Seed on:
[Wikipedia]
[Google]
[Amazon]
A seed is an

 After fertilization, the
After fertilization, the
 The main components of the embryo are:
* The
The main components of the embryo are:
* The

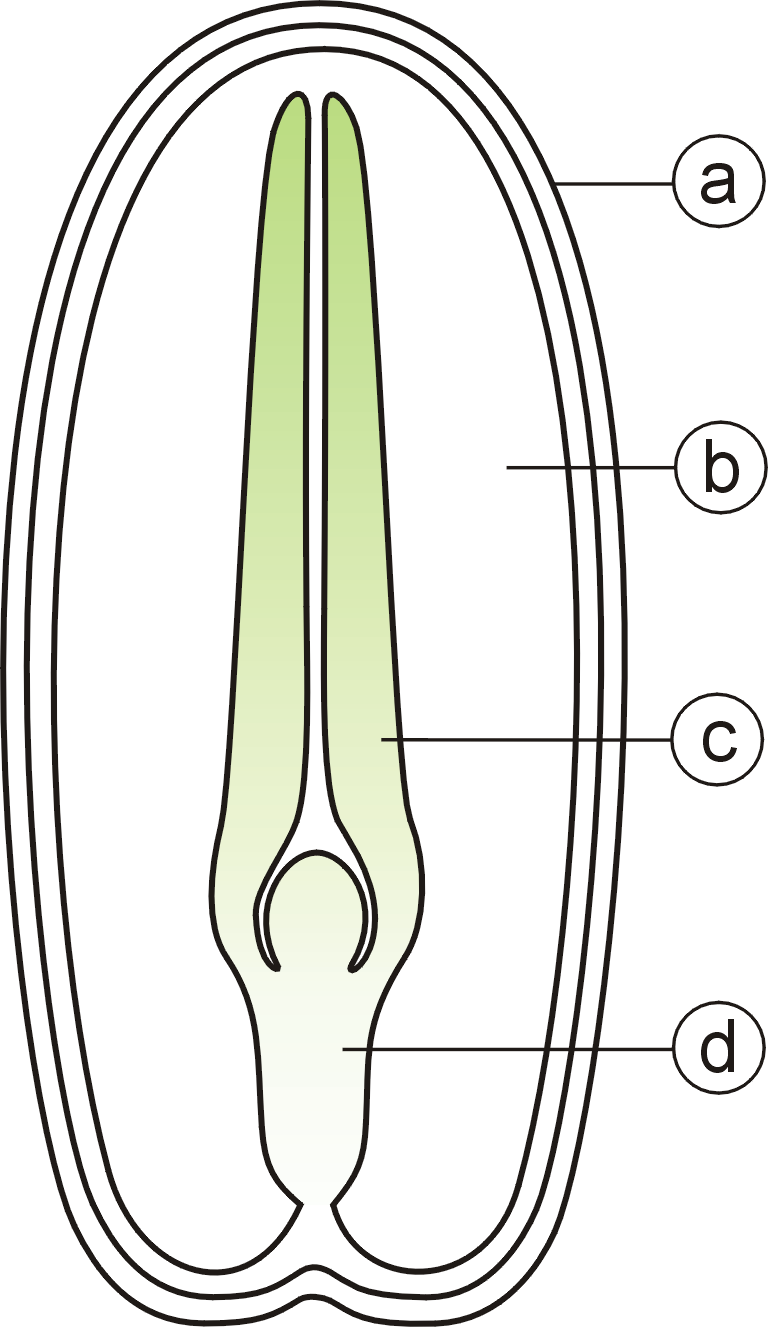 A typical seed includes two basic parts:
# an embryo;
# a seed coat.
In addition, the
A typical seed includes two basic parts:
# an embryo;
# a seed coat.
In addition, the

 The seed coat develops from the maternal tissue, the
The seed coat develops from the maternal tissue, the
 Seeds are very diverse in size. The dust-like orchid seeds are the smallest, with about one million seeds per gram; they are often embryonic seeds with immature embryos and no significant energy reserves. Orchids and a few other groups of plants are mycoheterotrophs which depend on
Seeds are very diverse in size. The dust-like orchid seeds are the smallest, with about one million seeds per gram; they are often embryonic seeds with immature embryos and no significant energy reserves. Orchids and a few other groups of plants are mycoheterotrophs which depend on

 * Some seeds (e.g.,
* Some seeds (e.g.,
 Seed germination is a process by which a seed embryo develops into a seedling. It involves the reactivation of the metabolic pathways that lead to growth and the emergence of the radicle or seed root and plumule or shoot. The emergence of the seedling above the soil surface is the next phase of the plant's growth and is called seedling establishment.
Three fundamental conditions must exist before germination can occur. (1) The embryo must be alive, called seed viability. (2) Any dormancy requirements that prevent germination must be overcome. (3) The proper environmental conditions must exist for germination.
Far red light can prevent germination.
Seed viability is the ability of the embryo to germinate and is affected by a number of different conditions. Some plants do not produce seeds that have functional complete embryos, or the seed may have no embryo at all, often called empty seeds. Predators and pathogens can damage or kill the seed while it is still in the fruit or after it is dispersed. Environmental conditions like flooding or heat can kill the seed before or during germination. The age of the seed affects its health and germination ability: since the seed has a living embryo, over time cells die and cannot be replaced. Some seeds can live for a long time before germination, while others can only survive for a short period after dispersal before they die.
Seed vigor is a measure of the quality of seed, and involves the viability of the seed, the germination percentage, germination rate, and the strength of the seedlings produced.
The germination percentage is simply the proportion of seeds that germinate from all seeds subject to the right conditions for growth. The germination rate is the length of time it takes for the seeds to germinate. Germination percentages and rates are affected by seed viability, dormancy and environmental effects that impact on the seed and seedling. In agriculture and horticulture quality seeds have high viability, measured by germination percentage plus the rate of germination. This is given as a percent of germination over a certain amount of time, 90% germination in 20 days, for example. 'Dormancy' is covered above; many plants produce seeds with varying degrees of dormancy, and different seeds from the same fruit can have different degrees of dormancy. It's possible to have seeds with no dormancy if they are dispersed right away and do not dry (if the seeds dry they go into physiological dormancy). There is great variation amongst plants and a dormant seed is still a viable seed even though the germination rate might be very low.
Environmental conditions affecting seed germination include; water, oxygen, temperature and light.
Three distinct phases of seed germination occur: water imbibition; lag phase; and
Seed germination is a process by which a seed embryo develops into a seedling. It involves the reactivation of the metabolic pathways that lead to growth and the emergence of the radicle or seed root and plumule or shoot. The emergence of the seedling above the soil surface is the next phase of the plant's growth and is called seedling establishment.
