Quantum mechanics on:
[Wikipedia]
[Google]
[Amazon]
 Quantum mechanics is a fundamental
Quantum mechanics is a fundamental
Chapter 8, p. 215
The time evolution of a quantum state is described by the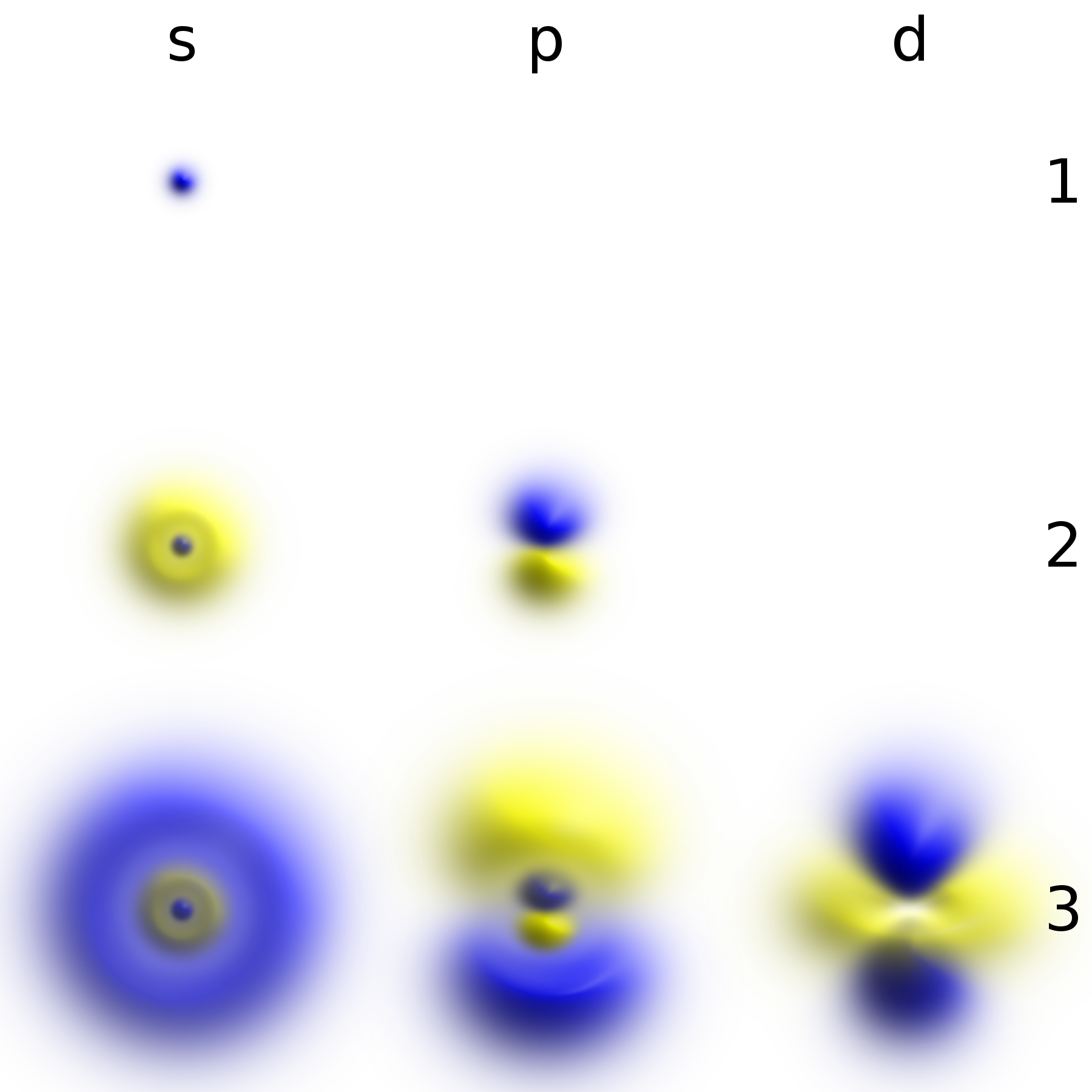 Some wave functions produce probability distributions that are independent of time, such as eigenstates of the Hamiltonian. Many systems that are treated dynamically in classical mechanics are described by such "static" wave functions. For example, a single
Some wave functions produce probability distributions that are independent of time, such as eigenstates of the Hamiltonian. Many systems that are treated dynamically in classical mechanics are described by such "static" wave functions. For example, a single
 The simplest example of a quantum system with a position degree of freedom is a free particle in a single spatial dimension. A free particle is one which is not subject to external influences, so that its Hamiltonian consists only of its kinetic energy:
:
The general solution of the Schrödinger equation is given by
:
which is a superposition of all possible
The simplest example of a quantum system with a position degree of freedom is a free particle in a single spatial dimension. A free particle is one which is not subject to external influences, so that its Hamiltonian consists only of its kinetic energy:
:
The general solution of the Schrödinger equation is given by
:
which is a superposition of all possible
 The particle in a one-dimensional potential energy box is the most mathematically simple example where restraints lead to the quantization of energy levels. The box is defined as having zero potential energy everywhere ''inside'' a certain region, and therefore infinite potential energy everywhere ''outside'' that region. For the one-dimensional case in the direction, the time-independent Schrödinger equation may be written
:
With the differential operator defined by
:
the previous equation is evocative of the classic kinetic energy analogue,
:
with state in this case having energy coincident with the kinetic energy of the particle.
The general solutions of the Schrödinger equation for the particle in a box are
:
or, from
The particle in a one-dimensional potential energy box is the most mathematically simple example where restraints lead to the quantization of energy levels. The box is defined as having zero potential energy everywhere ''inside'' a certain region, and therefore infinite potential energy everywhere ''outside'' that region. For the one-dimensional case in the direction, the time-independent Schrödinger equation may be written
:
With the differential operator defined by
:
the previous equation is evocative of the classic kinetic energy analogue,
:
with state in this case having energy coincident with the kinetic energy of the particle.
The general solutions of the Schrödinger equation for the particle in a box are
:
or, from
 As in the classical case, the potential for the quantum harmonic oscillator is given by
:
This problem can either be treated by directly solving the Schrödinger equation, which is not trivial, or by using the more elegant "ladder method" first proposed by Paul Dirac. The eigenstates are given by
:
:
where ''Hn'' are the Hermite polynomials
:
and the corresponding energy levels are
:
This is another example illustrating the discretization of energy for
As in the classical case, the potential for the quantum harmonic oscillator is given by
:
This problem can either be treated by directly solving the Schrödinger equation, which is not trivial, or by using the more elegant "ladder method" first proposed by Paul Dirac. The eigenstates are given by
:
:
where ''Hn'' are the Hermite polynomials
:
and the corresponding energy levels are
:
This is another example illustrating the discretization of energy for
 The
The
 Quantum mechanics was developed in the early decades of the 20th century, driven by the need to explain phenomena that, in some cases, had been observed in earlier times. Scientific inquiry into the wave nature of light began in the 17th and 18th centuries, when scientists such as Robert Hooke, Christiaan Huygens and
Quantum mechanics was developed in the early decades of the 20th century, driven by the need to explain phenomena that, in some cases, had been observed in earlier times. Scientific inquiry into the wave nature of light began in the 17th and 18th centuries, when scientists such as Robert Hooke, Christiaan Huygens and  This phase is known as the
This phase is known as the
Draft of 4th edition.
*
Online copy
* * Gunther Ludwig, 1968. ''Wave Mechanics''. London: Pergamon Press. *
online
* * Scerri, Eric R., 2006. ''The Periodic Table: Its Story and Its Significance''. Oxford University Press. Considers the extent to which chemistry and the periodic system have been reduced to quantum mechanics. * * * * Veltman, Martinus J.G. (2003), ''Facts and Mysteries in Elementary Particle Physics''. On Wikibooks
This Quantum World
Introduction to Quantum Theory at Quantiki.
Quantum Physics Made Relatively Simple
three video lectures by
Quantum Cook Book
an
PHYS 201: Fundamentals of Physics II
by
The Modern Revolution in Physics
– an online textbook. *
Chemistry
an
Physics
Se
8.04
an
5½ Examples in Quantum Mechanics
Imperial College Quantum Mechanics Course.
;Philosophy * * {{DEFAULTSORT:Quantum Mechanics
 Quantum mechanics is a fundamental
Quantum mechanics is a fundamental theory
A theory is a rational type of abstract thinking about a phenomenon, or the results of such thinking. The process of contemplative and rational thinking is often associated with such processes as observational study or research. Theories may be ...
in physics
Physics is the natural science that studies matter, its fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge which r ...
that provides a description of the physical properties of nature
Nature, in the broadest sense, is the physical world or universe. "Nature" can refer to the phenomena of the physical world, and also to life in general. The study of nature is a large, if not the only, part of science. Although humans are ...
at the scale of atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, ...
s and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, quantum field theory, quantum technology, and quantum information science
Quantum information science is an interdisciplinary field that seeks to understand the analysis, processing, and transmission of information using quantum mechanics principles. It combines the study of Information science with quantum effects in ...
.
Classical physics, the collection of theories that existed before the advent of quantum mechanics, describes many aspects of nature at an ordinary (macroscopic
The macroscopic scale is the length scale on which objects or phenomena are large enough to be visible with the naked eye, without magnifying optical instruments. It is the opposite of microscopic.
Overview
When applied to physical phenomena a ...
) scale, but is not sufficient for describing them at small (atomic and subatomic
In physical sciences, a subatomic particle is a particle that composes an atom. According to the Standard Model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a pro ...
) scales. Most theories in classical physics can be derived from quantum mechanics as an approximation valid at large (macroscopic) scale.
Quantum mechanics differs from classical physics in that energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of hea ...
, momentum, angular momentum
In physics, angular momentum (rarely, moment of momentum or rotational momentum) is the rotational analog of linear momentum. It is an important physical quantity because it is a conserved quantity—the total angular momentum of a closed syst ...
, and other quantities of a bound
Bound or bounds may refer to:
Mathematics
* Bound variable
* Upper and lower bounds, observed limits of mathematical functions
Physics
* Bound state, a particle that has a tendency to remain localized in one or more regions of space
Geography
*B ...
system are restricted to discrete values ( quantization); objects have characteristics of both particle
In the physical sciences, a particle (or corpuscule in older texts) is a small localized object which can be described by several physical or chemical properties, such as volume, density, or mass.
They vary greatly in size or quantity, from ...
s and wave
In physics, mathematics, and related fields, a wave is a propagating dynamic disturbance (change from equilibrium) of one or more quantities. Waves can be periodic, in which case those quantities oscillate repeatedly about an equilibrium (re ...
s (wave–particle duality
Wave–particle duality is the concept in quantum mechanics that every particle or quantum entity may be described as either a particle or a wave. It expresses the inability of the classical concepts "particle" or "wave" to fully describe the b ...
); and there are limits to how accurately the value of a physical quantity can be predicted prior to its measurement, given a complete set of initial conditions (the uncertainty principle
In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physic ...
