Reptiles, as commonly defined, are a group of
tetrapod
A tetrapod (; from Ancient Greek :wiktionary:τετρα-#Ancient Greek, τετρα- ''(tetra-)'' 'four' and :wiktionary:πούς#Ancient Greek, πούς ''(poús)'' 'foot') is any four-Limb (anatomy), limbed vertebrate animal of the clade Tetr ...
s with an
ectotherm
An ectotherm (), more commonly referred to as a "cold-blooded animal", is an animal in which internal physiological sources of heat, such as blood, are of relatively small or of quite negligible importance in controlling body temperature.Dav ...
ic metabolism and
amniotic development. Living traditional reptiles comprise four
orders:
Testudines
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
,
Crocodilia
Crocodilia () is an order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorph pseudosuchia ...
,
Squamata
Squamata (, Latin ''squamatus'', 'scaly, having scales') is the largest Order (biology), order of reptiles; most members of which are commonly known as Lizard, lizards, with the group also including Snake, snakes. With over 11,991 species, it i ...
, and
Rhynchocephalia. About 12,000 living species of reptiles are listed in the
Reptile Database
The Reptile Database is a scientific database that collects taxonomic information on all living reptile species (i.e. no fossil species such as dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared ...
. The study of the traditional reptile orders, customarily in combination with the study of modern
amphibian
Amphibians are ectothermic, anamniote, anamniotic, tetrapod, four-limbed vertebrate animals that constitute the class (biology), class Amphibia. In its broadest sense, it is a paraphyletic group encompassing all Tetrapod, tetrapods, but excl ...
s, is called
herpetology
Herpetology (from Ancient Greek ἑρπετόν ''herpetón'', meaning "reptile" or "creeping animal") is a branch of zoology concerned with the study of amphibians (including frogs, salamanders, and caecilians (Gymnophiona)) and reptiles (in ...
.
Reptiles have been subject to several conflicting
taxonomic definitions.
In
Linnaean taxonomy
Linnaean taxonomy can mean either of two related concepts:
# The particular form of biological classification (taxonomy) set up by Carl Linnaeus, as set forth in his ''Systema Naturae'' (1735) and subsequent works. In the taxonomy of Linnaeus th ...
, reptiles are gathered together under the
class
Class, Classes, or The Class may refer to:
Common uses not otherwise categorized
* Class (biology), a taxonomic rank
* Class (knowledge representation), a collection of individuals or objects
* Class (philosophy), an analytical concept used d ...
Reptilia ( ), which corresponds to common usage. Modern
cladistic taxonomy regards that group as
paraphyletic
Paraphyly is a taxonomic term describing a grouping that consists of the grouping's last common ancestor and some but not all of its descendant lineages. The grouping is said to be paraphyletic ''with respect to'' the excluded subgroups. In co ...
, since
genetic and
paleontological evidence has determined that
bird
Birds are a group of warm-blooded vertebrates constituting the class (biology), class Aves (), characterised by feathers, toothless beaked jaws, the Oviparity, laying of Eggshell, hard-shelled eggs, a high Metabolism, metabolic rate, a fou ...
s (class Aves), as members of
Dinosauria, are more closely related to living
crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s than to other reptiles, and are thus nested among reptiles from an evolutionary perspective. Many cladistic systems therefore redefine Reptilia as a
clade
In biology, a clade (), also known as a Monophyly, monophyletic group or natural group, is a group of organisms that is composed of a common ancestor and all of its descendants. Clades are the fundamental unit of cladistics, a modern approach t ...
(
monophyletic
In biological cladistics for the classification of organisms, monophyly is the condition of a taxonomic grouping being a clade – that is, a grouping of organisms which meets these criteria:
# the grouping contains its own most recent co ...
group) including birds, though the precise definition of this clade varies between authors.
Others prioritize the clade
Sauropsida
Sauropsida (Greek language, Greek for "lizard faces") is a clade of amniotes, broadly equivalent to the Class (biology), class Reptile, Reptilia, though typically used in a broader sense to also include extinct stem-group relatives of modern repti ...
, which typically refers to all
amniote
Amniotes are tetrapod vertebrate animals belonging to the clade Amniota, a large group that comprises the vast majority of living terrestrial animal, terrestrial and semiaquatic vertebrates. Amniotes evolution, evolved from amphibious Stem tet ...
s more closely related to modern reptiles than to
mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s.
The earliest known proto-reptiles originated from the
Carboniferous
The Carboniferous ( ) is a Geologic time scale, geologic period and System (stratigraphy), system of the Paleozoic era (geology), era that spans 60 million years, from the end of the Devonian Period Ma (million years ago) to the beginning of the ...
period, having evolved from advanced
reptiliomorph tetrapods which became increasingly adapted to life on dry land. The earliest known
eureptile ("true reptile") was ''
Hylonomus'', a small and superficially lizard-like animal which lived in
Nova Scotia
Nova Scotia is a Provinces and territories of Canada, province of Canada, located on its east coast. It is one of the three Maritime Canada, Maritime provinces and Population of Canada by province and territory, most populous province in Atlan ...
during the
Bashkirian
The Bashkirian is in the International Commission on Stratigraphy geologic timescale the lowest stage (stratigraphy), stage or oldest age (geology), age of the Pennsylvanian (geology), Pennsylvanian. The Bashkirian age lasted from to Mega annu ...
age of the
Late Carboniferous
Late or LATE may refer to:
Everyday usage
* Tardy, or late, not being on time
* Late (or the late) may refer to a person who is dead
Music
* Late (The 77s album), ''Late'' (The 77s album), 2000
* Late (Alvin Batiste album), 1993
* Late!, a pseudo ...
, around .
[ Genetic and fossil data argues that the two largest lineages of reptiles, Archosauromorpha (crocodilians, birds, and kin) and Lepidosauromorpha (lizards, and kin), diverged during the ]Permian
The Permian ( ) is a geologic period and System (stratigraphy), stratigraphic system which spans 47 million years, from the end of the Carboniferous Period million years ago (Mya), to the beginning of the Triassic Period 251.902 Mya. It is the s ...
period. In addition to the living reptiles, there are many diverse groups that are now extinct
Extinction is the termination of an organism by the death of its Endling, last member. A taxon may become Functional extinction, functionally extinct before the death of its last member if it loses the capacity to Reproduction, reproduce and ...
, in some cases due to mass extinction events. In particular, the Cretaceous–Paleogene extinction event
The Cretaceous–Paleogene (K–Pg) extinction event, also known as the K–T extinction, was the extinction event, mass extinction of three-quarters of the plant and animal species on Earth approximately 66 million years ago. The event cau ...
wiped out the pterosaur
Pterosaurs are an extinct clade of flying reptiles in the order Pterosauria. They existed during most of the Mesozoic: from the Late Triassic to the end of the Cretaceous (228 million to 66 million years ago). Pterosaurs are the earli ...
s, plesiosaurs
The Plesiosauria or plesiosaurs are an Order (biology), order or clade of extinct Mesozoic marine reptiles, belonging to the Sauropterygia.
Plesiosaurs first appeared in the latest Triassic Period (geology), Period, possibly in the Rhaetian st ...
, and all non-avian dinosaurs
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
alongside many species of crocodyliforms and squamates (e.g., mosasaur
Mosasaurs (from Latin ''Mosa'' meaning the 'Meuse', and Ancient Greek, Greek ' meaning 'lizard') are an extinct group of large aquatic reptiles within the family Mosasauridae that lived during the Late Cretaceous. Their first fossil remains wer ...
s). Modern non-bird reptiles inhabit all the continents except Antarctica.
Reptiles are tetrapod vertebrates
Vertebrates () are animals with a vertebral column (backbone or spine), and a cranium, or skull. The vertebral column surrounds and protects the spinal cord, while the cranium protects the brain.
The vertebrates make up the subphylum Vertebra ...
, creatures that either have four limbs or, like snakes, are descended from four-limbed ancestors. Unlike amphibian
Amphibians are ectothermic, anamniote, anamniotic, tetrapod, four-limbed vertebrate animals that constitute the class (biology), class Amphibia. In its broadest sense, it is a paraphyletic group encompassing all Tetrapod, tetrapods, but excl ...
s, reptiles do not have an aquatic larval stage. Most reptiles are oviparous
Oviparous animals are animals that reproduce by depositing fertilized zygotes outside the body (i.e., by laying or spawning) in metabolically independent incubation organs known as eggs, which nurture the embryo into moving offsprings kno ...
, although several species of squamates are viviparous, as were some extinct aquatic cladeseggshell
An eggshell is the outer covering of a hard-shelled egg (biology), egg and of some forms of eggs with soft outer coats.
Worm eggs
Nematode eggs present a two layered structure: an external vitellin layer made of chitin that confers mechanical ...
. As amniotes, reptile eggs are surrounded by membranes for protection and transport, which adapt them to reproduction on dry land. Many of the viviparous species feed their fetus
A fetus or foetus (; : fetuses, foetuses, rarely feti or foeti) is the unborn offspring of a viviparous animal that develops from an embryo. Following the embryonic development, embryonic stage, the fetal stage of development takes place. Pren ...
es through various forms of placenta analogous to those of mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s, with some providing initial care for their hatchlings. Extant
Extant or Least-concern species, least concern is the opposite of the word extinct. It may refer to:
* Extant hereditary titles
* Extant literature, surviving literature, such as ''Beowulf'', the oldest extant manuscript written in English
* Exta ...
reptiles range in size from a tiny gecko, ''Sphaerodactylus ariasae
''Sphaerodactylus ariasae'', commonly called the Jaragua sphaero or the Jaragua dwarf gecko, is the smallest species of lizard in the family Sphaerodactylidae.
Description
''Sphaerodactylus ariasae'' is the world's smallest known reptile. The ...
'', which can grow up to to the saltwater crocodile
The saltwater crocodile (''Crocodylus porosus'') is a crocodilian native to saltwater habitats, brackish wetlands and freshwater rivers from India's east coast across Southeast Asia and the Sundaland to northern Australia and Micronesia. It ha ...
, ''Crocodylus porosus'', which can reach over in length and weigh over .
Classification
Research history
 In the 13th century, the category of ''reptile'' was recognized in Europe as consisting of a miscellany of egg-laying creatures, including "snakes, various fantastic monsters, lizards, assorted amphibians, and worms", as recorded by
In the 13th century, the category of ''reptile'' was recognized in Europe as consisting of a miscellany of egg-laying creatures, including "snakes, various fantastic monsters, lizards, assorted amphibians, and worms", as recorded by Beauvais
Beauvais ( , ; ) is a town and Communes of France, commune in northern France, and prefecture of the Oise Departments of France, département, in the Hauts-de-France Regions of France, region, north of Paris.
The Communes of France, commune o ...
in his ''Mirror of Nature''.
In the 18th century, the reptiles were, from the outset of classification, grouped with the amphibian
Amphibians are ectothermic, anamniote, anamniotic, tetrapod, four-limbed vertebrate animals that constitute the class (biology), class Amphibia. In its broadest sense, it is a paraphyletic group encompassing all Tetrapod, tetrapods, but excl ...
s. Linnaeus
Carl Linnaeus (23 May 1707 – 10 January 1778), also known after ennoblement in 1761 as Carl von Linné,#Blunt, Blunt (2004), p. 171. was a Swedish biologist and physician who formalised binomial nomenclature, the modern system of naming o ...
, working from species-poor Sweden
Sweden, formally the Kingdom of Sweden, is a Nordic countries, Nordic country located on the Scandinavian Peninsula in Northern Europe. It borders Norway to the west and north, and Finland to the east. At , Sweden is the largest Nordic count ...
, where the common adder and grass snake are often found hunting in water, included all reptiles and amphibians in class
Class, Classes, or The Class may refer to:
Common uses not otherwise categorized
* Class (biology), a taxonomic rank
* Class (knowledge representation), a collection of individuals or objects
* Class (philosophy), an analytical concept used d ...
in his '' Systema Naturæ''.herpetology
Herpetology (from Ancient Greek ἑρπετόν ''herpetón'', meaning "reptile" or "creeping animal") is a branch of zoology concerned with the study of amphibians (including frogs, salamanders, and caecilians (Gymnophiona)) and reptiles (in ...
.
 It was not until the beginning of the 19th century that it became clear that reptiles and amphibians are, in fact, quite different animals, and P.A. Latreille erected the class ''Batracia'' (1825) for the latter, dividing the
It was not until the beginning of the 19th century that it became clear that reptiles and amphibians are, in fact, quite different animals, and P.A. Latreille erected the class ''Batracia'' (1825) for the latter, dividing the tetrapod
A tetrapod (; from Ancient Greek :wiktionary:τετρα-#Ancient Greek, τετρα- ''(tetra-)'' 'four' and :wiktionary:πούς#Ancient Greek, πούς ''(poús)'' 'foot') is any four-Limb (anatomy), limbed vertebrate animal of the clade Tetr ...
s into the four familiar classes of reptiles, amphibians, birds, and mammals. The British anatomist T.H. Huxley made Latreille's definition popular and, together with Richard Owen
Sir Richard Owen (20 July 1804 – 18 December 1892) was an English biologist, comparative anatomy, comparative anatomist and paleontology, palaeontologist. Owen is generally considered to have been an outstanding naturalist with a remarkabl ...
, expanded Reptilia to include the various fossil " antediluvian monsters", including dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s and the mammal-like (synapsid
Synapsida is a diverse group of tetrapod vertebrates that includes all mammals and their extinct relatives. It is one of the two major clades of the group Amniota, the other being the more diverse group Sauropsida (which includes all extant rept ...
) '' Dicynodon'' he helped describe. This was not the only possible classification scheme: In the Hunterian lectures delivered at the Royal College of Surgeons
The Royal College of Surgeons is an ancient college (a form of corporation) established in England to regulate the activity of surgeons. Derivative organisations survive in many present and former members of the Commonwealth. These organisations ...
in 1863, Huxley grouped the vertebrates into mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s, sauroids, and ichthyoids (the latter containing the fishes and amphibians). He subsequently proposed the names of Sauropsida
Sauropsida (Greek language, Greek for "lizard faces") is a clade of amniotes, broadly equivalent to the Class (biology), class Reptile, Reptilia, though typically used in a broader sense to also include extinct stem-group relatives of modern repti ...
and Ichthyopsida for the latter two groups. In 1866, Haeckel demonstrated that vertebrates could be divided based on their reproductive strategies, and that reptiles, birds, and mammals were united by the amniotic egg.
The terms ''Sauropsida'' ("lizard faces") and '' Theropsida'' ("beast faces") were used again in 1916 by E.S. Goodrich to distinguish between lizards, birds, and their relatives on the one hand (Sauropsida) and mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s and their extinct relatives (Theropsida) on the other. Goodrich supported this division by the nature of the hearts and blood vessels in each group, and other features, such as the structure of the forebrain. According to Goodrich, both lineages evolved from an earlier stem group, Protosauria ("first lizards") in which he included some animals today considered reptile-like amphibians, as well as early reptiles.Procolophonia
Procolophonia is an extinct suborder (clade) of herbivorous reptiles that lived from the Middle Permian till the end of the Triassic period. They were originally included as a suborder of the Cotylosauria (later renamed Captorhinida Carroll ...