Three fundamental conditions must exist before germination can occur. (1) The embryo must be alive, called seed viability. (2) Any dormancy requirements that prevent germination must be overcome. (3) The proper environmental conditions must exist for germination.
Far red light can prevent germination.
Seed viability is the ability of the embryo to germinate and is affected by a number of different conditions. Some plants do not produce seeds that have functional complete embryos, or the seed may have no embryo at all, often called empty seeds. Predators and pathogens can damage or kill the seed while it is still in the fruit or after it is dispersed. Environmental conditions like flooding or heat can kill the seed before or during germination. The age of the seed affects its health and germination ability: since the seed has a living embryo, over time cells die and cannot be replaced. Some seeds can live for a long time before germination, while others can only survive for a short period after dispersal before they die.
Seed vigor is a measure of the quality of seed, and involves the viability of the seed, the germination percentage, germination rate, and the strength of the seedlings produced.
The germination percentage is simply the proportion of seeds that germinate from all seeds subject to the right conditions for growth. The germination rate is the length of time it takes for the seeds to germinate. Germination percentages and rates are affected by seed viability, dormancy and environmental effects that impact on the seed and seedling. In agriculture and horticulture quality seeds have high viability, measured by germination percentage plus the rate of germination. This is given as a percent of germination over a certain amount of time, 90% germination in 20 days, for example. 'Dormancy' is covered above; many plants produce seeds with varying degrees of dormancy, and different seeds from the same fruit can have different degrees of dormancy. It's possible to have seeds with no dormancy if they are dispersed right away and do not dry (if the seeds dry they go into physiological dormancy). There is great variation amongst plants and a dormant seed is still a viable seed even though the germination rate might be very low.
Environmental conditions affecting seed germination include; water, oxygen, temperature and light.
Three distinct phases of seed germination occur: water imbibition; lag phase; and
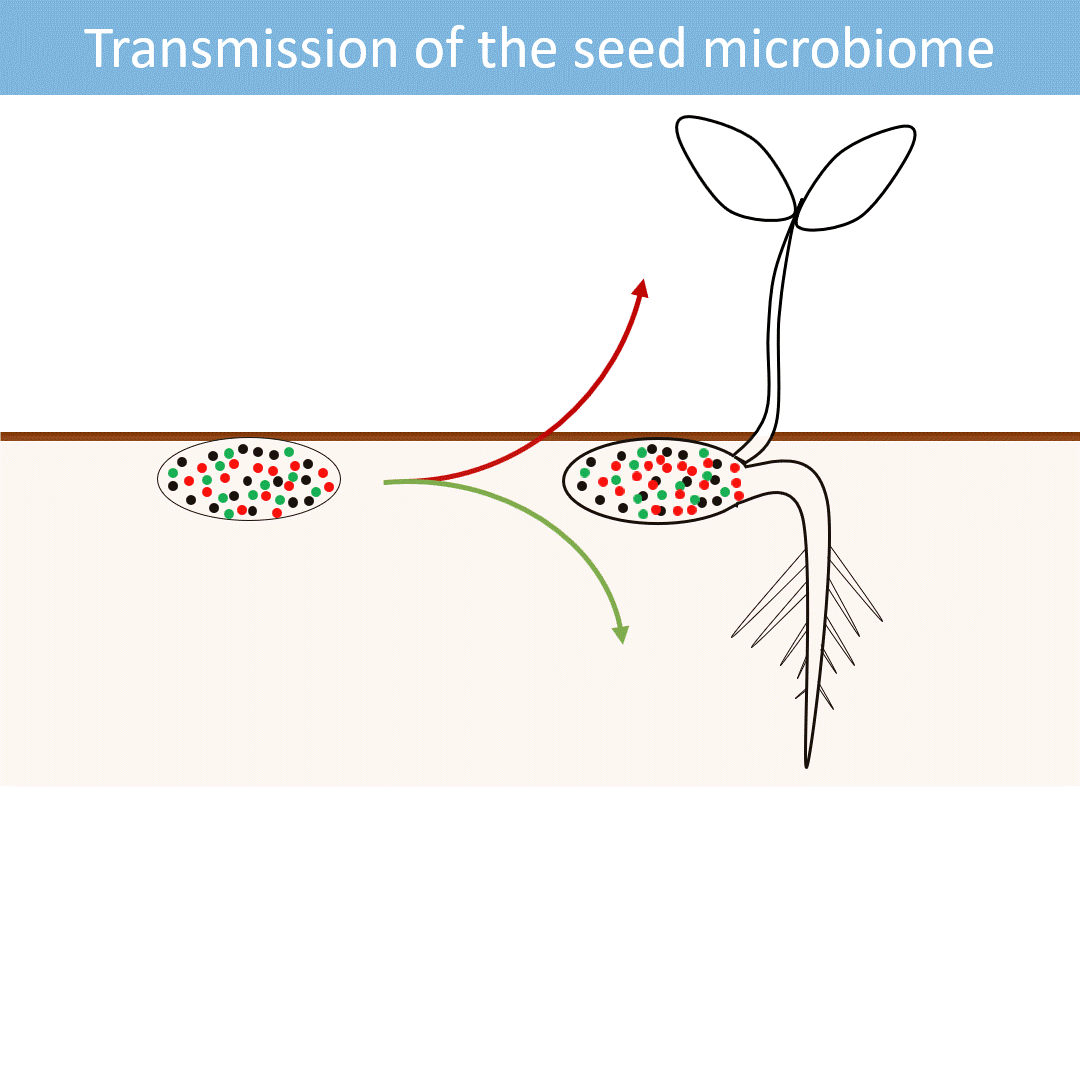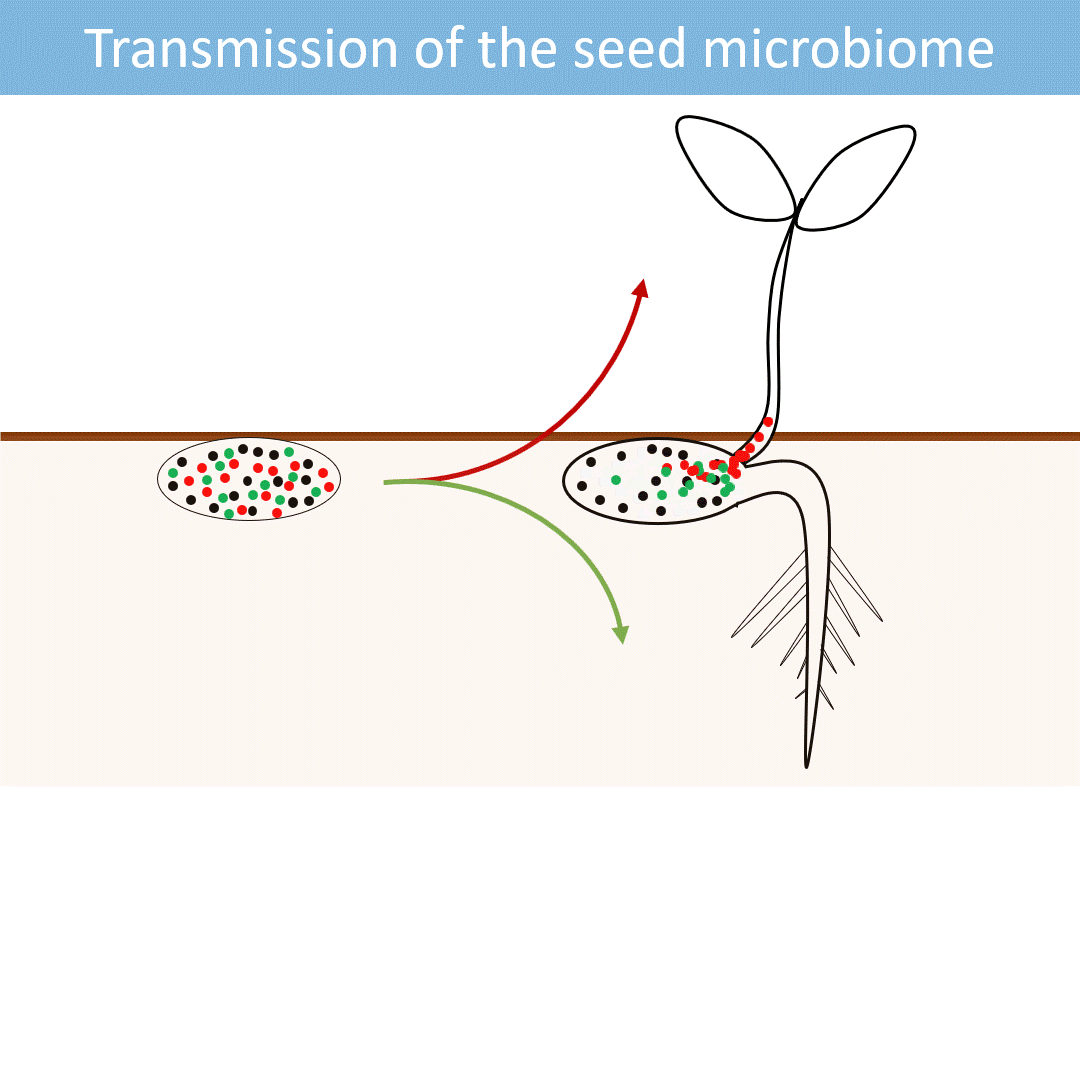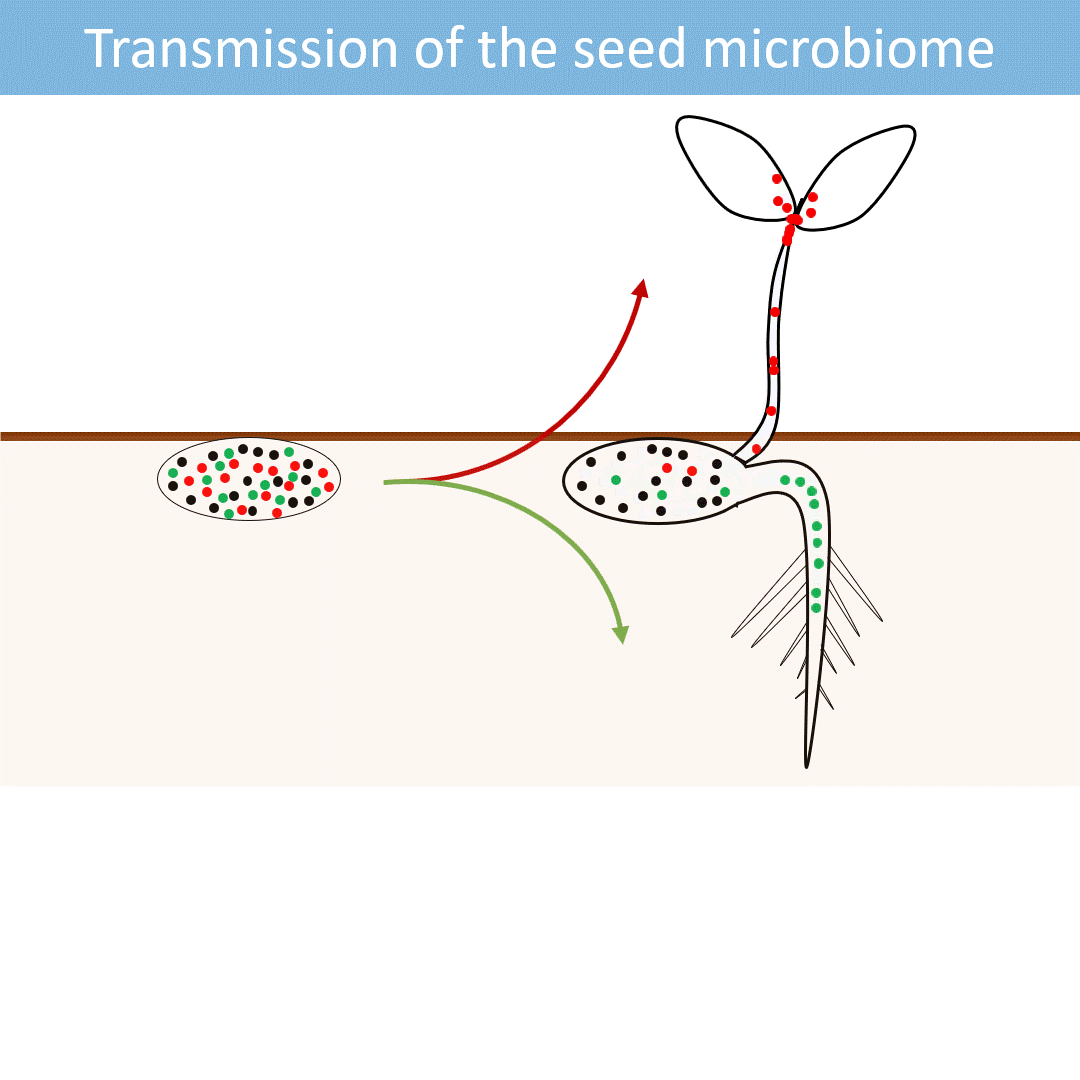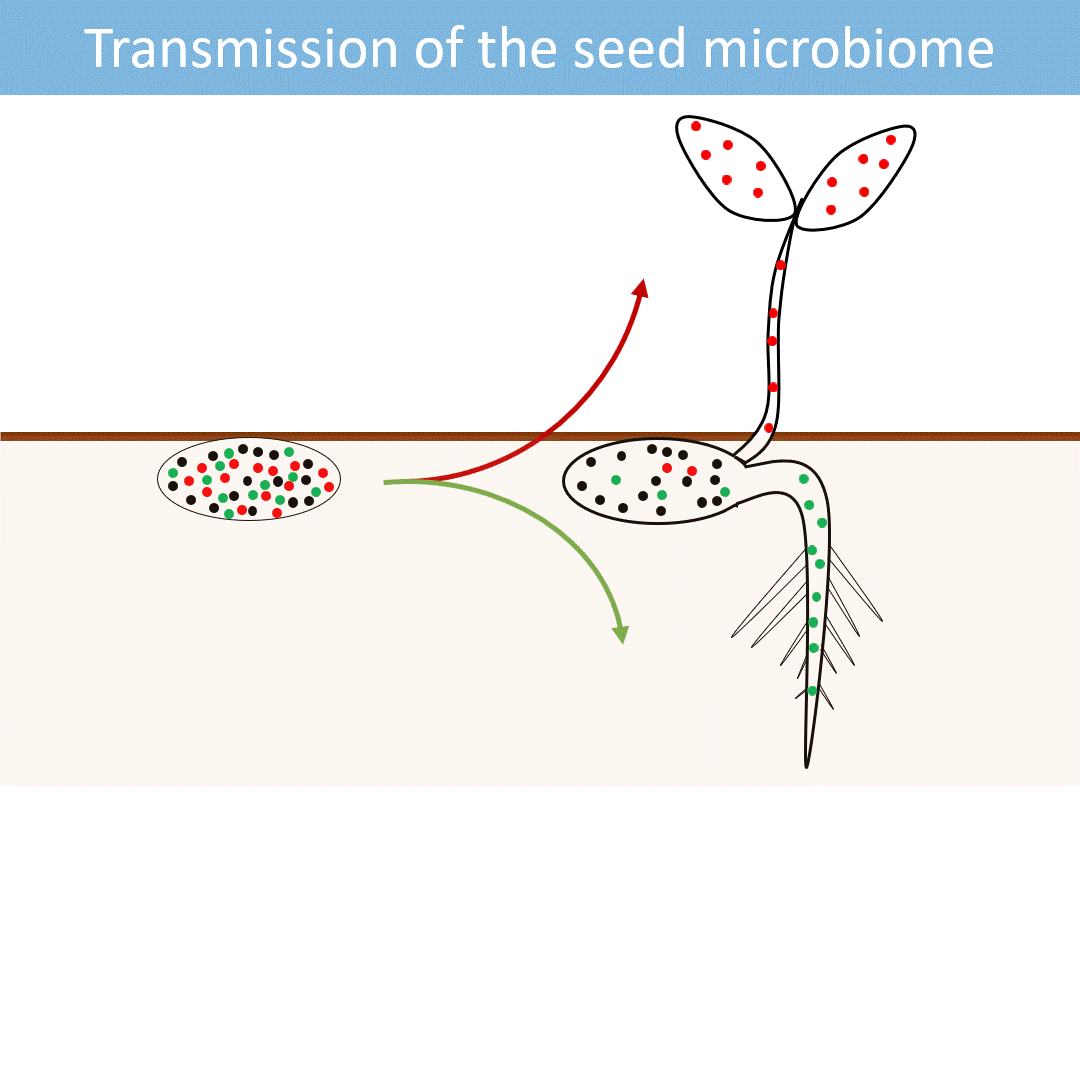 Seeds harbor a diverse microbial community. Most of these microorganisms are transmitted from the seed to the developing seedlings.
Seeds harbor a diverse microbial community. Most of these microorganisms are transmitted from the seed to the developing seedlings.

 * The Oldest viable seed, oldest viable carbon-14-dated seed that has grown into a plant was a Judean date palm seed about 2,000 years old, recovered from excavations at Herod the Great's palace on Masada in Israel. It was germinated in 2005. (A reported regeneration of ''Silene stenophylla'' (narrow-leafed campion) from material preserved for 31,800 years in the Siberian permafrost was achieved using fruit tissue, not seed.)
* The largest seed is produced by the
* The Oldest viable seed, oldest viable carbon-14-dated seed that has grown into a plant was a Judean date palm seed about 2,000 years old, recovered from excavations at Herod the Great's palace on Masada in Israel. It was germinated in 2005. (A reported regeneration of ''Silene stenophylla'' (narrow-leafed campion) from material preserved for 31,800 years in the Siberian permafrost was achieved using fruit tissue, not seed.)
* The largest seed is produced by the
A.C. Martin. The Comparative Internal Morphology of Seeds. American Midland Naturalist Vol. 36, No. 3 (Nov., 1946), pp. 513–660
M.B. McDonald, Francis Y. Kwong (eds.). Flower Seeds: Biology and Technology. CABI, 2005.
*
also available on lineVolume I
Edred John Henry Corner. The Seeds of Dicotyledons. Cambridge University Press, 1976.