).
Quantum mechanics arose gradually from theories to explain observations which could not be reconciled with classical physics, such as Max Planck
Max Karl Ernst Ludwig Planck (, ; 23 April 1858 – 4 October 1947) was a German theoretical physicist whose discovery of energy quanta won him the Nobel Prize in Physics in 1918.
Planck made many substantial contributions to theoretical p ...
's solution in 1900 to the black-body radiation problem, and the correspondence between energy and frequency in Albert Einstein
Albert Einstein ( ; ; 14 March 1879 – 18 April 1955) was a German-born theoretical physicist, widely acknowledged to be one of the greatest and most influential physicists of all time. Einstein is best known for developing the theory ...
's 1905 paper which explained the photoelectric effect
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, and solid sta ...
. These early attempts to understand microscopic phenomena, now known as the "old quantum theory
The old quantum theory is a collection of results from the years 1900–1925 which predate modern quantum mechanics. The theory was never complete or self-consistent, but was rather a set of heuristic corrections to classical mechanics. The theory ...
", led to the full development of quantum mechanics in the mid-1920s by Niels Bohr
Niels Henrik David Bohr (; 7 October 1885 – 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922 ...
, Erwin Schrödinger
Erwin Rudolf Josef Alexander Schrödinger (, ; ; 12 August 1887 – 4 January 1961), sometimes written as or , was a Nobel Prize-winning Austrian physicist with Irish citizenship who developed a number of fundamental results in quantum theo ...
, Werner Heisenberg
Werner Karl Heisenberg () (5 December 1901 – 1 February 1976) was a German theoretical physicist and one of the main pioneers of the theory of quantum mechanics. He published his work in 1925 in a breakthrough paper. In the subsequent serie ...
, Max Born, Paul Dirac
Paul Adrien Maurice Dirac (; 8 August 1902 – 20 October 1984) was an English theoretical physicist who is regarded as one of the most significant physicists of the 20th century. He was the Lucasian Professor of Mathematics at the Univer ...
and others. The modern theory is formulated in various specially developed mathematical formalisms. In one of them, a mathematical entity called the wave function
A wave function in quantum physics is a mathematical description of the quantum state of an isolated quantum system. The wave function is a complex-valued probability amplitude, and the probabilities for the possible results of measurements ...
provides information, in the form of probability amplitude
In quantum mechanics, a probability amplitude is a complex number used for describing the behaviour of systems. The modulus squared of this quantity represents a probability density.
Probability amplitudes provide a relationship between the qu ...
s, about what measurements of a particle's energy, momentum, and other physical properties may yield.
Overview and fundamental concepts
Quantum mechanics allows the calculation of properties and behaviour of physical systems. It is typically applied to microscopic systems: molecules, atoms and sub-atomic particles. It has been demonstrated to hold for complex molecules with thousands of atoms, but its application to human beings raises philosophical problems, such asWigner's friend
Wigner's friend is a thought experiment in theoretical quantum physics, first conceived by the physicist Eugene Wigner in 1961, Reprinted in and further developed by David Deutsch in 1985. The scenario involves an indirect observation of a Measure ...
, and its application to the universe as a whole remains speculative. Predictions of quantum mechanics have been verified experimentally to an extremely high degree of accuracy
Accuracy and precision are two measures of ''observational error''.
''Accuracy'' is how close a given set of measurements ( observations or readings) are to their ''true value'', while ''precision'' is how close the measurements are to each oth ...
.
A fundamental feature of the theory is that it usually cannot predict with certainty what will happen, but only give probabilities. Mathematically, a probability is found by taking the square of the absolute value of a complex number
In mathematics, a complex number is an element of a number system that extends the real numbers with a specific element denoted , called the imaginary unit and satisfying the equation i^= -1; every complex number can be expressed in the fo ...
, known as a probability amplitude. This is known as the Born rule
The Born rule (also called Born's rule) is a key postulate of quantum mechanics which gives the probability that a measurement of a quantum system will yield a given result. In its simplest form, it states that the probability density of findi ...
, named after physicist Max Born. For example, a quantum particle like an electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
can be described by a wave function
A wave function in quantum physics is a mathematical description of the quantum state of an isolated quantum system. The wave function is a complex-valued probability amplitude, and the probabilities for the possible results of measurements ...
, which associates to each point in space a probability amplitude. Applying the Born rule to these amplitudes gives a probability density function
In probability theory, a probability density function (PDF), or density of a continuous random variable, is a function whose value at any given sample (or point) in the sample space (the set of possible values taken by the random variable) ca ...
for the position that the electron will be found to have when an experiment is performed to measure it. This is the best the theory can do; it cannot say for certain where the electron will be found. The Schrödinger equation
The Schrödinger equation is a linear partial differential equation that governs the wave function of a quantum-mechanical system. It is a key result in quantum mechanics, and its discovery was a significant landmark in the development of th ...
relates the collection of probability amplitudes that pertain to one moment of time to the collection of probability amplitudes that pertain to another.
One consequence of the mathematical rules of quantum mechanics is a tradeoff in predictability between different measurable quantities. The most famous form of this uncertainty principle
In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physic ...
says that no matter how a quantum particle is prepared or how carefully experiments upon it are arranged, it is impossible to have a precise prediction for a measurement of its position and also at the same time for a measurement of its momentum.
Another consequence of the mathematical rules of quantum mechanics is the phenomenon of quantum interference
In physics, interference is a phenomenon in which two waves combine by adding their displacement together at every single point in space and time, to form a resultant wave of greater, lower, or the same amplitude. Constructive and destructive ...
, which is often illustrated with the double-slit experiment
In modern physics, the double-slit experiment is a demonstration that light and matter can display characteristics of both classically defined waves and particles; moreover, it displays the fundamentally probabilistic nature of quantum mechanics ...
. In the basic version of this experiment, a coherent light source, such as a laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The fi ...
beam, illuminates a plate pierced by two parallel slits, and the light passing through the slits is observed on a screen behind the plate. The wave nature of light causes the light waves passing through the two slits to interfere, producing bright and dark bands on the screen – a result that would not be expected if light consisted of classical particles. However, the light is always found to be absorbed at the screen at discrete points, as individual particles rather than waves; the interference pattern appears via the varying density of these particle hits on the screen. Furthermore, versions of the experiment that include detectors at the slits find that each detected photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
passes through one slit (as would a classical particle), and not through both slits (as would a wave). However, such experiments demonstrate that particles do not form the interference pattern if one detects which slit they pass through. Other atomic-scale entities, such as electrons
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
, are found to exhibit the same behavior when fired towards a double slit. This behavior is known as wave–particle duality
Wave–particle duality is the concept in quantum mechanics that every particle or quantum entity may be described as either a particle or a wave. It expresses the inability of the classical concepts "particle" or "wave" to fully describe the b ...
.
Another counter-intuitive phenomenon predicted by quantum mechanics is quantum tunnelling: a particle that goes up against a potential barrier
In quantum mechanics, the rectangular (or, at times, square) potential barrier is a standard one-dimensional problem that demonstrates the phenomena of wave-mechanical tunneling (also called "quantum tunneling") and wave-mechanical reflection. ...
can cross it, even if its kinetic energy is smaller than the maximum of the potential. In classical mechanics this particle would be trapped. Quantum tunnelling has several important consequences, enabling radioactive decay, nuclear fusion
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles ( neutrons or protons). The difference in mass between the reactants and products is manife ...
in stars, and applications such as scanning tunnelling microscopy
A scanning tunneling microscope (STM) is a type of microscope used for imaging surfaces at the atomic level. Its development in 1981 earned its inventors, Gerd Binnig and Heinrich Rohrer, then at IBM Zürich, the Nobel Prize in Physics in 1986. ...
and the tunnel diode
A tunnel diode or Esaki diode is a type of semiconductor diode that has effectively " negative resistance" due to the quantum mechanical effect called tunneling. It was invented in August 1957 by Leo Esaki, Yuriko Kurose, and Takashi Suz ...
.
When quantum systems interact, the result can be the creation of quantum entanglement
Quantum entanglement is the phenomenon that occurs when a group of particles are generated, interact, or share spatial proximity in a way such that the quantum state of each particle of the group cannot be described independently of the state of ...
: their properties become so intertwined that a description of the whole solely in terms of the individual parts is no longer possible. Erwin Schrödinger called entanglement "...''the'' characteristic trait of quantum mechanics, the one that enforces its entire departure from classical lines of thought". Quantum entanglement enables the counter-intuitive properties of quantum pseudo-telepathy
Quantum pseudo-telepathy is the fact that in certain Bayesian games with asymmetric information, players who have access to a shared physical system in an entangled quantum state, and who are able to execute strategies that are contingent upon m ...
, and can be a valuable resource in communication protocols, such as quantum key distribution
Quantum key distribution (QKD) is a secure communication method which implements a cryptographic protocol involving components of quantum mechanics. It enables two parties to produce a shared random secret key known only to them, which can then b ...
and superdense coding
In quantum information theory, superdense coding (also referred to as ''dense coding'') is a quantum communication protocol to communicate a number of classical bits of information by only transmitting a smaller number of qubits, under the assum ...
. Contrary to popular misconception, entanglement does not allow sending signals faster than light
Faster-than-light (also FTL, superluminal or supercausal) travel and communication are the conjectural propagation of matter or information faster than the speed of light (). The special theory of relativity implies that only particles with zero ...
, as demonstrated by the no-communication theorem
In physics, the no-communication theorem or no-signaling principle is a no-go theorem from quantum information theory which states that, during measurement of an entangled quantum state, it is not possible for one observer, by making a measurem ...
.
Another possibility opened by entanglement is testing for "hidden variables
Hidden variables may refer to:
* Confounding, in statistics, an extraneous variable in a statistical model that correlates (directly or inversely) with both the dependent variable and the independent variable
* Hidden transformation, in computer ...
", hypothetical properties more fundamental than the quantities addressed in quantum theory itself, knowledge of which would allow more exact predictions than quantum theory can provide. A collection of results, most significantly Bell's theorem, have demonstrated that broad classes of such hidden-variable theories are in fact incompatible with quantum physics. According to Bell's theorem, if nature actually operates in accord with any theory of ''local'' hidden variables, then the results of a Bell test
A Bell test, also known as Bell inequality test or Bell experiment, is a real-world physics experiment designed to test the theory of quantum mechanics in relation to Albert Einstein's concept of local realism. Named for John Stewart Bell, the e ...
will be constrained in a particular, quantifiable way. Many Bell tests have been performed, using entangled particles, and they have shown results incompatible with the constraints imposed by local hidden variables.
It is not possible to present these concepts in more than a superficial way without introducing the actual mathematics involved; understanding quantum mechanics requires not only manipulating complex numbers, but also linear algebra
Linear algebra is the branch of mathematics concerning linear equations such as:
:a_1x_1+\cdots +a_nx_n=b,
linear maps such as:
:(x_1, \ldots, x_n) \mapsto a_1x_1+\cdots +a_nx_n,
and their representations in vector spaces and through matrices ...