, Eosuchia, Millerosauria, Chelonia (turtles), Squamata
Squamata (, Latin ''squamatus'', 'scaly, having scales') is the largest Order (biology), order of reptiles; most members of which are commonly known as Lizard, lizards, with the group also including Snake, snakes. With over 11,991 species, it i ...
(lizards and snakes), Rhynchocephalia, Crocodilia
Crocodilia () is an order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorph pseudosuchia ...
, " thecodonts" (paraphyletic
Paraphyly is a taxonomic term describing a grouping that consists of the grouping's last common ancestor and some but not all of its descendant lineages. The grouping is said to be paraphyletic ''with respect to'' the excluded subgroups. In co ...
basal Archosaur
Archosauria () or archosaurs () is a clade of diapsid sauropsid tetrapods, with birds and crocodilians being the only extant taxon, extant representatives. Although broadly classified as reptiles, which traditionally exclude birds, the cladistics ...
ia), non- avian dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s, pterosaur
Pterosaurs are an extinct clade of flying reptiles in the order Pterosauria. They existed during most of the Mesozoic: from the Late Triassic to the end of the Cretaceous (228 million to 66 million years ago). Pterosaurs are the earli ...
s, ichthyosaurs, and sauropterygia
Sauropterygia ("lizard flippers") is an extinct taxon of diverse, aquatic diapsid reptiles that developed from terrestrial ancestors soon after the end-Permian extinction and flourished during the Triassic before all except for the Plesiosau ...
ns.vertebrae
Each vertebra (: vertebrae) is an irregular bone with a complex structure composed of bone and some hyaline cartilage, that make up the vertebral column or spine, of vertebrates. The proportions of the vertebrae differ according to their spinal ...
. The animals singled out by these formulations, the amniote
Amniotes are tetrapod vertebrate animals belonging to the clade Amniota, a large group that comprises the vast majority of living terrestrial animal, terrestrial and semiaquatic vertebrates. Amniotes evolution, evolved from amphibious Stem tet ...
s other than the mammals and the birds, are still those considered reptiles today. The synapsid/sauropsid division supplemented another approach, one that split the reptiles into four subclasses based on the number and position of temporal fenestrae, openings in the sides of the skull behind the eyes. This classification was initiated by Henry Fairfield Osborn and elaborated and made popular by Romer's classic ''
The synapsid/sauropsid division supplemented another approach, one that split the reptiles into four subclasses based on the number and position of temporal fenestrae, openings in the sides of the skull behind the eyes. This classification was initiated by Henry Fairfield Osborn and elaborated and made popular by Romer's classic ''Vertebrate Paleontology
Vertebrate paleontology is the subfield of paleontology that seeks to discover, through the study of fossilized remains, the behavior, reproduction and appearance of extinct vertebrates (animals with vertebrae and their descendants). It also t ...
''.turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s and relatives)
* Synapsida
Synapsida is a diverse group of tetrapod vertebrates that includes all mammals and their extinct relatives. It is one of the two major clades of the group Amniota, the other being the more diverse group Sauropsida (which includes all extant rep ...
– one low fenestra – pelycosaur
Pelycosaur ( ) is an older term for basal or primitive Late Paleozoic synapsids, excluding the therapsids and their descendants. Previously, the term mammal-like reptile was used, and Pelycosauria was considered an order, but this is now thoug ...
s and therapsids (the ' mammal-like reptiles')
* Euryapsida – one high fenestra (above the postorbital and squamosal) – protorosaurs (small, early lizard-like reptiles) and the marine sauropterygia
Sauropterygia ("lizard flippers") is an extinct taxon of diverse, aquatic diapsid reptiles that developed from terrestrial ancestors soon after the end-Permian extinction and flourished during the Triassic before all except for the Plesiosau ...
ns and ichthyosaurs
Ichthyosauria is an taxonomy (biology), order of large extinction, extinct marine reptiles sometimes referred to as "ichthyosaurs", although the term is also used for wider clades in which the order resides.
Ichthyosaurians thrived during much of ...
, the latter called Parapsida in Osborn's work.
* Diapsida – two fenestrae – most reptiles, including lizard
Lizard is the common name used for all Squamata, squamate reptiles other than snakes (and to a lesser extent amphisbaenians), encompassing over 7,000 species, ranging across all continents except Antarctica, as well as most Island#Oceanic isla ...
s, snake
Snakes are elongated limbless reptiles of the suborder Serpentes (). Cladistically squamates, snakes are ectothermic, amniote vertebrates covered in overlapping scales much like other members of the group. Many species of snakes have s ...
s, crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s, dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s and pterosaur
Pterosaurs are an extinct clade of flying reptiles in the order Pterosauria. They existed during most of the Mesozoic: from the Late Triassic to the end of the Cretaceous (228 million to 66 million years ago). Pterosaurs are the earli ...
s.
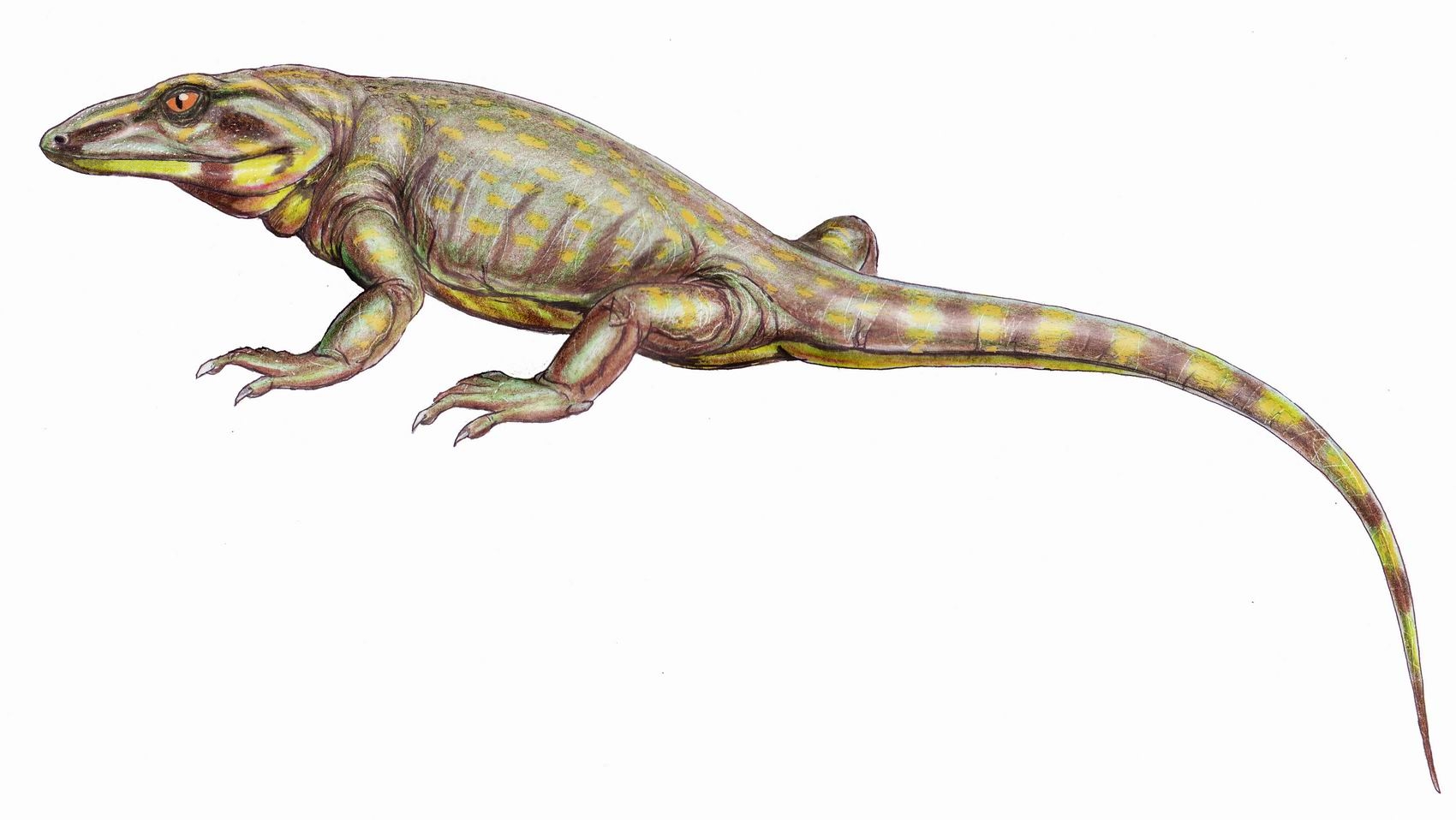 The composition of Euryapsida was uncertain.
The composition of Euryapsida was uncertain. Ichthyosaurs
Ichthyosauria is an taxonomy (biology), order of large extinction, extinct marine reptiles sometimes referred to as "ichthyosaurs", although the term is also used for wider clades in which the order resides.
Ichthyosaurians thrived during much of ...
were, at times, considered to have arisen independently of the other euryapsids, and given the older name Parapsida. Parapsida was later discarded as a group for the most part (ichthyosaurs being classified as ''incertae sedis
or is a term used for a taxonomy (biology), taxonomic group where its broader relationships are unknown or undefined. Alternatively, such groups are frequently referred to as "enigmatic taxa". In the system of open nomenclature, uncertainty ...
'' or with Euryapsida). However, four (or three if Euryapsida is merged into Diapsida) subclasses remained more or less universal for non-specialist work throughout the 20th century. It has largely been abandoned by recent researchers: In particular, the anapsid condition has been found to occur so variably among unrelated groups that it is not now considered a useful distinction.
Phylogenetics and modern definition
By the early 21st century, vertebrate paleontologists were beginning to adopt phylogenetic
In biology, phylogenetics () is the study of the evolutionary history of life using observable characteristics of organisms (or genes), which is known as phylogenetic inference. It infers the relationship among organisms based on empirical dat ...
taxonomy, in which all groups are defined in such a way as to be monophyletic
In biological cladistics for the classification of organisms, monophyly is the condition of a taxonomic grouping being a clade – that is, a grouping of organisms which meets these criteria:
# the grouping contains its own most recent co ...
; that is, groups which include all descendants of a particular ancestor. The reptiles as historically defined are paraphyletic
Paraphyly is a taxonomic term describing a grouping that consists of the grouping's last common ancestor and some but not all of its descendant lineages. The grouping is said to be paraphyletic ''with respect to'' the excluded subgroups. In co ...
, since they exclude both birds and mammals. These respectively evolved from dinosaurs and from early therapsids, both of which were traditionally called "reptiles".crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s than the latter are to the rest of extant reptiles. Colin Tudge wrote:
Mammals are a clade
In biology, a clade (), also known as a Monophyly, monophyletic group or natural group, is a group of organisms that is composed of a common ancestor and all of its descendants. Clades are the fundamental unit of cladistics, a modern approach t ...
, and therefore the cladists are happy to acknowledge the traditional taxon Mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
ia; and birds, too, are a clade, universally ascribed to the formal taxon Aves
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweight ...
. Mammalia and Aves are, in fact, subclades within the grand clade of the Amniota. But the traditional class Reptilia is not a clade. It is just a section of the clade Amniota
Amniotes are tetrapod vertebrate animals belonging to the clade Amniota, a large group that comprises the vast majority of living terrestrial and semiaquatic vertebrates. Amniotes evolved from amphibious stem tetrapod ancestors during the C ...
: The section that is left after the Mammalia and Aves have been hived off. It cannot be defined by synapomorphies
In phylogenetics, an apomorphy (or derived trait) is a novel character or character state that has evolved from its ancestral form (or plesiomorphy). A synapomorphy is an apomorphy shared by two or more taxa and is therefore hypothesized to ...
, as is the proper way. Instead, it is defined by a combination of the features it has and the features it lacks: reptiles are the amniotes that lack fur or feathers. At best, the cladists suggest, we could say that the traditional Reptilia are 'non-avian, non-mammalian amniotes'.
Despite the early proposals for replacing the paraphyletic Reptilia with a monophyletic Sauropsida
Sauropsida (Greek language, Greek for "lizard faces") is a clade of amniotes, broadly equivalent to the Class (biology), class Reptile, Reptilia, though typically used in a broader sense to also include extinct stem-group relatives of modern repti ...
, which includes birds, that term was never adopted widely or, when it was, was not applied consistently. When Sauropsida was used, it often had the same content or even the same definition as Reptilia. In 1988, Jacques Gauthier proposed a
When Sauropsida was used, it often had the same content or even the same definition as Reptilia. In 1988, Jacques Gauthier proposed a cladistic
Cladistics ( ; from Ancient Greek 'branch') is an approach to biological classification in which organisms are categorized in groups ("clades") based on hypotheses of most recent common ancestry. The evidence for hypothesized relationships is ...
definition of Reptilia as a monophyletic node-based crown group
In phylogenetics, the crown group or crown assemblage is a collection of species composed of the living representatives of the collection, the most recent common ancestor of the collection, and all descendants of the most recent common ancestor ...
containing turtles, lizards and snakes, crocodilians, and birds, their common ancestor and all its descendants. While Gauthier's definition was close to the modern consensus, nonetheless, it became considered inadequate because the actual relationship of turtles to other reptiles was not yet well understood at this time.[ Major revisions since have included the reassignment of synapsids as non-reptiles, and classification of turtles as diapsids.][ Gauthier 1994 and Laurin and Reisz 1995's definition of Sauropsida defined the scope of the group as distinct and broader than that of Reptilia, encompassing Mesosauridae as well as Reptilia ''sensu stricto''.]PhyloCode
The ''International Code of Phylogenetic Nomenclature'', known as the ''PhyloCode'' for short, is a formal set of rules governing phylogenetic nomenclature. Its current version is specifically designed to regulate the naming of clades, leaving the ...
, was published by Modesto and Anderson in 2004.[ Modesto and Anderson reviewed the many previous definitions and proposed a modified definition, which they intended to retain most traditional content of the group while keeping it stable and monophyletic. They defined Reptilia as all amniotes closer to '' Lacerta agilis'' and '' Crocodylus niloticus'' than to '']Homo sapiens
Humans (''Homo sapiens'') or modern humans are the most common and widespread species of primate, and the last surviving species of the genus ''Homo''. They are Hominidae, great apes characterized by their Prehistory of nakedness and clothing ...
''. This stem-based definition is equivalent to the more common definition of Sauropsida, which Modesto and Anderson synonymized with Reptilia, since the latter is better known and more frequently used. Unlike most previous definitions of Reptilia, however, Modesto and Anderson's definition includes birds, as they are within the clade that includes both lizards and crocodiles.[
]
Taxonomy
General classification of extinct and living reptiles, focusing on major groups.Sauropsida
Sauropsida (Greek language, Greek for "lizard faces") is a clade of amniotes, broadly equivalent to the Class (biology), class Reptile, Reptilia, though typically used in a broader sense to also include extinct stem-group relatives of modern repti ...
