Genomics on:
[Wikipedia]
[Google]
[Amazon]
Genomics is an interdisciplinary field of
 Most of the microorganisms whose genomes have been completely sequenced are problematic
Most of the microorganisms whose genomes have been completely sequenced are problematic

 The Illumina dye sequencing method is based on reversible dye-terminators and was developed in 1996 at the Geneva Biomedical Research Institute, by Pascal Mayer and Laurent Farinelli. In this method, DNA molecules and primers are first attached on a slide and amplified with
The Illumina dye sequencing method is based on reversible dye-terminators and was developed in 1996 at the Geneva Biomedical Research Institute, by Pascal Mayer and Laurent Farinelli. In this method, DNA molecules and primers are first attached on a slide and amplified with
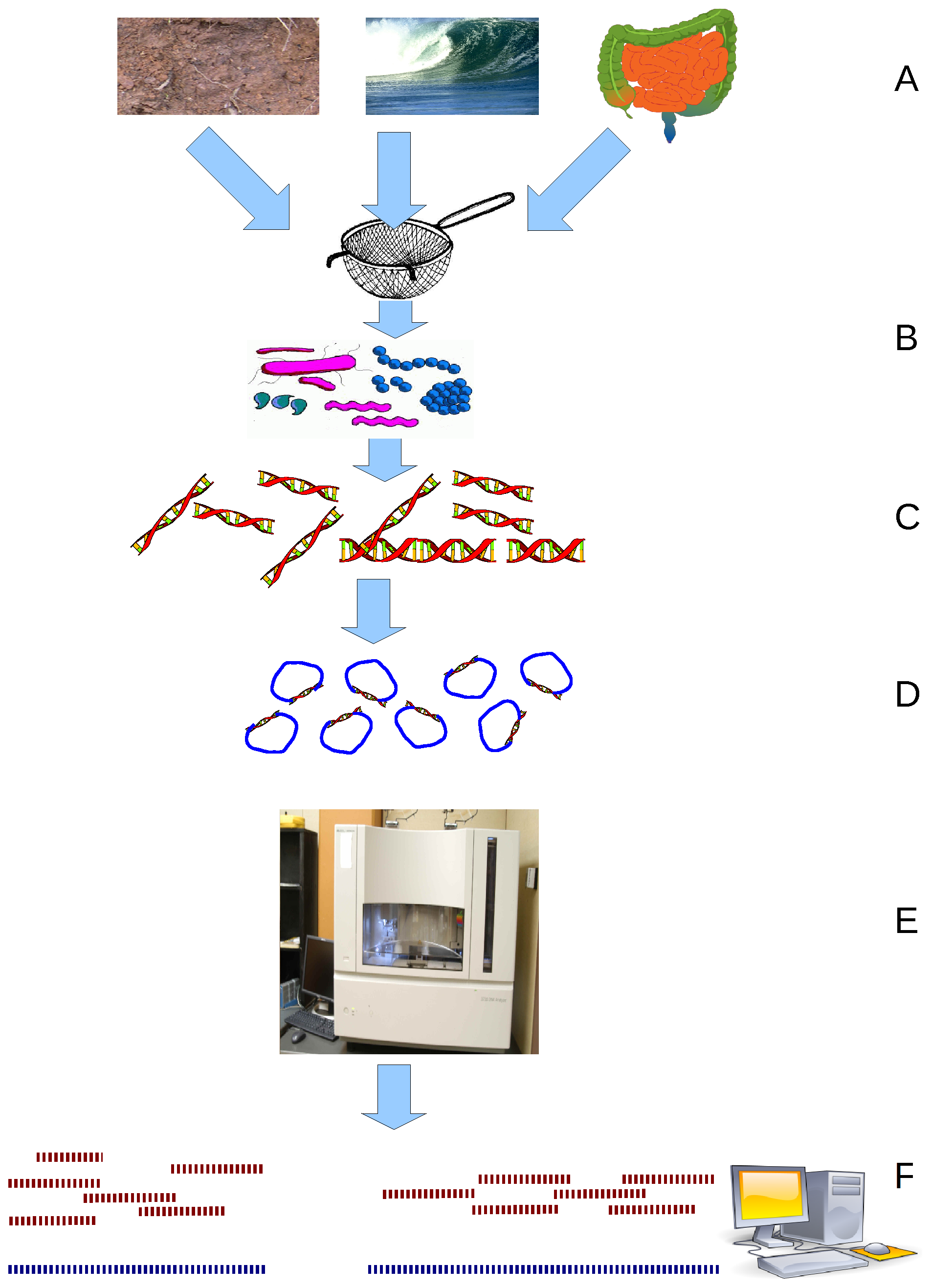 Metagenomics is the study of ''metagenomes'', genetic material recovered directly from environmental samples. The broad field may also be referred to as environmental genomics, ecogenomics or community genomics. While traditional
Metagenomics is the study of ''metagenomes'', genetic material recovered directly from environmental samples. The broad field may also be referred to as environmental genomics, ecogenomics or community genomics. While traditional
 Genomics has provided applications in many fields, including
Genomics has provided applications in many fields, including
Annual Review of Genomics and Human Genetics
BMC Genomics
A BMC journal on Genomics
Genomics journal
Genomics.org
An openfree genomics portal.
NHGRI
US government's genome institute
JCVI Comprehensive Microbial Resource
KoreaGenome.org
The first Korean Genome published and the sequence is available freely.
GenomicsNetwork
Looks at the development and use of the science and technologies of genomics.
Institute for Genome Sciences
Genomics research.
MIT OpenCourseWare HST.512 Genomic Medicine
A free, self-study course in genomic medicine. Resources include audio lectures and selected lecture notes.
ENCODE threads explorer
Machine learning approaches to genomics.
