The London Magazine, or, Gentleman's Monthly I ...
'' on 1 September 1768, and his letter to Carl Linnaeus
Carl Linnaeus (; 23 May 1707 – 10 January 1778), also known after his ennoblement in 1761 as Carl von Linné Blunt (2004), p. 171. (), was a Swedish botanist, zoologist, taxonomist, and physician who formalised binomial nomenclature, the ...
on 23 September 1768, in which he described the plant and proposed its English name ''Venus's Flytrap'' and scientific name ''Dionaea muscipula''.
Description
The Venus flytrap is a small plant whose structure can be described as a rosette of four to seven leaves, which arise from a short subterranean stem that is actually a bulb-like object. Each stem reaches a maximum size of about three to ten centimeters, depending on the time of year; longer leaves with robust traps are usually formed after flowering. Flytraps that have more than seven leaves arecolonies
In modern parlance, a colony is a territory subject to a form of foreign rule. Though dominated by the foreign colonizers, colonies remain separate from the administration of the original country of the colonizers, the '' metropolitan state'' ...
formed by rosettes that have divided beneath the ground.
 The leaf blade is divided into two regions: a flat, heart-shaped
The leaf blade is divided into two regions: a flat, heart-shaped photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored i ...
-capable petiole, and a pair of terminal lobes hinged at the midrib, forming the trap which is the true leaf. The upper surface of these lobes contains red anthocyanin
Anthocyanins (), also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart gave the name Anthokyan to a chemical compo ...
pigments and its edges secrete mucilage
Mucilage is a thick, gluey substance produced by nearly all plants and some microorganisms. These microorganisms include protists which use it for their locomotion. The direction of their movement is always opposite to that of the secretion of m ...
. The lobes exhibit rapid plant movement
Rapid plant movement encompasses plant movements, movement in plant structures occurring over a very short period, usually under one second. For example, the Venus flytrap closes its trap in about 100 milliseconds. The traps of Utricularia are muc ...
s, snapping shut when stimulated by prey. The trapping mechanism is tripped when prey contacts one of the three hair-like trichome
Trichomes (); ) are fine outgrowths or appendages on plants, algae, lichens, and certain protists. They are of diverse structure and function. Examples are hairs, glandular hairs, scales, and papillae. A covering of any kind of hair on a pla ...
s that are found on the upper surface of each of the lobes. The mechanism is so highly specialized that it can distinguish between living prey and non-prey stimuli, such as falling raindrops; two trigger hairs must be touched in succession within 20 seconds of each other or one hair touched twice in rapid succession, whereupon the lobes of the trap will snap shut, typically in about one-tenth of a second. The edges of the lobes are fringed by stiff hair-like protrusions or cilia, which mesh together and prevent large prey from escaping. These protrusions, and the trigger hairs (also known as sensitive hairs) are likely homologous with the tentacles found in this plant's close relatives, the sundew
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
s. Scientists have concluded that the snap trap evolved from a fly-paper trap similar to that of ''Drosera''.
The holes in the meshwork allow small prey to escape, presumably because the benefit that would be obtained from them would be less than the cost of digesting them. If the prey is too small and escapes, the trap will usually reopen within 12 hours. If the prey moves around in the trap, it tightens and digestion begins more quickly.
Speed of closing can vary depending on the amount of humidity, light, size of prey, and general growing conditions. The speed with which traps close can be used as an indicator of a plant's general health. Venus flytraps are not as humidity-dependent as are some other carnivorous plants, such as ''Nepenthes
''Nepenthes'' () is a genus of carnivorous plants, also known as tropical pitcher plants, or monkey cups, in the monotypic family Nepenthaceae. The genus includes about 170 species, and numerous natural and many cultivated hybrids. They are mos ...
'', ''Cephalotus
''Cephalotus'' ( or ; Greek: ''κεφαλή'' "head", and ''οὔς''/''ὠτός'' "ear", to describe the head of the anthers) is a genus which contains one species, ''Cephalotus follicularis'' the Albany pitcher plant, a small carnivorous pi ...
'', most ''Heliamphora
The genus ''Heliamphora'' ( or ; Greek: ''helos'' "marsh" and ''amphoreus'' " amphora") contains 23 species of pitcher plants endemic to South America.McPherson, S., A. Wistuba, A. Fleischmann & J. Nerz 2011. '' Sarraceniaceae of South America''. ...
'', and some ''Drosera
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
''.
The Venus flytrap exhibits variations in petiole shape and length and whether the leaf lies flat on the ground or extends up at an angle of about 40–60 degrees. The four major forms are: 'typica', the most common, with broad decumbent petioles; 'erecta', with leaves at a 45-degree angle; 'linearis', with narrow petioles and leaves at 45 degrees; and 'filiformis', with extremely narrow or linear petioles. Except for 'filiformis', all of these can be stages in leaf production of any plant depending on season (decumbent in summer versus short versus semi-erect in spring), length of photoperiod (long petioles in spring versus short in summer), and intensity of light (wide petioles in low light intensity versus narrow in brighter light).