**Parareptilia
Parareptilia ("near-reptiles") is an extinct group of basal sauropsids (" reptiles"), traditionally considered the sister taxon to Eureptilia (the group that likely contains all living reptiles and birds). Parareptiles first arose near the en ...
** Eureptilia
***Captorhinidae
Captorhinidae is an extinct family of tetrapods, traditionally considered primitive Reptile, reptiles, known from the late Carboniferous to the Late Permian. They had a cosmopolitan distribution across Pangea.
Description
Captorhinids are a cl ...
*** Diapsida
**** Araeoscelidia
**** Neodiapsida
***** Drepanosauromorpha (placement uncertain)
***** Younginiformes (paraphyletic
Paraphyly is a taxonomic term describing a grouping that consists of the grouping's last common ancestor and some but not all of its descendant lineages. The grouping is said to be paraphyletic ''with respect to'' the excluded subgroups. In co ...
)
*****Ichthyosauromorpha
The Ichthyosauromorpha are an extinct clade of Mesozoic marine reptiles consisting of the Ichthyosauriformes and the Hupehsuchia.
The node clade Ichthyosauromorpha was first defined by Ryosuke Motani ''et al.'' in 2014 as the group consisting ...
(placement uncertain)
***** Thalattosauria (placement uncertain)
***** Sauria
****** Lepidosauromorpha
******* Lepidosauriformes
******** Rhynchocephalia (tuatara)
********Squamata
Squamata (, Latin ''squamatus'', 'scaly, having scales') is the largest Order (biology), order of reptiles; most members of which are commonly known as Lizard, lizards, with the group also including Snake, snakes. With over 11,991 species, it i ...
(lizards and snakes)
****** Choristodera (placement uncertain)
******Sauropterygia
Sauropterygia ("lizard flippers") is an extinct taxon of diverse, aquatic diapsid reptiles that developed from terrestrial ancestors soon after the end-Permian extinction and flourished during the Triassic before all except for the Plesiosau ...
(placement uncertain)
******Pantestudines
Pantestudines or Pan-Testudines is the proposed group of all Reptile, reptiles more closely related to turtles than to any other living animal. It includes both modern turtles (crown group turtles, also known as Testudines) and all of their extin ...
(turtles and kin, placement uncertain)
****** Archosauromorpha
******* Protorosauria (paraphyletic)
******* Rhynchosauria
******* Allokotosauria
******* Archosauriformes
******** Phytosauria
******** Archosauria
*********Pseudosuchia
Pseudosuchia, from Ancient Greek ψεύδος (''pseúdos)'', meaning "false", and σούχος (''soúkhos''), meaning "crocodile" is one of two major divisions of Archosauria, including living crocodilians and all archosaurs more closely relat ...
**********Crocodilia
Crocodilia () is an order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorph pseudosuchia ...
(crocodilians)
*********Avemetatarsalia
Avemetatarsalia (meaning "bird metatarsals") is a clade of diapsid Reptile, reptiles containing all archosaurs more closely related to birds than to crocodilians. The two most successful groups of avemetatarsalians were the dinosaurs and pterosau ...
/Ornithodira
Avemetatarsalia (meaning "bird metatarsals") is a clade of diapsid reptiles containing all archosaurs more closely related to birds than to crocodilians. The two most successful groups of avemetatarsalians were the dinosaurs and pterosaurs. Di ...
**********Pterosaur
Pterosaurs are an extinct clade of flying reptiles in the order Pterosauria. They existed during most of the Mesozoic: from the Late Triassic to the end of the Cretaceous (228 million to 66 million years ago). Pterosaurs are the earli ...
ia
**********Dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
ia
***********Ornithischia
Ornithischia () is an extinct clade of mainly herbivorous dinosaurs characterized by a pelvic structure superficially similar to that of birds. The name ''Ornithischia'', or "bird-hipped", reflects this similarity and is derived from the Greek ...
*********** Saurischia (including birds (Aves
Birds are a group of warm-blooded vertebrates constituting the class Aves (), characterised by feathers, toothless beaked jaws, the laying of hard-shelled eggs, a high metabolic rate, a four-chambered heart, and a strong yet lightweight ...
))
Phylogeny
The cladogram
A cladogram (from Greek language, Greek ''clados'' "branch" and ''gramma'' "character") is a diagram used in cladistics to show relations among organisms. A cladogram is not, however, an Phylogenetic tree, evolutionary tree because it does not s ...
presented here illustrates the "family tree" of reptiles, and follows a simplified version of the relationships found by M.S. Lee, in 2013.[ though a few have recovered turtles as Lepidosauromorpha instead. The cladogram below used a combination of genetic (molecular) and fossil (morphological) data to obtain its results.][
]
The position of turtles
The placement of turtles has historically been highly variable. Classically, turtles were considered to be related to the primitive anapsid reptiles.sister clade
In phylogenetics, a sister group or sister taxon, also called an adelphotaxon, comprises the closest relative(s) of another given unit in an evolutionary tree.
Definition
The expression is most easily illustrated by a cladogram:
Taxon A and ...
to the archosaur
Archosauria () or archosaurs () is a clade of diapsid sauropsid tetrapods, with birds and crocodilians being the only extant taxon, extant representatives. Although broadly classified as reptiles, which traditionally exclude birds, the cladistics ...
s, the group that includes crocodilians, non-avian dinosaurs, and birds. However, in their comparative analysis of the timing of organogenesis
Organogenesis is the phase of embryonic development that starts at the end of gastrulation and continues until birth. During organogenesis, the three germ layers formed from gastrulation (the ectoderm, endoderm, and mesoderm) form the internal org ...
, Werneburg and Sánchez-Villagra (2009) found support for the hypothesis that turtles belong to a separate clade within Sauropsida
Sauropsida (Greek language, Greek for "lizard faces") is a clade of amniotes, broadly equivalent to the Class (biology), class Reptile, Reptilia, though typically used in a broader sense to also include extinct stem-group relatives of modern repti ...
, outside the saurian clade altogether.
Evolutionary history
Origin of the reptiles

 The origin of the reptiles lies about 310–320 million years ago, in the steaming swamps of the late
The origin of the reptiles lies about 310–320 million years ago, in the steaming swamps of the late Carboniferous
The Carboniferous ( ) is a Geologic time scale, geologic period and System (stratigraphy), system of the Paleozoic era (geology), era that spans 60 million years, from the end of the Devonian Period Ma (million years ago) to the beginning of the ...
period, when the first reptiles evolved from advanced reptiliomorphs.[
]
The oldest known animal that may have been an amniote
Amniotes are tetrapod vertebrate animals belonging to the clade Amniota, a large group that comprises the vast majority of living terrestrial animal, terrestrial and semiaquatic vertebrates. Amniotes evolution, evolved from amphibious Stem tet ...
is ''Casineria
''Casineria'' is an extinct genus of tetrapodomorph which lived about 340–334 million years ago in the Mississippian (geologic period), Mississippian epoch of the Carboniferous period. Its Generic name (biology), generic name, ''Casineria'', i ...
'' (though it may have been a temnospondyl
Temnospondyli (from Greek language, Greek τέμνειν, ''temnein'' 'to cut' and σπόνδυλος, ''spondylos'' 'vertebra') or temnospondyls is a diverse ancient order (biology), order of small to giant tetrapods—often considered Labyrinth ...
). A series of footprints from the fossil strata of Nova Scotia
Nova Scotia is a Provinces and territories of Canada, province of Canada, located on its east coast. It is one of the three Maritime Canada, Maritime provinces and Population of Canada by province and territory, most populous province in Atlan ...
dated to show typical reptilian toes and imprints of scales. These tracks are attributed to '' Hylonomus'', the oldest unquestionable reptile known.
It was a small, lizard-like animal, about long, with numerous sharp teeth indicating an insectivorous diet.amniote
Amniotes are tetrapod vertebrate animals belonging to the clade Amniota, a large group that comprises the vast majority of living terrestrial animal, terrestrial and semiaquatic vertebrates. Amniotes evolution, evolved from amphibious Stem tet ...
) and '' Paleothyris'', both of similar build and presumably similar habit.
However, microsaurs have been at times considered true reptiles, so an earlier origin is possible.
Rise of the reptiles
The earliest amniotes, including stem-reptiles (those amniotes closer to modern reptiles than to mammals), were largely overshadowed by larger stem-tetrapods, such as '' Cochleosaurus'', and remained a small, inconspicuous part of the fauna until the Carboniferous Rainforest Collapse
The Carboniferous rainforest collapse (CRC) was a minor extinction event that occurred around 305 million years ago in the Carboniferous period. The event occurred at the end of the Moscovian and continued into the early Kasimovian stages of th ...
.Mesosaurus
''Mesosaurus'' (meaning "middle lizard") is an extinct genus of reptile from the Early Permian of southern Africa and South America. Along with it, the genera '' Brazilosaurus'' and '' Stereosternum'', it is a member of the family Mesosauridae ...
'', a genus from the Early Permian 01 or 01 may refer to:
* The year 2001, or any year ending with 01
* The month of January
* 1 (number)
Music
* '01 (Richard Müller album), ''01'' (Richard Müller album), 2001
* 01 (Urban Zakapa album), ''01'' (Urban Zakapa album), 2011
* ''01011 ...
that had returned to water, feeding on fish.
A 2021 examination of reptile diversity in the Carboniferous and the Permian suggests a much higher degree of diversity than previously thought, comparable or even exceeding that of synapsids. Thus, the "First Age of Reptiles" was proposed.
Anapsids, synapsids, diapsids, and sauropsids
 It was traditionally assumed that the first reptiles retained an anapsid skull inherited from their ancestors.
It was traditionally assumed that the first reptiles retained an anapsid skull inherited from their ancestors.skull roof
The skull roof or the roofing bones of the skull are a set of bones covering the brain, eyes and nostrils in bony fishes, including land-living vertebrates. The bones are derived from dermal bone and are part of the dermatocranium.
In com ...
with only holes for the nostrils, eyes and a pineal eye.synapsid
Synapsida is a diverse group of tetrapod vertebrates that includes all mammals and their extinct relatives. It is one of the two major clades of the group Amniota, the other being the more diverse group Sauropsida (which includes all extant rept ...
-like openings (see below) in the skull roof of the skulls of several members of Parareptilia
Parareptilia ("near-reptiles") is an extinct group of basal sauropsids (" reptiles"), traditionally considered the sister taxon to Eureptilia (the group that likely contains all living reptiles and birds). Parareptiles first arose near the en ...
(the clade containing most of the amniotes traditionally referred to as "anapsids"), including lanthanosuchoids, millerettids, bolosaurids, some nycteroleterids, some procolophonoids and at least some mesosaur
Mesosaurs ("middle lizards") were a group of small aquatic reptiles that lived during the early Permian period ( Cisuralian), roughly 299 to 270 million years ago. Mesosaurs were the first known aquatic reptiles, having apparently returned to a ...
s[ These animals are traditionally referred to as "anapsids", and form a ]paraphyletic
Paraphyly is a taxonomic term describing a grouping that consists of the grouping's last common ancestor and some but not all of its descendant lineages. The grouping is said to be paraphyletic ''with respect to'' the excluded subgroups. In co ...
basic stock from which other groups evolved.[ Very shortly after the first amniotes appeared, a lineage called ]Synapsida
Synapsida is a diverse group of tetrapod vertebrates that includes all mammals and their extinct relatives. It is one of the two major clades of the group Amniota, the other being the more diverse group Sauropsida (which includes all extant rep ...
split off; this group was characterized by a temporal opening in the skull behind each eye giving room for the jaw muscle to move. These are the "mammal-like amniotes", or stem-mammals, that later gave rise to the true mammals
A mammal () is a vertebrate animal of the class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three middle e ...
. Soon after, another group evolved a similar trait, this time with a double opening behind each eye, earning them the name Diapsida ("two arches").[ The function of the holes in these groups was to lighten the skull and give room for the jaw muscles to move, allowing for a more powerful bite.]phylogenetic
In biology, phylogenetics () is the study of the evolutionary history of life using observable characteristics of organisms (or genes), which is known as phylogenetic inference. It infers the relationship among organisms based on empirical dat ...
studies with this in mind placed turtles firmly within Diapsida.archosaur
Archosauria () or archosaurs () is a clade of diapsid sauropsid tetrapods, with birds and crocodilians being the only extant taxon, extant representatives. Although broadly classified as reptiles, which traditionally exclude birds, the cladistics ...
s.
Permian reptiles
With the close of the Carboniferous
The Carboniferous ( ) is a Geologic time scale, geologic period and System (stratigraphy), system of the Paleozoic era (geology), era that spans 60 million years, from the end of the Devonian Period Ma (million years ago) to the beginning of the ...
, the amniotes became the dominant tetrapod fauna. While primitive, terrestrial reptiliomorphs still existed, the synapsid amniotes evolved the first truly terrestrial megafauna
In zoology, megafauna (from Ancient Greek, Greek μέγας ''megas'' "large" and Neo-Latin ''fauna'' "animal life") are large animals. The precise definition of the term varies widely, though a common threshold is approximately , this lower en ...
(giant animals) in the form of pelycosaurs, such as ''Edaphosaurus
''Edaphosaurus'' (, meaning "pavement lizard" for dense clusters of its teeth) is a genus of extinct edaphosaurid synapsids that lived in what is now North America and Europe around 303.4 to 272.5 million years ago, during the Late Carboniferous ...
'' and the carnivorous ''Dimetrodon
''Dimetrodon'' ( or ; ) is an extinct genus of sphenacodontid synapsid that lived during the Cisuralian (Early Permian) Epoch (geology), epoch of the Permian period, around 295–272 million years ago. With most species measuring long and ...
''. In the mid-Permian period, the climate became drier, resulting in a change of fauna: The pelycosaurs were replaced by the therapsids
Therapsida is a clade comprising a major group of eupelycosaurian synapsids that includes mammals and their ancestors and close relatives. Many of the traits today seen as unique to mammals had their origin within early therapsids, including li ...
.[ Colbert, E.H. & Morales, M. (2001): '' Colbert's Evolution of the Vertebrates: A History of the Backboned Animals Through Time''. 4th edition. John Wiley & Sons, Inc, New York. .]
The parareptiles, whose massive skull roof
The skull roof or the roofing bones of the skull are a set of bones covering the brain, eyes and nostrils in bony fishes, including land-living vertebrates. The bones are derived from dermal bone and are part of the dermatocranium.