, differential equation
In mathematics, a differential equation is an equation that relates one or more unknown functions and their derivatives. In applications, the functions generally represent physical quantities, the derivatives represent their rates of change, an ...
s, group theory
In abstract algebra, group theory studies the algebraic structures known as group (mathematics), groups.
The concept of a group is central to abstract algebra: other well-known algebraic structures, such as ring (mathematics), rings, field ...
, and other more advanced subjects. Accordingly, this article will present a mathematical formulation of quantum mechanics and survey its application to some useful and oft-studied examples.
Mathematical formulation
In the mathematically rigorous formulation of quantum mechanics, the state of a quantum mechanical system is a vector belonging to a ( separable) complex Hilbert space . This vector is postulated to be normalized under the Hilbert space inner product, that is, it obeys , and it is well-defined up to a complex number of modulus 1 (the global phase), that is, and represent the same physical system. In other words, the possible states are points in the projective space of a Hilbert space, usually called thecomplex projective space
In mathematics, complex projective space is the projective space with respect to the field of complex numbers. By analogy, whereas the points of a real projective space label the lines through the origin of a real Euclidean space, the points of a ...
. The exact nature of this Hilbert space is dependent on the system – for example, for describing position and momentum the Hilbert space is the space of complex square-integrable
In mathematics, a square-integrable function, also called a quadratically integrable function or L^2 function or square-summable function, is a real- or complex-valued measurable function for which the integral of the square of the absolute value ...
functions , while the Hilbert space for the spin of a single proton is simply the space of two-dimensional complex vectors with the usual inner product.
Physical quantities of interestposition, momentum, energy, spinare represented by observables, which are Hermitian {{Short description, none
Numerous things are named after the French mathematician Charles Hermite (1822–1901):
Hermite
* Cubic Hermite spline, a type of third-degree spline
* Gauss–Hermite quadrature, an extension of Gaussian quadrature m ...
(more precisely, self-adjoint
In mathematics, and more specifically in abstract algebra, an element ''x'' of a *-algebra is self-adjoint if x^*=x. A self-adjoint element is also Hermitian, though the reverse doesn't necessarily hold.
A collection ''C'' of elements of a st ...
) linear operators acting on the Hilbert space. A quantum state can be an eigenvector
In linear algebra, an eigenvector () or characteristic vector of a linear transformation is a nonzero vector that changes at most by a scalar factor when that linear transformation is applied to it. The corresponding eigenvalue, often denoted ...
of an observable, in which case it is called an eigenstate, and the associated eigenvalue
In linear algebra, an eigenvector () or characteristic vector of a linear transformation is a nonzero vector that changes at most by a scalar factor when that linear transformation is applied to it. The corresponding eigenvalue, often denoted ...
corresponds to the value of the observable in that eigenstate. More generally, a quantum state will be a linear combination of the eigenstates, known as a quantum superposition. When an observable is measured, the result will be one of its eigenvalues with probability given by the Born rule
The Born rule (also called Born's rule) is a key postulate of quantum mechanics which gives the probability that a measurement of a quantum system will yield a given result. In its simplest form, it states that the probability density of findi ...
: in the simplest case the eigenvalue is non-degenerate and the probability is given by , where is its associated eigenvector. More generally, the eigenvalue is degenerate and the probability is given by , where is the projector onto its associated eigenspace. In the continuous case, these formulas give instead the probability density
In probability theory, a probability density function (PDF), or density of a continuous random variable, is a function whose value at any given sample (or point) in the sample space (the set of possible values taken by the random variable) can ...
.
After the measurement, if result was obtained, the quantum state is postulated to collapse
Collapse or its variants may refer to:
Concepts
* Collapse (structural)
* Collapse (topology), a mathematical concept
* Collapsing manifold
* Collapse, the action of collapsing or telescoping objects
* Collapsing user interface elements
** ...
to , in the non-degenerate case, or to , in the general case. The probabilistic nature of quantum mechanics thus stems from the act of measurement. This is one of the most difficult aspects of quantum systems to understand. It was the central topic in the famous Bohr–Einstein debates, in which the two scientists attempted to clarify these fundamental principles by way of thought experiment
A thought experiment is a hypothetical situation in which a hypothesis, theory, or principle is laid out for the purpose of thinking through its consequences.
History
The ancient Greek ''deiknymi'' (), or thought experiment, "was the most anc ...
s. In the decades after the formulation of quantum mechanics, the question of what constitutes a "measurement" has been extensively studied. Newer interpretations of quantum mechanics
An interpretation of quantum mechanics is an attempt to explain how the mathematical theory of quantum mechanics might correspond to experienced reality. Although quantum mechanics has held up to rigorous and extremely precise tests in an extraord ...
have been formulated that do away with the concept of "wave function collapse
In quantum mechanics, wave function collapse occurs when a wave function—initially in a quantum superposition, superposition of several eigenstates—reduces to a single eigenstate due to interaction with the external world. This interaction is ...
" (see, for example, the many-worlds interpretation
The many-worlds interpretation (MWI) is an interpretation of quantum mechanics that asserts that the universal wavefunction is objectively real, and that there is no wave function collapse. This implies that all possible outcomes of quantum ...
). The basic idea is that when a quantum system interacts with a measuring apparatus, their respective wave functions become entangled so that the original quantum system ceases to exist as an independent entity. For details, see the article on measurement in quantum mechanics
In quantum physics, a measurement is the testing or manipulation of a physical system to yield a numerical result. The predictions that quantum physics makes are in general probabilistic. The mathematical tools for making predictions about what m ...
.Chapter 8, p. 215
The time evolution of a quantum state is described by the
Schrödinger equation
The Schrödinger equation is a linear partial differential equation that governs the wave function of a quantum-mechanical system. It is a key result in quantum mechanics, and its discovery was a significant landmark in the development of th ...
:
:
Here denotes the Hamiltonian
Hamiltonian may refer to:
* Hamiltonian mechanics, a function that represents the total energy of a system
* Hamiltonian (quantum mechanics), an operator corresponding to the total energy of that system
** Dyall Hamiltonian, a modified Hamiltonian ...
, the observable corresponding to the total energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of heat ...
of the system, and is the reduced Planck constant
The Planck constant, or Planck's constant, is a fundamental physical constant of foundational importance in quantum mechanics. The constant gives the relationship between the energy of a photon and its frequency, and by the mass-energy equivale ...
. The constant is introduced so that the Hamiltonian is reduced to the classical Hamiltonian in cases where the quantum system can be approximated by a classical system; the ability to make such an approximation in certain limits is called the correspondence principle.
The solution of this differential equation is given by
:
The operator is known as the time-evolution operator, and has the crucial property that it is unitary
Unitary may refer to:
Mathematics
* Unitary divisor
* Unitary element
* Unitary group
* Unitary matrix
* Unitary morphism
* Unitary operator
* Unitary transformation
* Unitary representation
* Unitarity (physics)
* ''E''-unitary inverse semigrou ...
. This time evolution is deterministic in the sense that – given an initial quantum state – it makes a definite prediction of what the quantum state will be at any later time.
 Some wave functions produce probability distributions that are independent of time, such as eigenstates of the Hamiltonian. Many systems that are treated dynamically in classical mechanics are described by such "static" wave functions. For example, a single
Some wave functions produce probability distributions that are independent of time, such as eigenstates of the Hamiltonian. Many systems that are treated dynamically in classical mechanics are described by such "static" wave functions. For example, a single electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
in an unexcited atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, ...
is pictured classically as a particle moving in a circular trajectory around the atomic nucleus
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron ...
, whereas in quantum mechanics, it is described by a static wave function surrounding the nucleus. For example, the electron wave function for an unexcited hydrogen atom is a spherically symmetric function known as an ''s'' orbital ( Fig. 1).
Analytic solutions of the Schrödinger equation are known for very few relatively simple model Hamiltonians including the quantum harmonic oscillator, the particle in a box
In quantum mechanics, the particle in a box model (also known as the infinite potential well or the infinite square well) describes a particle free to move in a small space surrounded by impenetrable barriers. The model is mainly used as a hypo ...
, the dihydrogen cation
The dihydrogen cation or hydrogen molecular ion is a cation (positive ion) with formula . It consists of two hydrogen nuclei ( protons) sharing a single electron. It is the simplest molecular ion.
The ion can be formed from the ionization of a ...
, and the hydrogen atom. Even the helium
Helium (from el, ἥλιος, helios, lit=sun) is a chemical element with the symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas and the first in the noble gas group in the periodic table. ...
atom – which contains just two electrons – has defied all attempts at a fully analytic treatment.
However, there are techniques for finding approximate solutions. One method, called perturbation theory
In mathematics and applied mathematics, perturbation theory comprises methods for finding an approximate solution to a problem, by starting from the exact solution of a related, simpler problem. A critical feature of the technique is a middl ...
, uses the analytic result for a simple quantum mechanical model to create a result for a related but more complicated model by (for example) the addition of a weak potential energy. Another method is called "semi-classical equation of motion", which applies to systems for which quantum mechanics produces only small deviations from classical behavior. These deviations can then be computed based on the classical motion. This approach is particularly important in the field of quantum chaos
Quantum chaos is a branch of physics which studies how chaotic classical dynamical systems can be described in terms of quantum theory. The primary question that quantum chaos seeks to answer is: "What is the relationship between quantum mech ...
.
Uncertainty principle
One consequence of the basic quantum formalism is theuncertainty principle
In quantum mechanics, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physic ...
. In its most familiar form, this states that no preparation of a quantum particle can imply simultaneously precise predictions both for a measurement of its position and for a measurement of its momentum. Both position and momentum are observables, meaning that they are represented by Hermitian operators. The position operator and momentum operator do not commute, but rather satisfy the canonical commutation relation
In quantum mechanics, the canonical commutation relation is the fundamental relation between canonical conjugate quantities (quantities which are related by definition such that one is the Fourier transform of another). For example,
hat x,\hat p_ ...
:
:
Given a quantum state, the Born rule lets us compute expectation values for both and , and moreover for powers of them. Defining
the uncertainty for an observable by a standard deviation, we have
:
and likewise for the momentum:
:
The uncertainty principle states that
:
Either standard deviation can in principle be made arbitrarily small, but not both simultaneously.Section 3.2 of . This fact is experimentally well-known for example in quantum optics; see e.g. chap. 2 and Fig. 2.1 This inequality generalizes to arbitrary pairs of self-adjoint operators and . The commutator of these two operators is
:
and this provides the lower bound on the product of standard deviations:
:
Another consequence of the canonical commutation relation is that the position and momentum operators are Fourier transforms of each other, so that a description of an object according to its momentum is the Fourier transform of its description according to its position. The fact that dependence in momentum is the Fourier transform of the dependence in position means that the momentum operator is equivalent (up to an factor) to taking the derivative according to the position, since in Fourier analysis differentiation corresponds to multiplication in the dual space. This is why in quantum equations in position space, the momentum is replaced by , and in particular in the non-relativistic Schrödinger equation in position space the momentum-squared term is replaced with a Laplacian times .