Global map of genomics laboratories
Genomics: Scitable by nature education
Learn All About Genetics Online
{{Authority control
molecular biology
Molecular biology is a branch of biology that seeks to understand the molecule, molecular basis of biological activity in and between Cell (biology), cells, including biomolecule, biomolecular synthesis, modification, mechanisms, and interactio ...
focusing on the structure, function, evolution, mapping, and editing of genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
s. A genome is an organism's complete set of DNA, including all of its genes as well as its hierarchical, three-dimensional structural configuration. In contrast to genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinians, Augustinian ...
, which refers to the study of ''individual'' genes and their roles in inheritance, genomics aims at the collective characterization and quantification of ''all'' of an organism's genes, their interrelations and influence on the organism. Genes may direct the production of proteins
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, re ...
with the assistance of enzymes and messenger molecules. In turn, proteins make up body structures such as organs and tissues as well as control chemical reactions and carry signals between cells. Genomics also involves the sequencing and analysis of genomes through uses of high throughput DNA sequencing
DNA sequencing is the process of determining the nucleic acid sequence – the order of nucleotides in DNA. It includes any method or technology that is used to determine the order of the four bases: adenine, thymine, cytosine, and guanine. The ...
and bioinformatics
Bioinformatics () is an interdisciplinary field of science that develops methods and Bioinformatics software, software tools for understanding biological data, especially when the data sets are large and complex. Bioinformatics uses biology, ...
to assemble and analyze the function and structure of entire genomes. Advances in genomics have triggered a revolution in discovery-based research and systems biology to facilitate understanding of even the most complex biological systems such as the brain.
The field also includes studies of intragenomic (within the genome) phenomena such as epistasis (effect of one gene on another), pleiotropy (one gene affecting more than one trait), heterosis (hybrid vigour), and other interactions between loci and allele
An allele is a variant of the sequence of nucleotides at a particular location, or Locus (genetics), locus, on a DNA molecule.
Alleles can differ at a single position through Single-nucleotide polymorphism, single nucleotide polymorphisms (SNP), ...
s within the genome.
History
Etymology
From the Greek ΓΕΝ ''gen'', "gene" (gamma, epsilon, nu, epsilon) meaning "become, create, creation, birth", and subsequent variants: genealogy, genesis, genetics, genic, genomere, genotype, genus etc. While the word ''genome'' (from the German ''Genom'', attributed to Hans Winkler) was in use in English as early as 1926, the term ''genomics'' was coined by Tom Roderick, a geneticist at the Jackson Laboratory (Bar Harbor, Maine
Bar Harbor () is a resort town on Mount Desert Island in Hancock County, Maine, United States. As of the 2020 census, its population is 5,089. The town is home to the College of the Atlantic, Jackson Laboratory, and MDI Biological Laborat ...
), over beers with James E. Womack, Tom Shows and Stephen O’Brien at a meeting held in Maryland
Maryland ( ) is a U.S. state, state in the Mid-Atlantic (United States), Mid-Atlantic region of the United States. It borders the states of Virginia to its south, West Virginia to its west, Pennsylvania to its north, and Delaware to its east ...
on the mapping of the human genome in 1986. First as the name for a new journal and then as a whole new science discipline.
Early sequencing efforts
Following Rosalind Franklin's confirmation of the helical structure of DNA, James D. Watson and Francis Crick's publication of the structure of DNA in 1953 and Fred Sanger's publication of theAmino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although over 500 amino acids exist in nature, by far the most important are the 22 α-amino acids incorporated into proteins. Only these 22 a ...
sequence of insulin in 1955, nucleic acid sequencing became a major target of early molecular biologists. In 1964, Robert W. Holley and colleagues published the first nucleic acid sequence ever determined, the ribonucleotide sequence of alanine transfer RNA. Extending this work, Marshall Nirenberg and Philip Leder revealed the triplet nature of the genetic code
Genetic code is a set of rules used by living cell (biology), cells to Translation (biology), translate information encoded within genetic material (DNA or RNA sequences of nucleotide triplets or codons) into proteins. Translation is accomplished ...
and were able to determine the sequences of 54 out of 64 codons in their experiments. In 1972, Walter Fiers and his team at the Laboratory of Molecular Biology of the University of Ghent (Ghent
Ghent ( ; ; historically known as ''Gaunt'' in English) is a City status in Belgium, city and a Municipalities of Belgium, municipality in the Flemish Region of Belgium. It is the capital and largest city of the Provinces of Belgium, province ...
, Belgium
Belgium, officially the Kingdom of Belgium, is a country in Northwestern Europe. Situated in a coastal lowland region known as the Low Countries, it is bordered by the Netherlands to the north, Germany to the east, Luxembourg to the southeas ...
) were the first to determine the sequence of a gene: the gene for Bacteriophage MS2 coat protein. Fiers' group expanded on their MS2 coat protein work, determining the complete nucleotide-sequence of bacteriophage MS2-RNA (whose genome encodes just four genes in 3569 base pair
A base pair (bp) is a fundamental unit of double-stranded nucleic acids consisting of two nucleobases bound to each other by hydrogen bonds. They form the building blocks of the DNA double helix and contribute to the folded structure of both DNA ...
s [bp]) and Simian virus 40 in 1976 and 1978, respectively.
DNA-sequencing technology developed
In addition to his seminal work on the amino acid sequence of insulin, Frederick Sanger and his colleagues played a key role in the development of DNA sequencing techniques that enabled the establishment of comprehensive genome sequencing projects. In 1975, he and Alan Coulson published a sequencing procedure using DNA polymerase with radiolabelled nucleotides that he called the ''Plus and Minus technique''. This involved two closely related methods that generated short oligonucleotides with defined 3' termini. These could be fractionated by electrophoresis on a polyacrylamide gel (called polyacrylamide gel electrophoresis) and visualised using autoradiography. The procedure could sequence up to 80 nucleotides in one go and was a big improvement, but was still very laborious. Nevertheless, in 1977 his group was able to sequence most of the 5,386 nucleotides of the single-strandedbacteriophage
A bacteriophage (), also known informally as a phage (), is a virus that infects and replicates within bacteria. The term is derived . Bacteriophages are composed of proteins that Capsid, encapsulate a DNA or RNA genome, and may have structu ...