In com ...
s had no postorbital holes, continued and flourished throughout the Permian. The pareiasaur
Pareiasaurs (meaning "cheek lizards") are an extinct clade of large, herbivorous parareptiles. Members of the group were armoured with osteoderms which covered large areas of the body. They first appeared in southern Pangea during the Middle Per ...
ian parareptiles reached giant proportions in the late Permian, eventually disappearing at the close of the period (the turtles being possible survivors).turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s, crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s, and dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s) and the Lepidosauromorpha (predecessors of modern lizard
Lizard is the common name used for all Squamata, squamate reptiles other than snakes (and to a lesser extent amphisbaenians), encompassing over 7,000 species, ranging across all continents except Antarctica, as well as most Island#Oceanic isla ...
s, snake
Snakes are elongated limbless reptiles of the suborder Serpentes (). Cladistically squamates, snakes are ectothermic, amniote vertebrates covered in overlapping scales much like other members of the group. Many species of snakes have s ...
s, and tuatara
The tuatara (''Sphenodon punctatus'') is a species of reptile endemic to New Zealand. Despite its close resemblance to lizards, it is actually the only extant member of a distinct lineage, the previously highly diverse order Rhynchocephal ...
s). Both groups remained lizard-like and relatively small and inconspicuous during the Permian.
Mesozoic reptiles
The close of the Permian saw the greatest mass extinction known (see the Permian–Triassic extinction event
The Permian–Triassic extinction event (also known as the P–T extinction event, the Late Permian extinction event, the Latest Permian extinction event, the End-Permian extinction event, and colloquially as the Great Dying,) was an extinction ...
), an event prolonged by the combination of two or more distinct extinction pulses.archosaur
Archosauria () or archosaurs () is a clade of diapsid sauropsid tetrapods, with birds and crocodilians being the only extant taxon, extant representatives. Although broadly classified as reptiles, which traditionally exclude birds, the cladistics ...
s became the dominant group during the Triassic
The Triassic ( ; sometimes symbolized 🝈) is a geologic period and system which spans 50.5 million years from the end of the Permian Period 251.902 million years ago ( Mya), to the beginning of the Jurassic Period 201.4 Mya. The Triassic is t ...
period, though it took 30 million years before their diversity was as great as the animals that lived in the Permian.dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s and pterosaur
Pterosaurs are an extinct clade of flying reptiles in the order Pterosauria. They existed during most of the Mesozoic: from the Late Triassic to the end of the Cretaceous (228 million to 66 million years ago). Pterosaurs are the earli ...
s, as well as the ancestors of crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s. Since reptiles, first rauisuchians and then dinosaurs, dominated the Mesozoic era, the interval is popularly known as the "Age of Reptiles". The dinosaurs also developed smaller forms, including the feather-bearing smaller theropods. In the Cretaceous
The Cretaceous ( ) is a geological period that lasted from about 143.1 to 66 mya (unit), million years ago (Mya). It is the third and final period of the Mesozoic Era (geology), Era, as well as the longest. At around 77.1 million years, it is the ...
period, these gave rise to the first true birds
Birds are a group of warm-blooded vertebrates constituting the class (biology), class Aves (), characterised by feathers, toothless beaked jaws, the Oviparity, laying of Eggshell, hard-shelled eggs, a high Metabolism, metabolic rate, a fou ...
.sister group
In phylogenetics, a sister group or sister taxon, also called an adelphotaxon, comprises the closest relative(s) of another given unit in an evolutionary tree.
Definition
The expression is most easily illustrated by a cladogram:
Taxon A and ...
to Archosauromorpha is Lepidosauromorpha, containing lizard
Lizard is the common name used for all Squamata, squamate reptiles other than snakes (and to a lesser extent amphisbaenians), encompassing over 7,000 species, ranging across all continents except Antarctica, as well as most Island#Oceanic isla ...
s and tuatara
The tuatara (''Sphenodon punctatus'') is a species of reptile endemic to New Zealand. Despite its close resemblance to lizards, it is actually the only extant member of a distinct lineage, the previously highly diverse order Rhynchocephal ...
s, as well as their fossil relatives. Lepidosauromorpha contained at least one major group of the Mesozoic sea reptiles: the mosasaurs, which lived during the Cretaceous
The Cretaceous ( ) is a geological period that lasted from about 143.1 to 66 mya (unit), million years ago (Mya). It is the third and final period of the Mesozoic Era (geology), Era, as well as the longest. At around 77.1 million years, it is the ...
period. The phylogenetic placement of other main groups of fossil sea reptiles – the ichthyopterygians (including ichthyosaurs) and the sauropterygia
Sauropterygia ("lizard flippers") is an extinct taxon of diverse, aquatic diapsid reptiles that developed from terrestrial ancestors soon after the end-Permian extinction and flourished during the Triassic before all except for the Plesiosau ...
ns, which evolved in the early Triassic – is more controversial. Different authors linked these groups either to lepidosauromorphs
Cenozoic reptiles

 The close of the
The close of the Cretaceous
The Cretaceous ( ) is a geological period that lasted from about 143.1 to 66 mya (unit), million years ago (Mya). It is the third and final period of the Mesozoic Era (geology), Era, as well as the longest. At around 77.1 million years, it is the ...
period saw the demise of the Mesozoic era reptilian megafauna (see the Cretaceous–Paleogene extinction event
The Cretaceous–Paleogene (K–Pg) extinction event, also known as the K–T extinction, was the extinction event, mass extinction of three-quarters of the plant and animal species on Earth approximately 66 million years ago. The event cau ...
, also known as K-T extinction event). Of the large marine reptile
Marine reptiles are reptiles which have become secondarily adapted for an aquatic or semiaquatic life in a marine environment. Only about 100 of the 12,000 extant reptile species and subspecies are classed as marine reptiles, including mari ...
s, only sea turtle
Sea turtles (superfamily Chelonioidea), sometimes called marine turtles, are reptiles of the order Testudines and of the suborder Cryptodira. The seven existing species of sea turtles are the flatback, green, hawksbill, leatherback, loggerh ...
s were left; and of the non-marine large reptiles, only the semi-aquatic crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
s and broadly similar choristoderes survived the extinction, with last members of the latter, the lizard-like '' Lazarussuchus'', becoming extinct in the Miocene
The Miocene ( ) is the first epoch (geology), geological epoch of the Neogene Period and extends from about (Ma). The Miocene was named by Scottish geologist Charles Lyell; the name comes from the Greek words (', "less") and (', "new") and mea ...
. Of the great host of dinosaurs dominating the Mesozoic, only the small beaked birds
Birds are a group of warm-blooded vertebrates constituting the class (biology), class Aves (), characterised by feathers, toothless beaked jaws, the Oviparity, laying of Eggshell, hard-shelled eggs, a high Metabolism, metabolic rate, a fou ...
survived. This dramatic extinction pattern at the end of the Mesozoic led into the Cenozoic. Mammals and birds filled the empty niches left behind by the reptilian megafauna and, while reptile diversification slowed, bird and mammal diversification took an exponential turn.[ However, reptiles were still important components of the megafauna, particularly in the form of large and giant ]tortoise
Tortoises ( ) are reptiles of the family Testudinidae of the order Testudines (Latin for "tortoise"). Like other turtles, tortoises have a shell to protect from predation and other threats. The shell in tortoises is generally hard, and like o ...
s.domesticated
Domestication is a multi-generational mutualistic relationship in which an animal species, such as humans or leafcutter ants, takes over control and care of another species, such as sheep or fungi, to obtain from them a steady supply of reso ...
species).
Morphology and physiology
Circulation
All lepidosaurs and turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s have a three-chambered heart
The heart is a muscular Organ (biology), organ found in humans and other animals. This organ pumps blood through the blood vessels. The heart and blood vessels together make the circulatory system. The pumped blood carries oxygen and nutrie ...
consisting of two atria, one variably partitioned ventricle, and two aortas that lead to the systemic circulation
In vertebrates, the circulatory system is a organ system, system of organs that includes the heart, blood vessels, and blood which is circulated throughout the body. It includes the cardiovascular system, or vascular system, that consists of ...
. The degree of mixing of oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
ated and deoxygenated blood in the three-chambered heart varies depending on the species and physiological state. Under different conditions, deoxygenated blood can be shunted back to the body or oxygenated blood can be shunted back to the lungs. This variation in blood flow has been hypothesized to allow more effective thermoregulation and longer diving times for aquatic species, but has not been shown to be a fitness advantage.
 For example,
For example, iguana
''Iguana'' (, ) is a genus of herbivorous lizards that are native to tropical areas of Mexico, Central America, South America, and the Caribbean. The genus was first described by Austrian naturalist Josephus Nicolaus Laurenti, J.N. Laurenti in ...
hearts, like the majority of the squamate
Squamata (, Latin ''squamatus'', 'scaly, having scales') is the largest Order (biology), order of reptiles; most members of which are commonly known as Lizard, lizards, with the group also including Snake, snakes. With over 11,991 species, it i ...
hearts, are composed of three chambers with two aorta and one ventricle, cardiac involuntary muscles. The main structures of the heart are the sinus venosus
The sinus venosus is a large quadrangular cavity which precedes the atrium on the venous side of the chordate heart.
In mammals, the sinus venosus exists distinctly only in the embryonic heart where it is found between the two venae cavae; i ...
, the pacemaker, the left atrium
The atrium (; : atria) is one of the two upper chambers in the heart that receives blood from the circulatory system. The blood in the atria is pumped into the heart ventricles through the atrioventricular mitral and tricuspid heart valves.
...
, the right atrium
The atrium (; : atria) is one of the two upper chambers in the heart that receives blood from the circulatory system. The blood in the atria is pumped into the heart ventricles through the atrioventricular mitral and tricuspid heart valves.
...
, the atrioventricular valve, the cavum venosum, cavum arteriosum, the cavum pulmonale, the muscular ridge, the ventricular ridge, pulmonary vein
The pulmonary veins are the veins that transfer Blood#Oxygen transport, oxygenated blood from the lungs to the heart. The largest pulmonary veins are the four ''main pulmonary veins'', two from each lung that drain into the left atrium of the h ...
s, and paired aortic arches.
Some squamate species (e.g., pythons and monitor lizards) have three-chambered hearts that become functionally four-chambered hearts during contraction. This is made possible by a muscular ridge that subdivides the ventricle during ventricular diastole and completely divides it during ventricular systole. Because of this ridge, some of these squamates are capable of producing ventricular pressure differentials that are equivalent to those seen in mammalian and avian hearts.
Crocodilia
Crocodilia () is an order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorph pseudosuchia ...
ns have an anatomically four-chambered heart, similar to bird
Birds are a group of warm-blooded vertebrates constituting the class (biology), class Aves (), characterised by feathers, toothless beaked jaws, the Oviparity, laying of Eggshell, hard-shelled eggs, a high Metabolism, metabolic rate, a fou ...
s, but also have two systemic aortas and are therefore capable of bypassing their pulmonary circulation
The pulmonary circulation is a division of the circulatory system in all vertebrates. The circuit begins with deoxygenated blood returned from the body to the right atrium of the heart where it is pumped out from the right ventricle to the lun ...
. In turtles, the ventricle is not perfectly divided, so a mix of aerated and nonaerated blood can occur.
Metabolism
 Modern non-avian reptiles exhibit some form of cold-bloodedness (i.e. some mix of poikilothermy,
Modern non-avian reptiles exhibit some form of cold-bloodedness (i.e. some mix of poikilothermy, ectotherm
An ectotherm (), more commonly referred to as a "cold-blooded animal", is an animal in which internal physiological sources of heat, such as blood, are of relatively small or of quite negligible importance in controlling body temperature.Dav ...
y, and bradymetabolism) so that they have limited physiological means of keeping the body temperature constant and often rely on external sources of heat. Due to a less stable core temperature than bird
Birds are a group of warm-blooded vertebrates constituting the class (biology), class Aves (), characterised by feathers, toothless beaked jaws, the Oviparity, laying of Eggshell, hard-shelled eggs, a high Metabolism, metabolic rate, a fou ...
s and mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s, reptilian biochemistry requires enzyme
An enzyme () is a protein that acts as a biological catalyst by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different mol ...
s capable of maintaining efficiency over a greater range of temperatures than in the case for warm-blooded
Warm-blooded is a term referring to animal species whose bodies maintain a temperature higher than that of their environment. In particular, homeothermic species (including birds and mammals) maintain a stable body temperature by regulating ...
animals. The optimum body temperature range varies with species, but is typically below that of warm-blooded animals; for many lizards, it falls in the range, while extreme heat-adapted species, like the American desert iguana ''Dipsosaurus dorsalis'', can have optimal physiological temperatures in the mammalian range, between . While the optimum temperature is often encountered when the animal is active, the low basal metabolism makes body temperature drop rapidly when the animal is inactive.
As in all animals, reptilian muscle action produces heat. In large reptiles, like leatherback turtles, the low surface-to-volume ratio allows this metabolically produced heat to keep the animals warmer than their environment even though they do not have a warm-blooded
Warm-blooded is a term referring to animal species whose bodies maintain a temperature higher than that of their environment. In particular, homeothermic species (including birds and mammals) maintain a stable body temperature by regulating ...
metabolism. This form of homeothermy is called gigantothermy; it has been suggested as having been common in large dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s and other extinct large-bodied reptiles.
The benefit of a low resting metabolism is that it requires far less fuel to sustain bodily functions. By using temperature variations in their surroundings, or by remaining cold when they do not need to move, reptiles can save considerable amounts of energy compared to endothermic animals of the same size. A crocodile needs from a tenth to a fifth of the food necessary for a lion
The lion (''Panthera leo'') is a large Felidae, cat of the genus ''Panthera'', native to Sub-Saharan Africa and India. It has a muscular, broad-chested body (biology), body; a short, rounded head; round ears; and a dark, hairy tuft at the ...
of the same weight and can live half a year without eating.calorie
The calorie is a unit of energy that originated from the caloric theory of heat. The large calorie, food calorie, dietary calorie, kilocalorie, or kilogram calorie is defined as the amount of heat needed to raise the temperature of one liter o ...
availability is too low to sustain large-bodied mammals and birds.
It is generally assumed that reptiles are unable to produce the sustained high energy output necessary for long distance chases or flying. Higher energetic capacity might have been responsible for the evolution of warm-blooded
Warm-blooded is a term referring to animal species whose bodies maintain a temperature higher than that of their environment. In particular, homeothermic species (including birds and mammals) maintain a stable body temperature by regulating ...
ness in birds and mammals. However, investigation of correlations between active capacity and thermophysiology show a weak relationship.
Respiratory system
All reptiles breathe using lung
The lungs are the primary Organ (biology), organs of the respiratory system in many animals, including humans. In mammals and most other tetrapods, two lungs are located near the Vertebral column, backbone on either side of the heart. Their ...
s. Aquatic turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s have developed more permeable skin, and some species have modified their cloaca
A cloaca ( ), : cloacae ( or ), or vent, is the rear orifice that serves as the only opening for the digestive (rectum), reproductive, and urinary tracts (if present) of many vertebrate animals. All amphibians, reptiles, birds, cartilagin ...
to increase the area for gas exchange
Gas exchange is the physical process by which gases move passively by diffusion across a surface. For example, this surface might be the air/water interface of a water body, the surface of a gas bubble in a liquid, a gas-permeable membrane, or a b ...