Composite systems and entanglement
When two different quantum systems are considered together, the Hilbert space of the combined system is thetensor product
In mathematics, the tensor product V \otimes W of two vector spaces and (over the same field) is a vector space to which is associated a bilinear map V\times W \to V\otimes W that maps a pair (v,w),\ v\in V, w\in W to an element of V \otime ...
of the Hilbert spaces of the two components. For example, let and be two quantum systems, with Hilbert spaces and , respectively. The Hilbert space of the composite system is then
:
If the state for the first system is the vector and the state for the second system is , then the state of the composite system is
:
Not all states in the joint Hilbert space can be written in this form, however, because the superposition principle implies that linear combinations of these "separable" or "product states" are also valid. For example, if and are both possible states for system , and likewise and are both possible states for system , then
:
is a valid joint state that is not separable. States that are not separable are called entangled.
If the state for a composite system is entangled, it is impossible to describe either component system or system by a state vector. One can instead define reduced density matrices that describe the statistics that can be obtained by making measurements on either component system alone. This necessarily causes a loss of information, though: knowing the reduced density matrices of the individual systems is not enough to reconstruct the state of the composite system. Just as density matrices specify the state of a subsystem of a larger system, analogously, positive operator-valued measures (POVMs) describe the effect on a subsystem of a measurement performed on a larger system. POVMs are extensively used in quantum information theory.
As described above, entanglement is a key feature of models of measurement processes in which an apparatus becomes entangled with the system being measured. Systems interacting with the environment in which they reside generally become entangled with that environment, a phenomenon known as quantum decoherence. This can explain why, in practice, quantum effects are difficult to observe in systems larger than microscopic.
Equivalence between formulations
There are many mathematically equivalent formulations of quantum mechanics. One of the oldest and most common is the " transformation theory" proposed byPaul Dirac
Paul Adrien Maurice Dirac (; 8 August 1902 – 20 October 1984) was an English theoretical physicist who is regarded as one of the most significant physicists of the 20th century. He was the Lucasian Professor of Mathematics at the Univer ...
, which unifies and generalizes the two earliest formulations of quantum mechanics – matrix mechanics
Matrix mechanics is a formulation of quantum mechanics created by Werner Heisenberg, Max Born, and Pascual Jordan in 1925. It was the first conceptually autonomous and logically consistent formulation of quantum mechanics. Its account of quantum j ...
(invented by Werner Heisenberg
Werner Karl Heisenberg () (5 December 1901 – 1 February 1976) was a German theoretical physicist and one of the main pioneers of the theory of quantum mechanics. He published his work in 1925 in a breakthrough paper. In the subsequent serie ...
) and wave mechanics Wave mechanics may refer to:
* the mechanics of waves
* the ''wave equation'' in quantum physics, see Schrödinger equation
See also
* Quantum mechanics
* Wave equation
The (two-way) wave equation is a second-order linear partial different ...
(invented by Erwin Schrödinger
Erwin Rudolf Josef Alexander Schrödinger (, ; ; 12 August 1887 – 4 January 1961), sometimes written as or , was a Nobel Prize-winning Austrian physicist with Irish citizenship who developed a number of fundamental results in quantum theo ...
). An alternative formulation of quantum mechanics is Feynman
Richard Phillips Feynman (; May 11, 1918 – February 15, 1988) was an American theoretical physicist, known for his work in the path integral formulation of quantum mechanics, the theory of quantum electrodynamics, the physics of the superflu ...
's path integral formulation
The path integral formulation is a description in quantum mechanics that generalizes the action principle of classical mechanics. It replaces the classical notion of a single, unique classical trajectory for a system with a sum, or functional i ...
, in which a quantum-mechanical amplitude is considered as a sum over all possible classical and non-classical paths between the initial and final states. This is the quantum-mechanical counterpart of the action principle
In physics, action is a scalar quantity describing how a physical system has changed over time. Action is significant because the equations of motion of the system can be derived through the principle of stationary action.
In the simple cas ...
in classical mechanics.
Symmetries and conservation laws
The Hamiltonian is known as the ''generator'' of time evolution, since it defines a unitary time-evolution operator for each value of . From this relation between and , it follows that any observable that commutes with will be ''conserved'': its expectation value will not change over time. This statement generalizes, as mathematically, any Hermitian operator can generate a family of unitary operators parameterized by a variable . Under the evolution generated by , any observable that commutes with will be conserved. Moreover, if is conserved by evolution under , then is conserved under the evolution generated by . This implies a quantum version of the result proven byEmmy Noether
Amalie Emmy NoetherEmmy is the '' Rufname'', the second of two official given names, intended for daily use. Cf. for example the résumé submitted by Noether to Erlangen University in 1907 (Erlangen University archive, ''Promotionsakt Emmy Noeth ...
in classical (Lagrangian
Lagrangian may refer to:
Mathematics
* Lagrangian function, used to solve constrained minimization problems in optimization theory; see Lagrange multiplier
** Lagrangian relaxation, the method of approximating a difficult constrained problem with ...
) mechanics: for every differentiable
In mathematics, a differentiable function of one real variable is a function whose derivative exists at each point in its domain. In other words, the graph of a differentiable function has a non-vertical tangent line at each interior point in its ...
symmetry of a Hamiltonian, there exists a corresponding conservation law.
Examples
Free particle
 The simplest example of a quantum system with a position degree of freedom is a free particle in a single spatial dimension. A free particle is one which is not subject to external influences, so that its Hamiltonian consists only of its kinetic energy:
:
The general solution of the Schrödinger equation is given by
:
which is a superposition of all possible
The simplest example of a quantum system with a position degree of freedom is a free particle in a single spatial dimension. A free particle is one which is not subject to external influences, so that its Hamiltonian consists only of its kinetic energy:
:
The general solution of the Schrödinger equation is given by
:
which is a superposition of all possible plane wave
In physics, a plane wave is a special case of wave or field: a physical quantity whose value, at any moment, is constant through any plane that is perpendicular to a fixed direction in space.
For any position \vec x in space and any time t, ...
s , which are eigenstates of the momentum operator with momentum . The coefficients of the superposition are , which is the Fourier transform of the initial quantum state .
It is not possible for the solution to be a single momentum eigenstate, or a single position eigenstate, as these are not normalizable quantum states. Instead, we can consider a Gaussian wave packet
In physics, a wave packet (or wave train) is a short "burst" or "envelope" of localized wave action that travels as a unit. A wave packet can be analyzed into, or can be synthesized from, an infinite set of component sinusoidal waves of diff ...
:
:
which has Fourier transform, and therefore momentum distribution
:
We see that as we make smaller the spread in position gets smaller, but the spread in momentum gets larger. Conversely, by making larger we make the spread in momentum smaller, but the spread in position gets larger. This illustrates the uncertainty principle.
As we let the Gaussian wave packet evolve in time, we see that its center moves through space at a constant velocity (like a classical particle with no forces acting on it). However, the wave packet will also spread out as time progresses, which means that the position becomes more and more uncertain. The uncertainty in momentum, however, stays constant.
Particle in a box
Euler's formula
Euler's formula, named after Leonhard Euler, is a mathematical formula in complex analysis that establishes the fundamental relationship between the trigonometric functions and the complex exponential function. Euler's formula states that fo ...
,
:
The infinite potential walls of the box determine the values of and at and where must be zero. Thus, at ,
:
and . At ,
:
in which cannot be zero as this would conflict with the postulate that has norm 1. Therefore, since , must be an integer multiple of ,
:
This constraint on implies a constraint on the energy levels, yielding
A finite potential well The finite potential well (also known as the finite square well) is a concept from quantum mechanics. It is an extension of the infinite potential well, in which a particle is confined to a "box", but one which has finite potential "walls". Unlike ...
is the generalization of the infinite potential well problem to potential wells having finite depth. The finite potential well problem is mathematically more complicated than the infinite particle-in-a-box problem as the wave function is not pinned to zero at the walls of the well. Instead, the wave function must satisfy more complicated mathematical boundary conditions as it is nonzero in regions outside the well. Another related problem is that of the rectangular potential barrier
In quantum mechanics, the rectangular (or, at times, square) potential barrier is a standard one-dimensional problem that demonstrates the phenomena of wave-mechanical tunneling (also called "quantum tunneling") and wave-mechanical reflection. ...
, which furnishes a model for the quantum tunneling
In physics, a quantum (plural quanta) is the minimum amount of any physical entity (physical property) involved in an interaction. The fundamental notion that a physical property can be "quantized" is referred to as "the hypothesis of quantizati ...
effect that plays an important role in the performance of modern technologies such as flash memory and scanning tunneling microscopy
A scanning tunneling microscope (STM) is a type of microscope used for imaging surfaces at the atomic level. Its development in 1981 earned its inventors, Gerd Binnig and Heinrich Rohrer, then at IBM Zürich, the Nobel Prize in Physics in 1986. ...
.
Harmonic oscillator
 As in the classical case, the potential for the quantum harmonic oscillator is given by
:
This problem can either be treated by directly solving the Schrödinger equation, which is not trivial, or by using the more elegant "ladder method" first proposed by Paul Dirac. The eigenstates are given by
:
:
where ''Hn'' are the Hermite polynomials
:
and the corresponding energy levels are
:
This is another example illustrating the discretization of energy for
As in the classical case, the potential for the quantum harmonic oscillator is given by
:
This problem can either be treated by directly solving the Schrödinger equation, which is not trivial, or by using the more elegant "ladder method" first proposed by Paul Dirac. The eigenstates are given by
:
:
where ''Hn'' are the Hermite polynomials
:
and the corresponding energy levels are
:
This is another example illustrating the discretization of energy for bound state
Bound or bounds may refer to:
Mathematics
* Bound variable
* Upper and lower bounds, observed limits of mathematical functions
Physics
* Bound state, a particle that has a tendency to remain localized in one or more regions of space
Geography
* ...
s.
Mach–Zehnder interferometer
Mach–Zehnder interferometer
The Mach–Zehnder interferometer is a device used to determine the relative phase shift variations between two collimated beams derived by splitting light from a single source. The interferometer has been used, among other things, to measure p ...
(MZI) illustrates the concepts of superposition and interference with linear algebra in dimension 2, rather than differential equations. It can be seen as a simplified version of the double-slit experiment, but it is of interest in its own right, for example in the delayed choice quantum eraser, the Elitzur–Vaidman bomb tester, and in studies of quantum entanglement.
We can model a photon going through the interferometer by considering that at each point it can be in a superposition of only two paths: the "lower" path which starts from the left, goes straight through both beam splitters, and ends at the top, and the "upper" path which starts from the bottom, goes straight through both beam splitters, and ends at the right. The quantum state of the photon is therefore a vector that is a superposition of the "lower" path and the "upper" path , that is, for complex . In order to respect the postulate that we require that .
