Melanoma on:
[Wikipedia]
[Google]
[Amazon]
Melanoma, also redundantly known as malignant melanoma, is a type of

 The earliest stage of melanoma starts when melanocytes begin out-of-control growth. Melanocytes are found between the outer layer of the skin (the epidermis) and the next layer (the dermis). This early stage of the disease is called the radial growth phase, when the tumor is less than 1 mm thick, and spreads at the level of the basal epidermis. Because the cancer cells have not yet reached the blood vessels deeper in the skin, it is very unlikely that this early-stage melanoma will spread to other parts of the body. If the melanoma is detected at this stage, then it can usually be completely removed with surgery.
When the tumor cells start to move in a different direction – vertically up into the epidermis and into the
The earliest stage of melanoma starts when melanocytes begin out-of-control growth. Melanocytes are found between the outer layer of the skin (the epidermis) and the next layer (the dermis). This early stage of the disease is called the radial growth phase, when the tumor is less than 1 mm thick, and spreads at the level of the basal epidermis. Because the cancer cells have not yet reached the blood vessels deeper in the skin, it is very unlikely that this early-stage melanoma will spread to other parts of the body. If the melanoma is detected at this stage, then it can usually be completely removed with surgery.
When the tumor cells start to move in a different direction – vertically up into the epidermis and into the
 Looking at the area in question is the most common method of suspecting a melanoma. Moles that are irregular in color or shape are typically treated as candidates. To detect melanomas (and increase survival rates), it is recommended to learn to recognize them (see "ABCDE" mnemonic), to regularly examine
Looking at the area in question is the most common method of suspecting a melanoma. Moles that are irregular in color or shape are typically treated as candidates. To detect melanomas (and increase survival rates), it is recommended to learn to recognize them (see "ABCDE" mnemonic), to regularly examine
File:Malignant melanoma (1) at thigh Case 01.jpg, Melanoma in skin biopsy with
 * Mucosal melanoma
* Mucosal melanoma
; When melanoma occurs on mucous membranes. * Desmoplastic melanoma * Melanoma with small nevus-like cells * Melanoma with features of a Spitz nevus * Uveal melanoma * Vaginal melanoma * Polypoid melanoma, a subclass of nodular melanoma.
 Metastatic melanomas can be detected by X-rays, CT scans, MRIs, PET and PET/CTs, ultrasound, LDH testing and photoacoustic detection. However, there is lack of evidence in the accuracy of staging of people with melanoma with various imaging methods.
Melanoma stages according to
Metastatic melanomas can be detected by X-rays, CT scans, MRIs, PET and PET/CTs, ultrasound, LDH testing and photoacoustic detection. However, there is lack of evidence in the accuracy of staging of people with melanoma with various imaging methods.
Melanoma stages according to
Amin MB, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. New York: Springer International Publishing; 2017:563‐585). * TX: Primary tumor thickness cannot be assessed (such as a diagnosis by curettage) * T0: No evidence of primary tumor (such as unknown primary or completely regressed melanoma) Stage 1 and 2 require an N (lymph node) class of: :*N0 – No regional metastases. Stage 1, 2 and 3 require an M (metastasis status) of: * M0: No evidence of distant metastasis Older systems include " Clark level" and "
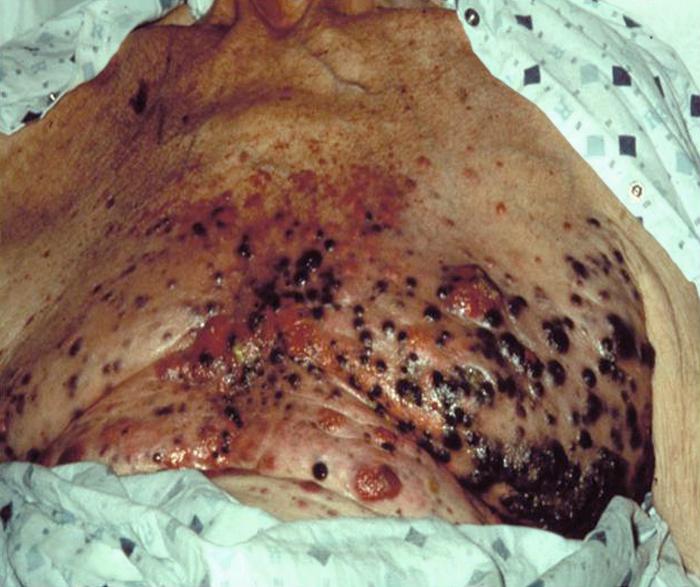 Confirmation of the clinical diagnosis is done with a
Confirmation of the clinical diagnosis is done with a

 Factors that affect prognosis include:
*
Factors that affect prognosis include:
*

 Globally, in 2012, melanoma occurred in 232,000 people and resulted in 55,000 deaths. Australia and New Zealand have the highest rates of melanoma in the world. It has become more common in the last 20 years in areas that are mostly
Globally, in 2012, melanoma occurred in 232,000 people and resulted in 55,000 deaths. Australia and New Zealand have the highest rates of melanoma in the world. It has become more common in the last 20 years in areas that are mostly
skin cancer
Skin cancers are cancers that arise from the skin. They are due to the development of abnormal cells that have the ability to invade or spread to other parts of the body. There are three main types of skin cancers: basal-cell skin cancer (BCC) ...
that develops from the pigment
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compo ...
-producing cells known as melanocytes. Melanomas typically occur in the skin, but may rarely occur in the mouth, intestines, or eye (uveal melanoma
Uveal melanoma is a type of eye cancer in the uvea of the eye. It is traditionally classed as originating in the iris, choroid, and ciliary body, but can also be divided into class I (low metastatic risk) and class II (high metastatic risk). Sym ...
). In women, they most commonly occur on the legs, while in men, they most commonly occur on the back. About 25% of melanomas develop from moles Moles can refer to:
* Moles de Xert, a mountain range in the Baix Maestrat comarca, Valencian Community, Spain
* The Moles (Australian band)
*The Moles, alter ego of Scottish band Simon Dupree and the Big Sound
People
*Abraham Moles, French engin ...
. Changes in a mole that can indicate melanoma include an increase in size, irregular edges, change in color, itchiness, or skin breakdown
Pressure ulcers, also known as pressure sores, bed sores or pressure injuries, are localised damage to the skin and/or underlying tissue that usually occur over a bony prominence as a result of usually long-term pressure, or pressure in combi ...
.
The primary cause of melanoma is ultraviolet light
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nanometer, nm (with a corresponding frequency around 30 Hertz, PHz) to 400 nm (750 Hertz, THz), shorter than that of visible light, but longer than ...
(UV) exposure in those with low levels of the skin pigment melanin
Melanin (; from el, μέλας, melas, black, dark) is a broad term for a group of natural pigments found in most organisms. Eumelanin is produced through a multistage chemical process known as melanogenesis, where the oxidation of the amino ...
. The UV light may be from the sun or other sources, such as tanning devices. Those with many moles, a history of affected family members, and poor immune function
Immunosuppression is a reduction of the activation or efficacy of the immune system. Some portions of the immune system itself have immunosuppressive effects on other parts of the immune system, and immunosuppression may occur as an adverse reacti ...
are at greater risk. A number of rare genetic conditions
A genetic disorder is a health problem caused by one or more abnormalities in the genome. It can be caused by a mutation in a single gene (monogenic) or multiple genes (polygenic) or by a chromosomal abnormality. Although polygenic disorders ...
, such as xeroderma pigmentosum
Xeroderma pigmentosum (XP) is a genetic disorder in which there is a decreased ability to repair DNA damage such as that caused by ultraviolet (UV) light. Symptoms may include a severe sunburn after only a few minutes in the sun, freckling in su ...
, also increase the risk. Diagnosis is by biopsy
A biopsy is a medical test commonly performed by a surgeon, interventional radiologist, or an interventional cardiologist. The process involves extraction of sample cells or tissues for examination to determine the presence or extent of a dise ...
and analysis of any skin lesion that has signs of being potentially cancerous.
Using sunscreen and avoiding UV light may prevent melanoma. Treatment is typically removal by surgery. In those with slightly larger cancers, nearby lymph nodes
A lymph node, or lymph gland, is a kidney-shaped organ of the lymphatic system and the adaptive immune system. A large number of lymph nodes are linked throughout the body by the lymphatic vessels. They are major sites of lymphocytes that inclu ...
may be tested for spread (metastasis
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
). Most people are cured if spread has not occurred. For those in whom melanoma has spread, immunotherapy
Immunotherapy or biological therapy is the treatment of disease by activating or suppressing the immune system. Immunotherapies designed to elicit or amplify an immune response are classified as ''activation immunotherapies,'' while immunotherap ...
, biologic therapy
A biopharmaceutical, also known as a biological medical product, or biologic, is any pharmaceutical drug product manufactured in, extracted from, or semisynthesized from biological sources. Different from totally synthesized pharmaceuticals, t ...
, radiation therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Radi ...
, or chemotherapy
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of cancer treatment that uses one or more anti-cancer drugs ( chemotherapeutic agents or alkylating agents) as part of a standardized chemotherapy regimen. Chemothe ...
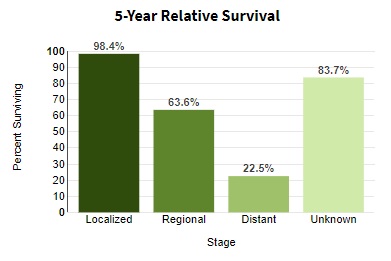
may improve survival. With treatment, the five-year survival rates
The five-year survival rate is a type of survival rate for estimating the prognosis of a particular disease, normally calculated from the point of diagnosis. Lead time bias from earlier diagnosis can affect interpretation of the five-year surviva ...
in the United States are 99% among those with localized disease, 65% when the disease has spread to lymph nodes, and 25% among those with distant spread. The likelihood that melanoma will reoccur or spread depends on its thickness
Thickness may refer to:
* Thickness (graph theory)
* Thickness (geology), the distance across a layer of rock
* Thickness (meteorology), the difference in height between two atmospheric pressure levels
* Thickness planer a woodworking machine
...
, how fast the cells are dividing, and whether or not the overlying skin has broken down.
Melanoma is the most dangerous type of skin cancer. Globally, in 2012, it newly occurred in 232,000 people. In 2015, 3.1 million people had active disease, which resulted in 59,800 deaths. Australia and New Zealand have the highest rates of melanoma in the world. High rates also occur in Northern Europe and North America, while it is less common in Asia, Africa, and Latin America. In the United States, melanoma occurs about 1.6 times more often in men than women. Melanoma has become more common since the 1960s in areas mostly populated by people of European descent
European emigration is the successive emigration waves from the European continent to other continents. The origins of the various European diasporas can be traced to the people who left the European nation states or stateless ethnic communities ...
.
Signs and symptoms
Early signs of melanoma are changes to the shape or color of existingmoles Moles can refer to:
* Moles de Xert, a mountain range in the Baix Maestrat comarca, Valencian Community, Spain
* The Moles (Australian band)
*The Moles, alter ego of Scottish band Simon Dupree and the Big Sound
People
*Abraham Moles, French engin ...
or, in the case of nodular melanoma
Nodular melanoma (NM) is the most aggressive form of melanoma. It tends to grow more rapidly in thickness (vertically penetrate the skin) than in diameter compared to other melanoma subtypes. Instead of arising from a pre-existing mole, it may app ...
, the appearance of a new lump anywhere on the skin. At later stages, the mole may itch
Itch (also known as pruritus) is a sensation that causes the desire or reflex to scratch. Itch has resisted many attempts to be classified as any one type of sensory experience. Itch has many similarities to pain, and while both are unpleasant ...
, ulcerate
Ulcerate is a New Zealand-based extreme metal band formed by guitarist Michael Hoggard and drummer Jamie Saint Merat in 2000. The band have released six studio albums to date. The band have been featured in numerous articles as one of New Zea ...