φX174, completing the first fully sequenced DNA-based genome. The refinement of the ''Plus and Minus'' method resulted in the chain-termination, or Sanger method (see below), which formed the basis of the techniques of DNA sequencing, genome mapping, data storage, and bioinformatic analysis most widely used in the following quarter-century of research. In the same year Walter Gilbert and Allan Maxam of Harvard University
Harvard University is a Private university, private Ivy League research university in Cambridge, Massachusetts, United States. Founded in 1636 and named for its first benefactor, the History of the Puritans in North America, Puritan clergyma ...
independently developed the Maxam-Gilbert method (also known as the ''chemical method'') of DNA sequencing, involving the preferential cleavage of DNA at known bases, a less efficient method. For their groundbreaking work in the sequencing of nucleic acids, Gilbert and Sanger shared half the 1980 Nobel Prize
The Nobel Prizes ( ; ; ) are awards administered by the Nobel Foundation and granted in accordance with the principle of "for the greatest benefit to humankind". The prizes were first awarded in 1901, marking the fifth anniversary of Alfred N ...
in chemistry with Paul Berg (recombinant DNA
Recombinant DNA (rDNA) molecules are DNA molecules formed by laboratory methods of genetic recombination (such as molecular cloning) that bring together genetic material from multiple sources, creating sequences that would not otherwise be fo ...
).
Complete genomes
The advent of these technologies resulted in a rapid intensification in the scope and speed of completion of genome sequencing projects. The first complete genome sequence of a eukaryotic organelle, the humanmitochondrion
A mitochondrion () is an organelle found in the cell (biology), cells of most eukaryotes, such as animals, plants and fungi. Mitochondria have a double lipid bilayer, membrane structure and use aerobic respiration to generate adenosine tri ...
(16,568 bp, about 16.6 kb [kilobase]), was reported in 1981, and the first chloroplast
A chloroplast () is a type of membrane-bound organelle, organelle known as a plastid that conducts photosynthesis mostly in plant cell, plant and algae, algal cells. Chloroplasts have a high concentration of chlorophyll pigments which captur ...
genomes followed in 1986. In 1992, the first eukaryotic chromosome
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most import ...
, chromosome III of brewer's yeast ''Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungal microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have be ...
'' (315 kb) was sequenced. The first free-living organism to be sequenced was that of ''Haemophilus influenzae
''Haemophilus influenzae'' (formerly called Pfeiffer's bacillus or ''Bacillus influenzae'') is a Gram-negative, Motility, non-motile, Coccobacillus, coccobacillary, facultative anaerobic organism, facultatively anaerobic, Capnophile, capnophili ...
'' (1.8 Mb [megabase]) in 1995. The following year a consortium of researchers from laboratories across North America
North America is a continent in the Northern Hemisphere, Northern and Western Hemisphere, Western hemispheres. North America is bordered to the north by the Arctic Ocean, to the east by the Atlantic Ocean, to the southeast by South Ameri ...
, Europe
Europe is a continent located entirely in the Northern Hemisphere and mostly in the Eastern Hemisphere. It is bordered by the Arctic Ocean to the north, the Atlantic Ocean to the west, the Mediterranean Sea to the south, and Asia to the east ...
, and Japan
Japan is an island country in East Asia. Located in the Pacific Ocean off the northeast coast of the Asia, Asian mainland, it is bordered on the west by the Sea of Japan and extends from the Sea of Okhotsk in the north to the East China Sea ...
announced the completion of the first complete genome sequence of a eukaryote, '' S. cerevisiae'' (12.1 Mb), and since then genomes have continued being sequenced at an exponentially growing pace. , the complete sequences are available for: 2,719 virus
A virus is a submicroscopic infectious agent that replicates only inside the living Cell (biology), cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea. Viruses are ...
es, 1,115 archaea
Archaea ( ) is a Domain (biology), domain of organisms. Traditionally, Archaea only included its Prokaryote, prokaryotic members, but this has since been found to be paraphyletic, as eukaryotes are known to have evolved from archaea. Even thou ...
and bacteria
Bacteria (; : bacterium) are ubiquitous, mostly free-living organisms often consisting of one Cell (biology), biological cell. They constitute a large domain (biology), domain of Prokaryote, prokaryotic microorganisms. Typically a few micr ...
, and 36 eukaryote
The eukaryotes ( ) constitute the Domain (biology), domain of Eukaryota or Eukarya, organisms whose Cell (biology), cells have a membrane-bound cell nucleus, nucleus. All animals, plants, Fungus, fungi, seaweeds, and many unicellular organisms ...
s, of which about half are fungi
A fungus (: fungi , , , or ; or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and mold (fungus), molds, as well as the more familiar mushrooms. These organisms are classified as one ...
.
pathogen
In biology, a pathogen (, "suffering", "passion" and , "producer of"), in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a Germ theory of d ...
s, such as ''Haemophilus influenzae
''Haemophilus influenzae'' (formerly called Pfeiffer's bacillus or ''Bacillus influenzae'') is a Gram-negative, Motility, non-motile, Coccobacillus, coccobacillary, facultative anaerobic organism, facultatively anaerobic, Capnophile, capnophili ...
'', which has resulted in a pronounced bias in their phylogenetic distribution compared to the breadth of microbial diversity. Of the other sequenced species, most were chosen because they were well-studied model organisms or promised to become good models. Yeast (''Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungal microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have be ...
'') has long been an important model organism
A model organism is a non-human species that is extensively studied to understand particular biological phenomena, with the expectation that discoveries made in the model organism will provide insight into the workings of other organisms. Mo ...
for the eukaryotic cell, while the fruit fly ''Drosophila melanogaster
''Drosophila melanogaster'' is a species of fly (an insect of the Order (biology), order Diptera) in the family Drosophilidae. The species is often referred to as the fruit fly or lesser fruit fly, or less commonly the "vinegar fly", "pomace fly" ...