The plant also has a flower on top of a long stem, about 6 inches long. The flower is pollinated from various flying insects such as sweat bees, longhorn beetle
The longhorn beetles (Cerambycidae), also known as long-horned or longicorns, are a large family of beetles, with over 35,000 species described. Most species are characterized by extremely long antennae, which are often as long as or longer than ...
s and checkered beetles.
Habitat and distribution
Habitat
 The Venus flytrap is found in
The Venus flytrap is found in nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
- and phosphorus-poor environments, such as bogs, wet savannahs, and canebrake
A canebrake or canebreak is a thicket of any of a variety of ''Arundinaria'' grasses: '' A. gigantea'', '' A. tecta'' and '' A. appalachiana''. As a bamboo, these giant grasses grow in thickets up to 24 ft tall. ''A. gigantea'' is generally ...
s. Small in stature and slow-growing, the Venus flytrap tolerates fire well and depends on periodic burning to suppress its competition. Fire suppression threatens its future in the wild. It survives in wet sandy and peaty soils. Although it has been successfully transplanted and grown in many locales around the world, it is native only to the coastal bogs of North and South Carolina in the United States, specifically within a 100-kilometer (60 mi) radius of Wilmington, North Carolina
Wilmington is a port city in and the county seat of New Hanover County in coastal southeastern North Carolina, United States.
With a population of 115,451 at the 2020 census, it is the eighth most populous city in the state. Wilmington is the ...
. One such place is North Carolina's Green Swamp. There also appears to be a naturalized population of Venus flytraps in northern Florida
Florida is a state located in the Southeastern region of the United States. Florida is bordered to the west by the Gulf of Mexico, to the northwest by Alabama, to the north by Georgia, to the east by the Bahamas and Atlantic Ocean, and to ...
as well as an introduced population in western Washington
Washington commonly refers to:
* Washington (state), United States
* Washington, D.C., the capital of the United States
** A metonym for the federal government of the United States
** Washington metropolitan area, the metropolitan area centered o ...
.Giblin, D. Nd''Dionaea muscipula''.
Burke Museum of Natural History and Culture. The nutritional poverty of the soil is the reason it relies on such elaborate traps: insect prey provide the nitrogen for
protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
formation that the soil cannot. They tolerate mild winters, and Venus flytraps that do not go through a period of winter dormancy will weaken and die after a period of time.
They are full sun plants, usually found only in areas with less than 10% canopy cover. The habitats where it thrives are typically either too nutrient-poor for many noncarnivorous plants to survive, or frequently disturbed by fires which regularly clear vegetation and prevent a shady overstory from developing. It can be found living alongside herbaceous plants, grasses, sphagnum, and fire-dependent Arundinaria
''Arundinaria'' is a genus of bamboo in the grass family the members of which are referred to generally as cane. ''Arundinaria'' is the only bamboo native to south and southeastern North America, with a native range from Maryland south to Florid ...
bamboos. Regular fire disturbance is an important part of its habitat, required every 3–5 years in most places for ''D. muscipula'' to thrive. After fire, ''D. muscipula'' seeds germinate well in ash and sandy soil, with seedlings growing well in the open post-fire conditions. The seeds germinate immediately without a dormant period.
Distribution
''Dionaea muscipula'' occurs naturally only along the coastal plain of North and South Carolina in the U.S, with all known current sites within 90 km ofWilmington, North Carolina
Wilmington is a port city in and the county seat of New Hanover County in coastal southeastern North Carolina, United States.
With a population of 115,451 at the 2020 census, it is the eighth most populous city in the state. Wilmington is the ...
. A 1958 survey of herbaria
A herbarium (plural: herbaria) is a collection of preserved plant specimens and associated data used for scientific study.
The specimens may be whole plants or plant parts; these will usually be in dried form mounted on a sheet of paper (called ...
specimens and old documents found 259 sites where the historical record documented the presence of ''D. muscipula'', within 21 counties in North and South Carolina. As of 2019, it was considered extirpated in North Carolina in the inland counties of Moore, Robeson, and Lenoir, as well as the South Carolina coastal counties of Charleston and Georgetown. Remaining extant populations exist in North Carolina in Beaufort, Craven, Pamlico, Carteret, Jones, Onslow, Duplin, Pender, New Hanover, Brunswick, Columbus, Bladen, Sampson, Cumberland, and Hoke counties, and in South Carolina in Horry county.