, or bleed. Early signs of melanoma are summarized by the mnemonic "ABCDEF":
* Asymmetry
* Borders (irregular with edges and corners)
* Colour (variegated
Variegation is the appearance of differently coloured zones in the leaves and sometimes the stems and fruit of plants. Species with variegated individuals are sometimes found in the understory of tropical rainforests, and this habitat is the s ...
)
* Diameter (greater than , about the size of a pencil eraser)
* Evolving over time
This classification does not apply to nodular melanoma, which has its own classifications:
* Elevated above the skin surface
* Firm to the touch
* Growing
Metastatic melanoma may cause nonspecific paraneoplastic symptoms, including loss of appetite, nausea
Nausea is a diffuse sensation of unease and discomfort, sometimes perceived as an urge to vomit. While not painful, it can be a debilitating symptom if prolonged and has been described as placing discomfort on the chest, abdomen, or back of the ...
, vomiting, and fatigue. Metastasis (spread) of early melanoma is possible, but relatively rare; less than a fifth of melanomas diagnosed early become metastatic. Brain metastases
A brain metastasis is a cancer that has metastasized (spread) to the brain from another location in the body and is therefore considered a secondary brain tumor. The metastasis typically shares a cancer cell type with the original site of the ...
are particularly common in patients with metastatic melanoma. It can also spread to the liver, bones, abdomen, or distant lymph nodes.
Cause
Melanomas are usually caused by DNA damage resulting from exposure to UV light from the sun.Genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinian friar wor ...
also play a role. Melanoma can also occur in skin areas with little sun exposure (i.e. mouth, soles of feet, palms of hands, genital areas). People with dysplastic nevus syndrome
Dysplastic nevus syndrome, also known as familial atypical multiple mole–melanoma (FAMMM) syndrome, is an inherited cutaneous condition described in certain families, and characterized by unusual nevi and multiple inherited melanomas. First des ...
, also known as familial atypical multiple mole melanoma, are at increased risk for the development of melanoma.
Having more than 50 moles indicates an increased risk melanoma might arise. A weakened immune system makes cancer development easier due to the body's weakened ability to fight cancer cells.
UV radiation
The UV radiation from tanning beds increases the risk of melanoma. TheInternational Agency for Research on Cancer
The International Agency for Research on Cancer (IARC; french: Centre International de Recherche sur le Cancer, CIRC) is an intergovernmental agency forming part of the World Health Organization of the United Nations.
Its role is to conduct and ...
finds that tanning beds are "carcinogenic to humans" and that people who begin using tanning devices before the age of thirty years are 75% more likely to develop melanoma.
Those who work in airplanes also appear to have an increased risk, believed to be due to greater exposure to UV.
UVB light (wavelengths between 315 and 280 nm) from the sun is absorbed by skin cell DNA and results in a type of direct DNA damage
Direct may refer to:
Mathematics
* Directed set, in order theory
* Direct limit of (pre), sheaves
* Direct sum of modules, a construction in abstract algebra which combines several vector spaces
Computing
* Direct access (disambiguation), ...
called cyclobutane pyrimidine dimers. Thymine
Thymine () ( symbol T or Thy) is one of the four nucleobases in the nucleic acid of DNA that are represented by the letters G–C–A–T. The others are adenine, guanine, and cytosine. Thymine is also known as 5-methyluracil, a pyrimidi ...
, cytosine
Cytosine () ( symbol C or Cyt) is one of the four nucleobases found in DNA and RNA, along with adenine, guanine, and thymine (uracil in RNA). It is a pyrimidine derivative, with a heterocyclic aromatic ring and two substituents attached (an ...
, or cytosine-thymine dimers are formed by the joining of two adjacent pyrimidine bases within a DNA strand. Somewhat similarly to UVB
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation i ...
, UVA light (longer wavelengths between 400 and 315 nm) from the sun or from tanning beds can also be directly absorbed by skin DNA (at about 1/100 to 1/1000 of the efficiency of UVB).
Exposure to radiation (UVA and UVB) is one of the major contributors to the development of melanoma. Occasional extreme sun exposure (resulting in "sunburn
Sunburn is a form of radiation burn that affects living tissue, such as skin, that results from an overexposure to ultraviolet (UV) radiation, usually from the Sun. Common symptoms in humans and animals include: red or reddish skin that is ho ...
") is causally related to melanoma. Melanoma is most common on the back in men and on legs in women (areas of intermittent sun exposure). The risk appears to be strongly influenced by socioeconomic conditions rather than indoor versus outdoor occupations; it is more common in professional and administrative workers than unskilled workers. Other factors are mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA replication, DNA or viral repl ...
s in or total loss of tumor suppressor gene
A tumor suppressor gene (TSG), or anti-oncogene, is a gene that regulates a cell during cell division and replication. If the cell grows uncontrollably, it will result in cancer. When a tumor suppressor gene is mutated, it results in a loss or re ...
s. Use of sunbeds (with deeply penetrating UVA rays) has been linked to the development of skin cancers, including melanoma.
Possible significant elements in determining risk include the intensity and duration of sun exposure, the age at which sun exposure occurs, and the degree of skin pigmentation
Human skin color ranges from the darkest brown to the lightest hues. Differences in skin color among individuals is caused by variation in pigmentation, which is the result of genetics (inherited from one's biological parents and or indiv ...
. Melanoma rates tend to be highest in countries settled by migrants from Northern Europe which have a large amount of direct, intense sunlight to which the skin of the settlers is not adapted, most notably Australia. Exposure during childhood is a more important risk factor than exposure in adulthood. This is seen in migration studies in Australia.
Having multiple severe sunburns increases the likelihood that future sunburns develop into melanoma due to cumulative damage. The sun and tanning beds are the main sources of UV radiation that increase the risk for melanoma and living close to the equator increases exposure to UV radiation.
Genetics
A number of rare mutations, which often run in families, greatly increase melanoma susceptibility. Severalgene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
s increase risks. Some rare genes have a relatively high risk of causing melanoma; some more common genes, such as a gene called ''MC1R'' that causes red hair, have a relatively lower elevated risk. Genetic testing
Genetic testing, also known as DNA testing, is used to identify changes in DNA sequence or chromosome structure. Genetic testing can also include measuring the results of genetic changes, such as RNA analysis as an output of gene expression, or ...
can be used to search for the mutations.
One class of mutations affects the gene ''CDKN2A''. An alternative reading frame
In molecular biology, a reading frame is a way of dividing the sequence of nucleotides in a nucleic acid ( DNA or RNA) molecule into a set of consecutive, non-overlapping triplets. Where these triplets equate to amino acids or stop signals during ...
mutation in this gene leads to the destabilization of p53
p53, also known as Tumor protein P53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory protein that is often mutated in human cancers. The p53 proteins (originally thought to be, and often s ...
, a transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The fu ...
involved in apoptosis
Apoptosis (from grc, ἀπόπτωσις, apóptōsis, 'falling off') is a form of programmed cell death that occurs in multicellular organisms. Biochemical events lead to characteristic cell changes (morphology) and death. These changes incl ...
and in 50% of human cancers. Another mutation in the same gene results in a nonfunctional inhibitor of CDK4, a cyclin-dependent kinase that promotes cell division
Cell division is the process by which a parent cell divides into two daughter cells. Cell division usually occurs as part of a larger cell cycle in which the cell grows and replicates its chromosome(s) before dividing. In eukaryotes, there ar ...
. Mutations that cause the skin condition xeroderma pigmentosum
Xeroderma pigmentosum (XP) is a genetic disorder in which there is a decreased ability to repair DNA damage such as that caused by ultraviolet (UV) light. Symptoms may include a severe sunburn after only a few minutes in the sun, freckling in su ...
(XP) also increase melanoma susceptibility. Scattered throughout the genome, these mutations reduce a cell's ability to repair DNA. Both CDKN2A and XP mutations are highly penetrant (the chances of a carrier to express the phenotype is high).
Familial melanoma is genetically heterogeneous, and loci for familial melanoma appear on the chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
arms 1p, 9p and 12q. Multiple genetic events have been related to melanoma's pathogenesis
Pathogenesis is the process by which a disease or disorder develops. It can include factors which contribute not only to the onset of the disease or disorder, but also to its progression and maintenance. The word comes from Greek πάθος ''pat ...
(disease development). The multiple tumor suppressor
A tumor suppressor gene (TSG), or anti-oncogene, is a gene that regulates a cell during cell division and replication. If the cell grows uncontrollably, it will result in cancer. When a tumor suppressor gene is mutated, it results in a loss or re ...
1 (CDKN2A/MTS1) gene encodes p16INK4a – a low-molecular weight
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
protein inhibitor of cyclin-dependent protein kinases (CDKs) – which has been localised to the p21 region of human chromosome 9. FAMMM is typically characterized by having 50 or more combined moles in addition to a family history of melanoma. It is transmitted autosomal dominantly and mostly associated with the ''CDKN2A'' mutations. People who have CDKN2A mutation associated FAMMM have a 38 fold increased risk of pancreatic cancer.
Other mutations confer lower risk, but are more common in the population. People with mutations in the ''MC1R
The melanocortin 1 receptor (MC1R), also known as melanocyte-stimulating hormone receptor (MSHR), melanin-activating peptide receptor, or melanotropin receptor, is a G protein–coupled receptor that binds to a class of pituitary peptide hormones ...
'' gene are two to four times more likely to develop melanoma than those with two wild-type (typical unaffected type) copies. ''MC1R'' mutations are very common, and all red-haired people have a mutated copy. Mutation of the '' MDM2'' SNP309 gene is associated with increased risks for younger women.
Fair- and red-haired people, persons with multiple atypical nevi
Nevus (plural nevi) is a nonspecific medical term for a visible, circumscribed, chronic lesion of the skin or mucosa. The term originates from ''nævus'', which is Latin for "birthmark"; however, a nevus can be either congenital (present at bir ...
or dysplastic nevi
A dysplastic nevus or atypical mole is a nevus (mole) whose appearance is different from that of common moles. In 1992, the NIH recommended that the term "dysplastic nevus" be avoided in favor of the term "atypical mole". An atypical mole may also ...
and persons born with giant congenital melanocytic nevi
The congenital melanocytic nevus is a type of melanocytic nevus (or mole) found in infants at birth. This type of birthmark occurs in an estimated 1% of infants worldwide; it is located in the area of the head and neck 15% of the time.
Signs an ...
are at increased risk.
A family history of melanoma greatly increases a person's risk, because mutations in several genes have been found in melanoma-prone families. People with a history of one melanoma are at increased risk of developing a second primary tumor.
Fair skin is the result of having less melanin in the skin, which means less protection from UV radiation exists. A family history could indicate a genetic predisposition to melanoma.
Pathophysiology
 The earliest stage of melanoma starts when melanocytes begin out-of-control growth. Melanocytes are found between the outer layer of the skin (the epidermis) and the next layer (the dermis). This early stage of the disease is called the radial growth phase, when the tumor is less than 1 mm thick, and spreads at the level of the basal epidermis. Because the cancer cells have not yet reached the blood vessels deeper in the skin, it is very unlikely that this early-stage melanoma will spread to other parts of the body. If the melanoma is detected at this stage, then it can usually be completely removed with surgery.
When the tumor cells start to move in a different direction – vertically up into the epidermis and into the
The earliest stage of melanoma starts when melanocytes begin out-of-control growth. Melanocytes are found between the outer layer of the skin (the epidermis) and the next layer (the dermis). This early stage of the disease is called the radial growth phase, when the tumor is less than 1 mm thick, and spreads at the level of the basal epidermis. Because the cancer cells have not yet reached the blood vessels deeper in the skin, it is very unlikely that this early-stage melanoma will spread to other parts of the body. If the melanoma is detected at this stage, then it can usually be completely removed with surgery.