Both beam splitters are modelled as the unitary matrix , which means that when a photon meets the beam splitter it will either stay on the same path with a probability amplitude of , or be reflected to the other path with a probability amplitude of . The phase shifter on the upper arm is modelled as the unitary matrix , which means that if the photon is on the "upper" path it will gain a relative phase of , and it will stay unchanged if it is in the lower path.
A photon that enters the interferometer from the left will then be acted upon with a beam splitter , a phase shifter , and another beam splitter , and so end up in the state
:
and the probabilities that it will be detected at the right or at the top are given respectively by
:
:
One can therefore use the Mach–Zehnder interferometer to estimate the phase shift
In physics and mathematics, the phase of a periodic function F of some real variable t (such as time) is an angle-like quantity representing the fraction of the cycle covered up to t. It is denoted \phi(t) and expressed in such a scale that it ...
by estimating these probabilities.
It is interesting to consider what would happen if the photon were definitely in either the "lower" or "upper" paths between the beam splitters. This can be accomplished by blocking one of the paths, or equivalently by removing the first beam splitter (and feeding the photon from the left or the bottom, as desired). In both cases there will be no interference between the paths anymore, and the probabilities are given by , independently of the phase . From this we can conclude that the photon does not take one path or another after the first beam splitter, but rather that it is in a genuine quantum superposition of the two paths.
Applications
Quantum mechanics has had enormous success in explaining many of the features of our universe, with regards to small-scale and discrete quantities and interactions which cannot be explained by classical methods. Quantum mechanics is often the only theory that can reveal the individual behaviors of the subatomic particles that make up all forms of matter (electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s, protons, neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons beh ...
s, photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s, and others). Solid-state physics and materials science are dependent upon quantum mechanics.
In many aspects modern technology operates at a scale where quantum effects are significant. Important applications of quantum theory include quantum chemistry, quantum optics
Quantum optics is a branch of atomic, molecular, and optical physics dealing with how individual quanta of light, known as photons, interact with atoms and molecules. It includes the study of the particle-like properties of photons. Photons have ...
, quantum computing, superconducting magnets, light-emitting diode
A light-emitting diode (LED) is a semiconductor device that emits light when current flows through it. Electrons in the semiconductor recombine with electron holes, releasing energy in the form of photons. The color of the light (co ...
s, the optical amplifier and the laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The fi ...
, the transistor
upright=1.4, gate (G), body (B), source (S) and drain (D) terminals. The gate is separated from the body by an insulating layer (pink).
A transistor is a semiconductor device used to Electronic amplifier, amplify or electronic switch, switch ...
and semiconductor
A semiconductor is a material which has an electrical conductivity value falling between that of a conductor, such as copper, and an insulator, such as glass. Its resistivity falls as its temperature rises; metals behave in the opposite way. ...
s such as the microprocessor
A microprocessor is a computer processor where the data processing logic and control is included on a single integrated circuit, or a small number of integrated circuits. The microprocessor contains the arithmetic, logic, and control circ ...
, medical and research imaging such as magnetic resonance imaging and electron microscopy. Explanations for many biological and physical phenomena are rooted in the nature of the chemical bond, most notably the macro-molecule DNA.
Relation to other scientific theories
Classical mechanics
The rules of quantum mechanics assert that the state space of a system is a Hilbert space and that observables of the system areHermitian operators
In mathematics, a self-adjoint operator on an infinite-dimensional complex vector space ''V'' with inner product \langle\cdot,\cdot\rangle (equivalently, a Hermitian operator in the finite-dimensional case) is a linear map ''A'' (from ''V'' to its ...
acting on vectors in that space – although they do not tell us which Hilbert space or which operators. These can be chosen appropriately in order to obtain a quantitative description of a quantum system, a necessary step in making physical predictions. An important guide for making these choices is the correspondence principle, a heuristic which states that the predictions of quantum mechanics reduce to those of classical mechanics in the regime of large quantum numbers. One can also start from an established classical model of a particular system, and then try to guess the underlying quantum model that would give rise to the classical model in the correspondence limit. This approach is known as quantization.
When quantum mechanics was originally formulated, it was applied to models whose correspondence limit was non-relativistic classical mechanics
Classical mechanics is a physical theory describing the motion of macroscopic objects, from projectiles to parts of machinery, and astronomical objects, such as spacecraft, planets, stars, and galaxies. For objects governed by classi ...
. For instance, the well-known model of the quantum harmonic oscillator uses an explicitly non-relativistic expression for the kinetic energy
In physics, the kinetic energy of an object is the energy that it possesses due to its motion.
It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acc ...
of the oscillator, and is thus a quantum version of the classical harmonic oscillator.
Complications arise with chaotic systems
Chaos theory is an interdisciplinary area of scientific study and branch of mathematics focused on underlying patterns and deterministic laws of dynamical systems that are highly sensitive to initial conditions, and were once thought to have ...
, which do not have good quantum numbers, and quantum chaos
Quantum chaos is a branch of physics which studies how chaotic classical dynamical systems can be described in terms of quantum theory. The primary question that quantum chaos seeks to answer is: "What is the relationship between quantum mech ...
studies the relationship between classical and quantum descriptions in these systems.
Quantum decoherence is a mechanism through which quantum systems lose coherence, and thus become incapable of displaying many typically quantum effects: quantum superpositions become simply probabilistic mixtures, and quantum entanglement
Quantum entanglement is the phenomenon that occurs when a group of particles are generated, interact, or share spatial proximity in a way such that the quantum state of each particle of the group cannot be described independently of the state of ...
becomes simply classical correlations. Quantum coherence is not typically evident at macroscopic scales, except maybe at temperatures approaching absolute zero at which quantum behavior may manifest macroscopically.
Many macroscopic properties of a classical system are a direct consequence of the quantum behavior of its parts. For example, the stability of bulk matter (consisting of atoms and molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioche ...
s which would quickly collapse under electric forces alone), the rigidity of solids, and the mechanical, thermal, chemical, optical and magnetic properties of matter are all results of the interaction of electric charge
Electric charge is the physical property of matter that causes charged matter to experience a force when placed in an electromagnetic field. Electric charge can be ''positive'' or ''negative'' (commonly carried by protons and electrons respe ...
s under the rules of quantum mechanics.
Special relativity and electrodynamics
Early attempts to merge quantum mechanics withspecial relativity
In physics, the special theory of relativity, or special relativity for short, is a scientific theory regarding the relationship between space and time. In Albert Einstein's original treatment, the theory is based on two postulates:
# The laws ...
involved the replacement of the Schrödinger equation with a covariant equation such as the Klein–Gordon equation
The Klein–Gordon equation (Klein–Fock–Gordon equation or sometimes Klein–Gordon–Fock equation) is a relativistic wave equation, related to the Schrödinger equation. It is second-order in space and time and manifestly Lorentz-covariant ...
or the Dirac equation
In particle physics, the Dirac equation is a relativistic wave equation derived by British physicist Paul Dirac in 1928. In its free form, or including electromagnetic interactions, it describes all spin- massive particles, called "Dirac par ...
. While these theories were successful in explaining many experimental results, they had certain unsatisfactory qualities stemming from their neglect of the relativistic creation and annihilation of particles. A fully relativistic quantum theory required the development of quantum field theory, which applies quantization to a field (rather than a fixed set of particles). The first complete quantum field theory, quantum electrodynamics
In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and spec ...
, provides a fully quantum description of the electromagnetic interaction
In physics, electromagnetism is an interaction that occurs between particles with electric charge. It is the second-strongest of the four fundamental interactions, after the strong force, and it is the dominant force in the interactions o ...
. Quantum electrodynamics is, along with general relativity
General relativity, also known as the general theory of relativity and Einstein's theory of gravity, is the geometric theory of gravitation published by Albert Einstein in 1915 and is the current description of gravitation in modern physics ...
, one of the most accurate physical theories ever devised.
The full apparatus of quantum field theory is often unnecessary for describing electrodynamic systems. A simpler approach, one that has been used since the inception of quantum mechanics, is to treat charged particles as quantum mechanical objects being acted on by a classical electromagnetic field. For example, the elementary quantum model of the hydrogen atom describes the electric field of the hydrogen atom using a classical Coulomb potential. This "semi-classical" approach fails if quantum fluctuations in the electromagnetic field play an important role, such as in the emission of photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s by charged particle
In physics, a charged particle is a particle with an electric charge. It may be an ion, such as a molecule or atom with a surplus or deficit of electrons relative to protons. It can also be an electron or a proton, or another elementary pa ...
s.
Quantum field
In theoretical physics, quantum field theory (QFT) is a theoretical framework that combines classical field theory, special relativity, and quantum mechanics. QFT is used in particle physics to construct physical models of subatomic particles ...
theories for the strong nuclear force
The strong interaction or strong force is a fundamental interaction that confines quarks into proton, neutron, and other hadron particles. The strong interaction also binds neutrons and protons to create atomic nuclei, where it is called the ...
and the weak nuclear force
In nuclear physics and particle physics, the weak interaction, which is also often called the weak force or weak nuclear force, is one of the four known fundamental interactions, with the others being electromagnetism, the strong interaction ...
have also been developed. The quantum field theory of the strong nuclear force is called quantum chromodynamics, and describes the interactions of subnuclear particles such as quarks and gluons. The weak nuclear force and the electromagnetic force were unified, in their quantized forms, into a single quantum field theory (known as electroweak theory
In particle physics, the electroweak interaction or electroweak force is the unified description of two of the four known fundamental interactions of nature: electromagnetism and the weak interaction. Although these two forces appear very differe ...
), by the physicists Abdus Salam
Mohammad Abdus Salam Salam adopted the forename "Mohammad" in 1974 in response to the anti-Ahmadiyya decrees in Pakistan, similarly he grew his beard. (; ; 29 January 192621 November 1996) was a Punjabi Pakistani theoretical physicist and a ...
, Sheldon Glashow
Sheldon Lee Glashow (, ; born December 5, 1932) is a Nobel Prize-winning American theoretical physicist. He is the Metcalf Professor of Mathematics and Physics at Boston University and Eugene Higgins Professor of Physics, Emeritus, at Harvard U ...
and Steven Weinberg
Steven Weinberg (; May 3, 1933 – July 23, 2021) was an American theoretical physicist and Nobel laureate in physics for his contributions with Abdus Salam and Sheldon Glashow to the unification of the weak force and electromagnetic interac ...
.
Relation to general relativity
Even though the predictions of both quantum theory and general relativity have been supported by rigorous and repeatedempirical evidence
Empirical evidence for a proposition is evidence, i.e. what supports or counters this proposition, that is constituted by or accessible to sense experience or experimental procedure. Empirical evidence is of central importance to the sciences ...