Population
A large-scale survey in 2019, conducted by the North Carolina Natural Heritage Program, counted a total of 163,951 individual Venus flytraps in North Carolina and 4,876 in South Carolina, estimating a total of 302,000 individuals remaining in the wild in its native range. This represents a reduction of more than 93% from a 1979 estimate of approximately 4,500,000 individuals. A 1958 study found 259 confirmed extant or historic sites. As of 2016, there were 71 known sites where the plant could be found in the wild. Of these 71 sites, only 20 were classified as having excellent or good long-term viability.Carnivory

Prey selectivity
Most carnivorous plants selectively feed on specific prey. This selection is due to the available prey and the type of trap used by the organism. With the Venus flytrap, prey is limited to beetles, spiders and other crawling arthropods. The ''Dionaea'' diet is 33% ants, 30% spiders, 10% beetles, and 10% grasshoppers, with fewer than 5% flying insects. Given that ''Dionaea'' evolved from an ancestral form of ''Drosera'' (carnivorous plants that use a sticky trap instead of a snap trap) the reason for this evolutionary branching becomes clear. ''Drosera'' consume smaller, aerial insects, whereas ''Dionaea'' consume larger terrestrial bugs. ''Dionaea'' are able to extract more nutrients from these larger bugs. This gives ''Dionaea'' an evolutionary advantage over their ancestral sticky trap form.Mechanism of trapping

 The Venus flytrap is one of a very small group of plants capable of rapid movement, such as ''
The Venus flytrap is one of a very small group of plants capable of rapid movement, such as ''Mimosa pudica
''Mimosa pudica'' (from la, pudica "shy, bashful or shrinking"; also called sensitive plant, sleepy plant, action plant, touch-me-not, shameplant) is a creeping annual or perennial flowering plant of the pea/legume family Fabaceae. It is often g ...
'', the Telegraph plant
''Codariocalyx motorius'' (though often placed in ''Desmodium''), known as the telegraph plant, dancing plant, or semaphore plant, is a tropical Asian shrub in the Pea Family (Papillionaceae), one of a few plants capable of rapid plant movement ...
, starfruit
Carambola, also known as star fruit, is the fruit of '' Averrhoa carambola'', a species of tree native to tropical Southeast Asia. The mildly poisonous fruit is commonly consumed in parts of Brazil, Southeast Asia, South Asia, the South Pacif ...
, sundew
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
s and bladderwort
''Utricularia'', commonly and collectively called the bladderworts, is a genus of carnivorous plants consisting of approximately 233 species (precise counts differ based on classification opinions; a 2001 publication lists 215 species).Salmon, Br ...
s.
The mechanism by which the trap snaps shut involves a complex interaction between elasticity, turgor
Turgor pressure is the force within the cell that pushes the plasma membrane against the cell wall.
It is also called ''hydrostatic pressure'', and is defined as the pressure in a fluid measured at a certain point within itself when at equilibri ...
and growth. The trap only shuts when there have been two stimulations of the trigger hairs; this is to avoid inadvertent triggering of the mechanism by dust and other wind-borne debris. In the open, untripped state, the lobes are convex
Convex or convexity may refer to:
Science and technology
* Convex lens, in optics
Mathematics
* Convex set, containing the whole line segment that joins points
** Convex polygon, a polygon which encloses a convex set of points
** Convex polytop ...
(bent outwards), but in the closed state, the lobes are concave
Concave or concavity may refer to:
Science and technology
* Concave lens
* Concave mirror
Mathematics
* Concave function, the negative of a convex function
* Concave polygon, a polygon which is not convex
* Concave set
* The concavity of a ...
(forming a cavity). It is the rapid flipping of this bistable state that closes the trap, but the mechanism by which this occurs is still poorly understood. When the trigger hairs are stimulated, an action potential
An action potential occurs when the membrane potential of a specific cell location rapidly rises and falls. This depolarization then causes adjacent locations to similarly depolarize. Action potentials occur in several types of animal cells, ...
(mostly involving calcium ions—see calcium in biology) is generated, which propagates across the lobes and stimulates cells in the lobes and in the midrib
This glossary of botanical terms is a list of definitions of terms and concepts relevant to botany and plants in general. Terms of plant morphology are included here as well as at the more specific Glossary of plant morphology and Glossary o ...
between them.
It is hypothesized that there is a threshold of ion buildup for the Venus flytrap to react to stimulation. The acid growth Acid growth refers to the ability of plant cells and plant cell walls to elongate or expand quickly at low (acidic) pH. The cell wall needs to be modified in order to maintain the turgor pressure. This modification is controlled by plant hormones ...
theory states that individual cells in the outer layers of the lobes and midrib rapidly move 1H+ (hydrogen ion
A hydrogen ion is created when a hydrogen atom loses or gains an electron. A positively charged hydrogen ion (or proton) can readily combine with other particles and therefore is only seen isolated when it is in a gaseous state or a nearly particle ...
s) into their cell walls, lowering the pH and loosening the extracellular components, which allows them to swell rapidly by osmosis
Osmosis (, ) is the spontaneous net movement or diffusion of solvent molecules through a selectively-permeable membrane from a region of high water potential (region of lower solute concentration) to a region of low water potential (region of ...
, thus elongating and changing the shape of the trap lobe. Alternatively, cells in the inner layers of the lobes and midrib may rapidly secrete other ion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
s, allowing water to follow by osmosis, and the cells to collapse. Both of these mechanisms may play a role and have some experimental evidence to support them.Williams, S. E. 2002Comparative physiology of the Droseraceae ''sensu stricto'' – How do tentacles bend and traps close?