'' has been a very important tool (notably in early pre-molecular genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinians, Augustinian ...
). The worm '' Caenorhabditis elegans'' is an often used simple model for multicellular organism
A multicellular organism is an organism that consists of more than one cell (biology), cell, unlike unicellular organisms. All species of animals, Embryophyte, land plants and most fungi are multicellular, as are many algae, whereas a few organism ...
s. The zebrafish '' Brachydanio rerio'' is used for many developmental studies on the molecular level, and the plant '' Arabidopsis thaliana'' is a model organism for flowering plants. The Japanese pufferfish ('' Takifugu rubripes'') and the spotted green pufferfish ('' Tetraodon nigroviridis'') are interesting because of their small and compact genomes, which contain very little noncoding DNA compared to most species. The mammals dog ('' Canis familiaris''), brown rat (''Rattus norvegicus
''Rattus'' is a genus of muroid rodents, all typically called rats. However, the term rat can also be applied to rodent species outside of this genus.
Species and description
The best-known ''Rattus'' species are the black rat (''R. rattus' ...
''), mouse (''Mus musculus
The house mouse (''Mus musculus'') is a small mammal of the rodent family Muridae, characteristically having a pointed snout, large rounded ears, and a long and almost hairless tail. It is one of the most abundant species of the genus ''Mus (genu ...
''), and chimpanzee ('' Pan troglodytes'') are all important model animals in medical research.
A rough draft of the human genome
The human genome is a complete set of nucleic acid sequences for humans, encoded as the DNA within each of the 23 distinct chromosomes in the cell nucleus. A small DNA molecule is found within individual Mitochondrial DNA, mitochondria. These ar ...
was completed by the Human Genome Project in early 2001, creating much fanfare. This project, completed in 2003, sequenced the entire genome for one specific person, and by 2007 this sequence was declared "finished" (less than one error in 20,000 bases and all chromosomes assembled). In the years since then, the genomes of many other individuals have been sequenced, partly under the auspices of the 1000 Genomes Project, which announced the sequencing of 1,092 genomes in October 2012. Completion of this project was made possible by the development of dramatically more efficient sequencing technologies and required the commitment of significant bioinformatics
Bioinformatics () is an interdisciplinary field of science that develops methods and Bioinformatics software, software tools for understanding biological data, especially when the data sets are large and complex. Bioinformatics uses biology, ...
resources from a large international collaboration. The continued analysis of human genomic data has profound political and social repercussions for human societies.
The "omics" revolution
The English-languageneologism
In linguistics, a neologism (; also known as a coinage) is any newly formed word, term, or phrase that has achieved popular or institutional recognition and is becoming accepted into mainstream language. Most definitively, a word can be considered ...
omics informally refers to a field of study in biology ending in ''-omics'', such as genomics, proteomics
Proteomics is the large-scale study of proteins. Proteins are vital macromolecules of all living organisms, with many functions such as the formation of structural fibers of muscle tissue, enzymatic digestion of food, or synthesis and replicatio ...
or metabolomics
Metabolomics is the scientific study of chemical processes involving metabolites, the small molecule substrates, intermediates, and products of cell metabolism. Specifically, metabolomics is the "systematic study of the unique chemical fingerpri ...
. The related suffix -ome is used to address the objects of study of such fields, such as the genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
, proteome
A proteome is the entire set of proteins that is, or can be, expressed by a genome, cell, tissue, or organism at a certain time. It is the set of expressed proteins in a given type of cell or organism, at a given time, under defined conditions. P ...
, or metabolome ( lipidome) respectively. The suffix ''-ome'' as used in molecular biology refers to a ''totality'' of some sort; similarly omics has come to refer generally to the study of large, comprehensive biological data sets. While the growth in the use of the term has led some scientists ( Jonathan Eisen, among others) to claim that it has been oversold, it reflects the change in orientation towards the quantitative analysis of complete or near-complete assortment of all the constituents of a system. In the study of symbioses, for example, researchers which were once limited to the study of a single gene product can now simultaneously compare the total complement of several types of biological molecules.
Genome analysis
After an organism has been selected, genome projects involve three components: the sequencing of DNA, the assembly of that sequence to create a representation of the original chromosome, and the annotation and analysis of that representation.Sequencing
Historically, sequencing was done in ''sequencing centers'', centralized facilities (ranging from large independent institutions such as Joint Genome Institute which sequence dozens of terabases a year, to local molecular biology core facilities) which contain research laboratories with the costly instrumentation and technical support necessary. As sequencing technology continues to improve, however, a new generation of effective fast turnaround benchtop sequencers has come within reach of the average academic laboratory. On the whole, genome sequencing approaches fall into two broad categories, ''shotgun'' and ''high-throughput'' (or ''next-generation'') sequencing.Shotgun sequencing
Shotgun sequencing is a sequencing method designed for analysis of DNA sequences longer than 1000 base pairs, up to and including entire chromosomes. It is named by analogy with the rapidly expanding, quasi-random firing pattern of a shotgun. Since gel electrophoresis sequencing can only be used for fairly short sequences (100 to 1000 base pairs), longer DNA sequences must be broken into random small segments which are then sequenced to obtain ''reads''. Multiple overlapping reads for the target DNA are obtained by performing several rounds of this fragmentation and sequencing. Computer programs then use the overlapping ends of different reads to assemble them into a continuous sequence. Shotgun sequencing is a random sampling process, requiring over-sampling to ensure a givennucleotide
Nucleotides are Organic compound, organic molecules composed of a nitrogenous base, a pentose sugar and a phosphate. They serve as monomeric units of the nucleic acid polymers – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA), both o ...
is represented in the reconstructed sequence; the average number of reads by which a genome is over-sampled is referred to as coverage.