United States Forest Service. Woody Plant Seed Manual. 1948
Stuppy, W. Glossary of Seed and Fruit Morphological Terms. Royal botanical gardens, Kew 2004
*[https://web.archive.org/web/20070528090503/http://www.kew.org/msbp/ The Millennium Seed Bank Project] Kew Garden's ambitious preservation project
The Svalbard Global Seed Vault
– a backup facility for the world's seed banks
Plant Physiology online: Types of Seed Dormancy and the Roles of Environmental Factors
*[https://www.blkmgroup.com/seedpaper] *[https://web.archive.org/web/20141218084446/http://theseedsite.co.uk/ The Seed Site]: collecting, storing, sowing, germinating, and exchanging seeds, with pictures of seeds, seedpods and seedlings.
Plant Fix: Check out various plant seeds and learn more information about them
{{Authority control Seeds, Botany Plant reproduction Plant sexuality
embryonic
Embryonic may refer to:
*Of or relating to an embryo
* ''Embryonic'' (album), a 2009 studio album by the Flaming Lips
*''Embryonics
''Embryonics'' is a double-CD compilation of tracks by the Australian progressive metal band, Alchemist. It w ...
plant enclosed in a protective outer covering, along with a food reserve. The formation of the seed is a part of the process of reproduction in seed plants, the spermatophytes, including the gymnosperm and angiosperm plants.
Seeds are the product of the ripened ovule, after the embryo sac is fertilized by sperm from pollen, forming a zygote. The embryo within a seed develops from the zygote, and grows within the mother plant to a certain size before growth is halted. The seed coat arises from the integuments of the ovule.
Seeds have been an important development in the reproduction and success of vegetable gymnosperm and angiosperm plants, relative to more primitive plants such as ferns, mosses and liverworts, which do not have seeds and use water-dependent means to propagate themselves. Seed plants now dominate biological niches on land, from forests to grasslands both in hot and cold climates
Climate is the long-term weather pattern in an area, typically averaged over 30 years. More rigorously, it is the mean and variability of meteorological variables over a time spanning from months to millions of years. Some of the meteorological ...
.
The term "seed" also has a general meaning that antedates the above – anything that can be sown
Sowing is the process of planting seeds. An area or object that has had seeds planted in it will be described as a sowed or sown area.
Plants which are usually sown
Among the major field crops, oats, wheat, and rye are sown, grasses and le ...
, e.g. "seed" potato
The potato is a starchy food, a tuber of the plant ''Solanum tuberosum'' and is a root vegetable native to the Americas. The plant is a perennial in the nightshade family Solanaceae.
Wild potato species can be found from the southern Unit ...
es, "seeds" of corn or sunflower "seeds". In the case of sunflower and corn "seeds", what is sown is the seed enclosed in a shell or husk, whereas the potato is a tuber
Tubers are a type of enlarged structure used as storage organs for nutrients in some plants. They are used for the plant's perennation (survival of the winter or dry months), to provide energy and nutrients for regrowth during the next growin ...
.
In the flowering plant
Flowering plants are plants that bear flowers and fruits, and form the clade Angiospermae (), commonly called angiosperms. The term "angiosperm" is derived from the Greek words ('container, vessel') and ('seed'), and refers to those plants th ...
s, the ovary ripens into a fruit
In botany, a fruit is the seed-bearing structure in flowering plants that is formed from the ovary after flowering.
Fruits are the means by which flowering plants (also known as angiosperms) disseminate their seeds. Edible fruits in particu ...
which contains the seed and serves to disseminate it. Many structures commonly referred to as "seeds" are actually dry fruits. Sunflower seeds are sometimes sold commercially while still enclosed within the hard wall of the fruit, which must be split open to reach the seed. Different groups of plants have other modifications, the so-called stone fruits (such as the peach
The peach (''Prunus persica'') is a deciduous tree first domesticated and cultivated in Zhejiang province of Eastern China. It bears edible juicy fruits with various characteristics, most called peaches and others (the glossy-skinned, non-fu ...
) have a hardened fruit layer (the endocarp
Fruit anatomy is the plant anatomy of the internal structure of fruit. Fruits are the mature ovary or ovaries of one or more flowers. They are found in three main anatomical categories: aggregate fruits, multiple fruits, and simple fruits. Aggr ...
) fused to and surrounding the actual seed. Nuts are the one-seeded, hard-shelled fruit of some plants with an indehiscent
Dehiscence is the splitting of a mature plant structure along a built-in line of weakness to release its contents. This is common among fruits, anthers and sporangia. Sometimes this involves the complete detachment of a part; structures that op ...
seed, such as an acorn or hazelnut.
History
The first land plants evolved around 468 million years ago, they reproduced using spores. The oldest seed bearing plants were gymnosperms, which had no ovaries to contain the seeds, arising sometime during the lateDevonian
The Devonian ( ) is a geologic period and system of the Paleozoic era, spanning 60.3 million years from the end of the Silurian, million years ago (Mya), to the beginning of the Carboniferous, Mya. It is named after Devon, England, whe ...
period (416 million to 358 million years ago) From these early gymnosperms, seed ferns evolved during the Carboniferous
The Carboniferous ( ) is a geologic period and system of the Paleozoic that spans 60 million years from the end of the Devonian Period million years ago ( Mya), to the beginning of the Permian Period, million years ago. The name ''Carbonifero ...
period (359 to 299 million years ago); they had ovules that were borne in a cupule, which were groups of enclosing branches likely used to protect the developing seed.
Seed production
Seeds are produced in several related groups of plants, and their manner of production distinguishes the angiosperms ("enclosed seeds") from the gymnosperms ("naked seeds"). Angiosperm seeds are produced in a hard or fleshy structure called afruit
In botany, a fruit is the seed-bearing structure in flowering plants that is formed from the ovary after flowering.
Fruits are the means by which flowering plants (also known as angiosperms) disseminate their seeds. Edible fruits in particu ...
that encloses the seeds for protection in order to secure healthy growth. Some fruits have layers of both hard and fleshy material. In gymnosperms, no special structure develops to enclose the seeds, which begin their development "naked" on the bracts of cones. However, the seeds do become covered by the cone
A cone is a three-dimensional geometric shape that tapers smoothly from a flat base (frequently, though not necessarily, circular) to a point called the apex or vertex.
A cone is formed by a set of line segments, half-lines, or lines con ...
scales as they develop in some species of conifer.
Seed production in natural plant populations varies widely from year to year in response to weather variables, insects and diseases, and internal cycles within the plants themselves. Over a 20-year period, for example, forests composed of loblolly pine
''Pinus taeda'', commonly known as loblolly pine, is one of several pines native to the Southeastern United States, from East Texas to Florida, and north to southern New Jersey. The wood industry classifies the species as a southern yellow pine. ...
and shortleaf pine
''Pinus echinata'', the shortleaf pine, is a species of pine native to the southeastern United States.
Description
The tree is variable in form, sometimes straight, sometimes crooked, with an irregular crown. The tree reaches heights of with a ...
produced from 0 to nearly 5.5 million sound pine seeds per hectare. Over this period, there were six bumper, five poor, and nine good seed crops, when evaluated for production of adequate seedling
A seedling is a young sporophyte developing out of a plant embryo from a seed. Seedling development starts with germination of the seed. A typical young seedling consists of three main parts: the radicle (embryonic root), the hypocotyl (embryo ...
s for natural forest reproduction.
Development
Angiosperm
Flowering plants are plants that bear flowers and fruits, and form the clade Angiospermae (), commonly called angiosperms. The term "angiosperm" is derived from the Greek words ('container, vessel') and ('seed'), and refers to those plants th ...
(flowering plants) seeds consist of three genetically distinct constituents: (1) the embryo formed from the zygote, (2) the endosperm, which is normally triploid, (3) the seed coat from tissue derived from the maternal tissue of the ovule. In angiosperms, the process of seed development begins with double fertilization
Double fertilization is a complex fertilization mechanism of flowering plants (angiosperms). This process involves the joining of a female gametophyte (megagametophyte, also called the embryo sac) with two male gametes (sperm). It begins when a ...
, which involves the fusion of two male gametes with the egg cell and the central cell to form the primary endosperm
The endosperm is a tissue produced inside the seeds of most of the flowering plants following double fertilization. It is triploid (meaning three chromosome sets per nucleus) in most species, which may be auxin-driven. It surrounds the embryo and ...
and the zygote. Right after fertilization, the zygote is mostly inactive, but the primary endosperm divides rapidly to form the endosperm tissue. This tissue becomes the food the young plant will consume until the roots have developed after germination.
Ovule
ovules
In seed plants, the ovule is the structure that gives rise to and contains the female reproductive cells. It consists of three parts: the ''integument'', forming its outer layer, the ''nucellus'' (or remnant of the megasporangium), and the fe ...
develop into the seeds. The ovule consists of a number of components:
* The funicle (''funiculus, funiculi'') or seed stalk which attaches the ovule to the placenta
The placenta is a temporary embryonic and later fetal organ that begins developing from the blastocyst shortly after implantation. It plays critical roles in facilitating nutrient, gas and waste exchange between the physically separate mater ...
and hence ovary or fruit wall, at the pericarp
Fruit anatomy is the plant anatomy of the internal structure of fruit. Fruits are the mature ovary or ovaries of one or more flowers. They are found in three main anatomical categories: aggregate fruits, multiple fruits, and simple fruits. Aggr ...
.
* The nucellus, the remnant of the megasporangium and main region of the ovule where the megagametophyte develops.
* The micropyle, a small pore or opening in the apex of the integument of the ovule where the pollen tube usually enters during the process of fertilization.
* The chalaza
The chalaza (; from Greek "hailstone"; plural ''chalazas'' or ''chalazae'', ) is a structure inside bird eggs and plant ovules. It attaches or suspends the yolk or nucellus within the larger structure.
In animals
In the eggs of most birds (n ...
, the base of the ovule opposite the micropyle, where integument and nucellus are joined together.
The shape of the ovules as they develop often affects the final shape of the seeds. Plants generally produce ovules of four shapes: the most common shape is called anatropous, with a curved shape. Orthotropous ovules are straight with all the parts of the ovule lined up in a long row producing an uncurved seed. Campylotropous ovules have a curved megagametophyte often giving the seed a tight "C" shape. The last ovule shape is called amphitropous, where the ovule is partly inverted and turned back 90 degrees on its stalk (the funicle or funiculus).
In the majority of flowering plants, the zygote's first division is transversely oriented in regards to the long axis, and this establishes the polarity of the embryo. The upper or chalazal pole becomes the main area of growth of the embryo, while the lower or micropylar pole produces the stalk-like suspensor that attaches to the micropyle. The suspensor absorbs and manufactures nutrients from the endosperm that are used during the embryo's growth.
Embryo
 The main components of the embryo are:
* The
The main components of the embryo are:
* The cotyledon
A cotyledon (; ; ; , gen. (), ) is a significant part of the embryo within the seed of a plant, and is defined as "the embryonic leaf in seed-bearing plants, one or more of which are the first to appear from a germinating seed." The num ...
s, the seed leaves, attached to the embryonic axis. There may be one (Monocotyledons
Monocotyledons (), commonly referred to as monocots, (Lilianae ''sensu'' Chase & Reveal) are grass and grass-like flowering plants (angiosperms), the seeds of which typically contain only one embryonic leaf, or cotyledon. They constitute one of t ...
), or two (Dicotyledons
The dicotyledons, also known as dicots (or, more rarely, dicotyls), are one of the two groups into which all the flowering plants (angiosperms) were formerly divided. The name refers to one of the typical characteristics of the group: namely, t ...
). The cotyledons are also the source of nutrients in the non-endospermic dicotyledons, in which case they replace the endosperm, and are thick and leathery. In endospermic seeds, the cotyledons are thin and papery. Dicotyledons have the point of attachment opposite one another on the axis.
* The epicotyl An epicotyl is important for the beginning stages of a plant's life. It is the region of a seedling stem above the stalks of the seed leaves of an embryo plant. It grows rapidly, showing hypogeal germination, and extends the stem above the soil surf ...
, the embryonic axis above the point of attachment of the cotyledon(s).
* The plumule, the tip of the epicotyl, and has a feathery appearance due to the presence of young leaf primordia at the apex, and will become the shoot upon germination.
* The hypocotyl
The hypocotyl (short for "hypocotyledonous stem", meaning "below seed leaf") is the stem of a germinating seedling, found below the cotyledons (seed leaves) and above the radicle ( root).
Eudicots
As the plant embryo grows at germination, it se ...
, the embryonic axis below the point of attachment of the cotyledon(s), connecting the epicotyl and the radicle, being the stem-root transition zone.
* The radicle
In botany, the radicle is the first part of a seedling (a growing plant embryo) to emerge from the seed during the process of germination. The radicle is the embryonic root of the plant, and grows downward in the soil (the shoot emerges from ...
, the basal tip of the hypocotyl, grows into the primary root.