Proceedings of the 4th International Carnivorous Plant Society Conference. Tokyo, Japan. pp. 77–81. Flytraps show an example of memory in plants; the plant knows if one of its trigger hairs have been touched, and remembers this for a few seconds. If a second touch occurs during that time frame, the flytrap closes. After closing, the flytrap
counts
Count (feminine: countess) is a historical title of nobility in certain European countries, varying in relative status, generally of middling rank in the hierarchy of nobility. Pine, L. G. ''Titles: How the King Became His Majesty''. New York: ...
additional stimulations of the trigger hairs, to five total, to start the production of digesting enzymes.


Digestion
If the prey is unable to escape, it will continue to stimulate the inner surface of the lobes, and this causes a further growth response that forces the edges of the lobes together, eventually sealing the trap hermetically and forming a "stomach" in whichdigestion
Digestion is the breakdown of large insoluble food molecules into small water-soluble food molecules so that they can be absorbed into the watery blood plasma. In certain organisms, these smaller substances are absorbed through the small intest ...
occurs. Release of the digestive enzymes is controlled by the hormone jasmonic acid, the same hormone that triggers the release of toxins as an anti-herbivore defense mechanism in non-carnivorous plants. (See Evolution
Evolution is change in the heritable characteristics of biological populations over successive generations. These characteristics are the expressions of genes, which are passed on from parent to offspring during reproduction. Variation ...
below) Once the digestive glands in the leaf lobes have been activated, digestion is catalysed
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
by hydrolase
Hydrolase is a class of enzyme that commonly perform as biochemical catalysts that use water to break a chemical bond, which typically results in dividing a larger molecule into smaller molecules. Some common examples of hydrolase enzymes are este ...
enzymes secreted by the glands. One of these enzymes includes GH18 chitinase, which breaks down chitin-containing exoskeleton of trapped insects. Synthesis of this enzyme begins with at least five action potentials, which will stimulate transcription of chitinase.
Oxidative protein modification is likely to be a pre-digestive mechanism used by ''Dionaea muscipula''. Aqueous leaf extracts have been found to contain quinone
The quinones are a class of organic compounds that are formally "derived from aromatic compounds uch as benzene or naphthaleneby conversion of an even number of –CH= groups into –C(=O)– groups with any necessary rearrangement of double ...
s such as the naphthoquinone
Naphthoquinones constitute a class of organic compounds structurally related to naphthalene. Two isomers are common for the parent naphthoquinones:
* 1,2-Naphthoquinone
* 1,4-Naphthoquinone
Natural products
* Alkannin
* Hexahydroxy-1,4-napht ...
plumbagin
Plumbagin or 5-hydroxy-2-methyl-1,4-naphthoquinone is an organic compound with the chemical formula . It is regarded as a toxin and it is genotoxic and mutagenic.
Plumbagin is a yellow dye,NADH
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an aden ...
-dependent diaphorases to produce superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of t ...
and hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ...
upon autoxidation
Autoxidation (sometimes auto-oxidation) refers to oxidations brought about by reactions with oxygen at normal temperatures, without the intervention of flame or electric spark. The term is usually used to describe the gradual degradation of organi ...
. Such oxidative modification could rupture animal cell membranes. Plumbagin
Plumbagin or 5-hydroxy-2-methyl-1,4-naphthoquinone is an organic compound with the chemical formula . It is regarded as a toxin and it is genotoxic and mutagenic.
Plumbagin is a yellow dye,apoptosis
Apoptosis (from grc, ἀπόπτωσις, apóptōsis, 'falling off') is a form of programmed cell death that occurs in multicellular organisms. Biochemical events lead to characteristic cell changes (morphology) and death. These changes incl ...
, associated with the regulation of the Bcl-2
Bcl-2 (B-cell lymphoma 2), encoded in humans by the ''BCL2'' gene, is the founding member of the Bcl-2 family of regulator proteins that regulate cell death (apoptosis), by either inhibiting (anti-apoptotic) or inducing (pro-apoptotic) apoptosis. ...
family of proteins. When the ''Dionaea'' extracts were pre-incubated with diaphorases and NADH in the presence of serum albumin
Serum albumin, often referred to simply as blood albumin, is an albumin (a type of globular protein) found in vertebrate blood. Human serum albumin is encoded by the ''ALB'' gene. Other mammalian forms, such as bovine serum albumin, are chemical ...
(SA), subsequent tryptic
Trypsin is an enzyme in the first section of the small intestine that starts the digestion of protein molecules by cutting these long chains of amino acids into smaller pieces. It is a serine protease from the PA clan superfamily, found in the dige ...
digestion of SA was facilitated. Since the secretory gland
Exocrine glands are glands that secrete substances on to an epithelial surface by way of a duct. Examples of exocrine glands include sweat, salivary, mammary, ceruminous, lacrimal, sebaceous, prostate and mucous. Exocrine glands are one of two ...
s of Droseraceae
Droseraceae is a family of carnivorous flowering plants, also known as the sundew family. It consists of approximately 180 species in three extant genera. Representatives of the Droseraceae are found on all continents except Antarctica.