, their abstract formalisms contradict each other and they have proven extremely difficult to incorporate into one consistent, cohesive model. Gravity is negligible in many areas of particle physics, so that unification between general relativity and quantum mechanics is not an urgent issue in those particular applications. However, the lack of a correct theory of quantum gravity is an important issue in physical cosmology
Physical cosmology is a branch of cosmology concerned with the study of cosmological models. A cosmological model, or simply cosmology, provides a description of the largest-scale structures and dynamics of the universe and allows study of f ...
and the search by physicists for an elegant "Theory of Everything
A theory of everything (TOE or TOE/ToE), final theory, ultimate theory, unified field theory or master theory is a hypothetical, singular, all-encompassing, coherent theoretical framework of physics that fully explains and links together all asp ...
" (TOE). Consequently, resolving the inconsistencies between both theories has been a major goal of 20th- and 21st-century physics. This TOE would combine not only the models of subatomic physics but also derive the four fundamental forces of nature from a single force or phenomenon.
One proposal for doing so is string theory, which posits that the point-like particles of particle physics
Particle physics or high energy physics is the study of fundamental particles and forces that constitute matter and radiation. The fundamental particles in the universe are classified in the Standard Model as fermions (matter particles) an ...
are replaced by one-dimensional
In physics and mathematics, a sequence of ''n'' numbers can specify a location in ''n''-dimensional space. When , the set of all such locations is called a one-dimensional space. An example of a one-dimensional space is the number line, where the ...
objects called strings
String or strings may refer to:
*String (structure), a long flexible structure made from threads twisted together, which is used to tie, bind, or hang other objects
Arts, entertainment, and media Films
* ''Strings'' (1991 film), a Canadian anim ...
. String theory describes how these strings propagate through space and interact with each other. On distance scales larger than the string scale, a string looks just like an ordinary particle, with its mass
Mass is an intrinsic property of a body. It was traditionally believed to be related to the quantity of matter in a physical body, until the discovery of the atom and particle physics. It was found that different atoms and different eleme ...
, charge
Charge or charged may refer to:
Arts, entertainment, and media Films
* '' Charge, Zero Emissions/Maximum Speed'', a 2011 documentary
Music
* ''Charge'' (David Ford album)
* ''Charge'' (Machel Montano album)
* ''Charge!!'', an album by The Aqu ...
, and other properties determined by the vibration
Vibration is a mechanical phenomenon whereby oscillations occur about an equilibrium point. The word comes from Latin ''vibrationem'' ("shaking, brandishing"). The oscillations may be periodic, such as the motion of a pendulum—or random, su ...
al state of the string. In string theory, one of the many vibrational states of the string corresponds to the graviton
In theories of quantum gravity, the graviton is the hypothetical quantum of gravity, an elementary particle that mediates the force of gravitational interaction. There is no complete quantum field theory of gravitons due to an outstanding mathem ...
, a quantum mechanical particle that carries gravitational force.
Another popular theory is loop quantum gravity (LQG), which describes quantum properties of gravity and is thus a theory of quantum spacetime
In mathematical physics, the concept of quantum spacetime is a generalization of the usual concept of spacetime in which some variables that ordinarily commute are assumed not to commute and form a different Lie algebra. The choice of that algebr ...
. LQG is an attempt to merge and adapt standard quantum mechanics and standard general relativity. This theory describes space as an extremely fine fabric "woven" of finite loops called spin network
In physics, a spin network is a type of diagram which can be used to represent states and interactions between particles and fields in quantum mechanics. From a mathematical perspective, the diagrams are a concise way to represent multilinear f ...
s. The evolution of a spin network over time is called a spin foam
In physics, the topological structure of spinfoam or spin foam consists of two-dimensional faces representing a configuration required by functional integration to obtain a Feynman's path integral description of quantum gravity. These structur ...
. The characteristic length scale of a spin foam is the Planck length, approximately 1.616×10−35 m, and so lengths shorter than the Planck length are not physically meaningful in LQG.
Philosophical implications
Since its inception, the many counter-intuitive aspects and results of quantum mechanics have provoked strongphilosophical
Philosophy (from , ) is the systematized study of general and fundamental questions, such as those about existence, reason, knowledge, values, mind, and language. Such questions are often posed as problems to be studied or resolved. Some ...
debates and many interpretations. The arguments centre on the probabilistic nature of quantum mechanics, the difficulties with wavefunction collapse
In quantum mechanics, wave function collapse occurs when a wave function—initially in a superposition of several eigenstates—reduces to a single eigenstate due to interaction with the external world. This interaction is called an ''observa ...
and the related measurement problem
In quantum mechanics, the measurement problem is the problem of how, or whether, wave function collapse occurs. The inability to observe such a collapse directly has given rise to different interpretations of quantum mechanics and poses a key se ...
, and quantum nonlocality
In theoretical physics, quantum nonlocality refers to the phenomenon by which the measurement statistics of a multipartite quantum system do not admit an interpretation in terms of a local realistic theory. Quantum nonlocality has been experime ...
. Perhaps the only consensus that exists about these issues is that there is no consensus. Richard Feynman
Richard Phillips Feynman (; May 11, 1918 – February 15, 1988) was an American theoretical physicist, known for his work in the path integral formulation of quantum mechanics, the theory of quantum electrodynamics, the physics of the superfl ...
once said, "I think I can safely say that nobody understands quantum mechanics." According to Steven Weinberg
Steven Weinberg (; May 3, 1933 – July 23, 2021) was an American theoretical physicist and Nobel laureate in physics for his contributions with Abdus Salam and Sheldon Glashow to the unification of the weak force and electromagnetic interac ...
, "There is now in my opinion no entirely satisfactory interpretation of quantum mechanics."
The views of Niels Bohr
Niels Henrik David Bohr (; 7 October 1885 – 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922 ...
, Werner Heisenberg
Werner Karl Heisenberg () (5 December 1901 – 1 February 1976) was a German theoretical physicist and one of the main pioneers of the theory of quantum mechanics. He published his work in 1925 in a breakthrough paper. In the subsequent serie ...
and other physicists are often grouped together as the "Copenhagen interpretation
The Copenhagen interpretation is a collection of views about the meaning of quantum mechanics, principally attributed to Niels Bohr and Werner Heisenberg. It is one of the oldest of numerous proposed interpretations of quantum mechanics, as feat ...
". According to these views, the probabilistic nature of quantum mechanics is not a ''temporary'' feature which will eventually be replaced by a deterministic theory, but is instead a ''final'' renunciation of the classical idea of "causality". Bohr in particular emphasized that any well-defined application of the quantum mechanical formalism must always make reference to the experimental arrangement, due to the complementary
A complement is something that completes something else.
Complement may refer specifically to:
The arts
* Complement (music), an interval that, when added to another, spans an octave
** Aggregate complementation, the separation of pitch-class ...
nature of evidence obtained under different experimental situations. Copenhagen-type interpretations remain popular in the 21st century.
Albert Einstein
Albert Einstein ( ; ; 14 March 1879 – 18 April 1955) was a German-born theoretical physicist, widely acknowledged to be one of the greatest and most influential physicists of all time. Einstein is best known for developing the theory ...
, himself one of the founders of quantum theory
Quantum theory may refer to:
Science
*Quantum mechanics, a major field of physics
*Old quantum theory, predating modern quantum mechanics
* Quantum field theory, an area of quantum mechanics that includes:
** Quantum electrodynamics
** Quantum ...
, was troubled by its apparent failure to respect some cherished metaphysical principles, such as determinism and locality
Locality may refer to:
* Locality (association), an association of community regeneration organizations in England
* Locality (linguistics)
* Locality (settlement)
* Suburbs and localities (Australia), in which a locality is a geographic subdivis ...
. Einstein's long-running exchanges with Bohr about the meaning and status of quantum mechanics are now known as the Bohr–Einstein debates. Einstein believed that underlying quantum mechanics must be a theory that explicitly forbids action at a distance
In physics, action at a distance is the concept that an object can be affected without being physically touched (as in mechanical contact) by another object. That is, it is the non-local interaction of objects that are separated in space.
Non- ...
. He argued that quantum mechanics was incomplete, a theory that was valid but not fundamental, analogous to how thermodynamics
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed by the four laws of th ...
is valid, but the fundamental theory behind it is statistical mechanics. In 1935, Einstein and his collaborators Boris Podolsky
Boris Yakovlevich Podolsky (russian: link=no, Бори́с Я́ковлевич Подо́льский; June 29, 1896 – November 28, 1966) was a Russian-American physicist of Jewish descent, noted for his work with Albert Einstein and Nathan ...
and Nathan Rosen
Nathan Rosen (Hebrew: נתן רוזן; March 22, 1909 – December 18, 1995) was an American-Israeli physicist noted for his study on the structure of the hydrogen atom and his work with Albert Einstein and Boris Podolsky on entangled wave functio ...
published an argument that the principle of locality implies the incompleteness of quantum mechanics, a thought experiment
A thought experiment is a hypothetical situation in which a hypothesis, theory, or principle is laid out for the purpose of thinking through its consequences.
History
The ancient Greek ''deiknymi'' (), or thought experiment, "was the most anc ...
later termed the Einstein–Podolsky–Rosen paradox
The Einstein–Podolsky–Rosen (EPR) paradox is a thought experiment proposed by physicists Albert Einstein, Boris Podolsky and Nathan Rosen, with which they argued that the description of physical reality provided by quantum mechanics was inco ...
. In 1964, John Bell showed that EPR's principle of locality, together with determinism, was actually incompatible with quantum mechanics: they implied constraints on the correlations produced by distance systems, now known as Bell inequalities, that can be violated by entangled particles. Since then several experiments have been performed to obtain these correlations, with the result that they do in fact violate Bell inequalities, and thus falsify the conjunction of locality with determinism.
Bohmian mechanics shows that it is possible to reformulate quantum mechanics to make it deterministic, at the price of making it explicitly nonlocal. It attributes not only a wave function to a physical system, but in addition a real position, that evolves deterministically under a nonlocal guiding equation. The evolution of a physical system is given at all times by the Schrödinger equation
The Schrödinger equation is a linear partial differential equation that governs the wave function of a quantum-mechanical system. It is a key result in quantum mechanics, and its discovery was a significant landmark in the development of th ...
together with the guiding equation; there is never a collapse of the wave function. This solves the measurement problem.
Everett's many-worlds interpretation
The many-worlds interpretation (MWI) is an interpretation of quantum mechanics that asserts that the universal wavefunction is objectively real, and that there is no wave function collapse. This implies that all possible outcomes of quantum ...
, formulated in 1956, holds that ''all'' the possibilities described by quantum theory ''simultaneously'' occur in a multiverse composed of mostly independent parallel universes. This is a consequence of removing the axiom of the collapse of the wave packet. All possible states of the measured system and the measuring apparatus, together with the observer, are present in a real physical quantum superposition. While the multiverse is deterministic, we perceive non-deterministic behavior governed by probabilities, because we don't observe the multiverse as a whole, but only one parallel universe at a time. Exactly how this is supposed to work has been the subject of much debate. Several attempts have been made to make sense of this and derive the Born rule, with no consensus on whether they have been successful.
Relational quantum mechanics
:''This article is intended for those already familiar with quantum mechanics and its attendant interpretational difficulties. Readers who are new to the subject may first want to read the introduction to quantum mechanics.''