Monocotyledonous plants have two additional structures in the form of sheaths. The plumule is covered with a coleoptile
Coleoptile is the pointed protective sheath covering the emerging shoot in monocotyledons such as grasses in which few leaf primordia and shoot apex of monocot embryo remain enclosed. The coleoptile protects the first leaf as well as the growing ...
that forms the first leaf while the radicle is covered with a coleorhiza The coleorhiza, coleorrhiza or root sheath is a protective layer of tissue that surrounds the radicle (the embryonic primary root) in monocotyledon seeds. During germination, the coleorhiza is the first part to grow out of the seed, growing through ...
that connects to the primary root and adventitious Important structures in plant development are buds, shoots, roots, leaves, and flowers; plants produce these tissues and structures throughout their life from meristems located at the tips of organs, or between mature tissues. Thus, a living plan ...
roots form the sides. Here the hypocotyl is a rudimentary axis between radicle and plumule. The seeds of corn are constructed with these structures; pericarp, scutellum (single large cotyledon) that absorbs nutrients from the endosperm, plumule, radicle, coleoptile, and coleorhiza – these last two structures are sheath-like and enclose the plumule and radicle, acting as a protective covering.
Seed coat
The maturing ovule undergoes marked changes in the integuments, generally a reduction and disorganization but occasionally a thickening. The seed coat forms from the two integuments or outer layers of cells of the ovule, which derive from tissue from the mother plant, the inner integument forms the tegmen and the outer forms the testa. (The seed coats of some monocotyledon plants, such as the grasses, are not distinct structures, but are fused with the fruit wall to form apericarp
Fruit anatomy is the plant anatomy of the internal structure of fruit. Fruits are the mature ovary or ovaries of one or more flowers. They are found in three main anatomical categories: aggregate fruits, multiple fruits, and simple fruits. Aggr ...
.) The testae of both monocots and dicots are often marked with patterns and textured markings, or have wings or tufts of hair. When the seed coat forms from only one layer, it is also called the testa, though not all such testae are homologous from one species to the next. The funiculus abscisses (detaches at fixed point – abscission zone), the scar forming an oval depression, the hilum. Anatropous ovules have a portion of the funiculus that is adnate (fused to the seed coat), and which forms a longitudinal ridge, or raphe, just above the hilum. In bitegmic ovules (e.g. ''Gossypium'' described here) both inner and outer integuments contribute to the seed coat formation. With continuing maturation the cells enlarge in the outer integument. While the inner epidermis may remain a single layer, it may also divide to produce two to three layers and accumulates starch, and is referred to as the colourless layer. By contrast, the outer epidermis becomes tanniferous. The inner integument may consist of eight to fifteen layers. (Kozlowski 1972)
As the cells enlarge, and starch is deposited in the outer layers of the pigmented zone below the outer epidermis, this zone begins to lignify, while the cells of the outer epidermis enlarge radially and their walls thicken, with nucleus and cytoplasm compressed into the outer layer. these cells which are broader on their inner surface are called palisade cells. In the inner epidermis, the cells also enlarge radially with plate like thickening of the walls. The mature inner integument has a palisade layer, a pigmented zone with 15–20 layers, while the innermost layer is known as the fringe layer. (Kozlowski 1972)
Gymnosperms
In gymnosperms, which do not form ovaries, the ovules and hence the seeds are exposed. This is the basis for their nomenclature – naked seeded plants. Two sperm cells transferred from the pollen do not develop the seed by double fertilization, but one sperm nucleus unites with the egg nucleus and the other sperm is not used. Sometimes each sperm fertilizes an egg cell and one zygote is then aborted or absorbed during early development. The seed is composed of the embryo (the result of fertilization) and tissue from the mother plant, which also form a cone around the seed in coniferous plants such aspine
A pine is any conifer tree or shrub in the genus ''Pinus'' () of the family Pinaceae. ''Pinus'' is the sole genus in the subfamily Pinoideae. The World Flora Online created by the Royal Botanic Gardens, Kew and Missouri Botanical Garden accep ...
and spruce.
Shape and appearance
A large number of terms are used to describe seed shapes, many of which are largely self-explanatory such as ''Bean-shaped'' (reniform) – resembling a kidney, with lobed ends on either side of the hilum, ''Square'' or ''Oblong'' – angular with all sides more or less equal or longer than wide, ''Triangular'' – three sided, broadest below middle, ''Elliptic'' or ''Ovate'' or ''Obovate'' – rounded at both ends, or egg shaped (ovate or obovate, broader at one end), being rounded but either symmetrical about the middle or broader below the middle or broader above the middle. Other less obvious terms include discoid (resembling a disc or plate, having both thickness and parallel faces and with a rounded margin), ellipsoid, globose (spherical
A sphere () is a geometrical object that is a three-dimensional analogue to a two-dimensional circle. A sphere is the set of points that are all at the same distance from a given point in three-dimensional space.. That given point is the ce ...
), or subglobose (Inflated, but less than spherical), lenticular, oblong, ovoid, reniform and sectoroid. Striate seeds are striped with parallel, longitudinal lines or ridges. The commonest colours are brown and black, other colours are infrequent. The surface varies from highly polished to considerably roughened. The surface may have a variety of appendages (see Seed coat), and be described by terms such as papillate
This glossary of botanical terms is a list of definitions of terms and concepts relevant to botany and plants in general. Terms of plant morphology are included here as well as at the more specific Glossary of plant morphology and Glossary o ...
or digitiform
This glossary of botanical terms is a list of definitions of terms and concepts relevant to botany and plants in general. Terms of plant morphology are included here as well as at the more specific Glossary of plant morphology and Glossary o ...
(finger-like). A seed coat with the consistency of cork
Cork or CORK may refer to:
Materials
* Cork (material), an impermeable buoyant plant product
** Cork (plug), a cylindrical or conical object used to seal a container
***Wine cork
Places Ireland
* Cork (city)
** Metropolitan Cork, also known as G ...
is referred to as suberose. Other terms include crustaceous (hard, thin or brittle).
Structure
 A typical seed includes two basic parts:
# an embryo;
# a seed coat.
In addition, the
A typical seed includes two basic parts:
# an embryo;
# a seed coat.
In addition, the endosperm
The endosperm is a tissue produced inside the seeds of most of the flowering plants following double fertilization. It is triploid (meaning three chromosome sets per nucleus) in most species, which may be auxin-driven. It surrounds the embryo and ...
forms a supply of nutrients for the embryo in most monocotyledons and the endospermic dicotyledons.
Seed types
Seeds have been considered to occur in many structurally different types (Martin 1946). These are based on a number of criteria, of which the dominant one is the embryo-to-seed size ratio. This reflects the degree to which the developing cotyledons absorb the nutrients of the endosperm, and thus obliterate it. Six types occur amongst the monocotyledons, ten in the dicotyledons, and two in the gymnosperms (linear and spatulate). This classification is based on three characteristics: embryo morphology, amount of endosperm and the position of the embryo relative to the endosperm.Embryo
In endospermic seeds, there are two distinct regions inside the seed coat, an upper and larger endosperm and a lower smaller embryo. The embryo is the fertilised ovule, an immature plant from which a new plant will grow under proper conditions. The embryo has onecotyledon
A cotyledon (; ; ; , gen. (), ) is a significant part of the embryo within the seed of a plant, and is defined as "the embryonic leaf in seed-bearing plants, one or more of which are the first to appear from a germinating seed." The num ...
or seed leaf in monocotyledon
Monocotyledons (), commonly referred to as monocots, (Lilianae ''sensu'' Chase & Reveal) are grass and grass-like flowering plants (angiosperms), the seeds of which typically contain only one embryonic leaf, or cotyledon. They constitute one of ...
s, two cotyledons in almost all dicotyledons and two or more in gymnosperms. In the fruit of grains
A grain is a small, hard, dry fruit (caryopsis) – with or without an attached hull layer – harvested for human or animal consumption. A grain crop is a grain-producing plant. The two main types of commercial grain crops are cereals and legumes ...
(caryopses) the single monocotyledon is shield shaped and hence called a scutellum. The scutellum is pressed closely against the endosperm from which it absorbs food and passes it to the growing parts. Embryo descriptors include small, straight, bent, curved, and curled.
Nutrient storage
Within the seed, there usually is a store of nutrients for theseedling
A seedling is a young sporophyte developing out of a plant embryo from a seed. Seedling development starts with germination of the seed. A typical young seedling consists of three main parts: the radicle (embryonic root), the hypocotyl (embryo ...
that will grow from the embryo. The form of the stored nutrition varies depending on the kind of plant. In angiosperms, the stored food begins as a tissue called the endosperm
The endosperm is a tissue produced inside the seeds of most of the flowering plants following double fertilization. It is triploid (meaning three chromosome sets per nucleus) in most species, which may be auxin-driven. It surrounds the embryo and ...
, which is derived from the mother plant and the pollen via double fertilization
Double fertilization is a complex fertilization mechanism of flowering plants (angiosperms). This process involves the joining of a female gametophyte (megagametophyte, also called the embryo sac) with two male gametes (sperm). It begins when a ...
. It is usually triploid
Polyploidy is a condition in which the cells of an organism have more than one pair of ( homologous) chromosomes. Most species whose cells have nuclei ( eukaryotes) are diploid, meaning they have two sets of chromosomes, where each set contain ...
, and is rich in oil
An oil is any nonpolar chemical substance that is composed primarily of hydrocarbons and is hydrophobic (does not mix with water) & lipophilic (mixes with other oils). Oils are usually flammable and surface active. Most oils are unsaturated ...
or starch, and protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
. In gymnosperms, such as conifers, the food storage tissue (also called endosperm) is part of the female gametophyte
A gametophyte () is one of the two alternation of generations, alternating multicellular organism, multicellular phases in the life cycles of plants and algae. It is a haploid multicellular organism that develops from a haploid spore that has on ...
, a haploid tissue. The endosperm is surrounded by the aleurone Aleurone (from Greek ''aleuron'', flour) is a protein found in protein granules of maturing seeds and tubers. The term also describes one of the two major cell types of the endosperm, the aleurone layer. The aleurone layer is the outermost layer of ...
layer (peripheral endosperm), filled with proteinaceous aleurone grains.
Originally, by analogy with the animal ovum
The egg cell, or ovum (plural ova), is the female reproductive cell, or gamete, in most anisogamous organisms (organisms that reproduce sexually with a larger, female gamete and a smaller, male one). The term is used when the female gamete is ...
, the outer nucellus layer (perisperm
In seed plants, the ovule is the structure that gives rise to and contains the female reproductive cells. It consists of three parts: the ''integument'', forming its outer layer, the ''nucellus'' (or remnant of the megasporangium), and the fe ...
) was referred to as albumen
Egg white is the clear liquid (also called the albumen or the glair/glaire) contained within an egg. In chickens it is formed from the layers of secretions of the anterior section of the hen's oviduct during the passage of the egg. It forms aro ...
, and the inner endosperm layer as vitellus. Although misleading, the term began to be applied to all the nutrient matter. This terminology persists in referring to endospermic seeds as "albuminous". The nature of this material is used in both describing and classifying seeds, in addition to the embryo to endosperm size ratio. The endosperm may be considered to be farinaceous (or mealy) in which the cells are filled with starch, as for instance cereal grains, or not (non-farinaceous). The endosperm may also be referred to as "fleshy" or "cartilaginous" with thicker soft cells such as coconut, but may also be oily as in ''Ricinus
''Ricinus communis'', the castor bean or castor oil plant, is a species of perennial flowering plant in the spurge family, Euphorbiaceae. It is the sole species in the monotypic genus, ''Ricinus'', and subtribe, Ricininae. The evolution of cas ...
'' (castor oil), '' Croton'' and Poppy
A poppy is a flowering plant in the subfamily Papaveroideae of the family Papaveraceae. Poppies are herbaceous plants, often grown for their colourful flowers. One species of poppy, ''Papaver somniferum'', is the source of the narcotic drug opi ...
. The endosperm is called "horny" when the cell walls are thicker such as date
Date or dates may refer to:
*Date (fruit), the fruit of the date palm (''Phoenix dactylifera'')
Social activity
*Dating, a form of courtship involving social activity, with the aim of assessing a potential partner
** Group dating
*Play date, a ...
and coffee
Coffee is a drink prepared from roasted coffee beans. Darkly colored, bitter, and slightly acidic, coffee has a stimulant, stimulating effect on humans, primarily due to its caffeine content. It is the most popular hot drink in the world.
S ...
, or "ruminated" if mottled, as in nutmeg
Nutmeg is the seed or ground spice of several species of the genus ''Myristica''. ''Myristica fragrans'' (fragrant nutmeg or true nutmeg) is a dark-leaved evergreen tree cultivated for two spices derived from its fruit: nutmeg, from its seed, an ...