When the tumor cells start to move in a different direction – vertically up into the epidermis and into the papillary dermis
The dermis or corium is a layer of skin between the epidermis (with which it makes up the cutis) and subcutaneous tissues, that primarily consists of dense irregular connective tissue and cushions the body from stress and strain. It is divided in ...
– cell behaviour changes dramatically.
The next step in the evolution is the invasive radial growth phase, in which individual cells start to acquire invasive potential. From this point on, melanoma is capable of spreading. The Breslow's depth
In medicine, Breslow's depth was used as a prognostic factor in melanoma of the skin. It is a description of how deeply tumor cells have invaded. Currently, the standard Breslow's depth has been replaced by the AJCC depth, in the AJCC staging sys ...
of the lesion is usually less than , while the Clark level is usually 2.
The vertical growth phase (VGP) following is invasive melanoma. The tumor becomes able to grow into the surrounding tissue and can spread around the body through blood or lymph vessels. The tumor thickness is usually more than , and the tumor involves the deeper parts of the dermis.
The host elicits an immunological reaction against the tumor during the VGP, which is judged by the presence and activity of the tumor infiltrating lymphocyte
Tumor-infiltrating lymphocytes (TIL) are white blood cells that have left the bloodstream and migrated towards a tumor. They include T cells and B cells and are part of the larger category of ‘tumor-infiltrating immune cells’ which consis ...
s (TILs). These cells sometimes completely destroy the primary tumor; this is called regression, which is the latest stage of development. In certain cases, the primary tumor is completely destroyed and only the metastatic tumor is discovered. About 40% of human melanomas contain activating mutations affecting the structure of the B-Raf protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respo ...
, resulting in constitutive signaling through the Raf to MAP kinase
A mitogen-activated protein kinase (MAPK or MAP kinase) is a type of protein kinase that is specific to the amino acids serine and threonine (i.e., a serine/threonine-specific protein kinase). MAPKs are involved in directing cellular responses ...
pathway.
An insult common to most cancers is damage to DNA. UVA light mainly causes thymine dimer
Pyrimidine dimers are molecular lesions formed from thymine or cytosine bases in DNA via photochemical reactions, commonly associated with direct DNA damage. Ultraviolet light (UV; particularly UVB) induces the formation of covalent linkages bet ...
s. UVA also produces reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
and these inflict other DNA damage, primarily single-strand breaks, oxidized pyrimidines
Pyrimidine (; ) is an aromatic, heterocyclic, organic compound similar to pyridine (). One of the three diazines (six-membered heterocyclics with two nitrogen atoms in the ring), it has nitrogen atoms at positions 1 and 3 in the ring. The other ...
and the oxidized purine
Purine is a heterocyclic aromatic organic compound that consists of two rings ( pyrimidine and imidazole) fused together. It is water-soluble. Purine also gives its name to the wider class of molecules, purines, which include substituted purines ...
8-oxoguanine
8-Oxoguanine (8-hydroxyguanine, 8-oxo-Gua, or OH8Gua) is one of the most common DNA lesions resulting from reactive oxygen species modifying guanine, and can result in a mismatched pairing with adenine resulting in G to T and C to A substitutions ...
(a mutagenic DNA change) at 1/10, 1/10, and 1/3rd the frequencies of UVA-induced thymine dimers, respectively.
If unrepaired, CPD photoproducts can lead to mutations by inaccurate translesion synthesis
DNA repair is a collection of processes by which a cell identifies and corrects damage to the DNA molecules that encode its genome. In human cells, both normal metabolic activities and environmental factors such as radiation can cause DNA dama ...
during DNA replication or repair. The most frequent mutations due to inaccurate synthesis past CPDs are cytosine to thymine (C>T) or CC>TT transition mutations. These are commonly referred to as UV fingerprint mutations, as they are the most specific mutation caused by UV, being frequently found in sun-exposed skin, but rarely found in internal organs. Errors in DNA repair of UV photoproducts, or inaccurate synthesis past these photoproducts, can also lead to deletions, insertions, and chromosomal translocation
In genetics, chromosome translocation is a phenomenon that results in unusual rearrangement of chromosomes. This includes balanced and unbalanced translocation, with two main types: reciprocal-, and Robertsonian translocation. Reciprocal translo ...
s.
The entire genomes of 25 melanomas were sequenced. On average, about 80,000 mutated bases (mostly C>T transitions) and about 100 structural rearrangements were found per melanoma genome. This is much higher than the roughly 70 mutations across generations (parent to child). Among the 25 melanomas, about 6,000 protein-coding genes had missense
In genetics, a missense mutation is a point mutation in which a single nucleotide change results in a codon that codes for a different amino acid. It is a type of nonsynonymous substitution.
Substitution of protein from DNA mutations
Missense mu ...
, nonsense
Nonsense is a communication, via speech, writing, or any other symbolic system, that lacks any coherent meaning. Sometimes in ordinary usage, nonsense is synonymous with absurdity or the ridiculous. Many poets, novelists and songwriters have u ...
, or splice site mutations. The transcriptomes of over 100 melanomas has also been sequenced and analyzed. Almost 70% of all human protein-coding genes are expressed in melanoma. Most of these genes are also expressed in other normal and cancer tissues, with some 200 genes showing a more specific expression pattern in melanoma compared to other forms of cancer. Examples of melanoma specific genes are tyrosinase
Tyrosinase is an oxidase that is the rate-limiting enzyme for controlling the production of melanin. The enzyme is mainly involved in two distinct reactions of melanin synthesis otherwise known as the Raper Mason pathway. Firstly, the hydroxy ...
, MLANA
Protein melan-A also known as melanoma antigen recognized by T cells 1 or MART-1 is a protein that in humans is encoded by the ''MLANA'' or "MALENA" gene. A fragment of the protein, usually consisting of the nine amino acids 27 to 35, is bound by ...
, and ''PMEL''.
UV radiation causes damage
Damage is any change in a thing, often a physical object, that degrades it away from its initial state. It can broadly be defined as "changes introduced into a system that adversely affect its current or future performance".Farrar, C.R., Sohn, H., ...
to the DNA of cells, typically thymine dimerization, which when unrepaired can create mutations in the cell's genes. This strong mutagenic factor makes cutaneous melanoma the tumor type with the highest number of mutations. When the cell divides
In mathematics, a divisor of an integer n, also called a factor of n, is an integer m that may be multiplied by some integer to produce n. In this case, one also says that n is a multiple of m. An integer n is divisible or evenly divisible by ...
, these mutations are propagated to new generations of cells. If the mutations occur in protooncogene
An oncogene is a gene that has the potential to cause cancer. In tumor cells, these genes are often mutated, or expressed at high levels.
s or tumor suppressor gene
A tumor suppressor gene (TSG), or anti-oncogene, is a gene that regulates a cell during cell division and replication. If the cell grows uncontrollably, it will result in cancer. When a tumor suppressor gene is mutated, it results in a loss or re ...
s, the rate of mitosis in the mutation-bearing cells can become uncontrolled, leading to the formation of a tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
. Data from patients suggest that aberrant levels of activating transcription factor in the nucleus of melanoma cells are associated with increased metastatic activity of melanoma cells; studies from mice on skin cancer tend to confirm a role for activating transcription factor-2 in cancer progression.
Cancer stem cells may also be involved.
Gene mutations
Large-scale studies, such asThe Cancer Genome Atlas
''The'' () is a grammatical article in English, denoting persons or things already mentioned, under discussion, implied or otherwise presumed familiar to listeners, readers, or speakers. It is the definite article in English. ''The'' is the m ...
, have characterized recurrent somatic alterations likely driving initiation and development of cutaneous melanoma. The Cancer Genome Atlas study has established four subtypes : ''BRAF'' mutant, ''RAS'' mutant, ''NF1'' mutant, and triple wild-type.
The most frequent mutation occurs in the 600th codon of ''BRAF'' (50% of cases). ''BRAF'' is normally involved in cell growth, and this specific mutation renders the protein constitutively active and independent of normal physiological regulation, thus fostering tumor growth. RAS genes ( NRAS, HRAS
GTPase HRas, from "Harvey Rat sarcoma virus", also known as transforming protein p21 is an enzyme that in humans is encoded by the gene. The ''HRAS'' gene is located on the short (p) arm of chromosome 11 at position 15.5, from base pair 522,241 ...
and KRAS
''KRAS'' (Kirsten rat sarcoma virus) is a gene that provides instructions for making a protein called K-Ras, a part of the RAS/MAPK pathway. The protein relays signals from outside the cell to the cell's nucleus. These signals instruct the cell ...
) are also recurrently mutated (30% of TCGA cases) and mutations in the 61st or 12th codons trigger oncogenic activity. Loss-of-function mutations often affect tumor suppressor genes
A tumor suppressor gene (TSG), or anti-oncogene, is a gene that regulates a cell during cell division and replication. If the cell grows uncontrollably, it will result in cancer. When a tumor suppressor gene is mutated, it results in a loss or red ...
such as NF1, TP53
p53, also known as Tumor protein P53, cellular tumor antigen p53 (UniProt name), or transformation-related protein 53 (TRP53) is a regulatory protein that is often mutated in human cancers. The p53 proteins (originally thought to be, and often ...
and CDKN2A
CDKN2A, also known as cyclin-dependent kinase inhibitor 2A, is a gene which in humans is located at chromosome 9, band p21.3. It is ubiquitously expressed in many tissues and cell types. The gene codes for two proteins, including the INK4 family ...
. Other oncogenic alterations include fusions involving various kinases such as BRAF, RAF1, ALK, RET, ROS1, NTRK1., NTRK3 and MET'' BRAF, RAS'', and ''NF1'' mutations and kinase fusions are remarkably mutually exclusive, as they occur in different subsets of patients. Assessment of mutation status can, therefore, improve patient stratification and inform targeted therapy with specific inhibitors.
In some cases (3-7%) mutated versions of ''BRAF'' and ''NRAS'' undergo copy-number amplification.
Metastasis
The research done by Sarna's team proved that heavily pigmented melanoma cells haveYoung's modulus
Young's modulus E, the Young modulus, or the modulus of elasticity in tension or compression (i.e., negative tension), is a mechanical property that measures the tensile or compressive stiffness of a solid material when the force is applied le ...
about 4.93, when in non-pigmented ones it was only 0.98. In another experiment they found that elasticity of melanoma cells is important for its metastasis and growth: non-pigmented tumors were bigger than pigmented and it was much easier for them to spread. They shown that there are both pigmented and non-pigmented cells in melanoma tumors
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
, so that they can both be drug-resistant and metastatic.
Diagnosis
 Looking at the area in question is the most common method of suspecting a melanoma. Moles that are irregular in color or shape are typically treated as candidates. To detect melanomas (and increase survival rates), it is recommended to learn to recognize them (see "ABCDE" mnemonic), to regularly examine
Looking at the area in question is the most common method of suspecting a melanoma. Moles that are irregular in color or shape are typically treated as candidates. To detect melanomas (and increase survival rates), it is recommended to learn to recognize them (see "ABCDE" mnemonic), to regularly examine moles Moles can refer to:
* Moles de Xert, a mountain range in the Baix Maestrat comarca, Valencian Community, Spain
* The Moles (Australian band)
*The Moles, alter ego of Scottish band Simon Dupree and the Big Sound
People
*Abraham Moles, French engin ...
for changes (shape, size, color, itching or bleeding) and to consult a qualified physician when a candidate appears.
However, many melanomas present as lesions smaller than 6 mm in diameter, and all melanomas are malignant when they first appear as a small dot. Physicians typically examine all moles, including those less than 6 mm in diameter. Seborrheic keratosis
A seborrheic keratosis is a non-cancerous (benign) skin tumour that originates from cells, namely keratinocytes, in the outer layer of the skin called the epidermis. Like liver spots, seborrheic keratoses are seen more often as people age.