Relational quantum m ...
appeared in the late 1990s as a modern derivative of Copenhagen-type ideas, and QBism
In physics and the philosophy of physics, quantum Bayesianism is a collection of related approaches to the interpretation of quantum mechanics, of which the most prominent is QBism (pronounced "cubism"). QBism is an interpretation that takes an a ...
was developed some years later.
History
 Quantum mechanics was developed in the early decades of the 20th century, driven by the need to explain phenomena that, in some cases, had been observed in earlier times. Scientific inquiry into the wave nature of light began in the 17th and 18th centuries, when scientists such as Robert Hooke, Christiaan Huygens and
Quantum mechanics was developed in the early decades of the 20th century, driven by the need to explain phenomena that, in some cases, had been observed in earlier times. Scientific inquiry into the wave nature of light began in the 17th and 18th centuries, when scientists such as Robert Hooke, Christiaan Huygens and Leonhard Euler
Leonhard Euler ( , ; 15 April 170718 September 1783) was a Swiss mathematician, physicist, astronomer, geographer, logician and engineer who founded the studies of graph theory and topology and made pioneering and influential discoveries in ma ...
proposed a wave theory of light based on experimental observations. In 1803 English polymath
A polymath ( el, πολυμαθής, , "having learned much"; la, homo universalis, "universal human") is an individual whose knowledge spans a substantial number of subjects, known to draw on complex bodies of knowledge to solve specific pro ...
Thomas Young described the famous double-slit experiment
In modern physics, the double-slit experiment is a demonstration that light and matter can display characteristics of both classically defined waves and particles; moreover, it displays the fundamentally probabilistic nature of quantum mechanics ...
. This experiment played a major role in the general acceptance of the wave theory of light
In physics, physical optics, or wave optics, is the branch of optics that studies Interference (wave propagation), interference, diffraction, Polarization (waves), polarization, and other phenomena for which the ray approximation of geometric opti ...
.
During the early 19th century, chemical
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., w ...
research by John Dalton and Amedeo Avogadro
Lorenzo Romano Amedeo Carlo Avogadro, Count of Quaregna and Cerreto (, also , ; 9 August 17769 July 1856) was an Italian scientist, most noted for his contribution to molecular theory now known as Avogadro's law, which states that equal volume ...
lent weight to the atomic theory
Atomic theory is the scientific theory that matter is composed of particles called atoms. Atomic theory traces its origins to an ancient philosophical tradition known as atomism. According to this idea, if one were to take a lump of matter ...
of matter, an idea that James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and li ...
, Ludwig Boltzmann
Ludwig Eduard Boltzmann (; 20 February 1844 – 5 September 1906) was an Austrian physicist and philosopher. His greatest achievements were the development of statistical mechanics, and the statistical explanation of the second law of ther ...
and others built upon to establish the kinetic theory of gases. The successes of kinetic theory gave further credence to the idea that matter is composed of atoms, yet the theory also had shortcomings that would only be resolved by the development of quantum mechanics. While the early conception of atoms from Greek philosophy had been that they were indivisible units the word "atom" deriving from the Greek for "uncuttable" the 19th century saw the formulation of hypotheses about subatomic structure. One important discovery in that regard was Michael Faraday
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English scientist who contributed to the study of electromagnetism and electrochemistry. His main discoveries include the principles underlying electromagnetic inducti ...
's 1838 observation of a glow caused by an electrical discharge inside a glass tube containing gas at low pressure. Julius Plücker
Julius Plücker (16 June 1801 – 22 May 1868) was a German mathematician and physicist. He made fundamental contributions to the field of analytical geometry and was a pioneer in the investigations of cathode rays that led eventually to the dis ...
, Johann Wilhelm Hittorf
Johann Wilhelm Hittorf (27 March 1824 – 28 November 1914) was a German physicist who was born in Bonn and died in Münster, Germany.
Hittorf was the first to compute the electricity-carrying capacity of charged atoms and molecules (ions), an ...
and Eugen Goldstein
Eugen Goldstein (; 5 September 1850 – 25 December 1930) was a German physicist. He was an early investigator of discharge tubes, the discoverer of anode rays or canal rays, later identified as positive ions in the gas phase including the h ...
carried on and improved upon Faraday's work, leading to the identification of cathode rays
Cathode rays or electron beam (e-beam) are streams of electrons observed in discharge tubes. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to el ...
, which J. J. Thomson
Sir Joseph John Thomson (18 December 1856 – 30 August 1940) was a British physicist and Nobel Laureate in Physics, credited with the discovery of the electron, the first subatomic particle to be discovered.
In 1897, Thomson showed that ...
found to consist of subatomic particles that would be called electrons.
The black-body radiation problem was discovered by Gustav Kirchhoff
Gustav Robert Kirchhoff (; 12 March 1824 – 17 October 1887) was a German physicist who contributed to the fundamental understanding of electrical circuits, spectroscopy, and the emission of black-body radiation by heated objects.
He ...
in 1859. In 1900, Max Planck
Max Karl Ernst Ludwig Planck (, ; 23 April 1858 – 4 October 1947) was a German theoretical physicist whose discovery of energy quanta won him the Nobel Prize in Physics in 1918.
Planck made many substantial contributions to theoretical p ...
proposed the hypothesis that energy is radiated and absorbed in discrete "quanta" (or energy packets), yielding a calculation that precisely matched the observed patterns of black-body radiation. The word ''quantum'' derives from the Latin
Latin (, or , ) is a classical language belonging to the Italic branch of the Indo-European languages. Latin was originally a dialect spoken in the lower Tiber area (then known as Latium) around present-day Rome, but through the power of the ...
, meaning "how great" or "how much". According to Planck, quantities of energy could be thought of as divided into "elements" whose size (''E'') would be proportional to their frequency
Frequency is the number of occurrences of a repeating event per unit of time. It is also occasionally referred to as ''temporal frequency'' for clarity, and is distinct from ''angular frequency''. Frequency is measured in hertz (Hz) which is eq ...
(''ν''):
:,
where ''h'' is Planck's constant. Planck cautiously insisted that this was only an aspect of the processes of absorption and emission of radiation and was not the ''physical reality'' of the radiation. In fact, he considered his quantum hypothesis a mathematical trick to get the right answer rather than a sizable discovery. However, in 1905 Albert Einstein
Albert Einstein ( ; ; 14 March 1879 – 18 April 1955) was a German-born theoretical physicist, widely acknowledged to be one of the greatest and most influential physicists of all time. Einstein is best known for developing the theory ...
interpreted Planck's quantum hypothesis realistically and used it to explain the photoelectric effect
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, and solid sta ...
, in which shining light on certain materials can eject electrons from the material. Niels Bohr
Niels Henrik David Bohr (; 7 October 1885 – 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922 ...
then developed Planck's ideas about radiation into a model of the hydrogen atom that successfully predicted the spectral line
A spectral line is a dark or bright line in an otherwise uniform and continuous spectrum, resulting from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. Spectral lines are often used to iden ...
s of hydrogen. Einstein further developed this idea to show that an electromagnetic wave
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visib ...
such as light could also be described as a particle (later called the photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
), with a discrete amount of energy that depends on its frequency. In his paper "On the Quantum Theory of Radiation," Einstein expanded on the interaction between energy and matter to explain the absorption and emission of energy by atoms. Although overshadowed at the time by his general theory of relativity, this paper articulated the mechanism underlying the stimulated emission of radiation, which became the basis of the laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The fi ...
.
 This phase is known as the
This phase is known as the old quantum theory
The old quantum theory is a collection of results from the years 1900–1925 which predate modern quantum mechanics. The theory was never complete or self-consistent, but was rather a set of heuristic corrections to classical mechanics. The theory ...
. Never complete or self-consistent, the old quantum theory was rather a set of heuristic
A heuristic (; ), or heuristic technique, is any approach to problem solving or self-discovery that employs a practical method that is not guaranteed to be optimal, perfect, or rational, but is nevertheless sufficient for reaching an immediate ...
corrections to classical mechanics
Classical mechanics is a physical theory describing the motion of macroscopic objects, from projectiles to parts of machinery, and astronomical objects, such as spacecraft, planets, stars, and galaxies. For objects governed by classi ...
. The theory is now understood as a semi-classical approximation to modern quantum mechanics. Notable results from this period include, in addition to the work of Planck, Einstein and Bohr mentioned above, Einstein and Peter Debye
Peter Joseph William Debye (; ; March 24, 1884 – November 2, 1966) was a Dutch-American physicist and physical chemist, and Nobel laureate in Chemistry.
Biography
Early life
Born Petrus Josephus Wilhelmus Debije in Maastricht, Netherlands, D ...
's work on the specific heat
In thermodynamics, the specific heat capacity (symbol ) of a substance is the heat capacity of a sample of the substance divided by the mass of the sample, also sometimes referred to as massic heat capacity. Informally, it is the amount of heat t ...
of solids, Bohr and Hendrika Johanna van Leeuwen's proof that classical physics cannot account for diamagnetism
Diamagnetic materials are repelled by a magnetic field; an applied magnetic field creates an induced magnetic field in them in the opposite direction, causing a repulsive force. In contrast, paramagnetic and ferromagnetic materials are attracte ...
, and Arnold Sommerfeld's extension of the Bohr model to include special-relativistic effects.
In the mid-1920s quantum mechanics was developed to become the standard formulation for atomic physics. In 1923, the French physicist Louis de Broglie
Louis Victor Pierre Raymond, 7th Duc de Broglie (, also , or ; 15 August 1892 – 19 March 1987) was a French physicist and aristocrat who made groundbreaking contributions to Old quantum theory, quantum theory. In his 1924 PhD thesis, he pos ...
put forward his theory of matter waves by stating that particles can exhibit wave characteristics and vice versa. Building on de Broglie's approach, modern quantum mechanics was born in 1925, when the German physicists Werner Heisenberg
Werner Karl Heisenberg () (5 December 1901 – 1 February 1976) was a German theoretical physicist and one of the main pioneers of the theory of quantum mechanics. He published his work in 1925 in a breakthrough paper. In the subsequent serie ...
, Max Born, and Pascual Jordan
Ernst Pascual Jordan (; 18 October 1902 – 31 July 1980) was a German theoretical and mathematical physicist who made significant contributions to quantum mechanics and quantum field theory. He contributed much to the mathematical form of matri ...
David Edwards,"The Mathematical Foundations of Quantum Mechanics", ''Synthese'', Volume 42, Number 1/September, 1979, pp. 1–70.D. Edwards, "The Mathematical Foundations of Quantum Field Theory: Fermions, Gauge Fields, and Super-symmetry, Part I: Lattice Field Theories", ''International J. of Theor. Phys.'', Vol. 20, No. 7 (1981). developed matrix mechanics
Matrix mechanics is a formulation of quantum mechanics created by Werner Heisenberg, Max Born, and Pascual Jordan in 1925. It was the first conceptually autonomous and logically consistent formulation of quantum mechanics. Its account of quantum j ...
and the Austrian physicist Erwin Schrödinger
Erwin Rudolf Josef Alexander Schrödinger (, ; ; 12 August 1887 – 4 January 1961), sometimes written as or , was a Nobel Prize-winning Austrian physicist with Irish citizenship who developed a number of fundamental results in quantum theo ...
invented wave mechanics Wave mechanics may refer to:
* the mechanics of waves
* the ''wave equation'' in quantum physics, see Schrödinger equation
See also
* Quantum mechanics
* Wave equation
The (two-way) wave equation is a second-order linear partial different ...