. Even with these adaptations, breathing is never fully accomplished without lungs. Lung ventilation is accomplished differently in each main reptile group. In squamates, the lungs are ventilated almost exclusively by the axial musculature. This is also the same musculature that is used during locomotion. Because of this constraint, most squamates are forced to hold their breath during intense runs. Some, however, have found a way around it. Varanids, and a few other lizard species, employ buccal pumping as a complement to their normal "axial breathing". This allows the animals to completely fill their lungs during intense locomotion, and thus remain aerobically active for a long time. Tegu lizards are known to possess a proto- diaphragm, which separates the pulmonary cavity from the visceral cavity. While not actually capable of movement, it does allow for greater lung inflation, by taking the weight of the viscera off the lungs.
Crocodilia
Crocodilia () is an order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorph pseudosuchia ...
ns actually have a muscular diaphragm that is analogous to the mammalian diaphragm. The difference is that the muscles for the crocodilian diaphragm pull the pubis (part of the pelvis, which is movable in crocodilians) back, which brings the liver down, thus freeing space for the lungs to expand. This type of diaphragmatic setup has been referred to as the "hepatic
The liver is a major metabolic organ (anatomy), organ exclusively found in vertebrates, which performs many essential biological Function (biology), functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of var ...
piston
A piston is a component of reciprocating engines, reciprocating pumps, gas compressors, hydraulic cylinders and pneumatic cylinders, among other similar mechanisms. It is the moving component that is contained by a cylinder (engine), cylinder a ...
". The airways form a number of double tubular chambers within each lung. On inhalation and exhalation air moves through the airways in the same direction, thus creating a unidirectional airflow through the lungs. A similar system is found in birds, monitor lizards and iguanas.
Most reptiles lack a secondary palate
The secondary palate is an anatomical structure that divides the nasal cavity from the oral cavity in many vertebrates.
In human embryology, it refers to that portion of the hard palate that is formed by the growth of the two palatine shelves med ...
, meaning that they must hold their breath while swallowing. Crocodilians have evolved a bony secondary palate that allows them to continue breathing while remaining submerged (and protect their brains against damage by struggling prey). Skinks (family Scincidae) also have evolved a bony secondary palate, to varying degrees. Snakes took a different approach and extended their trachea instead. Their tracheal extension sticks out like a fleshy straw, and allows these animals to swallow large prey without suffering from asphyxiation.
Turtles and tortoises
 How
How turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s breathe has been the subject of much study. To date, only a few species have been studied thoroughly enough to get an idea of how those turtles breathe. The varied results indicate that turtles have found a variety of solutions to this problem.
The difficulty is that most turtle shell
The turtle shell is a shield for the ventral and dorsal parts of turtles (the Order (biology), order Testudines), completely enclosing all the turtle's vital organs and in some cases even the head. It is constructed of modified bony elements such ...
s are rigid and do not allow for the type of expansion and contraction that other amniotes use to ventilate their lungs. Some turtles, such as the Indian flapshell ('' Lissemys punctata''), have a sheet of muscle that envelops the lungs. When it contracts, the turtle can exhale. When at rest, the turtle can retract the limbs into the body cavity and force air out of the lungs. When the turtle protracts its limbs, the pressure inside the lungs is reduced, and the turtle can suck air in. Turtle lungs are attached to the inside of the top of the shell (carapace), with the bottom of the lungs attached (via connective tissue) to the rest of the viscera. By using a series of special muscles (roughly equivalent to a diaphragm), turtles are capable of pushing their viscera up and down, resulting in effective respiration, since many of these muscles have attachment points in conjunction with their forelimbs (indeed, many of the muscles expand into the limb pockets during contraction).
Breathing during locomotion has been studied in three species, and they show different patterns. Adult female green sea turtles do not breathe as they crutch along their nesting beaches. They hold their breath during terrestrial locomotion and breathe in bouts as they rest. North American box turtles breathe continuously during locomotion, and the ventilation cycle is not coordinated with the limb movements.[
]
Sound production
Compared with frogs, birds, and mammals, reptiles are less vocal. Sound production is usually limited to hissing, which is produced merely by forcing air though a partly closed glottis
The glottis (: glottises or glottides) is the opening between the vocal folds (the rima glottidis). The glottis is crucial in producing sound from the vocal folds.
Etymology
From Ancient Greek ''γλωττίς'' (glōttís), derived from ''γ ...
and is not considered to be a true vocalization. The ability to vocalize exists in crocodilians, some lizards and turtles; and typically involves vibrating fold-like structures in the larynx
The larynx (), commonly called the voice box, is an organ (anatomy), organ in the top of the neck involved in breathing, producing sound and protecting the trachea against food aspiration. The opening of larynx into pharynx known as the laryngeal ...
or glottis. Some gecko
Geckos are small, mostly carnivorous lizards that have a wide distribution, found on every continent except Antarctica. Belonging to the infraorder Gekkota, geckos are found in warm climates. They range from .
Geckos are unique among lizards ...
s and turtles possess true vocal cords, which have elastin
Elastin is a protein encoded by the ''ELN'' gene in humans and several other animals. Elastin is a key component in the extracellular matrix of gnathostomes (jawed vertebrates). It is highly Elasticity (physics), elastic and present in connective ...
-rich connective tissue.
Hearing in snakes
Hearing in humans relies on 3 parts of the ear; the outer ear that directs sound waves into the ear canal, the middle ear that transmits incoming sound waves to the inner ear, and the inner ear that helps in hearing and keeping their balance. Unlike humans and other mammals, snakes do not possess an outer ear, a middle ear, and a tympanum but have an inner ear structure with cochlea
The cochlea is the part of the inner ear involved in hearing. It is a spiral-shaped cavity in the bony labyrinth, in humans making 2.75 turns around its axis, the modiolus (cochlea), modiolus. A core component of the cochlea is the organ of Cort ...
s directly connected to their jawbone. They are able to feel the vibrations generated from the sound waves in their jaw as they move on the ground. This is done by the use of mechanoreceptor
A mechanoreceptor, also called mechanoceptor, is a sensory receptor that responds to mechanical pressure or distortion. Mechanoreceptors are located on sensory neurons that convert mechanical pressure into action potential, electrical signals tha ...
s, sensory nerves that run along the body of snakes directing the vibrations along the spinal nerves to the brain. Snakes have a sensitive auditory perception and can tell which direction sound being made is coming from so that they can sense the presence of prey or predator but it is still unclear how sensitive snakes are to sound waves traveling through the air.
Skin
 Reptilian skin is covered in a horny
Reptilian skin is covered in a horny epidermis
The epidermis is the outermost of the three layers that comprise the skin, the inner layers being the dermis and Subcutaneous tissue, hypodermis. The epidermal layer provides a barrier to infection from environmental pathogens and regulates the ...
, making it watertight and enabling reptiles to live on dry land, in contrast to amphibians. Compared to mammalian skin, that of reptiles is rather thin and lacks the thick dermal
The dermis or corium is a layer of skin between the epidermis (with which it makes up the cutis) and subcutaneous tissues, that primarily consists of dense irregular connective tissue and cushions the body from stress and strain. It is divided i ...
layer that produces leather
Leather is a strong, flexible and durable material obtained from the tanning (leather), tanning, or chemical treatment, of animal skins and hides to prevent decay. The most common leathers come from cattle, sheep, goats, equine animals, buffal ...
in mammals.
Exposed parts of reptiles are protected by scales
Scale or scales may refer to:
Mathematics
* Scale (descriptive set theory), an object defined on a set of points
* Scale (ratio), the ratio of a linear dimension of a model to the corresponding dimension of the original
* Scale factor, a number ...
or scutes, sometimes with a bony base (osteoderm
Osteoderms are bony deposits forming scales, plates, or other structures based in the dermis. Osteoderms are found in many groups of extant and extinct reptiles and amphibians, including lizards, crocodilians, frogs, temnospondyls (extinct amph ...
s), forming armor
Armour (Commonwealth English) or armor (American English; see American and British English spelling differences#-our, -or, spelling differences) is a covering used to protect an object, individual, or vehicle from physical injury or damage, e ...
. In lepidosaurs, such as lizards and snakes, the whole skin is covered in overlapping epidermal scales. Such scales were once thought to be typical of the class Reptilia as a whole, but are now known to occur only in lepidosaurs. The scales found in turtles and crocodiles are of dermal
The dermis or corium is a layer of skin between the epidermis (with which it makes up the cutis) and subcutaneous tissues, that primarily consists of dense irregular connective tissue and cushions the body from stress and strain. It is divided i ...
, rather than epidermal, origin and are properly termed scutes. In turtles, the body is hidden inside a hard shell composed of fused scutes.
Lacking a thick dermis, reptilian leather is not as strong as mammalian leather. It is used in leather-wares for decorative purposes for shoes, belts and handbags, particularly crocodile skin.
Shedding
Reptiles shed their skin through a process called ecdysis
Ecdysis is the moulting of the cuticle in many invertebrates of the clade Ecdysozoa. Since the cuticle of these animals typically forms a largely inelastic exoskeleton, it is shed during growth and a new, larger covering is formed. The remnant ...
which occurs continuously throughout their lifetime. In particular, younger reptiles tend to shed once every five to six weeks while adults shed three to four times a year. Younger reptiles shed more because of their rapid growth rate. Once full size, the frequency of shedding drastically decreases. The process of ecdysis involves forming a new layer of skin under the old one. Proteolytic enzymes and lymphatic fluid is secreted between the old and new layers of skin. Consequently, this lifts the old skin from the new one allowing shedding to occur.
Excretion
Excretion
Excretion is elimination of metabolic waste, which is an essential process in all organisms. In vertebrates, this is primarily carried out by the lungs, Kidney (vertebrates), kidneys, and skin. This is in contrast with secretion, where the substa ...
is performed mainly by two small kidney
In humans, the kidneys are two reddish-brown bean-shaped blood-filtering organ (anatomy), organs that are a multilobar, multipapillary form of mammalian kidneys, usually without signs of external lobulation. They are located on the left and rig ...
s. In diapsids, uric acid
Uric acid is a heterocyclic compound of carbon, nitrogen, oxygen, and hydrogen with the Chemical formula, formula C5H4N4O3. It forms ions and salts known as urates and acid urates, such as ammonium acid urate. Uric acid is a product of the meta ...
is the main nitrogen
Nitrogen is a chemical element; it has Symbol (chemistry), symbol N and atomic number 7. Nitrogen is a Nonmetal (chemistry), nonmetal and the lightest member of pnictogen, group 15 of the periodic table, often called the Pnictogen, pnictogens. ...
ous waste product; turtles, like mammal
A mammal () is a vertebrate animal of the Class (biology), class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three ...
s, excrete mainly urea
Urea, also called carbamide (because it is a diamide of carbonic acid), is an organic compound with chemical formula . This amide has two Amine, amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest am ...
. Unlike the kidneys of mammals and birds, reptile kidneys are unable to produce liquid urine more concentrated than their body fluid. This is because they lack a specialized structure called a loop of Henle
In the kidney, the loop of Henle () (or Henle's loop, Henle loop, nephron loop or its Latin counterpart ''ansa nephroni'') is the portion of a nephron that leads from the proximal convoluted tubule to the distal convoluted tubule. Named after it ...
, which is present in the nephron
The nephron is the minute or microscopic structural and functional unit of the kidney. It is composed of a renal corpuscle and a renal tubule. The renal corpuscle consists of a tuft of capillaries called a glomerulus and a cup-shaped structu ...
s of birds and mammals. Because of this, many reptiles use the colon to aid in the reabsorption
In renal physiology, reabsorption, more specifically tubular reabsorption, is the process by which the nephron removes water and solutes from the tubular fluid (pre-urine) and returns them to the circulating blood. It is called ''reabsorption' ...
of water. Some are also able to take up water stored in the bladder
The bladder () is a hollow organ in humans and other vertebrates that stores urine from the kidneys. In placental mammals, urine enters the bladder via the ureters and exits via the urethra during urination. In humans, the bladder is a distens ...
. Excess salts are also excreted by nasal and lingual salt glands in some reptiles.
In all reptiles, the urinogenital ducts and the rectum
The rectum (: rectums or recta) is the final straight portion of the large intestine in humans and some other mammals, and the gut in others. Before expulsion through the anus or cloaca, the rectum stores the feces temporarily. The adult ...
both empty into an organ called a cloaca
A cloaca ( ), : cloacae ( or ), or vent, is the rear orifice that serves as the only opening for the digestive (rectum), reproductive, and urinary tracts (if present) of many vertebrate animals. All amphibians, reptiles, birds, cartilagin ...
. In some reptiles, a midventral wall in the cloaca may open into a urinary bladder, but not all. It is present in all turtles and tortoises as well as most lizards, but is lacking in the monitor lizard, the legless lizard
Legless lizard may refer to any of several groups of lizards that have independently lost limbs or reduced them to the point of being of no use in locomotion.Pough ''et al.'' 1992. Herpetology: Third Edition. Pearson Prentice Hall:Pearson Education ...
s. It is absent in the snakes, alligators, and crocodiles.
Many turtles and lizards have proportionally very large bladders. Charles Darwin
Charles Robert Darwin ( ; 12 February 1809 – 19 April 1882) was an English Natural history#Before 1900, naturalist, geologist, and biologist, widely known for his contributions to evolutionary biology. His proposition that all speci ...
noted that the Galapagos tortoise had a bladder which could store up to 20% of its body weight. Such adaptations are the result of environments such as remote islands and deserts where water is very scarce. Other desert-dwelling reptiles have large bladders that can store a long-term reservoir of water for up to several months and aid in osmoregulation
Osmoregulation is the active regulation of the osmotic pressure of an organism's body fluids, detected by osmoreceptors, to maintain the homeostasis of the organism's water content; that is, it maintains the fluid balance and the concentration ...
.
Turtles have two or more accessory urinary bladders, located lateral to the neck of the urinary bladder and dorsal to the pubis, occupying a significant portion of their body cavity. Their bladder is also usually bilobed with a left and right section. The right section is located under the liver, which prevents large stones from remaining in that side while the left section is more likely to have calculi.
Digestion

 Most reptiles are insectivorous or carnivorous and have simple and comparatively short digestive tracts due to meat being fairly simple to break down and digest.
Most reptiles are insectivorous or carnivorous and have simple and comparatively short digestive tracts due to meat being fairly simple to break down and digest. Digestion
Digestion is the breakdown of large insoluble food compounds into small water-soluble components so that they can be absorbed into the blood plasma. In certain organisms, these smaller substances are absorbed through the small intestine into th ...
is slower than in mammals
A mammal () is a vertebrate animal of the class Mammalia (). Mammals are characterised by the presence of milk-producing mammary glands for feeding their young, a broad neocortex region of the brain, fur or hair, and three middle e ...