The tum ...
may meet some or all of the ABCD criteria, and can lead to false alarm
A false alarm, also called a nuisance alarm, is the deceptive or erroneous report of an emergency, causing unnecessary panic and/or bringing resources (such as emergency services) to a place where they are not needed. False alarms may occur with ...
s. Doctors can generally distinguish seborrheic keratosis from melanoma upon examination or with dermatoscopy
Dermatoscopy also known as dermoscopy or epiluminescence microscopy, is the examination of skin lesions with a dermatoscope. It is a tool similar to a camera to allow for inspection of skin lesions unobstructed by skin surface reflections. The d ...
.
Some advocate replacing "enlarging" with "evolving": moles that change and evolve are a concern. Alternatively, some practitioners prefer "elevation". Elevation can help identify a melanoma, but lack of elevation does not mean that the lesion is not a melanoma. Most melanomas in the US are detected before they become elevated. By the time elevation is visible, they may have progressed to the more dangerous invasive stage.
In-person inspection of suspicious skin lesions is more accurate than visual inspection of images of suspicious skin lesions. When used by trained specialists, dermoscopy is more helpful to identify malignant lesions than use of the naked eye alone. Reflectance confocal microscopy may have better sensitivity and specificity than dermoscopy in diagnosing cutaneous melanoma but more studies are needed to confirm this result.
H&E stain
Hematoxylin and eosin stain ( or haematoxylin and eosin stain or hematoxylin-eosin stain; often abbreviated as H&E stain or HE stain) is one of the principal tissue stains used in histology. It is the most widely used stain in medical diagnos ...
– this case may represent superficial spreading melanoma.
File:Lymph node with almost complete replacement by metastatic melanoma.jpg, Lymph node with almost complete replacement by metastatic melanoma. The brown pigment is a focal deposition of melanin.
File:Dermatoscope1.JPG, A dermatoscope
Dermatoscopy also known as dermoscopy or epiluminescence microscopy, is the examination of skin lesions with a dermatoscope. It is a tool similar to a camera to allow for inspection of skin lesions unobstructed by skin surface reflections. The d ...
File:Malignant Melanoma, right posterior thigh.png, Malignant Melanoma, right posterior thigh
File:Melanoma in situ, vertex scalp.jpg, Melanoma in situ, vertex scalp marked for biopsy
File:Malignant Melanoma in situ evolving Right clavicle.jpg, Malignant Melanoma in situ, evolving, right clavicle marked for biopsy
File:Malignant Melanoma, vertex scalp.jpg, Malignant Melanoma, vertex scalp marked for biopsy
File:Malignant Melanoma right medial thigh.jpg, Malignant Melanoma, right medial thigh marked for biopsy
File:Malignant Melanoma Right Posterior Shoulder.jpg, Malignant Melanoma, right posterior shoulder circled for biopsy
File:Malignant Melanoma Left Forearm.jpg, Malignant Melanoma, left forearm marked for biopsy
File:Malignant Melanoma Left Forearm post excision.jpg, Malignant Melanoma left forearm post excision with purse-string closure
File:Melanoma in situ Right Forehead.jpg, Melanoma in situ, right forehead marked for biopsy
File:Melanoma in situ Right Forehead dermatoscope.jpg, Melanoma in situ, dermatoscope image, right forehead marked for biopsy
File:Malignant Melanoma right temple medial adjacent sebaceous hyperplasia right temple lateral.jpg, Malignant Melanoma in situ, evolving, a medial right temple with adjacent sebaceous hyperplasia, lateral
File:Malignant Melanoma in situ Left Anterior Shoulder.jpg, Malignant Melanoma in situ, left anterior shoulder marked for biopsy
File:Malignant Melanoma in situ Right Anterior Shoulder.jpg, Malignant Melanoma in situ, right anterior shoulder marked for biopsy
File:Malignant Melanoma in situ Left Upper Inner Arm.jpg, Malignant Melanoma in situ, left upper inner arm
File:Malignant Melanoma in situ Left Forearm.jpg, Malignant Melanoma in situ marked for biopsy, left forearm
File:Malignant Melanoma right upper medial back.jpg, Malignant Melanoma in situ, right upper medial back, marked for biopsy
File:Malignant Melanoma Mid Frontal Scalp.jpg, Malignant Melanoma, mid frontal scalp
File:Malignant Melanoma Left Mid Back.jpg, Malignant melanoma, left mid-back marked for biopsy
File:Malignant Melanoma Left Mid Back Dermatoscope.jpg, Malignant melanoma, left mid-back marked for biopsy, through dermatoscope
File:Gross pathology of melanoma metastasis.jpg, Gross pathology of melanoma metastasis, which is pigment-forming in a vast majority of cases, giving it a dark appearance.
Ugly duckling
One method is the "ugly duckling
"The Ugly Duckling" ( da, Den grimme ælling) is a Danish literary fairy tale by Danish poet and author Hans Christian Andersen (1805–1875). It was first published on 11 November 1843 in '' New Fairy Tales. First Volume. First Collection'' ...
sign".
Correlation of common lesion characteristics is made. Lesions that deviate from the common characteristics are labeled an "ugly duckling", and a further professional exam is required. The "Little Red Riding Hood
"Little Red Riding Hood" is a European fairy tale about a young girl and a sly wolf. Its origins can be traced back to several pre-17th century European folk tales. The two best known versions were written by Charles Perrault and the Brot ...
" sign suggests that individuals with fair skin and light-colored hair might have difficult-to-diagnose amelanotic melanomas. Extra care is required when examining such individuals, as they might have multiple melanomas and severely dysplastic nevi. A dermatoscope must be used to detect "ugly ducklings", as many melanomas in these individuals resemble nonmelanomas or are considered to be " wolves in sheep's clothing". These fair-skinned individuals often have lightly pigmented or amelanotic melanomas that do not present easy-to-observe color changes and variations. Their borders are often indistinct, complicating visual identification without a dermatoscope.
Amelanotic melanomas and melanomas arising in fair-skinned individuals are very difficult to detect, as they fail to show many of the characteristics in the ABCD rule, break the "ugly duckling" sign, and are hard to distinguish from acne scarring, insect bites, dermatofibromas, or lentigines
A lentigo () (plural lentigines, ) is a small pigmented spot on the skin with a clearly defined edge, surrounded by normal-appearing skin. It is a harmless (benign) hyperplasia of melanocytes which is linear in its spread. This means the hyperplasi ...
.
Biopsy
Following a visual examination and a dermatoscopic exam, or ''in vivo
Studies that are ''in vivo'' (Latin for "within the living"; often not italicized in English) are those in which the effects of various biological entities are tested on whole, living organisms or cells, usually animals, including humans, and ...
'' diagnostic tools such as a confocal microscope, the doctor may biopsy
A biopsy is a medical test commonly performed by a surgeon, interventional radiologist, or an interventional cardiologist. The process involves extraction of sample cells or tissues for examination to determine the presence or extent of a dise ...
the suspicious mole. A skin biopsy
Skin biopsy is a biopsy technique in which a skin lesion is removed to be sent to a pathologist to render a microscopic diagnosis. It is usually done under local anesthetic in a physician's office, and results are often available in 4 to 10 days. ...
performed under local anesthesia
Local anesthesia is any technique to induce the absence of sensation in a specific part of the body, generally for the aim of inducing local analgesia, that is, local insensitivity to pain, although other local senses may be affected as well. It ...
is often required to assist in making or confirming the diagnosis and in defining severity. Elliptical excisional biopsies may remove the tumor, followed by histological
Histology,
also known as microscopic anatomy or microanatomy, is the branch of biology which studies the microscopic anatomy of biological tissues. Histology is the microscopic counterpart to gross anatomy, which looks at larger structures vis ...
analysis and Breslow scoring. Incisional biopsies such as punch biopsies are usually contraindicated in suspected melanomas, because of the possibility of sampling error or local implantation causing misestimation of tumour thickness. However, fears that such biopsies may increase the risk of metastatic disease seem unfounded.
Total body photography, which involves photographic documentation of as much body surface as possible, is often used during follow-up for high-risk patients. The technique has been reported to enable early detection and provides a cost-effective approach (with any digital camera), but its efficacy has been questioned due to its inability to detect macroscopic changes. The diagnosis method should be used in conjunction with (and not as a replacement for) dermoscopic imaging, with a combination of both methods appearing to give extremely high rates of detection.
Histopathologic types
Melanoma is a type of neuroectodermal neoplasm. There are four main types of melanoma: * Mucosal melanoma"> Page 805 in: Other histopathologic types are:; When melanoma occurs on mucous membranes. * Desmoplastic melanoma * Melanoma with small nevus-like cells * Melanoma with features of a Spitz nevus * Uveal melanoma * Vaginal melanoma * Polypoid melanoma, a subclass of nodular melanoma.
In situ or invasive
A melanoma ''in situ
''In situ'' (; often not italicized in English) is a Latin phrase that translates literally to "on site" or "in position." It can mean "locally", "on site", "on the premises", or "in place" to describe where an event takes place and is used in ...
'' has not invaded beyond the basement membrane, whereas an ''invasive melanoma'' has spread beyond it.
Some histopathological types of melanoma are inherently invasive, including nodular melanoma
Nodular melanoma (NM) is the most aggressive form of melanoma. It tends to grow more rapidly in thickness (vertically penetrate the skin) than in diameter compared to other melanoma subtypes. Instead of arising from a pre-existing mole, it may app ...
and lentigo maligna melanoma
Lentigo maligna melanoma is a melanoma that has evolved from a lentigo maligna, as seen as a lentigo maligna with melanoma cells invading below the boundaries of the epidermis. Last Update: May 18, 2019. They are usually found on chronically sun d ...
, where the ''in situ'' counterpart to lentigo maligna melanoma is lentigo maligna
Lentigo maligna is where melanocyte cells have become malignant and grow continuously along the stratum basale of the skin, but have not invaded below the epidermis. Lentigo maligna is not the same as lentigo maligna melanoma, as detailed below. ...
. Lentigo maligna is sometimes classified as a very early melanoma, and sometimes a precursor to melanoma.
Superficial spreading melanomas and acral lentiginous melanoma
Acral lentiginous melanoma is an aggressive type of skin cancer that is not caused by sunlight. Melanoma is a group of serious skin cancers that arise from pigment cells (melanocytes); acral lentiginous melanoma is a kind of lentiginous skin me ...
s can be either ''in situ'' or invasive, but acral lentiginous melanomas are almost always invasive.
Staging
''Further context oncancer staging
Cancer staging is the process of determining the extent to which a cancer has developed by growing and spreading. Contemporary practice is to assign a number from I to IV to a cancer, with I being an isolated cancer and IV being a cancer that ha ...
is available at TNM.''
AJCC {{Short description, Organization standardising cancer staging
The American Joint Committee on Cancer (AJCC) is an organization best known for defining and popularizing cancer staging standards, officially the AJCC staging system.
The American Joi ...
, 8th edition:, citingAmin MB, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. New York: Springer International Publishing; 2017:563‐585). * TX: Primary tumor thickness cannot be assessed (such as a diagnosis by curettage) * T0: No evidence of primary tumor (such as unknown primary or completely regressed melanoma) Stage 1 and 2 require an N (lymph node) class of: :*N0 – No regional metastases. Stage 1, 2 and 3 require an M (metastasis status) of: * M0: No evidence of distant metastasis Older systems include " Clark level" and "
Breslow's depth
In medicine, Breslow's depth was used as a prognostic factor in melanoma of the skin. It is a description of how deeply tumor cells have invaded. Currently, the standard Breslow's depth has been replaced by the AJCC depth, in the AJCC staging sys ...
", quantifying microscopic depth of tumor invasion.