For much of its history, the technology underlying shotgun sequencing was the classical chain-termination method or ' Sanger method', which is based on the selective incorporation of chain-terminating dideoxynucleotides by DNA polymerase
A DNA polymerase is a member of a family of enzymes that catalyze the synthesis of DNA molecules from nucleoside triphosphates, the molecular precursors of DNA. These enzymes are essential for DNA replication and usually work in groups to create t ...
during in vitro
''In vitro'' (meaning ''in glass'', or ''in the glass'') Research, studies are performed with Cell (biology), cells or biological molecules outside their normal biological context. Colloquially called "test-tube experiments", these studies in ...
DNA replication
In molecular biology, DNA replication is the biological process of producing two identical replicas of DNA from one original DNA molecule. DNA replication occurs in all life, living organisms, acting as the most essential part of heredity, biolog ...
. Recently, shotgun sequencing has been supplanted by high-throughput sequencing methods, especially for large-scale, automated genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
analyses. However, the Sanger method remains in wide use, primarily for smaller-scale projects and for obtaining especially long contiguous DNA sequence reads (>500 nucleotides). Chain-termination methods require a single-stranded DNA template, a DNA primer, a DNA polymerase
A DNA polymerase is a member of a family of enzymes that catalyze the synthesis of DNA molecules from nucleoside triphosphates, the molecular precursors of DNA. These enzymes are essential for DNA replication and usually work in groups to create t ...
, normal deoxynucleosidetriphosphates (dNTPs), and modified nucleotides (dideoxyNTPs) that terminate DNA strand elongation. These chain-terminating nucleotides lack a 3'- OH group required for the formation of a phosphodiester bond
In chemistry, a phosphodiester bond occurs when exactly two of the hydroxyl groups () in phosphoric acid react with hydroxyl groups on other molecules to form two ester bonds. The "bond" involves this linkage . Discussion of phosphodiesters is d ...
between two nucleotides, causing DNA polymerase to cease extension of DNA when a ddNTP is incorporated. The ddNTPs may be radioactively or fluorescently labelled for detection in DNA sequencers. Typically, these machines can sequence up to 96 DNA samples in a single batch (run) in up to 48 runs a day.
High-throughput sequencing
The high demand for low-cost sequencing has driven the development of high-throughput sequencing technologies that parallelize the sequencing process, producing thousands or millions of sequences at once. High-throughput sequencing is intended to lower the cost of DNA sequencing beyond what is possible with standard dye-terminator methods. In ultra-high-throughput sequencing, as many as 500,000 sequencing-by-synthesis operations may be run in parallel. The Illumina dye sequencing method is based on reversible dye-terminators and was developed in 1996 at the Geneva Biomedical Research Institute, by Pascal Mayer and Laurent Farinelli. In this method, DNA molecules and primers are first attached on a slide and amplified with
The Illumina dye sequencing method is based on reversible dye-terminators and was developed in 1996 at the Geneva Biomedical Research Institute, by Pascal Mayer and Laurent Farinelli. In this method, DNA molecules and primers are first attached on a slide and amplified with polymerase
In biochemistry, a polymerase is an enzyme (Enzyme Commission number, EC 2.7.7.6/7/19/48/49) that synthesizes long chains of polymers or nucleic acids. DNA polymerase and RNA polymerase are used to assemble DNA and RNA molecules, respectively, by ...
so that local clonal colonies, initially coined "DNA colonies", are formed. To determine the sequence, four types of reversible terminator bases (RT-bases) are added and non-incorporated nucleotides are washed away. Unlike pyrosequencing, the DNA chains are extended one nucleotide at a time and image acquisition can be performed at a delayed moment, allowing for very large arrays of DNA colonies to be captured by sequential images taken from a single camera. Decoupling the enzymatic reaction and the image capture allows for optimal throughput and theoretically unlimited sequencing capacity; with an optimal configuration, the ultimate throughput of the instrument depends only on the A/D conversion rate of the camera. The camera takes images of the fluorescently labeled nucleotides, then the dye along with the terminal 3' blocker is chemically removed from the DNA, allowing the next cycle.
An alternative approach, ion semiconductor sequencing, is based on standard DNA replication chemistry. This technology measures the release of a hydrogen ion each time a base is incorporated. A microwell containing template DNA is flooded with a single nucleotide
Nucleotides are Organic compound, organic molecules composed of a nitrogenous base, a pentose sugar and a phosphate. They serve as monomeric units of the nucleic acid polymers – deoxyribonucleic acid (DNA) and ribonucleic acid (RNA), both o ...
, if the nucleotide is complementary to the template strand it will be incorporated and a hydrogen ion will be released. This release triggers an ISFET ion sensor. If a homopolymer is present in the template sequence multiple nucleotides will be incorporated in a single flood cycle, and the detected electrical signal will be proportionally higher.
Assembly
Sequence assembly refers to aligning and merging fragments of a much longer DNA sequence in order to reconstruct the original sequence. This is needed as currentDNA sequencing
DNA sequencing is the process of determining the nucleic acid sequence – the order of nucleotides in DNA. It includes any method or technology that is used to determine the order of the four bases: adenine, thymine, cytosine, and guanine. The ...
technology cannot read whole genomes as a continuous sequence, but rather reads small pieces of between 20 and 1000 bases, depending on the technology used. Third generation sequencing technologies such as PacBio or Oxford Nanopore routinely generate sequencing reads 10-100 kb in length; however, they have a high error rate at approximately 1 percent. Typically the short fragments, called reads, result from shotgun sequencing genomic DNA, or gene transcripts ( ESTs).