, reflecting their lower resting metabolism
Metabolism (, from ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run cellular processes; the co ...
and their inability to divide and masticate their food. Their poikilotherm metabolism has very low energy requirements, allowing large reptiles like crocodiles and large constrictors to live from a single large meal for months, digesting it slowly.[
While modern reptiles are predominantly carnivorous, during the early history of reptiles several groups produced some herbivorous ]megafauna
In zoology, megafauna (from Ancient Greek, Greek μέγας ''megas'' "large" and Neo-Latin ''fauna'' "animal life") are large animals. The precise definition of the term varies widely, though a common threshold is approximately , this lower en ...
: in the Paleozoic
The Paleozoic ( , , ; or Palaeozoic) Era is the first of three Era (geology), geological eras of the Phanerozoic Eon. Beginning 538.8 million years ago (Ma), it succeeds the Neoproterozoic (the last era of the Proterozoic Eon) and ends 251.9 Ma a ...
, the pareiasaur
Pareiasaurs (meaning "cheek lizards") are an extinct clade of large, herbivorous parareptiles. Members of the group were armoured with osteoderms which covered large areas of the body. They first appeared in southern Pangea during the Middle Per ...
s; and in the Mesozoic
The Mesozoic Era is the Era (geology), era of Earth's Geologic time scale, geological history, lasting from about , comprising the Triassic, Jurassic and Cretaceous Period (geology), Periods. It is characterized by the dominance of archosaurian r ...
several lines of dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
s.[ Today, ]turtle
Turtles are reptiles of the order (biology), order Testudines, characterized by a special turtle shell, shell developed mainly from their ribs. Modern turtles are divided into two major groups, the Pleurodira (side necked turtles) and Crypt ...
s are the only predominantly herbivorous reptile group, but several lines of agamas and iguanas have evolved to live wholly or partly on plants.gastrolith
A gastrolith, also called a stomach stone or gizzard stone, is a rock held inside a gastrointestinal tract. Gastroliths in some species are retained in the muscular gizzard and used to grind food in animals lacking suitable grinding teeth. In ...
s) to aid in digestion: The rocks are washed around in the stomach, helping to grind up plant matter.[ Fossil gastroliths have been found associated with both ornithopods and ]sauropods
Sauropoda (), whose members are known as sauropods (; from '' sauro-'' + '' -pod'', 'lizard-footed'), is a clade of saurischian ('lizard-hipped') dinosaurs. Sauropods had very long necks, long tails, small heads (relative to the rest of their b ...
, though whether they actually functioned as a gastric mill in the latter is disputed. Salt water crocodiles also use gastroliths as ballast
Ballast is dense material used as a weight to provide stability to a vehicle or structure. Ballast, other than cargo, may be placed in a vehicle, often a ship or the gondola of a balloon or airship, to provide stability. A compartment within ...
, stabilizing them in the water or helping them to dive. A dual function as both stabilizing ballast and digestion aid has been suggested for gastroliths found in plesiosaur
The Plesiosauria or plesiosaurs are an Order (biology), order or clade of extinct Mesozoic marine reptiles, belonging to the Sauropterygia.
Plesiosaurs first appeared in the latest Triassic Period (geology), Period, possibly in the Rhaetian st ...
s.
Nerves
The reptilian nervous system contains the same basic part of the amphibian
Amphibians are ectothermic, anamniote, anamniotic, tetrapod, four-limbed vertebrate animals that constitute the class (biology), class Amphibia. In its broadest sense, it is a paraphyletic group encompassing all Tetrapod, tetrapods, but excl ...
brain, but the reptile cerebrum
The cerebrum (: cerebra), telencephalon or endbrain is the largest part of the brain, containing the cerebral cortex (of the two cerebral hemispheres) as well as several subcortical structures, including the hippocampus, basal ganglia, and olfac ...
and cerebellum
The cerebellum (: cerebella or cerebellums; Latin for 'little brain') is a major feature of the hindbrain of all vertebrates. Although usually smaller than the cerebrum, in some animals such as the mormyrid fishes it may be as large as it or eve ...
are slightly larger. Most typical sense organs are well developed with certain exceptions, most notably the snake
Snakes are elongated limbless reptiles of the suborder Serpentes (). Cladistically squamates, snakes are ectothermic, amniote vertebrates covered in overlapping scales much like other members of the group. Many species of snakes have s ...
's lack of external ears (middle and inner ears are present). There are twelve pairs of cranial nerves
Cranial nerves are the nerves that emerge directly from the brain (including the brainstem), of which there are conventionally considered twelve pairs. Cranial nerves relay information between the brain and parts of the body, primarily to and f ...
. Due to their short cochlea, reptiles use electrical tuning to expand their range of audible frequencies.
Vision
Most reptiles are diurnal animals. The vision is typically adapted to daylight conditions, with color vision and more advanced visual depth perception
Depth perception is the ability to perceive distance to objects in the world using the visual system and visual perception. It is a major factor in perceiving the world in three dimensions.
Depth sensation is the corresponding term for non-hum ...
than in amphibians and most mammals.
Reptiles usually have excellent vision, allowing them to detect shapes and motions at long distances. They often have poor vision in low-light conditions. Birds, crocodiles and turtles have three types of photoreceptor: rods, single cones and double cones, which gives them sharp color vision and enables them to see ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
wavelengths.third eye
The third eye (also called the mind's eye or inner eye) is an invisible eye, usually depicted as located on the forehead, supposed to provide perception beyond ordinary sight. In Hinduism, the third eye refers to the ajna (or brow) chakra. In bot ...
, pineal eye or pineal gland
The pineal gland (also known as the pineal body or epiphysis cerebri) is a small endocrine gland in the brain of most vertebrates. It produces melatonin, a serotonin-derived hormone, which modulates sleep, sleep patterns following the diurnal c ...
. This "eye" does not work the same way as a normal eye does as it has only a rudimentary retina and lens and thus, cannot form images. It is, however, sensitive to changes in light and dark and can detect movement.[
Some snakes have extra sets of visual organs (in the loosest sense of the word) in the form of pits sensitive to ]infrared
Infrared (IR; sometimes called infrared light) is electromagnetic radiation (EMR) with wavelengths longer than that of visible light but shorter than microwaves. The infrared spectral band begins with the waves that are just longer than those ...
radiation (heat). Such heat-sensitive pits are particularly well developed in the pit vipers
The Crotalinae, commonly known as pit vipers,Mehrtens JM (1987). ''Living Snakes of the World in Color''. New York: Sterling Publishers. 480 pp. . or pit adders, are a subfamily of vipers found in Asia and the Americas. Like all other vipers, ...
, but are also found in boas and pythons. These pits allow the snakes to sense the body heat of birds and mammals, enabling pit vipers to hunt rodents in the dark.
Most reptiles, as well as birds, possess a nictitating membrane
The nictitating membrane (from Latin '' nictare'', to blink) is a transparent or translucent third eyelid present in some animals that can be drawn across the eye from the medial canthus to protect and moisten it while maintaining vision. Most ...
, a translucent third eyelid which is drawn over the eye from the inner corner. In crocodilians, it protects its eyeball surface while allowing a degree of vision underwater. However, many squamates, geckos and snakes in particular, lack eyelids, which are replaced by a transparent scale. This is called the brille, spectacle, or eyecap. The brille is usually not visible, except for when the snake molts, and it protects the eyes from dust and dirt.
Reproduction


 Reptiles generally
Reptiles generally reproduce sexually
Sexual reproduction is a type of reproduction that involves a complex Biological life cycle, life cycle in which a gamete (haploid reproductive cells, such as a sperm or egg cell) with a single set of chromosomes combines with another gamete to p ...
, though some are capable of asexual reproduction
Asexual reproduction is a type of reproduction that does not involve the fusion of gametes or change in the number of chromosomes. The offspring that arise by asexual reproduction from either unicellular or multicellular organisms inherit the f ...
. All reproductive activity occurs through the cloaca
A cloaca ( ), : cloacae ( or ), or vent, is the rear orifice that serves as the only opening for the digestive (rectum), reproductive, and urinary tracts (if present) of many vertebrate animals. All amphibians, reptiles, birds, cartilagin ...
, the single exit/entrance at the base of the tail where waste is also eliminated. Most reptiles have copulatory organs, which are usually retracted or inverted and stored inside the body. In turtles and crocodilians, the male has a single median penis
A penis (; : penises or penes) is a sex organ through which male and hermaphrodite animals expel semen during copulation (zoology), copulation, and through which male placental mammals and marsupials also Urination, urinate.
The term ''pen ...
, while squamates, including snakes and lizards, possess a pair of hemipenes, only one of which is typically used in each session. Tuatara, however, lack copulatory organs, and so the male and female simply press their cloacas together as the male discharges sperm.
Most reptiles lay amniotic eggs covered with leathery or calcareous shells. An amnion
The amnion (: amnions or amnia) is a membrane that closely covers human and various other embryos when they first form. It fills with amniotic fluid, which causes the amnion to expand and become the amniotic sac that provides a protective envir ...
(5), chorion
The chorion is the outermost fetal membrane around the embryo in mammals, birds and reptiles (amniotes). It is also present around the embryo of other animals, like insects and molluscs.
Structure
In humans and other therian mammals, the cho ...
(6), and allantois (8) are present during embryo
An embryo ( ) is the initial stage of development for a multicellular organism. In organisms that reproduce sexually, embryonic development is the part of the life cycle that begins just after fertilization of the female egg cell by the male sp ...
nic life. The eggshell (1) protects the crocodile embryo (11) and keeps it from drying out, but it is flexible to allow gas exchange. The chorion (6) aids in gas exchange between the inside and outside of the egg. It allows carbon dioxide to exit the egg and oxygen gas to enter the egg. The albumin (9) further protects the embryo and serves as a reservoir for water and protein. The allantois (8) is a sac that collects the metabolic waste produced by the embryo. The amniotic sac (10) contains amniotic fluid (12) which protects and cushions the embryo. The amnion (5) aids in osmoregulation and serves as a saltwater reservoir. The yolk sac (2) surrounding the yolk (3) contains protein and fat rich nutrients that are absorbed by the embryo via vessels (4) that allow the embryo to grow and metabolize. The air space (7) provides the embryo with oxygen while it is hatching. This ensures that the embryo will not suffocate while it is hatching. There are no larva
A larva (; : larvae ) is a distinct juvenile form many animals undergo before metamorphosis into their next life stage. Animals with indirect development such as insects, some arachnids, amphibians, or cnidarians typically have a larval phase ...
l stages of development. Viviparity
In animals, viviparity is development of the embryo inside the body of the mother, with the maternal circulation providing for the metabolic needs of the embryo's development, until the mother gives birth to a fully or partially developed juv ...
and ovoviviparity
Ovoviviparity, ovovivipary, ovivipary, or aplacental viviparity is a "bridging" form of reproduction between egg-laying oviparous and live-bearing viviparous reproduction. Ovoviviparous animals possess embryos that develop inside eggs that r ...
have evolved in squamates and many extinct clades of reptiles. Among squamates, many species, including all boas and most vipers, use this mode of reproduction. The degree of viviparity varies; some species simply retain the eggs until just before hatching, others provide maternal nourishment to supplement the yolk, and yet others lack any yolk and provide all nutrients via a structure similar to the mammalian placenta
The placenta (: placentas or placentae) is a temporary embryonic and later fetal organ that begins developing from the blastocyst shortly after implantation. It plays critical roles in facilitating nutrient, gas, and waste exchange between ...
. The earliest documented case of viviparity in reptiles is the Early Permian
The Permian ( ) is a geologic period and System (stratigraphy), stratigraphic system which spans 47 million years, from the end of the Carboniferous Period million years ago (Mya), to the beginning of the Triassic Period 251.902 Mya. It is the s ...
mesosaur
Mesosaurs ("middle lizards") were a group of small aquatic reptiles that lived during the early Permian period ( Cisuralian), roughly 299 to 270 million years ago. Mesosaurs were the first known aquatic reptiles, having apparently returned to a ...
s,mosasaur
Mosasaurs (from Latin ''Mosa'' meaning the 'Meuse', and Ancient Greek, Greek ' meaning 'lizard') are an extinct group of large aquatic reptiles within the family Mosasauridae that lived during the Late Cretaceous. Their first fossil remains wer ...
s, ichthyosaurs, and Sauropterygia
Sauropterygia ("lizard flippers") is an extinct taxon of diverse, aquatic diapsid reptiles that developed from terrestrial ancestors soon after the end-Permian extinction and flourished during the Triassic before all except for the Plesiosau ...
, a group that includes pachypleurosaurs and Plesiosauria
The Plesiosauria or plesiosaurs are an order or clade of extinct Mesozoic marine reptiles, belonging to the Sauropterygia.
Plesiosaurs first appeared in the latest Triassic Period, possibly in the Rhaetian stage, about 203 million year ...
.[
Asexual reproduction has been identified in squamates in six families of lizards and one snake. In some species of squamates, a population of females is able to produce a unisexual diploid clone of the mother. This form of asexual reproduction, called ]parthenogenesis
Parthenogenesis (; from the Greek + ) is a natural form of asexual reproduction in which the embryo develops directly from an egg without need for fertilization. In animals, parthenogenesis means the development of an embryo from an unfertiliz ...
, occurs in several species of gecko
Geckos are small, mostly carnivorous lizards that have a wide distribution, found on every continent except Antarctica. Belonging to the infraorder Gekkota, geckos are found in warm climates. They range from .
Geckos are unique among lizards ...
, and is particularly widespread in the teiids (especially ''Aspidocelis'') and lacertids ('' Lacerta''). In captivity, Komodo dragon
The Komodo dragon (''Varanus komodoensis''), also known as the Komodo monitor, is a large reptile of the monitor lizard family Varanidae that is endemic to the Indonesian islands of Komodo (island), Komodo, Rinca, Flores, Gili Dasami, and Gili ...
s (Varanidae) have reproduced by parthenogenesis
Parthenogenesis (; from the Greek + ) is a natural form of asexual reproduction in which the embryo develops directly from an egg without need for fertilization. In animals, parthenogenesis means the development of an embryo from an unfertiliz ...
.
Parthenogenetic species are suspected to occur among chameleon
Chameleons or chamaeleons (Family (biology), family Chamaeleonidae) are a distinctive and highly specialized clade of Old World lizards with 200 species described as of June 2015. The members of this Family (biology), family are best known for ...
s, agamids, xantusiids, and typhlopids.
Some reptiles exhibit temperature-dependent sex determination
Temperature-dependent sex determination (TSD) is a type of environmental sex determination in which the temperatures experienced during embryonic/larval development determine the sex of the offspring. It is observed in reptiles and teleost fish, ...
(TDSD), in which the incubation temperature determines whether a particular egg hatches as male or female. TDSD is most common in turtles and crocodiles, but also occurs in lizards and tuatara. To date, there has been no confirmation of whether TDSD occurs in snakes.