Descr ...
contain protease
A protease (also called a peptidase, proteinase, or proteolytic enzyme) is an enzyme that catalyzes (increases reaction rate or "speeds up") proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the ...
s and possibly other degradative enzyme A degradative enzyme is an enzyme (in a broader sense a protein) which degrades biological molecules. Some examples of degradative enzymes:
*Lipase, which digests lipids,
*Carbohydrases, which digest carbohydrates (e.g., sugars),
*Proteases, which d ...
s, it may be that the presence of oxygen-activating redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate (chemistry), substrate change. Oxidation is the loss of Electron, electrons or an increase in the oxidation state, while reduction ...
cofactors function as extracellular pre-digestive oxidant
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
s to render membrane-bound proteins of the prey (insect
Insects (from Latin ') are pancrustacean hexapod invertebrates of the class Insecta. They are the largest group within the arthropod phylum. Insects have a chitinous exoskeleton, a three-part body ( head, thorax and abdomen), three pairs ...
s) more susceptible to proteolytic
Proteolysis is the breakdown of proteins into smaller polypeptides or amino acids. Uncatalysed, the hydrolysis of peptide bonds is extremely slow, taking hundreds of years. Proteolysis is typically catalysed by cellular enzymes called protease ...
attacks.
Digestion takes about ten days, after which the prey is reduced to a husk of chitin
Chitin ( C8 H13 O5 N)n ( ) is a long-chain polymer of ''N''-acetylglucosamine, an amide derivative of glucose. Chitin is probably the second most abundant polysaccharide in nature (behind only cellulose); an estimated 1 billion tons of chit ...
. The trap then reopens, and is ready for reuse.
Evolution
 Carnivory in plants is a very specialized form of
Carnivory in plants is a very specialized form of foliar feeding Foliar feeding is a technique of feeding plants by applying liquid fertilizer directly to the leaves. Plants are able to absorb essential elements through their leaves. The absorption takes place through their stomata and also through their epidermi ...
, and is an adaptation found in several plants that grow in nutrient-poor soil. Carnivorous traps were naturally selected to allow these organisms to compensate for the nutrient deficiencies of their harsh environments and compensate for the reduced photosynthetic benefit. Phylogenetic studies have shown that carnivory in plants is a common adaptation in habitats with abundant sunlight and water but scarce nutrients. Carnivory has evolved independently six times in the angiosperms based on extant species, with likely many more carnivorous plant lineages now extinct.
The "snap trap" mechanism characteristic of ''Dionaea'' is shared with only one other carnivorous plant genus, ''Aldrovanda
''Aldrovanda'' is a genus of carnivorous plants encompassing one extant species (''Aldrovanda vesiculosa'', the waterwheel plant) and numerous extinct taxa. The genus is named in honor of the Italian naturalist Ulisse Aldrovandi, the founder of ...
''. For most of the 20th century, this relationship was thought to be coincidental, more precisely an example of convergent evolution
Convergent evolution is the independent evolution of similar features in species of different periods or epochs in time. Convergent evolution creates analogous structures that have similar form or function but were not present in the last com ...
. Some phylogenetic studies even suggested that the closest living relatives of ''Aldrovanda'' were the sundew
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
s. It was not until 2002 that a molecular evolutionary study, by analyzing combined nuclear
Nuclear may refer to:
Physics
Relating to the nucleus of the atom:
* Nuclear engineering
*Nuclear physics
*Nuclear power
*Nuclear reactor
*Nuclear weapon
*Nuclear medicine
*Radiation therapy
*Nuclear warfare
Mathematics
*Nuclear space
*Nuclear ...
and chloroplast DNA
Chloroplast DNA (cpDNA) is the DNA located in chloroplasts, which are photosynthetic organelles located within the cells of some eukaryotic organisms. Chloroplasts, like other types of plastid, contain a genome separate from that in the cell nu ...
sequences, indicated that ''Dionaea'' and ''Aldrovanda'' were closely related and that the snap trap mechanism evolved only once in a common ancestor
Common descent is a concept in evolutionary biology applicable when one species is the ancestor of two or more species later in time. All living beings are in fact descendants of a unique ancestor commonly referred to as the last universal comm ...
of the two genera.
A 2009 study presented evidence for the evolution of snap traps of ''Dionaea'' and ''Aldrovanda'' from a flypaper trap like ''Drosera regia'', based on molecular data
In genetics and biochemistry, sequencing means to determine the primary structure (sometimes incorrectly called the primary sequence) of an unbranched biopolymer. Sequencing results in a symbolic linear depiction known as a sequence which succi ...
. The molecular and physiological data imply that ''Dionaea'' and ''Aldrovanda'' snap traps evolved from the flypaper traps of a common ancestor with ''Drosera
''Drosera'', which is commonly known as the sundews, is one of the largest genera of carnivorous plants, with at least 194 species. 2 volumes. These members of the family Droseraceae lure, capture, and digest insects using stalked mucilaginou ...