Assembly approaches
Assembly can be broadly categorized into two approaches: ''de novo'' assembly, for genomes which are not similar to any sequenced in the past, and comparative assembly, which uses the existing sequence of a closely related organism as a reference during assembly. Relative to comparative assembly, ''de novo'' assembly is computationally difficult ( NP-hard), making it less favourable for short-read NGS technologies. Within the ''de novo'' assembly paradigm there are two primary strategies for assembly, Eulerian path strategies, and overlap-layout-consensus (OLC) strategies. OLC strategies ultimately try to create a Hamiltonian path through an overlap graph which is an NP-hard problem. Eulerian path strategies are computationally more tractable because they try to find a Eulerian path through a deBruijn graph.Finishing
Finished genomes are defined as having a single contiguous sequence with no ambiguities representing each replicon.Annotation
The DNA sequence assembly alone is of little value without additional analysis. Genome annotation is the process of attaching biological information to sequences, and consists of three main steps: # identifying portions of the genome that do not code for proteins # identifying elements on thegenome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
, a process called gene prediction, and
# attaching biological information to these elements.
Automatic annotation tools try to perform these steps '' in silico'', as opposed to manual annotation (a.k.a. curation) which involves human expertise and potential experimental verification. Ideally, these approaches co-exist and complement each other in the same annotation pipeline
A pipeline is a system of Pipe (fluid conveyance), pipes for long-distance transportation of a liquid or gas, typically to a market area for consumption. The latest data from 2014 gives a total of slightly less than of pipeline in 120 countries ...
(also see below).
Traditionally, the basic level of annotation is using BLAST for finding similarities, and then annotating genomes based on homologues. More recently, additional information is added to the annotation platform. The additional information allows manual annotators to deconvolute discrepancies between genes that are given the same annotation. Some databases use genome context information, similarity scores, experimental data, and integrations of other resources to provide genome annotations through their Subsystems approach. Other databases (e.g. Ensembl) rely on both curated data sources as well as a range of software tools in their automated genome annotation pipeline. ''Structural annotation'' consists of the identification of genomic elements, primarily ORFs and their localisation, or gene structure. ''Functional annotation'' consists of attaching biological information to genomic elements.
Sequencing pipelines and databases
The need for reproducibility and efficient management of the large amount of data associated with genome projects mean that computational pipelines have important applications in genomics.Research areas
Functional genomics
Functional genomics is a field ofmolecular biology
Molecular biology is a branch of biology that seeks to understand the molecule, molecular basis of biological activity in and between Cell (biology), cells, including biomolecule, biomolecular synthesis, modification, mechanisms, and interactio ...
that attempts to make use of the vast wealth of data produced by genomic projects (such as genome sequencing projects) to describe gene
In biology, the word gene has two meanings. The Mendelian gene is a basic unit of heredity. The molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protei ...
(and protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residue (biochemistry), residues. Proteins perform a vast array of functions within organisms, including Enzyme catalysis, catalysing metab ...
) functions and interactions. Functional genomics focuses on the dynamic aspects such as gene transcription, translation
Translation is the communication of the semantics, meaning of a #Source and target languages, source-language text by means of an Dynamic and formal equivalence, equivalent #Source and target languages, target-language text. The English la ...
, and protein–protein interactions, as opposed to the static aspects of the genomic information such as DNA sequence or structures. Functional genomics attempts to answer questions about the function of DNA at the levels of genes, RNA transcripts, and protein products. A key characteristic of functional genomics studies is their genome-wide approach to these questions, generally involving high-throughput methods rather than a more traditional "gene-by-gene" approach.
A major branch of genomics is still concerned with sequencing the genomes of various organisms, but the knowledge of full genomes has created the possibility for the field of functional genomics, mainly concerned with patterns of gene expression
Gene expression is the process (including its Regulation of gene expression, regulation) by which information from a gene is used in the synthesis of a functional gene product that enables it to produce end products, proteins or non-coding RNA, ...
during various conditions. The most important tools here are microarray
A microarray is a multiplex (assay), multiplex lab-on-a-chip. Its purpose is to simultaneously detect the expression of thousands of biological interactions. It is a two-dimensional array on a Substrate (materials science), solid substrate—usu ...
s and bioinformatics
Bioinformatics () is an interdisciplinary field of science that develops methods and Bioinformatics software, software tools for understanding biological data, especially when the data sets are large and complex. Bioinformatics uses biology, ...
.
Structural genomics
Structural genomics seeks to describe the 3-dimensional structure of every protein encoded by a givengenome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
. This genome-based approach allows for a high-throughput method of structure determination by a combination of experimental and modeling approaches. The principal difference between structural genomics and traditional structural prediction is that structural genomics attempts to determine the structure of every protein encoded by the genome, rather than focusing on one particular protein. With full-genome sequences available, structure prediction can be done more quickly through a combination of experimental and modeling approaches, especially because the availability of large numbers of sequenced genomes and previously solved protein structures allow scientists to model protein structure on the structures of previously solved homologs. Structural genomics involves taking a large number of approaches to structure determination, including experimental methods using genomic sequences or modeling-based approaches based on sequence or structural homology to a protein of known structure or based on chemical and physical principles for a protein with no homology to any known structure. As opposed to traditional structural biology
Structural biology deals with structural analysis of living material (formed, composed of, and/or maintained and refined by living cells) at every level of organization.
Early structural biologists throughout the 19th and early 20th centuries we ...
, the determination of a protein structure through a structural genomics effort often (but not always) comes before anything is known regarding the protein function. This raises new challenges in structural bioinformatics, i.e. determining protein function from its 3D structure.
Epigenomics
Epigenomics is the study of the complete set of epigenetic modifications on the genetic material of a cell, known as the epigenome. Epigenetic modifications are reversible modifications on a cell's DNA or histones that affect gene expression without altering the DNA sequence (Russell 2010 p. 475). Two of the most characterized epigenetic modifications are DNA methylation and histone modification. Epigenetic modifications play an important role in gene expression and regulation, and are involved in numerous cellular processes such as in differentiation/development and tumorigenesis. The study of epigenetics on a global level has been made possible only recently through the adaptation of genomic high-throughput assays.Metagenomics
 Metagenomics is the study of ''metagenomes'', genetic material recovered directly from environmental samples. The broad field may also be referred to as environmental genomics, ecogenomics or community genomics. While traditional
Metagenomics is the study of ''metagenomes'', genetic material recovered directly from environmental samples. The broad field may also be referred to as environmental genomics, ecogenomics or community genomics. While traditional microbiology
Microbiology () is the branches of science, scientific study of microorganisms, those being of unicellular organism, unicellular (single-celled), multicellular organism, multicellular (consisting of complex cells), or non-cellular life, acellula ...
and microbial genome sequencing rely upon cultivated clonal cultures
Culture ( ) is a concept that encompasses the social behavior, institutions, and Social norm, norms found in human societies, as well as the knowledge, beliefs, arts, laws, Social norm, customs, capabilities, Attitude (psychology), attitudes ...