, palms and Annonaceae
The Annonaceae are a family of flowering plants consisting of trees, shrubs, or rarely lianas commonly known as the custard apple family or soursop family. With 108 accepted genera and about 2400 known species, it is the largest family in the Ma ...
.
In most monocotyledons (such as grasses
Poaceae () or Gramineae () is a large and nearly ubiquitous family of monocotyledonous flowering plants commonly known as grasses. It includes the cereal grasses, bamboos and the grasses of natural grassland and species cultivated in lawns ...
and palms) and some (endospermic or albuminous) dicotyledons (such as castor bean
''Ricinus communis'', the castor bean or castor oil plant, is a species of perennial flowering plant in the spurge family, Euphorbiaceae. It is the sole species in the monotypic genus, ''Ricinus'', and subtribe, Ricininae. The evolution of ...
s) the embryo is embedded in the endosperm (and nucellus), which the seedling will use upon germination. In the non-endospermic dicotyledons the endosperm is absorbed by the embryo as the latter grows within the developing seed, and the cotyledons of the embryo become filled with stored food. At maturity, seeds of these species have no endosperm and are also referred to as exalbuminous seeds. The exalbuminous seeds include the legumes
A legume () is a plant in the family Fabaceae (or Leguminosae), or the fruit or seed of such a plant. When used as a dry grain, the seed is also called a pulse. Legumes are grown agriculturally, primarily for human consumption, for livestock fo ...
(such as bean
A bean is the seed of several plants in the family Fabaceae, which are used as vegetables for human or animal food. They can be cooked in many different ways, including boiling, frying, and baking, and are used in many traditional dishes th ...
s and pea
The pea is most commonly the small spherical seed or the seed-pod of the flowering plant species ''Pisum sativum''. Each pod contains several peas, which can be green or yellow. Botanically, pea pods are fruit, since they contain seeds and d ...
s), trees such as the oak
An oak is a tree or shrub in the genus ''Quercus'' (; Latin "oak tree") of the beech family, Fagaceae. There are approximately 500 extant species of oaks. The common name "oak" also appears in the names of species in related genera, notably ''L ...
and walnut
A walnut is the edible seed of a drupe of any tree of the genus ''Juglans'' (family Juglandaceae), particularly the Persian or English walnut, '' Juglans regia''.
Although culinarily considered a "nut" and used as such, it is not a true ...
, vegetables such as squash
Squash may refer to:
Sports
* Squash (sport), the high-speed racquet sport also known as squash racquets
* Squash (professional wrestling), an extremely one-sided match in professional wrestling
* Squash tennis, a game similar to squash but pla ...
and radish
The radish (''Raphanus raphanistrum'' subsp. ''sativus'') is an edible root vegetable of the family Brassicaceae that was domesticated in Asia prior to Roman times.
Radishes are grown and consumed throughout the world, being mostly eaten raw ...
, and sunflowers. According to Bewley and Black (1978), Brazil nut storage is in hypocotyl, this place of storage is uncommon among seeds. All gymnosperm seeds are albuminous.
Seed coat
 The seed coat develops from the maternal tissue, the
The seed coat develops from the maternal tissue, the integument
In biology, an integument is the tissue surrounding an organism's body or an organ within, such as skin, a husk, shell, germ or rind.
Etymology
The term is derived from ''integumentum'', which is Latin for "a covering". In a transferred, or ...
s, originally surrounding the ovule. The seed coat in the mature seed can be a paper-thin layer (e.g. peanut
The peanut (''Arachis hypogaea''), also known as the groundnut, goober (US), pindar (US) or monkey nut (UK), is a legume crop grown mainly for its edible Seed, seeds. It is widely grown in the tropics and subtropics, important to both small ...
) or something more substantial (e.g. thick and hard in honey locust
The honey locust (''Gleditsia triacanthos''), also known as the thorny locust or thorny honeylocust, is a deciduous tree in the family Fabaceae, native to central North America where it is mostly found in the moist soil of river valleys. Hon ...
and coconut), or fleshy as in the sarcotesta
The sarcotesta is a fleshy seedcoat, a type of testa. Examples of seeds with a sarcotesta are pomegranate and some cycad seeds. The sarcotesta of pomegranate seeds consists of epidermal cells derived from the integument
In biology, an integu ...
of pomegranate
The pomegranate (''Punica granatum'') is a fruit-bearing deciduous shrub in the family Lythraceae, subfamily Punicoideae, that grows between tall.
The pomegranate was originally described throughout the Mediterranean Basin, Mediterranean re ...
. The seed coat helps protect the embryo from mechanical injury, predators, and drying out. Depending on its development, the seed coat is either bitegmic or unitegmic. Bitegmic seeds form a testa from the outer integument and a tegmen from the inner integument while unitegmic seeds have only one integument. Usually, parts of the testa or tegmen form a hard protective mechanical layer. The mechanical layer may prevent water penetration and germination. Amongst the barriers may be the presence of lignified
Lignin is a class of complex organic polymers that form key structural materials in the support tissues of most plants. Lignins are particularly important in the formation of cell walls, especially in wood and bark, because they lend rigidity ...
sclereids
Sclereids are a reduced form of sclerenchyma cells with highly thickened, lignified cellular walls that form small bundles of durable layers of tissue in most plants.Evert, Ray F; Eichhorn, Susan E. Esau's Plant Anatomy: Meristems, Cells, and Ti ...
.
The outer integument has a number of layers, generally between four and eight organised into three layers: (a) outer epidermis, (b) outer pigmented zone of two to five layers containing tannin
Tannins (or tannoids) are a class of astringent, polyphenolic biomolecules that bind to and precipitate proteins and various other organic compounds including amino acids and alkaloids.
The term ''tannin'' (from Anglo-Norman ''tanner'' ...
and starch, and (c) inner epidermis. The endotegmen is derived from the inner epidermis of the inner integument, the exotegmen from the outer surface of the inner integument. The endotesta is derived from the inner epidermis of the outer integument, and the outer layer of the testa from the outer surface of the outer integument is referred to as the exotesta. If the exotesta is also the mechanical layer, this is called an exotestal seed, but if the mechanical layer is the endotegmen, then the seed is endotestal. The exotesta may consist of one or more rows of cells that are elongated and pallisade like (e.g. Fabaceae), hence 'palisade exotesta'.
In addition to the three basic seed parts, some seeds have an appendage, an aril
An aril (pronounced ), also called an arillus, is a specialized outgrowth from a seed that partly or completely covers the seed. An arillode or false aril is sometimes distinguished: whereas an aril grows from the attachment point of the see ...
, a fleshy outgrowth of the funicle ( funiculus), (as in yew and nutmeg
Nutmeg is the seed or ground spice of several species of the genus ''Myristica''. ''Myristica fragrans'' (fragrant nutmeg or true nutmeg) is a dark-leaved evergreen tree cultivated for two spices derived from its fruit: nutmeg, from its seed, an ...
) or an oily appendage, an elaiosome
Elaiosomes ( grc, ἔλαιον ''élaion'' "oil" + ''sóma'' "body") are fleshy structures that are attached to the seeds of many plant species. The elaiosome is rich in lipids and proteins, and may be variously shaped. Many plants have elaio ...
(as in '' Corydalis''), or hairs (trichomes). In the latter example these hairs are the source of the textile crop cotton
Cotton is a soft, fluffy staple fiber that grows in a boll, or protective case, around the seeds of the cotton plants of the genus ''Gossypium'' in the mallow family Malvaceae. The fiber is almost pure cellulose, and can contain minor perce ...
. Other seed appendages include the raphe (a ridge), wings, caruncles (a soft spongy outgrowth from the outer integument in the vicinity of the micropyle), spines, or tubercles.
A scar also may remain on the seed coat, called the hilum, where the seed was attached to the ovary wall by the funicle. Just below it is a small pore, representing the micropyle of the ovule.
Size and seed set
 Seeds are very diverse in size. The dust-like orchid seeds are the smallest, with about one million seeds per gram; they are often embryonic seeds with immature embryos and no significant energy reserves. Orchids and a few other groups of plants are mycoheterotrophs which depend on
Seeds are very diverse in size. The dust-like orchid seeds are the smallest, with about one million seeds per gram; they are often embryonic seeds with immature embryos and no significant energy reserves. Orchids and a few other groups of plants are mycoheterotrophs which depend on mycorrhizal fungi
A mycorrhiza (from Greek μύκης ', "fungus", and ῥίζα ', "root"; pl. mycorrhizae, mycorrhiza or mycorrhizas) is a symbiotic association between a fungus and a plant. The term mycorrhiza refers to the role of the fungus in the pla ...
for nutrition during germination and the early growth of the seedling. Some terrestrial orchid seedlings, in fact, spend the first few years of their lives deriving energy from the fungi and do not produce green leaves. At up to 55 pounds (25 kilograms) the largest seed is the ''coco de mer
Coco commonly refers to:
* Coco (folklore), a mythical bogeyman in many Hispano- and Lusophone nations
Coco may also refer to:
People
* Coco (given name), a first name, its shorthand, or unrelated nickname
* Coco (surname), a list of people w ...
''(Lodoicea maldivica). This indicates a 25 Billion fold difference in seed weight. Plants that produce smaller seeds can generate many more seeds per flower, while plants with larger seeds invest more resources into those seeds and normally produce fewer seeds. Small seeds are quicker to ripen and can be dispersed sooner, so autumn all blooming plants often have small seeds. Many annual plants produce great quantities of smaller seeds; this helps to ensure at least a few will end in a favorable place for growth. Herbaceous perennials and woody plants often have larger seeds; they can produce seeds over many years, and larger seeds have more energy reserves for germination and seedling growth and produce larger, more established seedlings after germination.
Functions
Seeds serve several functions for the plants that produce them. Key among these functions are nourishment of the embryo, dispersal to a new location, and dormancy during unfavorable conditions. Seeds fundamentally are means of reproduction, and most seeds are the product ofsexual reproduction
Sexual reproduction is a type of reproduction that involves a complex life cycle in which a gamete ( haploid reproductive cells, such as a sperm or egg cell) with a single set of chromosomes combines with another gamete to produce a zygote tha ...
which produces a remixing of genetic material and phenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology or physical form and structure, its developmental processes, its biochemical and physiological pr ...
variability on which natural selection
Natural selection is the differential survival and reproduction of individuals due to differences in phenotype. It is a key mechanism of evolution, the change in the heritable traits characteristic of a population over generations. Cha ...
acts. Plant seeds hold endophytic
An endophyte is an endosymbiont, often a bacterium or fungus, that lives within a plant for at least part of its life cycle without causing apparent disease. Endophytes are ubiquitous and have been found in all species of plants studied to date; h ...
microorganisms that can perform various functions, the most important of which is protection against disease.
Embryo nourishment
Seeds protect and nourish the embryo or young plant. They usually give a seedling a faster start than a sporeling from a spore, because of the larger food reserves in the seed and the multicellularity of the enclosed embryo.Dispersal
Unlike animals, plants are limited in their ability to seek out favorable conditions for life and growth. As a result, plants have evolved many ways to disperse their offspring by dispersing their seeds (see alsovegetative reproduction
Vegetative reproduction (also known as vegetative propagation, vegetative multiplication or cloning) is any form of asexual reproduction occurring in plants in which a new plant grows from a fragment or cutting of the parent plant or spec ...
). A seed must somehow "arrive" at a location and be there at a time favorable for germination and growth. When the fruits open and release their seeds in a regular way, it is called dehiscent, which is often distinctive for related groups of plants; these fruits include capsules, follicles, legume
A legume () is a plant in the family Fabaceae (or Leguminosae), or the fruit or seed of such a plant. When used as a dry grain, the seed is also called a pulse. Legumes are grown agriculturally, primarily for human consumption, for livestock f ...
s, silicle
A silique or siliqua (plural ''siliques'' or ''siliquae'') is a type of fruit (seed capsule) having two fused carpels with the length being more than three times the width. When the length is less than three times the width of the dried fruit i ...
s and silique
A silique or siliqua (plural ''siliques'' or ''siliquae'') is a type of fruit (seed capsule) having two fused carpels with the length being more than three times the width. When the length is less than three times the width of the dried fruit i ...
s. When fruits do not open and release their seeds in a regular fashion, they are called indehiscent, which include the fruits achene
An achene (; ), also sometimes called akene and occasionally achenium or achenocarp, is a type of simple dry fruit produced by many species of flowering plants. Achenes are monocarpellate (formed from one carpel) and indehiscent (they do not ope ...
s, caryopses, nuts, samaras, and utricles.