. Born introduced the probabilistic interpretation of Schrödinger's wave function in July 1926. Thus, the entire field of quantum physics emerged, leading to its wider acceptance at the Fifth Solvay Conference
The Solvay Conferences (french: Conseils Solvay) have been devoted to outstanding preeminent open problems in both physics and chemistry. They began with the historic invitation-only 1911 Solvay Conference on Physics, considered a turning point i ...
in 1927.
By 1930 quantum mechanics had been further unified and formalized by David Hilbert, Paul Dirac
Paul Adrien Maurice Dirac (; 8 August 1902 – 20 October 1984) was an English theoretical physicist who is regarded as one of the most significant physicists of the 20th century. He was the Lucasian Professor of Mathematics at the Univer ...
and John von Neumann
John von Neumann (; hu, Neumann János Lajos, ; December 28, 1903 – February 8, 1957) was a Hungarian-American mathematician, physicist, computer scientist, engineer and polymath. He was regarded as having perhaps the widest cove ...
with greater emphasis on measurement, the statistical nature of our knowledge of reality, and philosophical speculation about the 'observer'. It has since permeated many disciplines, including quantum chemistry, quantum electronics, quantum optics
Quantum optics is a branch of atomic, molecular, and optical physics dealing with how individual quanta of light, known as photons, interact with atoms and molecules. It includes the study of the particle-like properties of photons. Photons have ...
, and quantum information science
Quantum information science is an interdisciplinary field that seeks to understand the analysis, processing, and transmission of information using quantum mechanics principles. It combines the study of Information science with quantum effects in ...
. It also provides a useful framework for many features of the modern periodic table of elements
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of c ...
, and describes the behaviors of atoms during chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
ing and the flow of electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s in computer semiconductor
A semiconductor is a material which has an electrical conductivity value falling between that of a conductor, such as copper, and an insulator, such as glass. Its resistivity falls as its temperature rises; metals behave in the opposite way. ...
s, and therefore plays a crucial role in many modern technologies. While quantum mechanics was constructed to describe the world of the very small, it is also needed to explain some macroscopic
The macroscopic scale is the length scale on which objects or phenomena are large enough to be visible with the naked eye, without magnifying optical instruments. It is the opposite of microscopic.
Overview
When applied to physical phenomena a ...
phenomena such as superconductors
Superconductivity is a set of physical properties observed in certain materials where electrical resistance vanishes and magnetic flux fields are expelled from the material. Any material exhibiting these properties is a superconductor. Unlike ...
and superfluid
Superfluidity is the characteristic property of a fluid with zero viscosity which therefore flows without any loss of kinetic energy. When stirred, a superfluid forms vortices that continue to rotate indefinitely. Superfluidity occurs in two ...
s.
See also
* Bra–ket notation *Einstein's thought experiments
A hallmark of Albert Einstein's career was his use of visualized thought experiments (german: Gedankenexperiment) as a fundamental tool for understanding physical issues and for elucidating his concepts to others. Einstein's thought experiments too ...
* List of textbooks on classical and quantum mechanics
This is a list of notable textbooks on classical mechanics and quantum mechanics arranged according to level and surnames of the authors in alphabetical order.
Undergraduate
Classical mechanics *
*
*
*
*
*
*
*
Quantum mechanics
* Three ...
* Macroscopic quantum phenomena
Macroscopic quantum phenomena are processes showing quantum behavior at the macroscopic scale, rather than at the atomic scale where quantum effects are prevalent. The best-known examples of macroscopic quantum phenomena are superfluidity and su ...
* Phase-space formulation
The phase-space formulation of quantum mechanics places the position ''and'' momentum variables on equal footing in phase space. In contrast, the Schrödinger picture uses the position ''or'' momentum representations (see also position and mo ...
* Regularization (physics)
In physics, especially quantum field theory, regularization is a method of modifying observables which have singularities in order to make them finite by the introduction of a suitable parameter called the regulator. The regulator, also know ...
* Two-state quantum system
In quantum mechanics, a two-state system (also known as a two-level system) is a quantum system that can exist in any quantum superposition of two independent (physically distinguishable) quantum states. The Hilbert space describing such a sys ...
Explanatory notes
References
Further reading
The following titles, all by working physicists, attempt to communicate quantum theory to lay people, using a minimum of technical apparatus. * Chester, Marvin (1987). ''Primer of Quantum Mechanics''. John Wiley. * *Richard Feynman
Richard Phillips Feynman (; May 11, 1918 – February 15, 1988) was an American theoretical physicist, known for his work in the path integral formulation of quantum mechanics, the theory of quantum electrodynamics, the physics of the superfl ...
, 1985. '' QED: The Strange Theory of Light and Matter'', Princeton University Press. . Four elementary lectures on quantum electrodynamics
In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and spec ...
and quantum field theory, yet containing many insights for the expert.
* Ghirardi, GianCarlo, 2004. ''Sneaking a Look at God's Cards'', Gerald Malsbary, trans. Princeton Univ. Press. The most technical of the works cited here. Passages using algebra
Algebra () is one of the broad areas of mathematics. Roughly speaking, algebra is the study of mathematical symbols and the rules for manipulating these symbols in formulas; it is a unifying thread of almost all of mathematics.
Elementary ...
, trigonometry
Trigonometry () is a branch of mathematics that studies relationships between side lengths and angles of triangles. The field emerged in the Hellenistic world during the 3rd century BC from applications of geometry to astronomical studies ...
, and bra–ket notation can be passed over on a first reading.
* N. David Mermin
Nathaniel David Mermin (; born 30 March 1935) is a solid-state physicist at Cornell University best known for the eponymous Mermin–Wagner theorem, his application of the term " boojum" to superfluidity, his textbook with Neil Ashcroft on sol ...
, 1990, "Spooky actions at a distance: mysteries of the QT" in his ''Boojums All the Way Through''. Cambridge University Press: 110–76.
* Victor Stenger
Victor John Stenger (; January 29, 1935 – August 25, 2014) was an American particle physicist, philosopher, author, and religious skeptic.
Following a career as a research scientist in the field of particle physics, Stenger was associat ...
, 2000. ''Timeless Reality: Symmetry, Simplicity, and Multiple Universes''. Buffalo, NY: Prometheus Books. Chpts. 5–8. Includes cosmological
Cosmology () is a branch of physics and metaphysics dealing with the nature of the universe. The term ''cosmology'' was first used in English in 1656 in Thomas Blount's ''Glossographia'', and in 1731 taken up in Latin by German philosopher ...
and philosophical
Philosophy (from , ) is the systematized study of general and fundamental questions, such as those about existence, reason, knowledge, values, mind, and language. Such questions are often posed as problems to be studied or resolved. Some ...
considerations.
More technical:
*
*
*
*
* Bryce DeWitt
Bryce Seligman DeWitt (January 8, 1923 – September 23, 2004), was an American theoretical physicist noted for his work in gravitation and quantum field theory.
Life
He was born Carl Bryce Seligman, but he and his three brothers, including th ...
, R. Neill Graham, eds., 1973. ''The Many-Worlds Interpretation of Quantum Mechanics'', Princeton Series in Physics, Princeton University Press.
*
*
* D. Greenberger, K. Hentschel, F. Weinert, eds., 2009. ''Compendium of quantum physics, Concepts, experiments, history and philosophy'', Springer-Verlag, Berlin, Heidelberg.
* A standard undergraduate text.
* Max Jammer, 1966. ''The Conceptual Development of Quantum Mechanics''. McGraw Hill.
* Hagen Kleinert
Hagen Kleinert (born 15 June 1941) is professor of theoretical physics at the Free University of Berlin, Germany (since 1968)Honorary Doctorat the
West University of Timișoaraandat thin Bishkek. He is alsHonorary Memberof th For his contributio ...
, 2004. ''Path Integrals in Quantum Mechanics, Statistics, Polymer Physics, and Financial Markets'', 3rd ed. Singapore: World ScientificDraft of 4th edition.
*
Online copy
* * Gunther Ludwig, 1968. ''Wave Mechanics''. London: Pergamon Press. *
George Mackey
George Whitelaw Mackey (February 1, 1916 – March 15, 2006) was an American mathematician known for his contributions to quantum logic, representation theory, and noncommutative geometry.
Career
Mackey earned his bachelor of arts at Rice Unive ...
(2004). ''The mathematical foundations of quantum mechanics''. Dover Publications. .
*
* Albert Messiah
Albert Messiah (23 September 1921, Nice – 17 April 2013, Paris) was a French physicist.
He studied at the Ecole Polytechnique.
He spent the Second World War in the Free France forces: he embarked on 22 June 1940 at Saint-Jean-de-Luz for Engla ...
, 1966. ''Quantum Mechanics'' (Vol. I), English translation from French by G.M. Temmer. North Holland, John Wiley & Sons. Cf. chpt. IV, section IIIonline
* * Scerri, Eric R., 2006. ''The Periodic Table: Its Story and Its Significance''. Oxford University Press. Considers the extent to which chemistry and the periodic system have been reduced to quantum mechanics. * * * * Veltman, Martinus J.G. (2003), ''Facts and Mysteries in Elementary Particle Physics''. On Wikibooks
This Quantum World
External links
* J. O'Connor and E. F. RobertsonIntroduction to Quantum Theory at Quantiki.
Quantum Physics Made Relatively Simple
three video lectures by
Hans Bethe
Hans Albrecht Bethe (; July 2, 1906 – March 6, 2005) was a German-American theoretical physicist who made major contributions to nuclear physics, astrophysics, quantum electrodynamics, and solid-state physics, and who won the 1967 Nobel ...
; Course material
Quantum Cook Book
an
PHYS 201: Fundamentals of Physics II
by
Ramamurti Shankar
Ramamurti Shankar (born April 28, 1947) is the Josiah Willard Gibbs professor of Physics at Yale University, in New Haven, Connecticut.
Education
He received his B. Tech in electrical engineering from the Indian Institute of Technology Madras, I ...
, Yale OpenCourseware
The Modern Revolution in Physics
– an online textbook. *
MIT OpenCourseWare
MIT OpenCourseWare (MIT OCW) is an initiative of the Massachusetts Institute of Technology (MIT) to publish all of the educational materials from its undergraduate- and graduate-level courses online, freely and openly available to anyone, anyw ...
Chemistry
an
Physics
Se
8.04
an
5½ Examples in Quantum Mechanics
Imperial College Quantum Mechanics Course.
;Philosophy * * {{DEFAULTSORT:Quantum Mechanics