Laboratory
Lactate dehydrogenase
Lactate dehydrogenase (LDH or LD) is an enzyme found in nearly all living cells. LDH catalyzes the conversion of lactate to pyruvate and back, as it converts NAD+ to NADH and back. A dehydrogenase is an enzyme that transfers a hydride from on ...
(LDH) tests are often used to screen for metastases
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
, although many patients with metastases (even end-stage) have a normal LDH; extraordinarily high LDH often indicates the metastatic spread of the disease to the liver.
It is common for patients diagnosed with melanoma to have chest X-rays and an LDH test, and in some cases CT, MRI
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to form pictures of the anatomy and the physiological processes of the body. MRI scanners use strong magnetic fields, magnetic field gradients, and radio waves ...
, and/or PET
A pet, or companion animal, is an animal kept primarily for a person's company or entertainment rather than as a working animal, livestock, or a laboratory animal. Popular pets are often considered to have attractive appearances, intelligence ...
scans. Although controversial, sentinel lymph node
The sentinel lymph node is the hypothetical first lymph node or group of nodes draining a cancer. In case of established cancerous dissemination it is postulated that the sentinel lymph nodes are the target organs primarily reached by metastasiz ...
biopsies and examination of the lymph nodes are also performed in patients to assess spread to the lymph nodes. A diagnosis of melanoma is supported by the presence of the S-100 protein
The S100 proteins are a family of low molecular-weight proteins found in vertebrates characterized by two calcium-binding sites that have helix-loop-helix ("EF-hand-type") conformation. At least 21 different S100 proteins are known. They are en ...
marker.
HMB-45
HMB-45 is a monoclonal antibody that reacts against an antigen present in melanocyte, melanocytic tumors such as melanomas, and stands for Human Melanoma Black. It is used in anatomic pathology as a marker for such tumors. The specific antigen reco ...
is a monoclonal antibody that reacts against an antigen present in melanocytic tumors such as melanomas. It is used in anatomic pathology as a marker for such tumors. The antibody was generated to an extract of melanoma. It reacts positively against melanocytic tumors but not other tumors, thus demonstrating specificity and sensitivity. The antibody also reacts positively against junctional nevus cells but not intradermal nevi, and against fetal melanocytes but not normal adult melanocytes.
HMB-45 is nonreactive with almost all non-melanoma human malignancies, with the exception of rare tumors showing evidence of melanogenesis (e.g., pigmented schwannoma, clear cell sarcoma) or tumors associated with tuberous sclerosis complex (angiomyolipoma and lymphangiomyoma).
Prevention
There is no evidence to support or refute adult population screening for malignant melanoma.Ultraviolet radiation
Minimizing exposure to sources of ultraviolet radiation (the sun and sunbeds), following sun protection measures and wearingsun protective clothing
Sun protective clothing is clothing specifically designed for sun protection and is produced from a fabric rated for its level of ultraviolet (UV) protection. A novel weave structure and denier (related to thread count per inch) may produce sun p ...
(long-sleeved shirts, long trousers, and broad-brimmed hats) can offer protection.
Using artificial light for tanning was once believed to help prevent skin cancers, but it can actually lead to an increased incidence of melanomas.
UV nail lamps, which are used in nail salons to dry nail polish, are another common and widespread source of UV radiation that could be avoided. Although the risk of developing skin cancer through UV nail lamp use is low, it is still recommended to wear fingerless gloves and/or apply SPF 30 or greater sunscreen to the hands before using a UV nail lamp.
The body uses UV light to generate vitamin D
Vitamin D is a group of fat-soluble secosteroids responsible for increasing intestinal absorption of calcium, magnesium, and phosphate, and many other biological effects. In humans, the most important compounds in this group are vitamin D3 (c ...
so there is a need to balance getting enough sunlight to maintain healthy vitamin D levels and reducing the risk of melanoma; it takes around a half-hour of sunlight for the body to generate its vitamin D for the day and this is about the same amount of time it takes for fair-skinned people to get a sunburn. Exposure to sunlight can be intermittent instead of all at one time.
Sunscreen
Sunscreen appears to be effective in preventing melanoma. In the past, use of sunscreens with a sun protection factor (SPF) rating of 50 or higher on exposed areas were recommended; as older sunscreens more effectively blocked UVA with higher SPF. Currently, newer sunscreen ingredients (avobenzone
Avobenzone ( trade names Parsol 1789, Milestab 1789, Eusolex 9020, Escalol 517, Neo Heliopan 357 and others, INCI Butyl Methoxydibenzoylmethane) is an oil-soluble ingredient used in sunscreen products to absorb the full spectrum of UVA rays.
Hi ...
, zinc oxide
Zinc oxide is an inorganic compound with the formula . It is a white powder that is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, food supplements, rubbers, plastics, ceramics, glass, cement ...
, and titanium dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insolub ...
) effectively block both UVA and UVB even at lower SPFs. Sunscreen also protects against squamous cell carcinoma, another skin cancer.
Concerns have been raised that sunscreen might create a false sense of security against sun damage.
Medications
A 2005 review found tentative evidence thatstatin
Statins, also known as HMG-CoA reductase inhibitors, are a class of lipid-lowering medications that reduce illness and mortality in those who are at high risk of cardiovascular disease. They are the most common cholesterol-lowering drugs.
Low ...
and fibrate
In pharmacology, the fibrates are a class of amphipathic carboxylic acids and esters. They are derivatives of fibric acid (phenoxyisobutyric acid). They are used for a range of metabolic disorders, mainly hypercholesterolemia (high cholesterol), ...
medication may decrease the risk of melanoma. A 2006 review however did not support any benefit.
Treatment
 Confirmation of the clinical diagnosis is done with a
Confirmation of the clinical diagnosis is done with a skin biopsy
Skin biopsy is a biopsy technique in which a skin lesion is removed to be sent to a pathologist to render a microscopic diagnosis. It is usually done under local anesthetic in a physician's office, and results are often available in 4 to 10 days. ...
. This is usually followed up with a wider excision of the scar or tumor. Depending on the stage, a sentinel lymph node
The sentinel lymph node is the hypothetical first lymph node or group of nodes draining a cancer. In case of established cancerous dissemination it is postulated that the sentinel lymph nodes are the target organs primarily reached by metastasiz ...
biopsy may be performed. Controversy exists around trial evidence for sentinel lymph node biopsy; with unclear evidence of benefit as of 2015. Treatment of advanced malignant melanoma is performed from a multidisciplinary approach.
Surgery
Excisional biopsies may remove the tumor, but further surgery is often necessary to reduce the risk of recurrence. Complete surgical excision with adequatesurgical margin
A resection margin or surgical margin is the margin of apparently non-tumorous tissue around a tumor that has been surgically removed, called " resected", in surgical oncology. The resection is an attempt to remove a cancer tumor so that no portio ...
s and assessment for the presence of detectable metastatic disease along with short- and long-term followup is standard. Often this is done by a wide local excision
A wide local excision (WLE) is a surgical procedure to remove a small area of diseased or problematic tissue with a margin of normal tissue. This procedure is commonly performed on the breast and to skin lesions, but can be used on any area of the ...
(WLE) with margins. Melanoma-in-situ and lentigo malignas are treated with narrower surgical margins, usually . Many surgeons consider the standard of care for standard excision of melanoma-in-situ, but margin might be acceptable for margin controlled surgery (Mohs surgery
Mohs surgery, developed in 1938 by a general surgeon, Frederic E. Mohs, is microscopically controlled surgery used to treat both common and rare types of skin cancer. During the surgery, after each removal of tissue and while the patient waits, ...
, or the double-bladed technique with margin control). The wide excision aims to reduce the rate of tumor recurrence at the site of the original lesion. This is a common pattern of treatment failure in melanoma. Considerable research has aimed to elucidate appropriate margins for excision with a general trend toward less aggressive treatment during the last decades. A 2009 meta-analysis of randomized controlled trials found a small difference in survival rates favoring wide excision of primary cutaneous melanomas, but these results were not statistically significant.
Mohs surgery has been reported with cure rate as low as 77% and as high as 98.0% for melanoma-in-situ. CCPDMA
Complete circumferential peripheral and deep margin assessment (CCPDMA) is the preferred method for the removal of certain cancers, especially skin cancers.
There are two forms of CCPDMA surgery: Mohs surgery and surgical excision coupled with mar ...
and the "double scalpel" peripheral margin controlled surgery is equivalent to Mohs surgery in effectiveness on this "intra-epithelial" type of melanoma.
Melanomas that spread usually do so to the lymph nodes
A lymph node, or lymph gland, is a kidney-shaped organ of the lymphatic system and the adaptive immune system. A large number of lymph nodes are linked throughout the body by the lymphatic vessels. They are major sites of lymphocytes that inclu ...
in the area of the tumor before spreading elsewhere. Attempts to improve survival by removing lymph nodes surgically (lymphadenectomy
Lymphadenectomy or lymph node dissection is the surgical removal of one or more groups of lymph nodes. It is almost always performed as part of the surgical management of cancer. In a regional lymph node dissection, some of the lymph nodes in the ...
) were associated with many complications, but no overall survival benefit. Recently, the technique of sentinel lymph node
The sentinel lymph node is the hypothetical first lymph node or group of nodes draining a cancer. In case of established cancerous dissemination it is postulated that the sentinel lymph nodes are the target organs primarily reached by metastasiz ...
biopsy has been developed to reduce the complications of lymph node surgery while allowing assessment of the involvement of nodes with tumor.
Biopsy of sentinel lymph nodes is a widely used procedure when treating cutaneous melanoma.
Neither sentinel lymph node biopsy nor other diagnostic tests should be performed to evaluate early, thin melanoma, including melanoma in situ, T1a melanoma or T1b melanoma ≤ 0.5mm., which cites:
*
*
* People with these conditions are unlikely to have the cancer spread to their lymph nodes or anywhere else and have a 5-year survival rate of 97%. Because of these considerations, sentinel lymph node biopsy is considered unnecessary health care
Unnecessary health care (overutilization, overuse, or overtreatment) is health care provided with a higher volume or cost than is appropriate.
In the United States, where health care costs are the highest as a percentage of GDP, overuse was the ...
for them. Furthermore, baseline blood tests and radiographic studies should not be performed only based on identifying this kind of melanoma, as there are more accurate tests for detecting cancer and these tests have high false-positive rates. To potentially correct false positives, gene expression profiling may be used as auxiliary testing for ambiguous and small lesions.
Sentinel lymph node biopsy is often performed, especially for T1b/T2+ tumors, mucosal tumors, ocular melanoma and tumors of the limbs. A process called lymphoscintigraphy is performed in which a radioactive tracer is injected at the tumor site to localize the sentinel node(s). Further precision is provided using a blue tracer dye, and surgery is performed to biopsy the node(s). Routine hematoxylin and eosin (H&E) and immunoperoxidase Immunoperoxidase is a type of immunostain used in molecular biology, medical research, and clinical diagnostics. In particular, immunoperoxidase reactions refer to a sub-class of immunohistochemical or immunocytochemical procedures in which the ant ...
staining will be adequate to rule out node involvement. Polymerase chain reaction
The polymerase chain reaction (PCR) is a method widely used to rapidly make millions to billions of copies (complete or partial) of a specific DNA sample, allowing scientists to take a very small sample of DNA and amplify it (or a part of it) ...
(PCR) tests on nodes, usually performed to test for entry into clinical trials, now demonstrate that many patients with a negative sentinel lymph node actually had a small number of positive cells in their nodes. Alternatively, a fine-needle aspiration
Fine-needle aspiration (FNA) is a diagnostic procedure used to investigate lumps or masses. In this technique, a thin (23–25 gauge (0.52 to 0.64 mm outer diameter)), hollow needle is inserted into the mass for sampling of cells that, aft ...
biopsy may be performed and is often used to test masses.
If a lymph node is positive, depending on the extent of lymph node spread, a radical lymph node dissection will often be performed. If the disease is completely resected, the patient will be considered for adjuvant therapy.