Longevity
Giant tortoise
Tortoises ( ) are reptiles of the family Testudinidae of the order Testudines (Latin for "tortoise"). Like other turtles, tortoises have a shell to protect from predation and other threats. The shell in tortoises is generally hard, and like o ...
s are among the longest-lived vertebrate animals (over 100 years by some estimates) and have been used as a model for studying longevity
Longevity may refer to especially long-lived members of a population, whereas ''life expectancy'' is defined Statistics, statistically as the average number of years remaining at a given age. For example, a population's life expectancy at birth ...
.[Quesada V, Freitas-Rodríguez S, Miller J, Pérez-Silva JG, Jiang ZF, Tapia W, Santiago-Fernández O, Campos-Iglesias D, Kuderna LFK, Quinzin M, Álvarez MG, Carrero D, Beheregaray LB, Gibbs JP, Chiari Y, Glaberman S, Ciofi C, Araujo-Voces M, Mayoral P, Arango JR, Tamargo-Gómez I, Roiz-Valle D, Pascual-Torner M, Evans BR, Edwards DL, Garrick RC, Russello MA, Poulakakis N, Gaughran SJ, Rueda DO, Bretones G, Marquès-Bonet T, White KP, Caccone A, López-Otín C. Giant tortoise genomes provide insights into longevity and age-related disease. Nat Ecol Evol. 2019 Jan;3(1):87-95. doi: 10.1038/s41559-018-0733-x. Epub 2018 Dec 3. PMID 30510174; PMCID: PMC6314442] DNA analysis of the genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
s of Lonesome George, the iconic last member of '' Chelonoidis abingdonii'', and the Aldabra giant tortoise
The Aldabra giant tortoise (''Aldabrachelys gigantea'') is a species of tortoise in the family Testudinidae and genus '' Aldabrachelys''. The species is endemic to the Seychelles, with the nominate subspecies, ''A. g. gigantea'' native to Alda ...
''Aldabrachelys gigantea'' led to the detection of lineage-specific variants affecting DNA repair
DNA repair is a collection of processes by which a cell (biology), cell identifies and corrects damage to the DNA molecules that encode its genome. A weakened capacity for DNA repair is a risk factor for the development of cancer. DNA is cons ...
genes that might contribute to our understanding of increased lifespan.[
]
Cognition
Reptiles are generally considered less intelligent than mammals and birds.encephalization quotient
Encephalization quotient (EQ), encephalization level (EL), or just encephalization is a relative brain size measure that is defined as the ratio between observed and predicted brain mass for an animal of a given size, based on nonlinear regre ...
being about one tenth of that of mammals, though larger reptiles can show more complex brain development. Larger lizards, like the monitors, are known to exhibit complex behavior, including cooperation and cognitive abilities allowing them to optimize their foraging
Foraging is searching for wild food resources. It affects an animal's fitness because it plays an important role in an animal's ability to survive and reproduce. Foraging theory is a branch of behavioral ecology that studies the foraging behavi ...
and territoriality over time. Crocodiles have relatively larger brains and show a fairly complex social structure. The Komodo dragon
The Komodo dragon (''Varanus komodoensis''), also known as the Komodo monitor, is a large reptile of the monitor lizard family Varanidae that is endemic to the Indonesian islands of Komodo (island), Komodo, Rinca, Flores, Gili Dasami, and Gili ...
is even known to engage in play,operant conditioning
Operant conditioning, also called instrumental conditioning, is a learning process in which voluntary behaviors are modified by association with the addition (or removal) of reward or aversive stimuli. The frequency or duration of the behavior ma ...
, visual discrimination and retained learned behaviors with long-term memory. Sea turtles have been regarded as having simple brains, but their flippers are used for a variety of foraging tasks (holding, bracing, corralling) in common with marine mammals.
There is evidence that reptiles are sentient and able to feel emotions including anxiety
Anxiety is an emotion characterised by an unpleasant state of inner wikt:turmoil, turmoil and includes feelings of dread over Anticipation, anticipated events. Anxiety is different from fear in that fear is defined as the emotional response ...
and pleasure
Pleasure is experience that feels good, that involves the enjoyment of something. It contrasts with pain or suffering, which are forms of feeling bad. It is closely related to value, desire and action: humans and other conscious animals find ...
.
Defense mechanisms
Many small reptiles, such as snakes and lizards, that live on the ground or in the water are vulnerable to being preyed on by all kinds of carnivorous animals. Thus, avoidance is the most common form of defense in reptiles. At the first sign of danger, most snakes and lizards crawl away into the undergrowth, and turtles and crocodiles will plunge into water and sink out of sight.
Camouflage and warning
 Reptiles tend to avoid confrontation through
Reptiles tend to avoid confrontation through camouflage
Camouflage is the use of any combination of materials, coloration, or illumination for concealment, either by making animals or objects hard to see, or by disguising them as something else. Examples include the leopard's spotted coat, the b ...
. Two major groups of reptile predators are birds and other reptiles, both of which have well-developed color vision. Thus the skins of many reptiles have cryptic coloration of plain or mottled gray, green, and brown to allow them to blend into the background of their natural environment. Aided by the reptiles' capacity for remaining motionless for long periods, the camouflage of many snakes is so effective that people or domestic animals are most typically bitten because they accidentally step on them.
When camouflage fails to protect them, blue-tongued skink
Blue-tongued skinks comprise the Australasian genus ''Tiliqua'', which contains some of the largest members of the skink family (biology), family (Scincidae). They are commonly called blue-tongued lizards or simply blue-tongues or blueys in Aust ...
s will try to ward off attackers by displaying their blue tongues, and the frill-necked lizard will display its brightly colored frill. These same displays are used in territorial disputes and during courtship.Gila monster
The Gila monster (''Heloderma suspectum'', ) is a species of venomous lizard native to the Southwestern United States and the northwestern Mexico, Mexican state of Sonora. It is a heavy, slow-moving reptile, up to long, and it is the only ve ...
and the beaded lizard) and many of the coral snake
Coral snakes are a large group of elapid snakes that can be divided into two distinct groups, the Old World coral snakes and New World coral snakes. There are 27 species of Old World coral snakes, in three genera ('' Calliophis'', '' Hemibungar ...
s have high-contrast warning coloration, warning potential predators they are venomous. A number of non-venomous North American snake species have colorful markings similar to those of the coral snake, an oft cited example of Batesian mimicry
Batesian mimicry is a form of mimicry where a harmless species has evolved to imitate the warning signals of a harmful species directed at a predator of them both. It is named after the English naturalist Henry Walter Bates, who worked on butt ...
.
Alternative defense in snakes
Camouflage does not always fool a predator. When caught out, snake species adopt different defensive tactics and use a complicated set of behaviors when attacked. Some species, like cobras or hognose snakes, first elevate their head and spread out the skin of their neck in an effort to look large and threatening. Failure of this strategy may lead to other measures practiced particularly by cobras, vipers, and closely related species, which use venom
Venom or zootoxin is a type of toxin produced by an animal that is actively delivered through a wound by means of a bite, sting, or similar action. The toxin is delivered through a specially evolved ''venom apparatus'', such as fangs or a sti ...
to attack. The venom is modified saliva, delivered through fangs from a venom gland. Some non-venomous snakes, such as American hognose snakes or European grass snake, play dead when in danger; some, including the grass snake, exude a foul-smelling liquid to deter attackers.
Defense in crocodilians
When a crocodilian
Crocodilia () is an Order (biology), order of semiaquatic, predatory reptiles that are known as crocodilians. They first appeared during the Late Cretaceous and are the closest living relatives of birds. Crocodilians are a type of crocodylomorp ...
is concerned about its safety, it will gape to expose the teeth and tongue. If this does not work, the crocodilian gets a little more agitated and typically begins to make hissing sounds. After this, the crocodilian will start to change its posture dramatically to make itself look more intimidating. The body is inflated to increase apparent size. If absolutely necessary, it may decide to attack an enemy.
 Some species try to bite immediately. Some will use their heads as sledgehammers and literally smash an opponent, some will rush or swim toward the threat from a distance, even chasing the opponent onto land or galloping after it. The main weapon in all crocodiles is the bite, which can generate very high bite force. Many species also possess canine-like teeth. These are used primarily for seizing prey, but are also used in fighting and display.
Some species try to bite immediately. Some will use their heads as sledgehammers and literally smash an opponent, some will rush or swim toward the threat from a distance, even chasing the opponent onto land or galloping after it. The main weapon in all crocodiles is the bite, which can generate very high bite force. Many species also possess canine-like teeth. These are used primarily for seizing prey, but are also used in fighting and display.
Shedding and regenerating tails
Geckos
Geckos are small, mostly carnivorous lizards that have a wide distribution, found on every continent except Antarctica. Belonging to the infraorder Gekkota, geckos are found in warm climates. They range from .
Geckos are unique among lizards f ...
, skinks, and some other lizards that are captured by the tail will shed part of the tail structure through a process called autotomy and thus be able to flee. The detached tail will continue to thrash, creating a deceptive sense of continued struggle and distracting the predator's attention from the fleeing prey animal. The detached tails of leopard gecko
The leopard gecko or common leopard gecko (''Eublepharis macularius'') is a ground-dwelling gecko native to the rocky dry grassland and desert regions of Afghanistan, Iran, Pakistan, India, and Nepal. The leopard gecko is a popular pet, and due ...
s can wiggle for up to 20 minutes. The tail grows back in most species, but some, like crested geckos, lose their tails for the rest of their lives. In many species the tails are of a separate and dramatically more intense color than the rest of the body so as to encourage potential predators to strike for the tail first. In the shingleback skink and some species of geckos, the tail is short and broad and resembles the head, so that the predators may attack it rather than the more vulnerable front part.
Relations with humans
In cultures and religions
 Dinosaurs have been widely depicted in culture since the English palaeontologist
Dinosaurs have been widely depicted in culture since the English palaeontologist Richard Owen
Sir Richard Owen (20 July 1804 – 18 December 1892) was an English biologist, comparative anatomy, comparative anatomist and paleontology, palaeontologist. Owen is generally considered to have been an outstanding naturalist with a remarkabl ...
coined the name ''dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared during the Triassic Geological period, period, between 243 and 233.23 million years ago (mya), although the exact origin and timing of the #Evolutio ...
'' in 1842. As soon as 1854, the Crystal Palace Dinosaurs were on display to the public in south London.Charles Dickens
Charles John Huffam Dickens (; 7 February 1812 – 9 June 1870) was an English novelist, journalist, short story writer and Social criticism, social critic. He created some of literature's best-known fictional characters, and is regarded by ...
placed a ''Megalosaurus
''Megalosaurus'' (meaning "great lizard", from Ancient Greek, Greek , ', meaning 'big', 'tall' or 'great' and , ', meaning 'lizard') is an extinct genus of large carnivorous theropod dinosaurs of the Middle Jurassic Epoch (Bathonian stage, 166 ...
'' in the first chapter of his novel ''Bleak House
''Bleak House'' is a novel by English author Charles Dickens, first published as a 20-episode Serial (literature), serial between 12 March 1852 and 12 September 1853. The novel has many characters and several subplots, and is told partly by th ...
'' in 1852.
The dinosaurs featured in books, films, television programs, artwork, and other media have been used for both education and entertainment. The depictions range from the realistic, as in the television documentaries
A documentary film (often described simply as a documentary) is a nonfiction motion picture intended to "document reality, primarily for instruction, education or maintaining a historical record". The American author and media analyst Bill ...
of the 1990s and first decade of the 21st century, to the fantastic, as in the monster movie
A monster movie, monster film, creature feature or giant monster film is a film that focuses on one or more characters struggling to survive attacks by one or more antagonistic monsters, often abnormally megafauna, large ones. The film may also ...
s of the 1950s and 1960s.pharaoh
Pharaoh (, ; Egyptian language, Egyptian: ''wikt:pr ꜥꜣ, pr ꜥꜣ''; Meroitic language, Meroitic: 𐦲𐦤𐦧, ; Biblical Hebrew: ''Parʿō'') was the title of the monarch of ancient Egypt from the First Dynasty of Egypt, First Dynasty ( ...
. It was worshipped as one of the gods and was also used for sinister purposes: murder of an adversary and ritual suicide (Cleopatra
Cleopatra VII Thea Philopator (; The name Cleopatra is pronounced , or sometimes in both British and American English, see and respectively. Her name was pronounced in the Greek dialect of Egypt (see Koine Greek phonology). She was ...
). In Greek mythology
Greek mythology is the body of myths originally told by the Ancient Greece, ancient Greeks, and a genre of ancient Greek folklore, today absorbed alongside Roman mythology into the broader designation of classical mythology. These stories conc ...
, snakes are associated with deadly antagonists, as a chthonic
In Greek mythology, deities referred to as chthonic () or chthonian () were gods or spirits who inhabited the underworld or existed in or under the earth, and were typically associated with death or fertility. The terms "chthonic" and "chthonian" ...
symbol, roughly translated as ''earthbound''. The nine-headed Lernaean Hydra
The Lernaean Hydra or Hydra of Lerna (), more often known simply as the Hydra, is a serpentine lake monster in Greek mythology and Roman mythology. Its lair was the lake of Lerna in the Argolid, which was also the site of the myth of the Dan ...
that Hercules
Hercules (, ) is the Roman equivalent of the Greek divine hero Heracles, son of Jupiter and the mortal Alcmena. In classical mythology, Hercules is famous for his strength and for his numerous far-ranging adventures.
The Romans adapted the Gr ...
defeated and the three Gorgon
The Gorgons ( ; ), in Greek mythology, are three monstrous sisters, Stheno, Euryale, and Medusa, said to be the daughters of Phorcys and Ceto. They lived near their sisters the Graeae, and were able to turn anyone who looked at them to sto ...
sisters are children of Gaia
In Greek mythology, Gaia (; , a poetic form of ('), meaning 'land' or 'earth'),, , . also spelled Gaea (), is the personification of Earth. Gaia is the ancestral mother—sometimes parthenogenic—of all life. She is the mother of Uranus (S ...
, the earth. Medusa
In Greek mythology, Medusa (; ), also called Gorgo () or the Gorgon, was one of the three Gorgons. Medusa is generally described as a woman with living snakes in place of hair; her appearance was so hideous that anyone who looked upon her wa ...
was one of the three Gorgon sisters who Perseus
In Greek mythology, Perseus (, ; Greek language, Greek: Περσεύς, Romanization of Greek, translit. Perseús) is the legendary founder of the Perseid dynasty. He was, alongside Cadmus and Bellerophon, the greatest Greek hero and slayer of ...
defeated. Medusa is described as a hideous mortal, with snakes instead of hair and the power to turn men to stone with her gaze. After killing her, Perseus gave her head to Athena
Athena or Athene, often given the epithet Pallas, is an ancient Greek religion, ancient Greek goddess associated with wisdom, warfare, and handicraft who was later syncretism, syncretized with the Roman goddess Minerva. Athena was regarde ...
who fixed it to her shield called the Aegis
The aegis ( ; ''aigís''), as stated in the ''Iliad'', is a device carried by Athena and Zeus, variously interpreted as an animal skin or a shield and sometimes featuring the head of a Gorgon. There may be a connection with a deity named Aex, a ...