''. Pre-adaptations to the evolution of snap traps were identified in several species of ''Drosera'', such as rapid leaf and tentacle movement. The model proposes that plant carnivory by snap trap evolved from the flypaper traps, driven by increasing prey size. Bigger prey provides greater nutritional value, but large insects can easily escape the sticky mucilage of flypaper traps; the evolution of snap traps would therefore prevent escape and kleptoparasitism
Kleptoparasitism (etymologically, parasitism by theft) is a form of feeding in which one animal deliberately takes food from another. The strategy is evolutionarily stable when stealing is less costly than direct feeding, which can mean when f ...
(theft of prey captured by the plant before it can derive benefit from it), and would also permit a more complete digestion.
In 2016, a study of the expression of genes in the plant's leaves as they captured and digested prey was published in the journal, ''Genome Research
''Genome Research'' is a peer-reviewed scientific journal published by Cold Spring Harbor Laboratory Press. Disregarding review journals, Genome Research ranks 2nd in the category 'Genetics and Genomics' after Nature Genetics. The focus of the jo ...
''. The gene activation observed in the leaves of the plants gives support to the hypothesis that the carnivorous mechanisms present in the flytrap are a specially adapted version of mechanisms used by non-carnivorous plants to defend against herbivorous insects. In many non-carnivorous plants, jasmonic acid serves as a signaling molecule for the activation of defense mechanisms, such as the production of hydrolases, which can destroy chitin
Chitin ( C8 H13 O5 N)n ( ) is a long-chain polymer of ''N''-acetylglucosamine, an amide derivative of glucose. Chitin is probably the second most abundant polysaccharide in nature (behind only cellulose); an estimated 1 billion tons of chit ...
and other molecular components of insect and microbial pests. In the Venus flytrap, this same molecule has been found to be responsible for the activation of the plant's digestive glands. A few hours after the capture of prey, another set of genes is activated inside the glands, the same set of genes that is active in the roots of other plants, allowing them to absorb nutrients. The use of similar biological pathways in the traps as non-carnivorous plants use for other purposes indicates that somewhere in its evolutionary history, the Venus flytrap repurposed these genes to facilitate carnivory.
Proposed evolutionary history
Carnivorous plants are generallyherbaceous
Herbaceous plants are vascular plants that have no persistent woody stems above ground. This broad category of plants includes many perennials, and nearly all annuals and biennials.
Definitions of "herb" and "herbaceous"
The fourth edition of t ...
, and their traps the result of primary growth
In botany, secondary growth is the growth that results from cell division in the cambium (botany), cambia or lateral meristems and that causes the Plant stem, stems and roots to thicken, while primary growth is growth that occurs as a result of ...
. They generally do not form readily fossilizable structures such as thick bark or wood. As a result, there is no fossil evidence of the steps that might link ''Dionaea'' and ''Aldrovanda'', or either genus with their common ancestor, ''Drosera''. Nevertheless, it is possible to infer an evolutionary history based on phylogenetic studies of both genera. Researchers have proposed a series of steps that would ultimately result in the complex snap-trap mechanism:
* Larger insects usually walk over the plant, instead of flying to it, and are more likely to break free from sticky glands alone. Therefore, a plant with wider leaves, like '' Drosera falconeri'', must have adapted to move the trap and its stalks in directions that maximized its chance of capturing and retaining such prey—in this particular case, longitudinally. Once adequately "wrapped", escape would be more difficult.
* Evolutionary pressure then selected for plants with shorter response time, in a manner similar to ''Drosera burmannii
''Drosera burmanni'', the tropical sundew,Erickson, Rica. 1968. ''Plants of Prey in Australia''. Lamb Paterson Pty. Ltd.: Osborne Park, Western Australia. is a small, compact species in the carnivorous plant genus '' Drosera''. Its natural geogra ...
'' or '' Drosera glanduligera''. The faster the closing, the less reliant on the flypaper model the plant would be.
* As the trap became more and more active, the energy required to "wrap" the prey increased. Plants that could somehow differentiate between actual insects and random detritus/rain droplets would have an advantage, thus explaining the specialization of inner tentacles into trigger hairs.
* Ultimately, as the plant relied more on closing around the insect rather than gluing them to the leaf surface, the tentacles so evident in ''Drosera'' would lose their original function altogether, becoming the "teeth" and trigger hairs—an example of natural selection utilizing pre-existing structures for new functions.
* Completing the transition, the plant eventually developed the depressed digestive glands found inside the trap, rather than using the dews in the stalks, further differentiating it from genus ''Drosera''.
Phylogenetic studies using molecular characters place the emergence of carnivory in the ancestors of ''Dionaea muscipula'' to 85.6 million years ago, and the development of the snap-trap in the ancestors of ''Dionaea'' and its sister genus ''Aldrovanda'' to approximately 48 million years ago.
Cultivation
 Plants can be propagated by seed, taking around four to five years to reach maturity. More commonly, they are propagated by clonal division in spring or summer. Venus flytraps can also be propagated ''in vitro'' using
Plants can be propagated by seed, taking around four to five years to reach maturity. More commonly, they are propagated by clonal division in spring or summer. Venus flytraps can also be propagated ''in vitro'' using plant tissue culture
Plant tissue culture is a collection of techniques used to maintain or grow plant cells, tissues or organs under sterile conditions on a nutrient culture medium of known composition. It is widely used to produce clones of a plant in a method known ...