Excisional skin biopsy
Skin biopsy is a biopsy technique in which a skin lesion is removed to be sent to a pathologist to render a microscopic diagnosis. It is usually done under local anesthetic in a physician's office, and results are often available in 4 to 10 days. ...
is the management of choice. Here, the suspect lesion is totally removed with an adequate (but minimal, usually 1 or 2 mm) ellipse of surrounding skin and tissue. To avoid disruption of the local lymphatic drainage, the preferred surgical margin for the initial biopsy should be narrow (1 mm). The biopsy should include the epidermal, dermal, and subcutaneous layers of the skin. This enables the histopathologist
Histopathology (compound of three Greek words: ''histos'' "tissue", πάθος ''pathos'' "suffering", and -λογία ''-logia'' "study of") refers to the microscopic examination of tissue in order to study the manifestations of disease. Spe ...
to determine the thickness of the melanoma by microscopic examination. This is described by Breslow's thickness (measured in millimeters). However, for large lesions, such as suspected lentigo maligna, or for lesions in surgically difficult areas (face, toes, fingers, eyelids), a small punch biopsy in representative areas will give adequate information and will not disrupt the final staging or depth determination. In no circumstances should the initial biopsy include the final surgical margin (0.5 cm, 1.0 cm, or 2 cm), as a misdiagnosis can result in excessive scarring and morbidity
A disease is a particular abnormal condition that negatively affects the structure or function of all or part of an organism, and that is not immediately due to any external injury. Diseases are often known to be medical conditions that a ...
from the procedure. A large initial excision will disrupt the local lymphatic drainage and can affect further lymphangiogram-directed lymphnode dissection. A small punch biopsy can be used at any time where for logistical and personal reasons a patient refuses more invasive excisional biopsy. Small punch biopsies are minimally invasive and heal quickly, usually without noticeable scarring.
Add on treatment
High-risk melanomas may requireadjuvant treatment In pharmacology, an adjuvant is a drug or other substance, or a combination of substances, that is used to increase the efficacy or potency of certain drugs. Specifically, the term can refer to:
* Adjuvant therapy in cancer management
* Analgesi ...
, although attitudes to this vary in different countries. In the United States, most patients in otherwise good health will begin up to a year of high-dose interferon treatment, which has severe side effects, but may improve the patient's prognosis slightly. However, the British Association of Dermatologists
The British Association of Dermatologists is a charity established in 1920 whose charitable objects are the practice, teaching, training, and research of dermatology. It produces the ''British Journal of Dermatology
The ''British Journal of De ...
guidelines on melanoma state that interferon is not recommended as a standard adjuvant treatment for melanoma. A 2013 meta-analysis suggested that the addition of interferon alpha increased disease-free and overall survival for people with AJCC TNM stage II-III cutaneous melanoma. A 2011 meta-analysis showed that interferon could lengthen the time before a melanoma comes back but increased survival by only 3% at 5 years. The unpleasant side effects also greatly decrease quality of life.
In the European Union, interferon is usually not used outside the scope of clinical trials.
Chemotherapy
Chemotherapy drugs such as Dacarbazine have been the backbone of metastatic melanoma treatment since FDA approval in 1975; however, its efficacy in terms of survival has never been proven in an RCT. In people with locally advanced cutaneous malignancies and sarcoma, isolated limb infusion (ILI) has been found to be a minimally invasive and well-tolerated procedure for delivering regional chemotherapy.Targeted therapy
Melanoma cells have mutations that allow them to survive and grow indefinitely in the body. Small-molecule targeted therapies work by blocking the genes involved in pathways for tumor proliferation and survival. The main treatments are BRAF,C-Kit
Proto-oncogene c-KIT is the gene encoding the receptor tyrosine kinase protein known as tyrosine-protein kinase KIT, CD117 (cluster of differentiation 117) or mast/stem cell growth factor receptor (SCFR). Multiple transcript variants encoding dif ...
and NRAS inhibitors. These inhibitors work to inhibit the downstream pathways involved in cell proliferation and tumour development due to specific gene mutations. People can be treated with small-molecule targeted inhibitors if they are positive for the specific mutation. BRAF inhibitor
BRAF is a human gene that encodes a protein called B-Raf. The gene is also referred to as proto-oncogene B-Raf and v-Raf murine sarcoma viral oncogene homolog B, while the protein is more formally known as serine/threonine-protein kinase B-Raf.
T ...
s, such as vemurafenib
Vemurafenib ( INN, marketed as Zelboraf) is an inhibitor of the B-Raf enzyme developed by Plexxikon (now part of Daiichi-Sankyo) and Genentech for the treatment of late-stage melanoma.; The name "vemurafenib" comes from V600E mutated BRAF in ...
and dabrafenib
Dabrafenib, sold under the brand name Tafinlar & Rafinlar ( both by Novartis) among others, is a medication for the treatment of cancers associated with a mutated version of the gene BRAF. Dabrafenib acts as an inhibitor of the associated enzyme ...
and a MEK inhibitor A MEK inhibitor is a chemical or drug that inhibits the mitogen-activated protein kinase kinase enzymes MEK1 and/or MEK2.
They can be used to affect the MAPK/ERK pathway which is often overactive in some cancers. (See MAPK/ERK pathway#Clinical sign ...
trametinib
Trametinib, sold under the brand name Mekinist among others, is an anticancer medication used for the treatment of melanoma. It is a MEK inhibitor drug with anti-cancer activity. It inhibits MEK1 and MEK2.
Trametinib had good results for metas ...
are the most effective, approved treatments for BRAF positive melanoma. Melanoma tumors can develop resistance during therapy which can make therapy no longer effective, but combining the use of BRAF and MEK inhibitors may create a fast and lasting melanoma therapy response.
A number of treatments improve survival over traditional chemotherapy. Biochemotherapy (chemotherapy with cytokines IL-2 and IFN-α) combined with BRAF inhibitors improved survival for people with BRAF positive melanoma. Biochemotherapy alone did not improve overall survival and had higher toxicity than chemotherapy. Combining multiple chemotherapy agents (polychemotherapy) did not improve survival over monochemotherapy. Targeted therapies result in relatively short progression-free survival Progression-free survival (PFS) is "the length of time during and after the treatment of a disease, such as cancer, that a patient lives with the disease but it does not get worse". In oncology, PFS usually refers to situations in which a tumor is p ...
(PFS) times. The therapy combination of dabrafenib and trametinib has a 3-year PFS of 23%, and 5-year PFS of 13%.
Immunotherapy
Immunotherapy
Immunotherapy or biological therapy is the treatment of disease by activating or suppressing the immune system. Immunotherapies designed to elicit or amplify an immune response are classified as ''activation immunotherapies,'' while immunotherap ...
is aimed at stimulating the person's immune system against the tumor, by enhancing the body's own ability to recognize and kill cancer cells. The current approach to treating melanoma with immunotherapy includes three broad categories of treatments including cytokine
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in autocrin ...
s, immune check point inhibitors, and adoptive cell transfer
Adoptive cell transfer (ACT) is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system with the goal of improving immune functiona ...
. These treatment options are most often used in people with metastatic melanoma and significantly improves overall survival. However, these treatments are often costly. For example, one immune check point inhibitor treatment, pembrolizumab
Pembrolizumab, sold under the brand name Keytruda, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer, cervical cancer, and certain types of breast canc ...
, costs $10,000 to US$12,000 for a single dose administered every 3 weeks.
Cytokine therapies used for melanoma include IFN-a
The type-I interferons (IFN) are cytokines which play essential roles in inflammation, immunoregulation, tumor cells recognition, and T-cell responses. In the human genome, a cluster of thirteen functional IFN genes is located at the 9p21.3 cyto ...
and IL-2
The Ilyushin Il-2 (Russian: Илью́шин Ил-2) is a ground-attack plane that was produced by the Soviet Union in large numbers during the Second World War. The word ''shturmovík'' ( Cyrillic: штурмовик), the generic Russian term ...
. IL-2 (Proleukin
Interleukin-2 (IL-2) is an interleukin, a type of cytokine signaling molecule in the immune system. It is a 15.5–16 kDa protein that regulates the activities of white blood cells (leukocytes, often lymphocytes) that are responsible for ...
) was the first new therapy approved (1990 EU, 1992 US) for the treatment of metastatic melanoma in 20 years. IL-2 may offer the possibility of a complete and long-lasting remission in this disease in a small percentage of people with melanoma. Intralesional IL-2 for in-transit metastases has a high complete response rate ranging from 40 to 100%. Similarly, IFN-a
The type-I interferons (IFN) are cytokines which play essential roles in inflammation, immunoregulation, tumor cells recognition, and T-cell responses. In the human genome, a cluster of thirteen functional IFN genes is located at the 9p21.3 cyto ...
has shown only modest survival benefits and high toxicity, limiting its use as a stand-alone therapy.
Immune check point inhibitors include anti-CTLA-4
CTLA-4 or CTLA4 (cytotoxic T-lymphocyte-associated protein 4), also known as CD152 ( cluster of differentiation 152), is a protein receptor that functions as an immune checkpoint and downregulates immune responses. CTLA-4 is constitutively exp ...
monoclonal antibodies (ipilimumab
Ipilimumab, sold under the brand name Yervoy, is a monoclonal antibody medication that works to activate the immune system by targeting CTLA-4, a protein receptor that downregulates the immune system.
Cytotoxic T lymphocytes (CTLs) can recogniz ...
and tremelimumab
Tremelimumab, sold under the brand name Imjudo, is a fully human monoclonal antibody against CTLA-4. It is an immune checkpoint blocker.
Tremelimumab was approved for unresectable hepatocellular carcinoma medical use in the United States in Oc ...
), toll-like receptor
Toll-like receptors (TLRs) are a class of proteins that play a key role in the innate immune system. They are single-pass membrane-spanning receptors usually expressed on sentinel cells such as macrophages and dendritic cells, that recognize ...
(TLR) agonists, CD40
Cluster of differentiation 40, CD40 is a costimulatory protein found on antigen-presenting cells and is required for their activation. The binding of CD154 ( CD40L) on TH cells to CD40 activates antigen presenting cells and induces a variety of d ...
agonists, anti-PD-1
Programmed cell death protein 1, also known as PD-1 and CD279 (cluster of differentiation 279), is a protein on the surface of T and B cells that has a role in regulating the immune system's response to the cells of the human body by down-regula ...
(pembrolizumab
Pembrolizumab, sold under the brand name Keytruda, is a humanized antibody used in cancer immunotherapy that treats melanoma, lung cancer, head and neck cancer, Hodgkin lymphoma, stomach cancer, cervical cancer, and certain types of breast canc ...
, pidilizumab, and nivolumab
Nivolumab, sold under the brand name Opdivo, is a medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urotheli ...
) and PD-L1
Programmed death-ligand 1 (PD-L1) also known as cluster of differentiation 274 (CD274) or B7 homolog 1 (B7-H1) is a protein that in humans is encoded by the ''CD274'' gene.
Programmed death-ligand 1 (PD-L1) is a 40kDa type 1 transmembrane protei ...
antibodies. Evidence suggests that anti-PD-1
Programmed cell death protein 1, also known as PD-1 and CD279 (cluster of differentiation 279), is a protein on the surface of T and B cells that has a role in regulating the immune system's response to the cells of the human body by down-regula ...
antibodies are more effective than anti- CTLA4 antibodies with less systemic toxicity. The five-year progression-free survival for immunotherapy with pembrolizumab is 21%. A therapeutic approach that includes the combination of different therapies improves overall survival and progression-free survival compared to treatment with the separate immunotherapy drugs alone.
Ongoing research is looking at treatment by adoptive cell transfer
Adoptive cell transfer (ACT) is the transfer of cells into a patient. The cells may have originated from the patient or from another individual. The cells are most commonly derived from the immune system with the goal of improving immune functiona ...