. The Titans
In Greek mythology, the Titans ( ; ) were the pre-Twelve Olympians, Olympian gods. According to the ''Theogony'' of Hesiod, they were the twelve children of the primordial parents Uranus (mythology), Uranus (Sky) and Gaia (Earth). The six male ...
are depicted in art with their legs replaced by bodies of snakes for the same reason: They are children of Gaia, so they are bound to the earth.Shiva
Shiva (; , ), also known as Mahadeva (; , , Help:IPA/Sanskrit, ɐɦaːd̪eːʋɐh and Hara, is one of the Hindu deities, principal deities of Hinduism. He is the God in Hinduism, Supreme Being in Shaivism, one of the major traditions w ...
, while Vishnu
Vishnu (; , , ), also known as Narayana and Hari, is one of the Hindu deities, principal deities of Hinduism. He is the supreme being within Vaishnavism, one of the major traditions within contemporary Hinduism, and the god of preservation ( ...
is depicted often as sleeping on a seven-headed snake or within the coils of a serpent. There are temples in India solely for cobras sometimes called ''Nagraj'' (King of Snakes), and it is believed that snakes are symbols of fertility. In the annual Hindu festival of Nag Panchami
Naga Panchami (Sanskrit: नागपञ्चमी, International Alphabet of Sanskrit Transliteration, IAST: ''Nāgapañcamī'') is a day of traditional worship of Nāg, ''naga''s (or najas or nags) or snakes (which are associated with the my ...
, snakes are venerated and prayed to.jaguar
The jaguar (''Panthera onca'') is a large felidae, cat species and the only extant taxon, living member of the genus ''Panthera'' that is native to the Americas. With a body length of up to and a weight of up to , it is the biggest cat spe ...
are arguably the most important animals in ancient Mesoamerica
Mesoamerica is a historical region and cultural area that begins in the southern part of North America and extends to the Pacific coast of Central America, thus comprising the lands of central and southern Mexico, all of Belize, Guatemala, El S ...
. "In states of ecstasy, lords dance a serpent dance; great descending snakes adorn and support buildings from Chichen Itza
Chichén Itzá , , often with the emphasis reversed in English to ; from () "at the mouth of the well of the Itza people, Itza people" (often spelled ''Chichen Itza'' in English and traditional Yucatec Maya) was a large Pre-Columbian era, ...
to Tenochtitlan
, also known as Mexico-Tenochtitlan, was a large Mexican in what is now the historic center of Mexico City. The exact date of the founding of the city is unclear, but the date 13 March 1325 was chosen in 1925 to celebrate the 600th annivers ...
, and the Nahuatl
Nahuatl ( ; ), Aztec, or Mexicano is a language or, by some definitions, a group of languages of the Uto-Aztecan language family. Varieties of Nahuatl are spoken by about Nahuas, most of whom live mainly in Central Mexico and have smaller popul ...
word ''coatl'' meaning serpent or twin, forms part of primary deities such as Mixcoatl, Quetzalcoatl, and Coatlicue." In Christianity and Judaism, a serpent appears in Genesis to tempt Adam and Eve
Adam and Eve, according to the creation myth of the Abrahamic religions, were the first man and woman. They are central to the belief that humanity is in essence a single family, with everyone descended from a single pair of original ancestors. ...
with the forbidden fruit
In Abrahamic religions, forbidden fruit is a name given to the fruit growing in the Garden of Eden that God commands mankind Taboo#In religion and mythology, not to eat. In the biblical story, Adam and Eve eat the fruit from the tree of the know ...
from the Tree of Knowledge of Good and Evil.
The turtle has a prominent position as a symbol of steadfastness and tranquility in religion, mythology, and folklore from around the world.World Turtle
The World Turtle, also called the Cosmic Turtle or the World-Bearing Turtle, is a mytheme of a giant turtle (or tortoise) supporting or containing Religious cosmology, the world. It occurs in Hinduism, Chinese mythology, and the mythologies of th ...
'' carries the world upon its back or supports the heavens.
Medicine
 Deaths from snakebites are uncommon in many parts of the world, but are still counted in tens of thousands per year in India.
Deaths from snakebites are uncommon in many parts of the world, but are still counted in tens of thousands per year in India.antivenom
Antivenom, also known as antivenin, venom antiserum, and antivenom immunoglobulin, is a specific treatment for envenomation. It is composed of antibodies and used to treat certain venomous bites and stings. Antivenoms are recommended only if ...
made from the venom of the snake. To produce antivenom, a mixture of the venoms of different species of snake is injected into the body of a horse in ever-increasing dosages until the horse is immunized. Blood is then extracted; the serum is separated, purified and freeze-dried. The cytotoxic effect of snake venom is being researched as a potential treatment for cancers.
Gila monsters produce compounds that reduce plasma glucose; one of these substances is now used in the anti-diabetes
Diabetes mellitus, commonly known as diabetes, is a group of common endocrine diseases characterized by sustained high blood sugar levels. Diabetes is due to either the pancreas not producing enough of the hormone insulin, or the cells of th ...
drug exenatide (Byetta), a glucagon-like peptide-1
Glucagon-like peptide-1 (GLP-1) is a 30- or 31-amino-acid-long peptide hormone deriving from tissue-specific posttranslational processing of the proglucagon peptide. It is produced and secreted by intestinal enteroendocrine L-cells and cer ...
(GLP-1) receptor agonist like semiglutide (Ozempic).Geckos
Geckos are small, mostly carnivorous lizards that have a wide distribution, found on every continent except Antarctica. Belonging to the infraorder Gekkota, geckos are found in warm climates. They range from .
Geckos are unique among lizards f ...
have also been used as folk medicine, especially in China, without any evidence that they have any active compounds. Turtles have been used in Chinese traditional medicine for thousands of years, with every part of the turtle believed to have medical benefits (again, without scientific evidence). Growing demand for turtle meat has placed pressure on vulnerable wild populations of turtles.
Commercial farming
Crocodiles are protected in many parts of the world, and are farmed commercially. Their hides are tanned and used to make leather goods such as shoes and handbags; crocodile meat is also considered a delicacy. The most commonly farmed species are the saltwater and Nile crocodiles. Farming has resulted in an increase in the saltwater crocodile population in Australia
Australia, officially the Commonwealth of Australia, is a country comprising mainland Australia, the mainland of the Australia (continent), Australian continent, the island of Tasmania and list of islands of Australia, numerous smaller isl ...
, as eggs are usually harvested from the wild, so landowners have an incentive to conserve their habitat. Crocodile leather is made into wallets, briefcases, purses, handbags, belts, hats, and shoes. Crocodile oil has been used for various purposes.
Snakes are also farmed, primarily in East
East is one of the four cardinal directions or points of the compass. It is the opposite direction from west and is the direction from which the Sun rises on the Earth.
Etymology
As in other languages, the word is formed from the fact that ea ...
and Southeast Asia
Southeast Asia is the geographical United Nations geoscheme for Asia#South-eastern Asia, southeastern region of Asia, consisting of the regions that are situated south of China, east of the Indian subcontinent, and northwest of the Mainland Au ...
, and their production has become more intensive in the last decade. Snake farm
A snake farm is a facility that houses and breeds a wide variety of snakes, often for the purpose of research and the collection of snake venom, venom for the creation of antivenom. Many snake farms are primarily tourist attractions. Notable sna ...
ing has been troubling for conservation in the past as it can lead to overexploitation
Overexploitation, also called overharvesting or ecological overshoot, refers to harvesting a renewable resource to the point of diminishing returns. Continued overexploitation can lead to the destruction of the resource, as it will be unable to ...
of wild snakes and their natural prey to supply the farms. However, farming snakes can limit the hunting of wild snakes, while reducing the slaughter of higher-order vertebrates like cows. The energy efficiency of snakes is higher than expected for carnivores, due to their ectothermy and low metabolism. Waste protein from the poultry and pig industries is used as feed in snake farms. Snake farms produce meat, snake skin, and antivenom.
Turtle farming is another known but controversial practice. Turtles have been farmed for a variety of reasons, ranging from food to traditional medicine, the pet trade, and scientific conservation. Demand for turtle meat and medicinal products is one of the main threats to turtle conservation in Asia. Though commercial breeding would seem to insulate wild populations, it can stoke the demand for them and increase wild captures.[ Even the potentially appealing concept of raising turtles at a farm to release into the wild is questioned by some veterinarians who have had some experience with farm operations. They caution that this may introduce into the wild populations infectious diseases that occur on the farm, but have not (yet) been occurring in the wild.
]
Reptiles in captivity
A herpetarium is a zoological exhibition space for reptiles and amphibians.
In the Western world, some snakes (especially relatively docile species such as the ball python and corn snake
The corn snake (''Pantherophis guttatus''), sometimes called red rat snake is a species of North American rat snake in the Family (biology), family Colubridae. The species subdues its small prey by constriction. It is found throughout the sout ...
) are sometimes kept as pets. Numerous species of lizard are kept as pet
A pet, or companion animal, is an animal kept primarily for a person's company or entertainment rather than as a working animal, livestock, or a laboratory animal. Popular pets are often considered to have attractive/ cute appearances, inte ...
s, including bearded dragons,iguana
''Iguana'' (, ) is a genus of herbivorous lizards that are native to tropical areas of Mexico, Central America, South America, and the Caribbean. The genus was first described by Austrian naturalist Josephus Nicolaus Laurenti, J.N. Laurenti in ...
s, anole
Dactyloidae are a family of lizards commonly known as anoles (singular anole ) and native to warmer parts of the Americas, ranging from southeastern United States to Paraguay. Instead of treating it as a family, some authorities prefer to treat ...
s, and gecko
Geckos are small, mostly carnivorous lizards that have a wide distribution, found on every continent except Antarctica. Belonging to the infraorder Gekkota, geckos are found in warm climates. They range from .
Geckos are unique among lizards ...
s (such as the popular leopard gecko
The leopard gecko or common leopard gecko (''Eublepharis macularius'') is a ground-dwelling gecko native to the rocky dry grassland and desert regions of Afghanistan, Iran, Pakistan, India, and Nepal. The leopard gecko is a popular pet, and due ...
and the crested gecko).Salmonella
''Salmonella'' is a genus of bacillus (shape), rod-shaped, (bacillus) Gram-negative bacteria of the family Enterobacteriaceae. The two known species of ''Salmonella'' are ''Salmonella enterica'' and ''Salmonella bongori''. ''S. enterica'' ...
'' and other pathogens.
See also
* Amphibian and reptile tunnel
* List of reptiles
* Lists of reptiles by region
A list is a set of discrete items of information collected and set forth in some format for utility, entertainment, or other purposes. A list may be memorialized in any number of ways, including existing only in the mind of the list-maker, bu ...
* Reptile Database
The Reptile Database is a scientific database that collects taxonomic information on all living reptile species (i.e. no fossil species such as dinosaur
Dinosaurs are a diverse group of reptiles of the clade Dinosauria. They first appeared ...
*
Notes
References
Further reading
*
* Duellman, William E., Berg, Barbara (1962),
Type Specimens of Amphibians and Reptiles in the Museum of Natural History, the University of Kansas
'
*
*
*
External links
*
*
*
*
*
*
* — an online full text copy of a 22 volume 13,000 page summary of the state of reptile research.
{{Portal bar, Reptiles
Extant Pennsylvanian first appearances
Paraphyletic groups
Articles containing video clips
 In the 13th century, the category of ''reptile'' was recognized in Europe as consisting of a miscellany of egg-laying creatures, including "snakes, various fantastic monsters, lizards, assorted amphibians, and worms", as recorded by
In the 13th century, the category of ''reptile'' was recognized in Europe as consisting of a miscellany of egg-laying creatures, including "snakes, various fantastic monsters, lizards, assorted amphibians, and worms", as recorded by  It was not until the beginning of the 19th century that it became clear that reptiles and amphibians are, in fact, quite different animals, and P.A. Latreille erected the class ''Batracia'' (1825) for the latter, dividing the
It was not until the beginning of the 19th century that it became clear that reptiles and amphibians are, in fact, quite different animals, and P.A. Latreille erected the class ''Batracia'' (1825) for the latter, dividing the  The synapsid/sauropsid division supplemented another approach, one that split the reptiles into four subclasses based on the number and position of temporal fenestrae, openings in the sides of the skull behind the eyes. This classification was initiated by Henry Fairfield Osborn and elaborated and made popular by Romer's classic ''
The synapsid/sauropsid division supplemented another approach, one that split the reptiles into four subclasses based on the number and position of temporal fenestrae, openings in the sides of the skull behind the eyes. This classification was initiated by Henry Fairfield Osborn and elaborated and made popular by Romer's classic '' The composition of Euryapsida was uncertain.
The composition of Euryapsida was uncertain.  When Sauropsida was used, it often had the same content or even the same definition as Reptilia. In 1988, Jacques Gauthier proposed a
When Sauropsida was used, it often had the same content or even the same definition as Reptilia. In 1988, Jacques Gauthier proposed a 
 The origin of the reptiles lies about 310–320 million years ago, in the steaming swamps of the late
The origin of the reptiles lies about 310–320 million years ago, in the steaming swamps of the late  It was traditionally assumed that the first reptiles retained an anapsid skull inherited from their ancestors. This type of skull has a
It was traditionally assumed that the first reptiles retained an anapsid skull inherited from their ancestors. This type of skull has a 
 The close of the
The close of the  For example,
For example,  Modern non-avian reptiles exhibit some form of cold-bloodedness (i.e. some mix of poikilothermy,
Modern non-avian reptiles exhibit some form of cold-bloodedness (i.e. some mix of poikilothermy,  How
How  Reptilian skin is covered in a horny
Reptilian skin is covered in a horny 
 Most reptiles are insectivorous or carnivorous and have simple and comparatively short digestive tracts due to meat being fairly simple to break down and digest.
Most reptiles are insectivorous or carnivorous and have simple and comparatively short digestive tracts due to meat being fairly simple to break down and digest. 
 Reptiles generally
Reptiles generally  Reptiles tend to avoid confrontation through
Reptiles tend to avoid confrontation through  Some species try to bite immediately. Some will use their heads as sledgehammers and literally smash an opponent, some will rush or swim toward the threat from a distance, even chasing the opponent onto land or galloping after it. The main weapon in all crocodiles is the bite, which can generate very high bite force. Many species also possess canine-like teeth. These are used primarily for seizing prey, but are also used in fighting and display.
Some species try to bite immediately. Some will use their heads as sledgehammers and literally smash an opponent, some will rush or swim toward the threat from a distance, even chasing the opponent onto land or galloping after it. The main weapon in all crocodiles is the bite, which can generate very high bite force. Many species also possess canine-like teeth. These are used primarily for seizing prey, but are also used in fighting and display.
 Dinosaurs have been widely depicted in culture since the English palaeontologist
Dinosaurs have been widely depicted in culture since the English palaeontologist