, early environmental gene sequencing cloned specific genes (often the 16S rRNA
16S ribosomal RNA (or 16Svedberg, S rRNA) is the RNA component of the 30S subunit of a prokaryotic ribosome (SSU rRNA). It binds to the Shine-Dalgarno sequence and provides most of the SSU structure.
The genes coding for it are referred to as ...
gene) to produce a profile of diversity in a natural sample. Such work revealed that the vast majority of microbial biodiversity had been missed by cultivation-based methods. Recent studies use "shotgun" Sanger sequencing or massively parallel pyrosequencing to get largely unbiased samples of all genes from all the members of the sampled communities. Because of its power to reveal the previously hidden diversity of microscopic life, metagenomics offers a powerful lens for viewing the microbial world that has the potential to revolutionize understanding of the entire living world.
Model systems
Viruses and bacteriophages
Bacteriophage
A bacteriophage (), also known informally as a phage (), is a virus that infects and replicates within bacteria. The term is derived . Bacteriophages are composed of proteins that Capsid, encapsulate a DNA or RNA genome, and may have structu ...
s have played and continue to play a key role in bacterial genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinians, Augustinian ...
and molecular biology
Molecular biology is a branch of biology that seeks to understand the molecule, molecular basis of biological activity in and between Cell (biology), cells, including biomolecule, biomolecular synthesis, modification, mechanisms, and interactio ...
. Historically, they were used to define gene
In biology, the word gene has two meanings. The Mendelian gene is a basic unit of heredity. The molecular gene is a sequence of nucleotides in DNA that is transcribed to produce a functional RNA. There are two types of molecular genes: protei ...
structure and gene regulation. Also the first genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
to be sequenced was a bacteriophage
A bacteriophage (), also known informally as a phage (), is a virus that infects and replicates within bacteria. The term is derived . Bacteriophages are composed of proteins that Capsid, encapsulate a DNA or RNA genome, and may have structu ...
. However, bacteriophage research did not lead the genomics revolution, which is clearly dominated by bacterial genomics. Only very recently has the study of bacteriophage genomes become prominent, thereby enabling researchers to understand the mechanisms underlying phage evolution. Bacteriophage genome sequences can be obtained through direct sequencing of isolated bacteriophages, but can also be derived as part of microbial genomes. Analysis of bacterial genomes has shown that a substantial amount of microbial DNA consists of prophage
A prophage is a bacteriophage (often shortened to "phage") genome that is integrated into the circular bacterial chromosome or exists as an extrachromosomal plasmid within the bacterial cell (biology), cell. Integration of prophages into the bacte ...
sequences and prophage-like elements. A detailed database mining of these sequences offers insights into the role of prophages in shaping the bacterial genome: Overall, this method verified many known bacteriophage groups, making this a useful tool for predicting the relationships of prophages from bacterial genomes.
Cyanobacteria
At present there are 24cyanobacteria
Cyanobacteria ( ) are a group of autotrophic gram-negative bacteria that can obtain biological energy via oxygenic photosynthesis. The name "cyanobacteria" () refers to their bluish green (cyan) color, which forms the basis of cyanobacteri ...
for which a total genome sequence is available. 15 of these cyanobacteria come from the marine environment. These are six '' Prochlorococcus'' strains, seven marine '' Synechococcus'' strains, '' Trichodesmium erythraeum'' IMS101 and '' Crocosphaera watsonii'' WH8501. Several studies have demonstrated how these sequences could be used very successfully to infer important ecological and physiological characteristics of marine cyanobacteria. However, there are many more genome projects currently in progress, amongst those there are further '' Prochlorococcus'' and marine '' Synechococcus'' isolates, '' Acaryochloris'' and '' Prochloron'', the N2-fixing filamentous cyanobacteria '' Nodularia spumigena'', '' Lyngbya aestuarii'' and '' Lyngbya majuscula'', as well as bacteriophage
A bacteriophage (), also known informally as a phage (), is a virus that infects and replicates within bacteria. The term is derived . Bacteriophages are composed of proteins that Capsid, encapsulate a DNA or RNA genome, and may have structu ...
s infecting marine cyanobaceria. Thus, the growing body of genome information can also be tapped in a more general way to address global problems by applying a comparative approach. Some new and exciting examples of progress in this field are the identification of genes for regulatory RNAs, insights into the evolutionary origin of photosynthesis
Photosynthesis ( ) is a system of biological processes by which photosynthetic organisms, such as most plants, algae, and cyanobacteria, convert light energy, typically from sunlight, into the chemical energy necessary to fuel their metabo ...
, or estimation of the contribution of horizontal gene transfer
Horizontal gene transfer (HGT) or lateral gene transfer (LGT) is the movement of genetic material between organisms other than by the ("vertical") transmission of DNA from parent to offspring (reproduction). HGT is an important factor in the e ...
to the genomes that have been analyzed.
Applications
 Genomics has provided applications in many fields, including
Genomics has provided applications in many fields, including medicine
Medicine is the science and Praxis (process), practice of caring for patients, managing the Medical diagnosis, diagnosis, prognosis, Preventive medicine, prevention, therapy, treatment, Palliative care, palliation of their injury or disease, ...
, biotechnology
Biotechnology is a multidisciplinary field that involves the integration of natural sciences and Engineering Science, engineering sciences in order to achieve the application of organisms and parts thereof for products and services. Specialists ...