. Adoptive cell transfer refers to the application of pre-stimulated, modified T cell
A T cell is a type of lymphocyte. T cells are one of the important white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell r ...
s or dendritic cells and is presently used to minimize complications from graft-versus-host disease.
The combination nivolumab/relatlimab (Opdualag) was approved for medical use in the United States in March 2022.
Lentigo maligna
Standard excision is still being done by most surgeons. Unfortunately, the recurrence rate is exceedingly high (up to 50%). This is due to the ill-defined visible surgical margin, and the facial location of the lesions (often forcing the surgeon to use a narrow surgical margin). The narrow surgical margin used, combined with the limitation of the standard "bread-loafing" technique of fixed tissue histology – result in a high "false negative" error rate, and frequent recurrences. Margin control (peripheral margins) is necessary to eliminate the false negative errors. Ifbread loafing
Bread loafing is a common method of processing surgical specimens for histopathology. The process involves cutting the specimen into 3 or more sections. The cut sections are mounted by embedding in paraffin or frozen medium. The cut edge is then t ...
is used, distances from sections should approach 0.1 mm to assure that the method approaches complete margin control. A meta-analysis of the literature in 2014 found no randomized controlled trials of surgical interventions to treat lentigo maligna or melanoma in-situ, even though surgery is the most widely used treatment.
Mohs surgery
Mohs surgery, developed in 1938 by a general surgeon, Frederic E. Mohs, is microscopically controlled surgery used to treat both common and rare types of skin cancer. During the surgery, after each removal of tissue and while the patient waits, ...
has been done with cure rate reported to be as low as 77%, and as high as 95% by another author. The "double scalpel" peripheral margin controlled excision method approximates the Mohs method in margin control, but requires a pathologist intimately familiar with the complexity of managing the vertical margin on the thin peripheral sections and staining methods.
Some melanocytic nevi, and melanoma-in-situ (lentigo maligna
Lentigo maligna is where melanocyte cells have become malignant and grow continuously along the stratum basale of the skin, but have not invaded below the epidermis. Lentigo maligna is not the same as lentigo maligna melanoma, as detailed below. ...
) have resolved with an experimental treatment, imiquimod
Imiquimod, sold under the brand name Aldara among others, is a medication that acts as an immune response modifier that is used to treat genital warts, superficial basal cell carcinoma, and actinic keratosis. Scientists at 3M's pharmaceuticals ...
(Aldara) topical cream, an immune enhancing agent. Some derma-surgeons are combining the 2 methods: surgically excising the cancer and then treating the area with Aldara cream postoperatively for three months. While some studies have suggested the adjuvant use of topical tazarotene, the current evidence is insufficient to recommend it and suggests that it increases topical inflammation, leading to lower patient compliance.
Radiation
Radiation therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Radi ...
is often used after surgical resection for patients with locally or regionally advanced melanoma or for patients with un-resectable distant metastases. Kilovoltage x-ray beams are often used for these treatments and have the property of the maximum radiation dose occurring close to the skin surface. It may reduce the rate of local recurrence but does not prolong survival. Radioimmunotherapy
Radioimmunotherapy (RIT) uses an antibody labeled with a radionuclide to deliver cytotoxic radiation to a target cell. It is a form of unsealed source radiotherapy. In cancer therapy, an antibody with specificity for a tumor-associated antigen i ...
of metastatic melanoma is currently under investigation.
Radiotherapy has a role in the palliation of metastatic melanoma.
Prognosis
 Factors that affect prognosis include:
*
Factors that affect prognosis include:
* tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
thickness in millimeters (Breslow's depth
In medicine, Breslow's depth was used as a prognostic factor in melanoma of the skin. It is a description of how deeply tumor cells have invaded. Currently, the standard Breslow's depth has been replaced by the AJCC depth, in the AJCC staging sys ...
),
* depth related to skin structures ( Clark level),
* type of melanoma,
* presence of ulceration,
* presence of lymphatic/perineural invasion
In pathology, perineural invasion, abbreviated PNI, refers to the invasion of cancer to the space surrounding a nerve. It is common in head and neck cancer, prostate cancer and colorectal cancer.
Unlike perineurial spread (PNS), which is defined ...
,
* presence of tumor-infiltrating lymphocyte
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic ad ...
s (if present, prognosis is better),
* location of lesion,
* presence of satellite lesions, and
* presence of regional or distant metastasis
Metastasis is a pathogenic agent's spread from an initial or primary site to a different or secondary site within the host's body; the term is typically used when referring to metastasis by a cancerous tumor. The newly pathological sites, then ...
.
Certain types of melanoma have worse prognoses but this is explained by their thickness
Thickness may refer to:
* Thickness (graph theory)
* Thickness (geology), the distance across a layer of rock
* Thickness (meteorology), the difference in height between two atmospheric pressure levels
* Thickness planer a woodworking machine
...
. Less invasive melanomas even with lymph node metastases carry a better prognosis than deep melanomas without regional metastasis at time of staging. Local recurrences tend to behave similarly to a primary unless they are at the site of a wide local excision
A wide local excision (WLE) is a surgical procedure to remove a small area of diseased or problematic tissue with a margin of normal tissue. This procedure is commonly performed on the breast and to skin lesions, but can be used on any area of the ...
(as opposed to a staged excision or punch/shave excision) since these recurrences tend to indicate lymphatic invasion.
When melanomas have spread to the lymph nodes, one of the most important factors is the number of nodes with malignancy. Extent of malignancy within a node is also important; micro-metastases in which malignancy is only microscopic have a more favorable prognosis than macrometastases. In some cases micrometastases may only be detected by special staining, and if malignancy is only detectable by a rarely employed test known as the polymerase chain reaction
The polymerase chain reaction (PCR) is a method widely used to rapidly make millions to billions of copies (complete or partial) of a specific DNA sample, allowing scientists to take a very small sample of DNA and amplify it (or a part of it) ...
(PCR), the prognosis is better. Macro-metastases in which malignancy is clinically apparent (in some cases cancer completely replaces a node) have a far worse prognosis, and if nodes are matted or if there is extracapsular extension, the prognosis is worse still. In addition to these variables, expression levels and copy number variations of a number of relevant genes may be used to support assessment of malignant melanoma prognosis.
Stage IV melanoma, in which it has metastasized, is the most deadly skin malignancy: five-year survival is 22.5%. When there is distant metastasis, the cancer is generally considered incurable. The five-year survival rate is less than 10%. The median survival is 6–12 months. Treatment is palliative, focusing on life extension and quality of life
Quality of life (QOL) is defined by the World Health Organization as "an individual's perception of their position in life in the context of the culture and value systems in which they live and in relation to their goals, expectations, standards ...
. In some cases, patients may live many months or even years with metastatic melanoma (depending on the aggressiveness of the treatment). Metastases to skin and lungs have a better prognosis. Metastases to brain, bone and liver are associated with a worse prognosis. Survival is better with metastasis in which the location of the primary tumor is unknown.
There is not enough definitive evidence to adequately stage, and thus give a prognosis for, ocular melanoma and melanoma of soft parts, or mucosal melanoma (e.g., rectal melanoma), although these tend to metastasize more easily. Even though regression may increase survival, when a melanoma has regressed, it is impossible to know its original size and thus the original tumor is often worse than a pathology report might indicate.
About 200 genes are prognostic in melanoma, with both unfavorable genes where high expression is correlated to poor survival and favorable genes where high expression is associated with longer survival times. Examples of unfavorable genes are MCM6
DNA replication licensing factor MCM6 is a protein that in humans is encoded by the ''MCM6'' gene. MCM6 is one of the highly conserved mini-chromosome maintenance proteins ( MCM) that are essential for the initiation of eukaryotic genome replicati ...
and TIMELESS; an example of a favorable gene is WIPI1
WD repeat domain phosphoinositide-interacting protein 1 (WIPI-1), also known as Atg18 protein homolog (ATG18) and WD40 repeat protein interacting with phosphoinositides of 49 kDa (WIPI 49 kDa), is a protein that in humans is encoded by the ''WIPI1 ...
.
An increased neutorphil-to-lymphocyte ratio is associated with worse outcomes.
Epidemiology
Caucasian
Caucasian may refer to:
Anthropology
*Anything from the Caucasus region
**
**
** ''Caucasian Exarchate'' (1917–1920), an ecclesiastical exarchate of the Russian Orthodox Church in the Caucasus region
*
*
*
Languages
* Northwest Caucasian l ...
.
The rate of melanoma has increased in the recent years, but it is not clear to what extent changes in behavior, in the environment, or in early detection are involved.
Australia
Australia
Australia, officially the Commonwealth of Australia, is a Sovereign state, sovereign country comprising the mainland of the Australia (continent), Australian continent, the island of Tasmania, and numerous List of islands of Australia, sma ...
has a very high – and increasing – rate of melanoma. In 2012, deaths from melanoma occurred in 7.3–9.8 per 100,000 population. In Australia, melanoma is the third most common cancer in either sex; indeed, its incidence is higher than for lung cancer
Lung cancer, also known as lung carcinoma (since about 98–99% of all lung cancers are carcinomas), is a malignant lung tumor characterized by uncontrolled cell growth in tissue (biology), tissues of the lung. Lung carcinomas derive from tran ...
, although the latter accounts for more deaths. It is estimated that in 2012, more than 12,000 Australians were diagnosed with melanoma: given Australia's modest population, this is better expressed as 59.6 new cases per 100,000 population per year; >1 in 10 of all new cancer cases were melanomas. Melanoma incidence in Australia is matter of significance, for the following reasons:
* Australian melanoma incidence has increased by more than 30 per cent between 1991 and 2009.
* Australian melanoma age-standardized incidence rates were, as of 2008, at least 12 times higher than the world average.
* Australian melanoma incidence is, by some margin, the highest in the world.
* Overall age-standardized cancer incidence in Australia is the highest in the world, and this is attributable to melanoma alone. Age-standardized overall cancer incidence is similar to New Zealand, but there is a statistically significant difference between Australia and all other parts of the developed world including North America, Western Europe, and the Mediterranean.
United States
In the United States, about 9,000 people die from melanoma a year. In 2011, it affected 19.7 per 100,000, and resulted in death in 2.7 per 100,000. In 2013: * 71,943 people in the United States were diagnosed with melanomas of the skin, including 42,430 men and 29,513 women. * 9,394 people in the United States died from melanomas of the skin, including 6,239 men and 3,155 women. The American Cancer Society's estimates for melanoma incidence in the United States for 2017 are: * About 87,110 new melanomas will be diagnosed (about 52,170 in men and 34,940 in women). * About 9,730 people are expected to die of melanoma (about 6,380 men and 3,350 women). Melanoma is more than 20 times more common in whites than in African Americans. Overall, the lifetime risk of getting melanoma is about 2.5% (1 in 40) for whites, 0.1% (1 in 1,000) for African Americans, and 0.5% (1 in 200) for Hispanics. The risk of melanoma increases as people age. The average age of people when the disease is diagnosed is 63.History
Although melanoma is not a new disease, evidence for its occurrence in antiquity is rather scarce. However, one example lies in a 1960s examination of ninePeru
, image_flag = Flag of Peru.svg
, image_coat = Escudo nacional del Perú.svg
, other_symbol = Great Seal of the State
, other_symbol_type = Seal (emblem), National seal
, national_motto = "Fi ...
vian mummies, radiocarbon
Carbon-14, C-14, or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and c ...
dated to be approximately 2400 years old, which showed apparent signs of melanoma: melanotic masses in the skin and diffuse metastases to the bones.