. Most Venus flytraps found for sale in nurseries garden centers have been produced using this method, as this is the most cost-effective way to propagate them on a large scale. Regardless of the propagation method used, the plants will live for 20 to 30 years if cultivated in the right conditions.
Cultivars
Venus flytraps are by far the most commonly recognized and cultivated carnivorous plant, and they are frequently sold as houseplants. Variouscultivar
A cultivar is a type of cultivated plant that people have selected for desired traits and when propagated retain those traits. Methods used to propagate cultivars include: division, root and stem cuttings, offsets, grafting, tissue culture, ...
s (cultivated varieties) have come into the market through tissue culture of selected genetic mutations, and these plants are raised in large quantities for commercial markets. The cultivars 'Akai Ryu and 'South West Giant' have gained the Royal Horticultural Society
The Royal Horticultural Society (RHS), founded in 1804 as the Horticultural Society of London, is the UK's leading gardening charity.
The RHS promotes horticulture through its five gardens at Wisley (Surrey), Hyde Hall (Essex), Harlow Carr (Nort ...
's Award of Garden Merit
The Award of Garden Merit (AGM) is a long-established annual award for plants by the British Royal Horticultural Society (RHS). It is based on assessment of the plants' performance under UK growing conditions.
History
The Award of Garden Merit ...
.
Conservation
Although widely cultivated for sale as a houseplant, ''D. muscipula'' has suffered a significant decline in its population in the wild. The population in its native range is estimated to have decreased 93% since 1979.Status
The species is underEndangered Species Act
The Endangered Species Act of 1973 (ESA or "The Act"; 16 U.S.C. § 1531 et seq.) is the primary law in the United States for protecting imperiled species. Designed to protect critically imperiled species from extinction as a "consequence of ec ...
review by the U.S. Fish & Wildlife Service. The current review commenced in 2018, after an initial "90-day" review found that action may be warranted. A previous review in 1993 resulted in a determination that the plant was a "Potential candidate without sufficient data on vulnerability". The IUCN Red List
The International Union for Conservation of Nature (IUCN) Red List of Threatened Species, also known as the IUCN Red List or Red Data Book, founded in 1964, is the world's most comprehensive inventory of the global conservation status of biol ...
classifies the species as "vulnerable". The State of North Carolina lists ''Dionaea muscipula'' as a species of "Special Concern–Vulnerable". The species is protected under Appendix II of the Convention on International Trade in Endangered Species
CITES (shorter name for the Convention on International Trade in Endangered Species of Wild Fauna and Flora, also known as the Washington Convention) is a multilateral treaty to protect endangered plants and animals from the threats of interna ...
(CITES) meaning international trade (including in parts and derivatives) is regulated by the CITES permitting system. NatureServe
NatureServe, Inc. is a non-profit organization based in Arlington County, Virginia, US, that provides proprietary wildlife conservation-related data, tools, and services to private and government clients, partner organizations, and the public. Nat ...
classified it as "Imperiled" (G2) in a 2018 review.
The U.S. Fish and Wildlife Service has not indicated a timeline to conclude its current review of ''Dionaea muscipula''. The Endangered Species Act specifies a two-year timeline for a species review. However, the species listing process takes 12.1 years on average.
Threats
The Venus flytrap is only found in the wild in a very particular set of conditions, requiring flat land with moist, acidic, nutrient-poor soils that receive full sun and burn frequently in forest fires, and is therefore sensitive to many types of disturbance. A 2011 review identified five categories of threats for the species: agriculture, road-building, biological resource use (poaching and lumber activities), natural systems modifications (drainage and fire suppression), and pollution (fertilizer).Habitat loss
Habitat destruction (also termed habitat loss and habitat reduction) is the process by which a natural habitat becomes incapable of supporting its native species. The organisms that previously inhabited the site are displaced or dead, thereby ...
is a major threat to the species. The human population of the coastal Carolinas is rapidly expanding. For example, Brunswick County, North Carolina, which has the largest number of Venus flytrap populations, has seen a 27% increase in its human population from 2010 to 2018. As the population grows, residential and commercial development and road building directly eliminate flytrap habitat, while site preparation that entails ditching and draining can dry out soil in surrounding areas, destroying the viability of the species. Additionally, increased recreational use of natural areas in populated areas directly destroys the plants by crushing or uprooting them.
Fire suppression
Wildfire suppression is a range of firefighting tactics used to suppress wildfires. Firefighting efforts in wild land areas require different techniques, equipment, and training from the more familiar structure fire fighting found in populated ...
is another threat to the Venus flytrap. In the absence of regular fires, shrubs and trees encroach, outcompeting the species and leading to local extirpations. ''D. muscipula'' requires fire every 3–5 years, and best thrives with annual brush fires. Although flytraps and their seeds are typically killed alongside their competition in fires, seeds from flytraps adjacent to the burnt zone propagate quickly in the ash and full sun conditions that occur post a fire disturbance. Because the mature plants and new seedlings are typically destroyed in the regular fires that are necessary to maintain their habitat, ''D. muscipulas survival relies upon adequate seed production and dispersal from outside the burnt patches back into the burnt habitat, requiring a critical mass of populations, and exposing the success of any one population to metapopulation dynamics. These dynamics make small, isolated populations particularly vulnerable to extirpation, for if there are no mature plants adjacent to the fire zone, there is no source of seeds post-fire.