By wind (anemochory)

 * Some seeds (e.g.,
* Some seeds (e.g., pine
A pine is any conifer tree or shrub in the genus ''Pinus'' () of the family Pinaceae. ''Pinus'' is the sole genus in the subfamily Pinoideae. The World Flora Online created by the Royal Botanic Gardens, Kew and Missouri Botanical Garden accep ...
) have a wing that aids in wind dispersal.
* The dustlike seeds of orchid
Orchids are plants that belong to the family Orchidaceae (), a diverse and widespread group of flowering plants with blooms that are often colourful and fragrant.
Along with the Asteraceae, they are one of the two largest families of flowerin ...
s are carried efficiently by the wind.
* Some seeds (e.g. milkweed
''Asclepias'' is a genus of herbaceous, perennial, flowering plants known as milkweeds, named for their latex, a milky substance containing cardiac glycosides termed cardenolides, exuded where cells are damaged. Most species are toxic to humans ...
, poplar) have hairs that aid in wind dispersal.
Other seeds are enclosed in fruit structures that aid wind dispersal in similar ways:
* Dandelion
''Taraxacum'' () is a large genus of flowering plants in the family Asteraceae, which consists of species commonly known as dandelions. The scientific and hobby study of the genus is known as taraxacology. The genus is native to Eurasia and Nor ...
achenes have hairs.
* Maple
''Acer'' () is a genus of trees and shrubs commonly known as maples. The genus is placed in the family Sapindaceae.Stevens, P. F. (2001 onwards). Angiosperm Phylogeny Website. Version 9, June 2008 nd more or less continuously updated since http ...
samaras have two wings.
By water (hydrochory)
* Some plants, such as ''Mucuna
''Mucuna'' is a genus of around 100 accepted species of climbing lianas (vines) and shrubs of the family Fabaceae: tribe Phaseoleae, typically found in tropical forests.
The leaves are trifoliolate, alternate, or spiraled, and the flowers are pe ...
'' and '' Dioclea'', produce buoyant seeds termed sea-beans or drift seeds because they float in rivers to the oceans and wash up on beaches.
By animals (zoochory)
* Seeds ( burrs) with barbs or hooks (e.g.acaena
''Acaena'' is a genus of about 60 species of mainly evergreen, creeping herbaceous perennial plants and subshrubs in the family Rosaceae, native mainly to the Southern Hemisphere, notably New Zealand, Australia and South America, but with a fe ...
, burdock, dock) which attach to animal fur or feathers, and then drop off later.
* Seeds with a fleshy covering (e.g. apple
An apple is an edible fruit produced by an apple tree (''Malus domestica''). Apple fruit tree, trees are agriculture, cultivated worldwide and are the most widely grown species in the genus ''Malus''. The tree originated in Central Asia, wh ...
, cherry, juniper) are eaten by animals (bird
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweig ...
s, mammals, reptiles, fish
Fish are aquatic, craniate, gill-bearing animals that lack limbs with digits. Included in this definition are the living hagfish, lampreys, and cartilaginous and bony fish as well as various extinct related groups. Approximately 95% of li ...
) which then disperse these seeds in their droppings
Feces ( or faeces), known colloquially and in slang as poo and poop, are the solid or semi-solid remains of food that was not digested in the small intestine, and has been broken down by bacteria in the large intestine. Feces contain a relati ...
.
* Seeds ( nuts) are attractive long-term storable food resources for animals (e.g. acorns, hazelnut, walnut
A walnut is the edible seed of a drupe of any tree of the genus ''Juglans'' (family Juglandaceae), particularly the Persian or English walnut, '' Juglans regia''.
Although culinarily considered a "nut" and used as such, it is not a true ...
); the seeds are stored some distance from the parent plant, and some escape being eaten if the animal forgets them.
Myrmecochory
Myrmecochory ( (sometimes myrmechory); from grc, μύρμηξ, mýrmēks ("ant") and ''khoreíā'' ("circular dance") is seed dispersal by ants, an ecologically significant ant–plant interaction with worldwide distribution. Most myrmeco ...
is the dispersal of seeds by ant
Ants are eusocial insects of the family Formicidae and, along with the related wasps and bees, belong to the order Hymenoptera. Ants evolved from vespoid wasp ancestors in the Cretaceous period. More than 13,800 of an estimated total of ...
s. Foraging ants disperse seeds which have appendages called elaiosome
Elaiosomes ( grc, ἔλαιον ''élaion'' "oil" + ''sóma'' "body") are fleshy structures that are attached to the seeds of many plant species. The elaiosome is rich in lipids and proteins, and may be variously shaped. Many plants have elaio ...
s (e.g. bloodroot
''Sanguinaria canadensis'', bloodroot, is a perennial, herbaceous flowering plant native to eastern North America. It is the only species in the genus ''Sanguinaria'', included in the poppy family Papaveraceae, and is most closely related to ' ...
, trillium
''Trillium'' (trillium, wakerobin, toadshade, tri flower, birthroot, birthwort, and sometimes "wood lily") is a genus of about fifty flowering plant species in the family Melanthiaceae. ''Trillium'' species are native to temperate regions of No ...
s, acacias, and many species of Proteaceae). Elaiosomes are soft, fleshy structures that contain nutrients for animals that eat them. The ants carry such seeds back to their nest, where the elaiosomes are eaten. The remainder of the seed, which is hard and inedible to the ants, then germinates either within the nest or at a removal site where the seed has been discarded by the ants.Ricklefs, Robert E. (1993) ''The Economy of Nature'', 3rd ed., p. 396. (New York: W.H. Freeman). . This dispersal relationship is an example of mutualism, since the plants depend upon the ants to disperse seeds, while the ants depend upon the plants seeds for food. As a result, a drop in numbers of one partner can reduce success of the other. In South Africa
South Africa, officially the Republic of South Africa (RSA), is the southernmost country in Africa. It is bounded to the south by of coastline that stretch along the South Atlantic and Indian Oceans; to the north by the neighbouring countri ...
, the Argentine ant
The Argentine ant (''Linepithema humile''), formerly ''Iridomyrmex humilis'', is an ant native to northern Argentina, Uruguay, Paraguay, Bolivia and southern Brazil. It is an invasive species that has been established in many Mediterranean c ...
(''Linepithema humile'') has invaded
An invasion is a military offensive in which large numbers of combatants of one geopolitical entity aggressively enter territory owned by another such entity, generally with the objective of either: conquering; liberating or re-establishing con ...
and displaced native species of ants. Unlike the native ant species, Argentine ants do not collect the seeds of ''Mimetes cucullatus
''Mimetes cucullatus'' is an evergreen shrub with several, mostly not branching, upright stems of 1–2 m (3–7 ft) high, that has been assigned to the family Proteaceae. It is the most widespread and most common pagoda species that ca ...
'' or eat the elaiosomes. In areas where these ants have invaded, the numbers of ''Mimetes'' seedlings have dropped.
Dormancy
Seed dormancy has two main functions: the first is synchronizing germination with the optimal conditions for survival of the resulting seedling; the second is spreading germination of a batch of seeds over time so a catastrophe (e.g. late frosts, drought,herbivory
A herbivore is an animal anatomically and physiologically adapted to eating plant material, for example foliage or marine algae, for the main component of its diet. As a result of their plant diet, herbivorous animals typically have mouthpart ...
) does not result in the death of all offspring of a plant ( bet-hedging). Seed dormancy is defined as a seed failing to germinate under environmental conditions optimal for germination, normally when the environment is at a suitable temperature with proper soil moisture. This true dormancy or innate dormancy is therefore caused by conditions within the seed that prevent germination. Thus dormancy is a state of the seed, not of the environment. Induced dormancy, enforced dormancy or seed quiescence occurs when a seed fails to germinate because the external environmental conditions are inappropriate for germination, mostly in response to conditions being too dark or light, too cold or hot, or too dry.
Seed dormancy is not the same as seed persistence in the soil or on the plant, though even in scientific publications dormancy and persistence are often confused or used as synonyms.
Often, seed dormancy is divided into four major categories: exogenous; endogenous; combinational; and secondary. A more recent system distinguishes five classes: morphological, physiological, morphophysiological, physical, and combinational dormancy.
Exogenous dormancy is caused by conditions outside the embryo, including:
* Physical dormancy or hard seed coats occurs when seeds are impermeable to water. At dormancy break, a specialized structure, the 'water gap', is disrupted in response to environmental cues, especially temperature, so water can enter the seed and germination can occur. Plant families where physical dormancy occurs include Anacardiaceae
The Anacardiaceae, commonly known as the cashew family or sumac family, are a family of flowering plants, including about 83 genera with about 860 known species. Members of the Anacardiaceae bear fruits that are drupes and in some cases produce ...
, Cannaceae
''Canna'' or canna lily is the only genus of flowering plants in the family Cannaceae, consisting of 10 species.The Cannaceae of the World, H. Maas-van der Kamer & P.J.M. Maas, BLUMEA 53: 247-318 Cannas are not true lilies, but have been ass ...
, Convulvulaceae, Fabaceae and Malvaceae
Malvaceae, or the mallows, is a family of flowering plants estimated to contain 244 genera with 4225 known species. Well-known members of economic importance include okra, cotton, cacao and durian. There are also some genera containing familiar ...
.
* Chemical dormancy considers species that lack physiological dormancy, but where a chemical prevents germination. This chemical can be leached out of the seed by rainwater or snow melt or be deactivated somehow. Leaching of chemical inhibitors from the seed by rain water is often cited as an important cause of dormancy release in seeds of desert plants, but little evidence exists to support this claim.
Endogenous dormancy is caused by conditions within the embryo itself, including:
* In morphological dormancy, germination is prevented due to morphological characteristics of the embryo. In some species, the embryo is just a mass of cells when seeds are dispersed; it is not differentiated. Before germination can take place, both differentiation and growth of the embryo have to occur. In other species, the embryo is differentiated but not fully grown (underdeveloped) at dispersal, and embryo growth up to a species specific length is required before germination can occur. Examples of plant families where morphological dormancy occurs are Apiaceae
Apiaceae or Umbelliferae is a family of mostly aromatic flowering plants named after the type genus ''Apium'' and commonly known as the celery, carrot or parsley family, or simply as umbellifers. It is the 16th-largest family of flowering plants ...
, Cycadaceae
''Cycas'' is a genus of plants belonging to a very ancient lineage, the Cycadophyta, which are not closely related to palms, ferns, trees or any other modern group of plants. They are evergreen perennials which achieved their maximum diversity in ...
, Liliaceae
The lily family, Liliaceae, consists of about 15 genera and 610 species of flowering plants within the order Liliales. They are monocotyledonous, perennial, herbaceous, often bulbous geophytes. Plants in this family have evolved with a fair a ...
, Magnoliaceae
The Magnoliaceae () are a flowering plant family, the magnolia family, in the order Magnoliales. It consists of two genera: ''Magnolia'' and '' Liriodendron'' (tulip trees).
Unlike most angiosperms, whose flower parts are in whorls (rings), t ...
and Ranunculaceae
Ranunculaceae (buttercup or crowfoot family; Latin "little frog", from "frog") is a family of over 2,000 known species of flowering plants in 43 genera, distributed worldwide.
The largest genera are ''Ranunculus'' (600 species), ''Delphinium' ...
.Baskin, C.C. and Baskin, J.M. (1998) Seeds: Ecology, biogeography, and evolution of dormancy and germination.San Diego, Academic Press.Baskin, J.M. and Baskin, C.C. (2004) A classification system for seed dormancy. Seed Science Research 14:1–16.
* Morphophysiological dormancy includes seeds with underdeveloped embryos, and also have physiological components to dormancy. These seeds, therefore, require a dormancy-breaking treatments, as well as a period of time to develop fully grown embryos. Plant families where morphophysiological dormancy occurs include Apiaceae
Apiaceae or Umbelliferae is a family of mostly aromatic flowering plants named after the type genus ''Apium'' and commonly known as the celery, carrot or parsley family, or simply as umbellifers. It is the 16th-largest family of flowering plants ...
, Aquifoliaceae
''Ilex'' (), or holly, is a genus of over 570 species of flowering plants in the family Aquifoliaceae, and the only living genus in that family. ''Ilex'' has the most species of any woody dioecious angiosperm genus. The species are evergree ...
, Liliaceae
The lily family, Liliaceae, consists of about 15 genera and 610 species of flowering plants within the order Liliales. They are monocotyledonous, perennial, herbaceous, often bulbous geophytes. Plants in this family have evolved with a fair a ...