John Hunter is reported to be the first to operate on metastatic melanoma in 1787. Although not knowing precisely what it was, he described it as a "cancerous fungous excrescence". The excised tumor was preserved in the Hunterian Museum
The Hunterian is a complex of museums located in and operated by the University of Glasgow in Glasgow, Scotland. It is the oldest museum in Scotland. It covers the Hunterian Museum, the Hunterian Art Gallery, the Mackintosh House, the Zoology M ...
of the Royal College of Surgeons of England
The Royal College of Surgeons of England (RCS England) is an independent professional body and registered charity that promotes and advances standards of surgical care for patients, and regulates surgery and dentistry in England and Wales. T ...
. It was not until 1968 that microscopic examination of the specimen revealed it to be an example of metastatic melanoma.
The French physician René Laennec
René-Théophile-Hyacinthe Laennec (; 17 February 1781 – 13 August 1826) was a French physician and musician. His skill at carving his own wooden flutes led him to invent the stethoscope in 1816, while working at the Hôpital Necker ...
was the first to describe melanoma as a disease entity. His report was initially presented during a lecture for the Faculté de Médecine de Paris in 1804 and then published as a bulletin in 1806.
The first English-language report of melanoma was presented by an English general practitioner from Stourbridge, William Norris in 1820. In his later work in 1857 he remarked that there is a familial predisposition for development of melanoma (''Eight Cases of Melanosis
Melanosis is a form of hyperpigmentation associated with increased melanin.
It can also refer to:
* Melanism
* Ocular melanosis
* Smoker's melanosis
* Oral melanosis
* Riehl melanosis
Riehl melanosis is a form of contact dermatitis, beginning wi ...
with Pathological and Therapeutical Remarks on That Disease''). Norris was also a pioneer in suggesting a link between nevi and melanoma and the possibility of a relationship between melanoma and environmental exposures, by observing that most of his patients had pale complexions. He also described that melanomas could be amelanotic and later showed the metastatic nature of melanoma by observing that they can disseminate to other visceral organs.
The first formal acknowledgment of advanced melanoma as untreatable came from Samuel Cooper Samuel or Sam Cooper may refer to:
*Samuel Cooper (painter) (1609–1672), English miniature painter
*Samuel Cooper (clergyman) (1725–1783), Congregationalist minister in Boston, Massachusetts
* Samuel Cooper (surgeon) (1780–1848), English surge ...
in 1840. He stated that the only chance for a cure depends upon the early removal of the disease (i.e., early excision of the malignant mole) ...'
More than one and a half centuries later this situation remains largely unchanged.
Terminology
The word ''melanoma'' came to English from 19th-centuryNew Latin
New Latin (also called Neo-Latin or Modern Latin) is the revival of Literary Latin used in original, scholarly, and scientific works since about 1500. Modern scholarly and technical nomenclature, such as in zoological and botanical taxonomy ...
and uses combining forms derived from ancient Greek
Ancient Greek includes the forms of the Greek language used in ancient Greece and the ancient world from around 1500 BC to 300 BC. It is often roughly divided into the following periods: Mycenaean Greek (), Dark Ages (), the Archaic peri ...
roots: '' melano-'' (denoting melanin
Melanin (; from el, μέλας, melas, black, dark) is a broad term for a group of natural pigments found in most organisms. Eumelanin is produced through a multistage chemical process known as melanogenesis, where the oxidation of the amino ...
) + '' -oma'' (denoting a tissue mass and especially a neoplasm
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
), in turn from Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
μέλας ''melas'', "dark", and -ωμα ''oma'', "process". The word ''melanoma'' has a long history of being used in a broader sense to refer to any melanocytic tumor
Melanocytic tumors are neoplasm, tumors developed from melanocytes.
Types
* Melanocytic nevus
* Melanocytic tumors of uncertain malignant potential
* Melanoma
Tumor
{{med-sign-stub ...
, typically, but not always malignant, but today the narrower sense referring only to malignant types has become so dominant that benign tumors are usually not called melanomas anymore and the word ''melanoma'' is now usually taken to mean malignant melanoma unless otherwise specified. Terms such as "benign melanocytic tumor
Melanocytic tumors are neoplasm, tumors developed from melanocytes.
Types
* Melanocytic nevus
* Melanocytic tumors of uncertain malignant potential
* Melanoma
Tumor
{{med-sign-stub ...
" unequivocally label the benign types, and modern histopathologic tumor classifications used in medicine do not use the word for benign tumors.
Research
Pharmacotherapy research for un-resectable or metastatic malignant melanoma is ongoing.Targeted therapies
In clinical research, adoptive cell therapy andgene therapy
Gene therapy is a medical field which focuses on the genetic modification of cells to produce a therapeutic effect or the treatment of disease by repairing or reconstructing defective genetic material. The first attempt at modifying human DN ...
, are being tested.
Two kinds of experimental treatments developed at the National Cancer Institute
The National Cancer Institute (NCI) coordinates the United States National Cancer Program and is part of the National Institutes of Health (NIH), which is one of eleven agencies that are part of the U.S. Department of Health and Human Services. ...
(NCI), have been used in metastatic melanoma with tentative success.
The first treatment involves adoptive cell therapy (ACT) using TILs immune cells (tumor-infiltrating lymphocytes) isolated from a person's own melanoma tumor. These cells are grown in large numbers in a laboratory and returned to the patient after a treatment that temporarily reduces normal T cells in the patient's body. TIL therapy following lymphodepletion can result in durable complete response in a variety of setups.
The second treatment, adoptive transfer of genetically altered autologous lymphocytes, depends on delivering genes that encode so called T cell receptor
The T-cell receptor (TCR) is a protein complex found on the surface of T cells, or T lymphocytes, that is responsible for recognizing fragments of antigen as peptides bound to major histocompatibility complex (MHC) molecules. The binding ...
s (TCRs), into patient's lymphocytes. After that manipulation lymphocytes recognize and bind to certain molecules found on the surface of melanoma cells and kill them.
A cancer vaccine showed modest benefit in late-stage testing in 2009 against melanoma.
BRAF inhibitors
About 60% of melanomas contain a mutation in the B-Raf gene. Early clinical trials suggested that B-Raf inhibitors including Plexxicon'svemurafenib
Vemurafenib ( INN, marketed as Zelboraf) is an inhibitor of the B-Raf enzyme developed by Plexxikon (now part of Daiichi-Sankyo) and Genentech for the treatment of late-stage melanoma.; The name "vemurafenib" comes from V600E mutated BRAF in ...
could lead to substantial tumor regression in a majority of patients if their tumor contain the B-Raf mutation. In June 2011, a large clinical trial
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, diet ...
confirmed the positive findings from those earlier trials.
In August 2011, Vemurafenib received FDA approval for the treatment of late-stage melanoma. In May 2013 the US FDA
The United States Food and Drug Administration (FDA or US FDA) is a federal agency of the Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food s ...
approved dabrafenib as a single agent treatment for patients with BRAF V600E mutation-positive advanced melanoma.
Some researchers believe that combination therapies that simultaneously block multiple pathways may improve efficacy by making it more difficult for the tumor cells to mutate before being destroyed. In October 2012 a study reported that combining Dabrafenib with a MEK inhibitor A MEK inhibitor is a chemical or drug that inhibits the mitogen-activated protein kinase kinase enzymes MEK1 and/or MEK2.
They can be used to affect the MAPK/ERK pathway which is often overactive in some cancers. (See MAPK/ERK pathway#Clinical sign ...
trametinib
Trametinib, sold under the brand name Mekinist among others, is an anticancer medication used for the treatment of melanoma. It is a MEK inhibitor drug with anti-cancer activity. It inhibits MEK1 and MEK2.
Trametinib had good results for metas ...
led to even better outcomes. Compared to Dabrafenib alone, progression-free survival was increased to 41% from 9%, and the median progression-free survival Progression-free survival (PFS) is "the length of time during and after the treatment of a disease, such as cancer, that a patient lives with the disease but it does not get worse". In oncology, PFS usually refers to situations in which a tumor is p ...
increased to 9.4 months versus 5.8 months. Some side effects were, however, increased in the combined study.
In January 2014, the FDA approved the combination of dabrafenib and trametinib for the treatment of people with BRAF V600E/K-mutant metastatic melanoma. In June 2018, the FDA approved the combination of a BRAF inhibitor encorafenib and a MEK inhibitor binimetinib for the treatment of un-resectable or metastatic melanoma with a BRAF V600E or V600K mutation.
Eventual resistance to BRAF and MEK inhibitors may be due to a cell surface protein known as EphA2
EPH receptor A2 (ephrin type-A receptor 2) is a protein that in humans is encoded by the ''EPHA2'' gene.
Function
This gene belongs to the ephrin receptor subfamily of the protein- tyrosine kinase family. EPH and EPH-related receptors have be ...
which is now being investigated.
Ipilimumab
At the American Society of Clinical Oncology Conference in June 2010, theBristol-Myers Squibb
The Bristol Myers Squibb Company (BMS) is an American multinational pharmaceutical company. Headquartered in New York City, BMS is one of the world's largest pharmaceutical companies and consistently ranks on the ''Fortune'' 500 list of the lar ...
pharmaceutical company reported the clinical findings of their drug ipilimumab
Ipilimumab, sold under the brand name Yervoy, is a monoclonal antibody medication that works to activate the immune system by targeting CTLA-4, a protein receptor that downregulates the immune system.
Cytotoxic T lymphocytes (CTLs) can recogniz ...
. The study found an increase in median survival from 6.4 to 10 months in patients with advanced melanomas treated with the monoclonal ipilimumab, versus an experimental vaccine. It also found a one-year survival rate of 25% in the control group using the vaccine, 44% in the vaccine and ipilimumab group, and 46% in the group treated with ipilimumab alone. However, some have raised concerns about this study for its use of the unconventional control arm, rather than comparing the drug against a placebo or standard treatment. The criticism was that although Ipilimumab performed better than the vaccine, the vaccine has not been tested before and may be causing toxicity, making the drug appear better by comparison.
Ipilimumab was approved by the FDA in March 2011 to treat patients with late-stage melanoma that has spread or cannot be removed by surgery.
In June 2011, a clinical trial of ipilimumab plus dacarbazine
Dacarbazine (DTIC), also known as imidazole carboxamide, is a chemotherapy medication used in the treatment of melanoma and Hodgkin's lymphoma. For Hodgkin's it is often used together with vinblastine, bleomycin, and doxorubicin. It is given ...
combined this immune system booster with the standard chemotherapy drug that targets cell division. It showed an increase in median survival for these late stage patients to 11 months instead of the 9 months normally seen. Researchers were also hopeful of improving the five year survival rate, though serious adverse side-effects were seen in some patients. A course of treatment costs $120,000. The drug's brandname is Yervoy.
Surveillance methods
Advances in high resolution ultrasound scanning have enabled surveillance of metastatic burden to the sentinel lymph nodes. The Screening and Surveillance of Ultrasound in Melanoma trial (SUNMEL) is evaluating ultrasound as an alternative to invasive surgical methods.Oncolytic virotherapy
In some countries oncolytic virotherapy methods are studied and used to treat melanoma. Oncolytic virotherapy is a promising branch ofvirotherapy
Virotherapy is a treatment using biotechnology to convert viruses into therapeutic agents by reprogramming viruses to treat diseases. There are three main branches of virotherapy: anti-cancer oncolytic viruses, viral vectors for gene therapy and ...
, where oncolytic virus
An oncolytic virus is a virus that preferentially infects and kills cancer cells. As the infected cancer cells are destroyed by oncolysis, they release new infectious virus particles or virions to help destroy the remaining tumour. Oncolytic vir ...
es are used to treat diseases; viruses can increase metabolism, reduce anti-tumor immunity and disorganize vasculature. Talimogene laherparepvec (T-VEC) (which is a herpes simplex virus type 1–derived oncolytic immunotherapy), was shown to be useful against metastatic melanoma in 2015 with an increased survival of 4.4 months.
Notes
References
External links
* {{Authority control Cancer Medical mnemonics Wikipedia medicine articles ready to translate