Poaching
Poaching has been defined as the illegal hunting or capturing of wild animals, usually associated with land use rights.
Poaching was once performed by impoverished peasants for subsistence purposes and to supplement meager diets. It was set a ...
has been another cause of population decline. Harvesting Venus flytraps on public land became illegal in North Carolina in 1958, and since then a legal cultivation industry has formed, growing tens of thousands of flytraps in commercial greenhouse
A greenhouse (also called a glasshouse, or, if with sufficient heating, a hothouse) is a structure with walls and roof made chiefly of Transparent ceramics, transparent material, such as glass, in which plants requiring regulated climatic condit ...
s for sale as household plants. Yet in 2016, the ''New York Times'' reported that demand for wild plants still exists, which "has led to a 'Venus flytrap crime ring. In 2014, the state of North Carolina made Venus flytrap poaching a felony
A felony is traditionally considered a crime of high seriousness, whereas a misdemeanor is regarded as less serious. The term "felony" originated from English common law (from the French medieval word "félonie") to describe an offense that resu ...
. Since then, several poachers have been charged, with one man receiving 17 months in prison for poaching 970 Venus flytraps, and another man charged with 73 felony counts in 2019. Poachers may do greater harm to the wild populations than a simple count of individuals taken would indicate, as they may selectively harvest the largest plants at a site, which have more flowers and fruit and therefore generate more seeds than smaller plants.
Additionally, the species is particularly vulnerable to catastrophic climate events. Most Venus flytrap sites are only 2–4 meters (6.5 –13 feet) above sea level and are located in a region prone to hurricanes, making storm surges and rising sea levels
Rising may refer to:
* Rising, a stage in baking - see Proofing (baking technique)
*Elevation
* Short for Uprising, a rebellion
Film and TV
* "Rising" (''Stargate Atlantis''), the series premiere of the science fiction television program ''Starga ...
a long-term threat.
Designations
In 2005, the Venus flytrap was designated as the state carnivorous plant of North Carolina.In alternative medicine
Venus flytrap extract is available on the market as anherbal remedy
Herbal medicine (also herbalism) is the study of pharmacognosy and the use of medicinal plants, which are a basis of traditional medicine. With worldwide research into pharmacology, some herbal medicines have been translated into modern remedies ...
, sometimes as the prime ingredient of a patent medicine
A patent medicine, sometimes called a proprietary medicine, is an over-the-counter (nonprescription) medicine or medicinal preparation that is typically protected and advertised by a trademark and trade name (and sometimes a patent) and claimed ...
named "Carnivora". According to the American Cancer Society
The American Cancer Society (ACS) is a nationwide voluntary health organization dedicated to eliminating cancer. Established in 1913, the society is organized into six geographical regions of both medical and lay volunteers operating in more than ...
, these products are promoted in alternative medicine
Alternative medicine is any practice that aims to achieve the healing effects of medicine despite lacking biological plausibility, testability, repeatability, or evidence from clinical trials. Complementary medicine (CM), complementary and alt ...
as a treatment for a variety of human ailments including HIV
The human immunodeficiency viruses (HIV) are two species of ''Lentivirus'' (a subgroup of retrovirus) that infect humans. Over time, they cause acquired immunodeficiency syndrome (AIDS), a condition in which progressive failure of the immune ...
, Crohn's disease and skin cancer
Skin cancers are cancers that arise from the skin. They are due to the development of abnormal cells that have the ability to invade or spread to other parts of the body. There are three main types of skin cancers: basal-cell skin cancer (BCC) ...
, but available scientific evidence does not support the health claims made for Venus flytrap extract.
See also
*Carnivorous plants of North America
The North American continent is home to a wide variety of carnivorous plant species. Species from seven genera are native to the continent, and three of these genera are found nowhere else on the planet.
Genera and species
;''Catopsis''
*'' ...
*List of ineffective cancer treatments
This is a non-exhaustive list of alternative treatments that have been promoted to treat or prevent cancer in humans but which lack scientific and medical evidence of effectiveness. In many cases, there is scientific evidence that the alleged tr ...
References
External links
Images and movies of the Venus flytrap ''(Dionaea muscipula)''
at
ARKive
ARKive was a global initiative with the mission of "promoting the conservation of the world's threatened species, through the power of wildlife imagery", which it did by locating and gathering films, photographs and audio recordings of the worl ...
Venus Flytrap Growing Guide and Distribution Map
Venus flytrap origins uncovered – BBC
Criminal Podcast Episode Five: Dropping Like Flies
{{Authority control Articles containing video clips Carnivorous plants of North America Dionaea (plant) Endemic flora of the United States Flora of North Carolina Flora of South Carolina Plant cognition Plants described in 1768 Symbols of North Carolina Vulnerable plants