, Magnoliaceae
The Magnoliaceae () are a flowering plant family, the magnolia family, in the order Magnoliales. It consists of two genera: ''Magnolia'' and '' Liriodendron'' (tulip trees).
Unlike most angiosperms, whose flower parts are in whorls (rings), t ...
, Papaveraceae
The Papaveraceae are an economically important family of about 42 genera and approximately 775 known species of flowering plants in the order Ranunculales, informally known as the poppy family. The family is cosmopolitan, occurring in temperat ...
and Ranunculaceae
Ranunculaceae (buttercup or crowfoot family; Latin "little frog", from "frog") is a family of over 2,000 known species of flowering plants in 43 genera, distributed worldwide.
The largest genera are ''Ranunculus'' (600 species), ''Delphinium' ...
. Some plants with morphophysiological dormancy, such as '' Asarum'' or ''Trillium
''Trillium'' (trillium, wakerobin, toadshade, tri flower, birthroot, birthwort, and sometimes "wood lily") is a genus of about fifty flowering plant species in the family Melanthiaceae. ''Trillium'' species are native to temperate regions of No ...
'' species, have multiple types of dormancy, one affects radicle (root) growth, while the other affects plumule (shoot) growth. The terms "double dormancy" and "two-year seeds" are used for species whose seeds need two years to complete germination or at least two winters and one summer. Dormancy of the radicle (seedling root) is broken during the first winter after dispersal while dormancy of the shoot bud is broken during the second winter.
* Physiological dormancy means the embryo, due to physiological causes, cannot generate enough power to break through the seed coat, endosperm or other covering structures. Dormancy is typically broken at cool wet, warm wet, or warm dry conditions. Abscisic acid is usually the growth inhibitor in seeds, and its production can be affected by light.
**Drying, in some plants, including a number of grasses and those from seasonally arid regions, is needed before they will germinate. The seeds are released, but need to have a lower moisture content before germination can begin. If the seeds remain moist after dispersal, germination can be delayed for many months or even years. Many herbaceous plants from temperate climate zones have physiological dormancy that disappears with drying of the seeds. Other species will germinate after dispersal only under very narrow temperature ranges, but as the seeds dry, they are able to germinate over a wider temperature range.
* In seeds with combinational dormancy, the seed or fruit coat is impermeable to water and the embryo has physiological dormancy. Depending on the species, physical dormancy can be broken before or after physiological dormancy is broken.
* Secondary dormancy* is caused by conditions after the seed has been dispersed and occurs in some seeds when nondormant seed is exposed to conditions that are not favorable to germination, very often high temperatures. The mechanisms of secondary dormancy are not yet fully understood, but might involve the loss of sensitivity in receptors in the plasma membrane.
The following types of seed dormancy do not involve seed dormancy, strictly speaking, as lack of germination is prevented by the environment, not by characteristics of the seed itself (see Germination):
* Photodormancy or light sensitivity affects germination of some seeds. These photoblastic seeds need a period of darkness or light to germinate. In species with thin seed coats, light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 te ...
may be able to penetrate into the dormant embryo. The presence of light or the absence of light may trigger the germination process, inhibiting germination in some seeds buried too deeply or in others not buried in the soil.
* Thermodormancy is seed sensitivity to heat or cold. Some seeds, including cocklebur and amaranth, germinate only at high temperatures (30 °C or 86 °F); many plants that have seeds that germinate in early to midsummer have thermodormancy, so germinate only when the soil temperature is warm. Other seeds need cool soils to germinate, while others, such as celery, are inhibited when soil temperatures are too warm. Often, thermodormancy requirements disappear as the seed ages or dries.
Not all seeds undergo a period of dormancy. Seeds of some mangrove
A mangrove is a shrub or tree that grows in coastal saline water, saline or brackish water. The term is also used for tropical coastal vegetation consisting of such species. Mangroves are taxonomically diverse, as a result of convergent evoluti ...
s are viviparous; they begin to germinate while still attached to the parent. The large, heavy root allows the seed to penetrate into the ground when it falls. Many garden plant seeds will germinate readily as soon as they have water and are warm enough; though their wild ancestors may have had dormancy, these cultivated plants lack it. After many generations of selective pressure by plant breeders and gardeners, dormancy has been selected out.
For annuals, seeds are a way for the species to survive dry or cold seasons. Ephemeral plants are usually annuals that can go from seed to seed in as few as six weeks.
Persistence and seed banks
Germination
 Seed germination is a process by which a seed embryo develops into a seedling. It involves the reactivation of the metabolic pathways that lead to growth and the emergence of the radicle or seed root and plumule or shoot. The emergence of the seedling above the soil surface is the next phase of the plant's growth and is called seedling establishment.
Three fundamental conditions must exist before germination can occur. (1) The embryo must be alive, called seed viability. (2) Any dormancy requirements that prevent germination must be overcome. (3) The proper environmental conditions must exist for germination.
Far red light can prevent germination.
Seed viability is the ability of the embryo to germinate and is affected by a number of different conditions. Some plants do not produce seeds that have functional complete embryos, or the seed may have no embryo at all, often called empty seeds. Predators and pathogens can damage or kill the seed while it is still in the fruit or after it is dispersed. Environmental conditions like flooding or heat can kill the seed before or during germination. The age of the seed affects its health and germination ability: since the seed has a living embryo, over time cells die and cannot be replaced. Some seeds can live for a long time before germination, while others can only survive for a short period after dispersal before they die.
Seed vigor is a measure of the quality of seed, and involves the viability of the seed, the germination percentage, germination rate, and the strength of the seedlings produced.
The germination percentage is simply the proportion of seeds that germinate from all seeds subject to the right conditions for growth. The germination rate is the length of time it takes for the seeds to germinate. Germination percentages and rates are affected by seed viability, dormancy and environmental effects that impact on the seed and seedling. In agriculture and horticulture quality seeds have high viability, measured by germination percentage plus the rate of germination. This is given as a percent of germination over a certain amount of time, 90% germination in 20 days, for example. 'Dormancy' is covered above; many plants produce seeds with varying degrees of dormancy, and different seeds from the same fruit can have different degrees of dormancy. It's possible to have seeds with no dormancy if they are dispersed right away and do not dry (if the seeds dry they go into physiological dormancy). There is great variation amongst plants and a dormant seed is still a viable seed even though the germination rate might be very low.
Environmental conditions affecting seed germination include; water, oxygen, temperature and light.
Three distinct phases of seed germination occur: water imbibition; lag phase; and
Seed germination is a process by which a seed embryo develops into a seedling. It involves the reactivation of the metabolic pathways that lead to growth and the emergence of the radicle or seed root and plumule or shoot. The emergence of the seedling above the soil surface is the next phase of the plant's growth and is called seedling establishment.
Three fundamental conditions must exist before germination can occur. (1) The embryo must be alive, called seed viability. (2) Any dormancy requirements that prevent germination must be overcome. (3) The proper environmental conditions must exist for germination.
Far red light can prevent germination.
Seed viability is the ability of the embryo to germinate and is affected by a number of different conditions. Some plants do not produce seeds that have functional complete embryos, or the seed may have no embryo at all, often called empty seeds. Predators and pathogens can damage or kill the seed while it is still in the fruit or after it is dispersed. Environmental conditions like flooding or heat can kill the seed before or during germination. The age of the seed affects its health and germination ability: since the seed has a living embryo, over time cells die and cannot be replaced. Some seeds can live for a long time before germination, while others can only survive for a short period after dispersal before they die.
Seed vigor is a measure of the quality of seed, and involves the viability of the seed, the germination percentage, germination rate, and the strength of the seedlings produced.
The germination percentage is simply the proportion of seeds that germinate from all seeds subject to the right conditions for growth. The germination rate is the length of time it takes for the seeds to germinate. Germination percentages and rates are affected by seed viability, dormancy and environmental effects that impact on the seed and seedling. In agriculture and horticulture quality seeds have high viability, measured by germination percentage plus the rate of germination. This is given as a percent of germination over a certain amount of time, 90% germination in 20 days, for example. 'Dormancy' is covered above; many plants produce seeds with varying degrees of dormancy, and different seeds from the same fruit can have different degrees of dormancy. It's possible to have seeds with no dormancy if they are dispersed right away and do not dry (if the seeds dry they go into physiological dormancy). There is great variation amongst plants and a dormant seed is still a viable seed even though the germination rate might be very low.
Environmental conditions affecting seed germination include; water, oxygen, temperature and light.
Three distinct phases of seed germination occur: water imbibition; lag phase; and radicle
In botany, the radicle is the first part of a seedling (a growing plant embryo) to emerge from the seed during the process of germination. The radicle is the embryonic root of the plant, and grows downward in the soil (the shoot emerges from ...
emergence.
In order for the seed coat to split, the embryo must imbibe (soak up water), which causes it to swell, splitting the seed coat. However, the nature of the seed coat determines how rapidly water can penetrate and subsequently initiate germination. The rate of imbibition is dependent on the permeability of the seed coat, amount of water in the environment and the area of contact the seed has to the source of water. For some seeds, imbibing too much water too quickly can kill the seed. For some seeds, once water is imbibed the germination process cannot be stopped, and drying then becomes fatal. Other seeds can imbibe and lose water a few times without causing ill effects, but drying can cause secondary dormancy.
Repair of DNA damage
During seed dormancy, often associated with unpredictable and stressful environments,DNA damage
DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA d ...
accumulates as the seeds age. In rye seeds, the reduction of DNA integrity due to damage is associated with loss of seed viability during storage. Upon germination, seeds of ''Vicia faba
''Vicia faba'', commonly known as the broad bean, fava bean, or faba bean, is a species of vetch, a flowering plant in the pea and bean family Fabaceae. It is widely cultivated as a crop for human consumption, and also as a cover crop. Varieti ...
'' undergo DNA repair
DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA da ...
. A plant DNA ligase
In biochemistry, a ligase is an enzyme that can catalyze the joining (ligation) of two large molecules by forming a new chemical bond. This is typically via hydrolysis of a small pendant chemical group on one of the larger molecules or the enzym ...
that is involved in repair of single- and double-strand breaks during seed germination is an important determinant of seed longevity. Also, in Arabidopsis seeds, the activities of the DNA repair enzymes Poly ADP ribose polymerase
Poly (ADP-ribose) polymerase (PARP) is a family of proteins involved in a number of cellular processes such as DNA repair, genomic stability, and programmed cell death.
Members of PARP family
The PARP family comprises 17 members (10 putative) ...
s (PARP) are likely needed for successful germination. Thus DNA damages that accumulate during dormancy appear to be a problem for seed survival, and the enzymatic repair of DNA damages during germination appears to be important for seed viability.
Inducing germination
A number of different strategies are used by gardeners and horticulturists to breakseed dormancy
Seed dormancy is an evolutionary adaptation that prevents seeds from germinating during unsuitable ecological conditions that would typically lead to a low probability of seedling survival. Dormant seeds do not germinate in a specified period of ...
.
Scarification allows water and gases to penetrate into the seed; it includes methods to physically break the hard seed coats or soften them by chemicals, such as soaking in hot water or poking holes in the seed with a pin or rubbing them on sandpaper or cracking with a press or hammer. Sometimes fruits are harvested while the seeds are still immature and the seed coat is not fully developed and sown right away before the seed coat become impermeable. Under natural conditions, seed coats are worn down by rodents chewing on the seed, the seeds rubbing against rocks (seeds are moved by the wind or water currents), by undergoing freezing and thawing of surface water, or passing through an animal's digestive tract. In the latter case, the seed coat protects the seed from digestion
Digestion is the breakdown of large insoluble food molecules into small water-soluble food molecules so that they can be absorbed into the watery blood plasma. In certain organisms, these smaller substances are absorbed through the small intest ...
, while often weakening the seed coat such that the embryo is ready to sprout when it is deposited, along with a bit of fecal matter that acts as fertilizer, far from the parent plant. Microorganism
A microorganism, or microbe,, ''mikros'', "small") and ''organism'' from the el, ὀργανισμός, ''organismós'', "organism"). It is usually written as a single word but is sometimes hyphenated (''micro-organism''), especially in olde ...
s are often effective in breaking down hard seed coats and are sometimes used by people as a treatment; the seeds are stored in a moist warm sandy medium for several months under nonsterile conditions.
Stratification (botany), Stratification, also called moist-chilling, breaks down physiological dormancy, and involves the addition of moisture to the seeds so they absorb water, and they are then subjected to a period of moist chilling to after-ripen the embryo. Sowing in late summer and fall and allowing to overwinter under cool conditions is an effective way to stratify seeds; some seeds respond more favorably to periods of oscillating temperatures which are a part of the natural environment.
Leaching or the soaking in water removes chemical inhibitors in some seeds that prevent germination. Rain and melting snow naturally accomplish this task. For seeds planted in gardens, running water is best – if soaked in a container, 12 to 24 hours of soaking is sufficient. Soaking longer, especially in stagnant water, can result in oxygen starvation and seed death. Seeds with hard seed coats can be soaked in hot water to break open the impermeable cell layers that prevent water intake.
Other methods used to assist in the germination of seeds that have dormancy include prechilling, predrying, daily alternation of temperature, light exposure, potassium nitrate, the use of plant growth regulators, such as gibberellins, cytokinins, ethylene, thiourea, sodium hypochlorite, and others. Some seeds germinate best after a fire. For some seeds, fire cracks hard seed coats, while in others, chemical dormancy is broken in reaction to the presence of smoke. Liquid smoke is often used by gardeners to assist in the germination of these species.
Sterile seeds
Seeds may be sterile for few reasons: they may have been irradiated, unpollinated, cells lived past expectancy, or bred for the purpose.Evolution and origin of seeds
The issue of the origin of seed plants remains unsolved. However, more and more data tends to place this origin in the middleDevonian
The Devonian ( ) is a geologic period and system of the Paleozoic era, spanning 60.3 million years from the end of the Silurian, million years ago (Mya), to the beginning of the Carboniferous, Mya. It is named after Devon, England, whe ...
. The description in 2004 of the proto-seed ''Runcaria heinzelinii'' in the Givetian of Belgium is an indication of that ancient origin of seed-plants. As with modern ferns, most land plants before this time reproduced by sending into the air spores that would land and become whole new plants.
Taxonomists have described early "true" seeds from the upper Devonian, which probably became the theater of their true first evolutionary radiation. With this radiation came an evolution of seed size, shape, dispersal and eventually the radiation of gymnosperms and angiosperms and monocotyledon
Monocotyledons (), commonly referred to as monocots, (Lilianae ''sensu'' Chase & Reveal) are grass and grass-like flowering plants (angiosperms), the seeds of which typically contain only one embryonic leaf, or cotyledon. They constitute one of ...
s and dicotyledons. Seed plants progressively became one of the major elements of nearly all ecosystems.
True to the seed
Also called growing true, refers to plants whose seed will yield the same type of plant as the original plant. Open pollinated plants, which include heirlooms, will almost always grow true to seed if another variety does not cross-pollinate them.Seed microbiome
 Seeds harbor a diverse microbial community. Most of these microorganisms are transmitted from the seed to the developing seedlings.
Seeds harbor a diverse microbial community. Most of these microorganisms are transmitted from the seed to the developing seedlings.
Economic importance

Seed market
In the United States farmers spent $22 billion on seeds in 2018, a 35 percent increase since 2010. DowDuPont and Monsanto account for 72 percent of corn and soybean seed sales in the U.S. with the average price of a bag of GMO Sweet corn, corn seed is priced at $270.Edible seeds
Many seeds are edible and the majority of human calories comes from seeds, especially from cereals,legume
A legume () is a plant in the family Fabaceae (or Leguminosae), or the fruit or seed of such a plant. When used as a dry grain, the seed is also called a pulse. Legumes are grown agriculturally, primarily for human consumption, for livestock f ...
s and nuts. Seeds also provide most cooking oils, many beverages and spices and some important food additives. In different seeds the Embryo#Embryos of plants and animals, seed embryo or the endosperm
The endosperm is a tissue produced inside the seeds of most of the flowering plants following double fertilization. It is triploid (meaning three chromosome sets per nucleus) in most species, which may be auxin-driven. It surrounds the embryo and ...
dominates and provides most of the nutrients. The storage protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
s of the embryo and endosperm differ in their amino acid content and physical properties. For example, the gluten of wheat, important in providing the Elasticity (physics), elastic property to bread dough is strictly an endosperm protein.
Seeds are used to propagate many crops such as cereals, legumes, forestry, forest trees, turfgrasses, and pasture grasses. Particularly in developing countries, a major constraint faced is the inadequacy of the marketing channels to get the seed to poor farmers. Thus the use of farmer-retained seed remains quite common.
Seeds are also eaten by animals (seed predation), and are also fed to livestock or provided as birdseed.
Poison and food safety
While some seeds are edible, others are harmful, poisonous or deadly. Plants and seeds often contain chemical compounds to discourage herbivores and seed predation, seed predators. In some cases, these compounds simply taste bad (such as in Mustard (condiment), mustard), but other compounds are toxic or break down into toxic compounds within the digestive system. Children, being smaller than adults, are more susceptible to poisoning by plants and seeds. A deadly poison, ricin, comes from seeds of thecastor bean
''Ricinus communis'', the castor bean or castor oil plant, is a species of perennial flowering plant in the spurge family, Euphorbiaceae. It is the sole species in the monotypic genus, ''Ricinus'', and subtribe, Ricininae. The evolution of ...
. Reported lethal doses are anywhere from two to eight seeds,
though only a few deaths have been reported when castor beans have been ingested by animals.
In addition, seeds containing amygdalin – apple
An apple is an edible fruit produced by an apple tree (''Malus domestica''). Apple fruit tree, trees are agriculture, cultivated worldwide and are the most widely grown species in the genus ''Malus''. The tree originated in Central Asia, wh ...
, apricot, bitter almond, peach
The peach (''Prunus persica'') is a deciduous tree first domesticated and cultivated in Zhejiang province of Eastern China. It bears edible juicy fruits with various characteristics, most called peaches and others (the glossy-skinned, non-fu ...
, plum, cherry, quince, and others – when consumed in sufficient amounts, may cause cyanide poisoning.
Other seeds that contain poisons include annona, cotton
Cotton is a soft, fluffy staple fiber that grows in a boll, or protective case, around the seeds of the cotton plants of the genus ''Gossypium'' in the mallow family Malvaceae. The fiber is almost pure cellulose, and can contain minor perce ...
, custard apple, datura, uncooked durian, golden chain, Horse-chestnut (tree), horse-chestnut, Delphinium, larkspur, locoweed, lychee, nectarine, rambutan, rosary pea, sour sop, sugar apple, wisteria, and yew. The seeds of the strychnine tree are also poisonous, containing the poison strychnine.
The seeds of many legumes, including the common bean (''Phaseolus vulgaris''), contain proteins called lectins which can cause gastric distress if the beans are eaten without cooking. The common bean and many others, including the soybean, also contain trypsin inhibitors which interfere with the action of the digestive enzyme trypsin. Normal cooking processes degrade lectins and trypsin inhibitors to harmless forms.
Other uses
Cotton fiber grows attached to Gossypium, cotton plant seeds. Other seed fibers are from kapok tree, kapok andmilkweed
''Asclepias'' is a genus of herbaceous, perennial, flowering plants known as milkweeds, named for their latex, a milky substance containing cardiac glycosides termed cardenolides, exuded where cells are damaged. Most species are toxic to humans ...
.
Many important nonfood oils are extracted from seeds. Linseed oil is used in paints. Oil from jojoba and crambe are similar to whale oil.
Seeds are the source of some medicines including castor oil, tea tree oil and the quack cancer drug Amygdalin, Laetrile.
Many seeds have been used as beads in necklaces and rosaries including Job's tears, Chinaberry, Abrus precatorius, rosary pea, and castor bean
''Ricinus communis'', the castor bean or castor oil plant, is a species of perennial flowering plant in the spurge family, Euphorbiaceae. It is the sole species in the monotypic genus, ''Ricinus'', and subtribe, Ricininae. The evolution of ...
. However, the latter three are also poisonous.
Other seed uses include:
* Seeds once used as weights for Weighing scale, balances.
* Seeds used as toys by children, such as for the game Conkers.
* Resin from ''Clusia rosea'' seeds used to caulk boats.
* Nematicide from milkweed
''Asclepias'' is a genus of herbaceous, perennial, flowering plants known as milkweeds, named for their latex, a milky substance containing cardiac glycosides termed cardenolides, exuded where cells are damaged. Most species are toxic to humans ...
seeds.
* Cottonseed meal used as animal feed and fertilizer.
Seed records
 * The Oldest viable seed, oldest viable carbon-14-dated seed that has grown into a plant was a Judean date palm seed about 2,000 years old, recovered from excavations at Herod the Great's palace on Masada in Israel. It was germinated in 2005. (A reported regeneration of ''Silene stenophylla'' (narrow-leafed campion) from material preserved for 31,800 years in the Siberian permafrost was achieved using fruit tissue, not seed.)
* The largest seed is produced by the
* The Oldest viable seed, oldest viable carbon-14-dated seed that has grown into a plant was a Judean date palm seed about 2,000 years old, recovered from excavations at Herod the Great's palace on Masada in Israel. It was germinated in 2005. (A reported regeneration of ''Silene stenophylla'' (narrow-leafed campion) from material preserved for 31,800 years in the Siberian permafrost was achieved using fruit tissue, not seed.)
* The largest seed is produced by the coco de mer
Coco commonly refers to:
* Coco (folklore), a mythical bogeyman in many Hispano- and Lusophone nations
Coco may also refer to:
People
* Coco (given name), a first name, its shorthand, or unrelated nickname
* Coco (surname), a list of people w ...
, or "double coconut palm", ''Lodoicea maldivica''. The entire fruit
In botany, a fruit is the seed-bearing structure in flowering plants that is formed from the ovary after flowering.
Fruits are the means by which flowering plants (also known as angiosperms) disseminate their seeds. Edible fruits in particu ...
may weigh up to 23 kilograms (50 pounds) and usually contains a single seed.
* The smallest seeds are produced by epiphytic orchids. They are only 85 micrometers long, and weigh 0.81 micrograms. They have no endosperm
The endosperm is a tissue produced inside the seeds of most of the flowering plants following double fertilization. It is triploid (meaning three chromosome sets per nucleus) in most species, which may be auxin-driven. It surrounds the embryo and ...
and contain underdeveloped embryos.
* The earliest fossil seeds are around 365 million years old from the Famennian, Late Devonian
The Devonian ( ) is a geologic period and system of the Paleozoic era, spanning 60.3 million years from the end of the Silurian, million years ago (Mya), to the beginning of the Carboniferous, Mya. It is named after Devon, England, whe ...
of West Virginia. The seeds are preserved immature ovules of the plant ''Elkinsia polymorpha''.
In religion
The Book of Genesis in the Old Testament begins with an explanation of how all plant forms began:And God said, Let the earth bring forth grass, the herb yielding seed, and the fruit tree yielding fruit after his kind, whose seed is in itself, upon the earth: and it was so. And the earth brought forth grass, and herb yielding seed after its kind, and the tree yielding fruit, whose seed was in itself, after its kind: and God saw that it was good. And the evening and the morning were the third day.The Quran speaks of seed germination thus:
It is Allah Who causeth the seed-grain and the date-stone to split and sprout. He causeth the living to issue from the dead, and He is the one to cause the dead to issue from the living. That is Allah: then how are ye deluded away from the truth?Quran, Translation: Abdullah Yusuf Ali, Al-An'aam 95:6
See also
* Biological dispersal * Carpology * Genetically modified crops * List of world's largest seeds * Recalcitrant seed * Seed company * Seed enhancement * Seed library * Seed orchard * Seed paper * Seed saving * Seed testing * Seed trap * Seedbed * Soil seed bank * Selective embryo abortionReferences
Bibliography
* *A.C. Martin. The Comparative Internal Morphology of Seeds. American Midland Naturalist Vol. 36, No. 3 (Nov., 1946), pp. 513–660
M.B. McDonald, Francis Y. Kwong (eds.). Flower Seeds: Biology and Technology. CABI, 2005.
*
also available on line
Edred John Henry Corner. The Seeds of Dicotyledons. Cambridge University Press, 1976.
United States Forest Service. Woody Plant Seed Manual. 1948
Stuppy, W. Glossary of Seed and Fruit Morphological Terms. Royal botanical gardens, Kew 2004
External links
*[https://web.archive.org/web/20070528090503/http://www.kew.org/msbp/ The Millennium Seed Bank Project] Kew Garden's ambitious preservation project
The Svalbard Global Seed Vault
– a backup facility for the world's seed banks
Plant Physiology online: Types of Seed Dormancy and the Roles of Environmental Factors
*[https://www.blkmgroup.com/seedpaper] *[https://web.archive.org/web/20141218084446/http://theseedsite.co.uk/ The Seed Site]: collecting, storing, sowing, germinating, and exchanging seeds, with pictures of seeds, seedpods and seedlings.
Plant Fix: Check out various plant seeds and learn more information about them
{{Authority control Seeds, Botany Plant reproduction Plant sexuality