, anthropology
Anthropology is the scientific study of humanity, concerned with human behavior, human biology, cultures, society, societies, and linguistics, in both the present and past, including archaic humans. Social anthropology studies patterns of behav ...
and other social sciences
Social science (often rendered in the plural as the social sciences) is one of the branches of science, devoted to the study of society, societies and the Social relation, relationships among members within those societies. The term was former ...
.
Genomic medicine
Next-generation genomic technologies allow clinicians and biomedical researchers to drastically increase the amount of genomic data collected on large study populations. When combined with new informatics approaches that integrate many kinds of data with genomic data in disease research, this allows researchers to better understand the genetic bases of drug response and disease. Early efforts to apply the genome to medicine included those by a Stanford team led by Euan Ashley who developed the first tools for the medical interpretation of a human genome. The Genomes2People research program at Brigham and Women’s Hospital, Broad Institute and Harvard Medical School was established in 2012 to conduct empirical research in translating genomics into health. Brigham and Women's Hospital opened a Preventive Genomics Clinic in August 2019, with Massachusetts General Hospital following a month later. The ''All of Us'' research program aims to collect genome sequence data from 1 million participants to become a critical component of the precision medicine research platform and the ''UK Biobank'' initiative has studied more than 500.000 individuals with deep genomic and phenotypic data.Synthetic biology and bioengineering
The growth of genomic knowledge has enabled increasingly sophisticated applications ofsynthetic biology
Synthetic biology (SynBio) is a multidisciplinary field of science that focuses on living systems and organisms. It applies engineering principles to develop new biological parts, devices, and systems or to redesign existing systems found in nat ...
. In 2010 researchers at the J. Craig Venter Institute announced the creation of a partially synthetic species of bacterium, '' Mycoplasma laboratorium'', derived from the genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
of '' Mycoplasma genitalium''.
Population and conservation genomics
Population genomics has developed as a popular field of research, where genomic sequencing methods are used to conduct large-scale comparisons of DNA sequences among populations - beyond the limits of genetic markers such as short-range PCR products or microsatellites traditionally used inpopulation genetics
Population genetics is a subfield of genetics that deals with genetic differences within and among populations, and is a part of evolutionary biology. Studies in this branch of biology examine such phenomena as Adaptation (biology), adaptation, s ...
. Population genomics studies genome
A genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding genes, other functional regions of the genome such as ...
-wide effects to improve our understanding of microevolution so that we may learn the phylogenetic
In biology, phylogenetics () is the study of the evolutionary history of life using observable characteristics of organisms (or genes), which is known as phylogenetic inference. It infers the relationship among organisms based on empirical dat ...
history and demography
Demography () is the statistical study of human populations: their size, composition (e.g., ethnic group, age), and how they change through the interplay of fertility (births), mortality (deaths), and migration.
Demographic analysis examine ...
of a population. Population genomic methods are used for many different fields including evolutionary biology
Evolutionary biology is the subfield of biology that studies the evolutionary processes such as natural selection, common descent, and speciation that produced the diversity of life on Earth. In the 1930s, the discipline of evolutionary biolo ...
, ecology
Ecology () is the natural science of the relationships among living organisms and their Natural environment, environment. Ecology considers organisms at the individual, population, community (ecology), community, ecosystem, and biosphere lev ...
, biogeography
Biogeography is the study of the species distribution, distribution of species and ecosystems in geography, geographic space and through evolutionary history of life, geological time. Organisms and biological community (ecology), communities o ...
, conservation biology
Conservation biology is the study of the conservation of nature and of Earth's biodiversity with the aim of protecting species, their habitats, and ecosystems from excessive rates of extinction and the erosion of biotic interactions. It is an i ...
and fisheries management. Similarly, landscape genomics has developed from landscape genetics to use genomic methods to identify relationships between patterns of environmental and genetic variation.
Conservationists can use the information gathered by genomic sequencing in order to better evaluate genetic factors key to species conservation, such as the genetic diversity
Genetic diversity is the total number of genetic characteristics in the genetic makeup of a species. It ranges widely, from the number of species to differences within species, and can be correlated to the span of survival for a species. It is d ...
of a population or whether an individual is heterozygous for a recessive inherited genetic disorder. By using genomic data to evaluate the effects of evolutionary processes and to detect patterns in variation throughout a given population, conservationists can formulate plans to aid a given species without as many variables left unknown as those unaddressed by standard genetic approaches.
See also
* Hi-C (genomic analysis technique) * Cognitive genomics * Computational genomics * Epigenomics * Functional genomics * GeneCalling, an mRNA profiling technology * Genomics of domestication * Genetics in fiction * Glycomics * Immunomics * Metagenomics * Pathogenomics * Personal genomics *Proteomics
Proteomics is the large-scale study of proteins. Proteins are vital macromolecules of all living organisms, with many functions such as the formation of structural fibers of muscle tissue, enzymatic digestion of food, or synthesis and replicatio ...
* Transcriptomics
* Venomics
Venomics is the study of proteins associated with ''venom'', a toxic substance secreted by animals, which is typically injected either offensively or defensively into prey or aggressors, respectively.
Background
Venom is produced in a speciali ...
* Psychogenomics
* Whole genome sequencing
Whole genome sequencing (WGS), also known as full genome sequencing or just genome sequencing, is the process of determining the entirety of the DNA sequence of an organism's genome at a single time. This entails sequencing all of an organism's ...
* Thomas Roderick
References
Further reading
* * * * * electronic-book electronic-External links
Annual Review of Genomics and Human Genetics
BMC Genomics
A BMC journal on Genomics
Genomics journal
Genomics.org
An openfree genomics portal.
NHGRI
US government's genome institute
JCVI Comprehensive Microbial Resource
KoreaGenome.org
The first Korean Genome published and the sequence is available freely.
GenomicsNetwork
Looks at the development and use of the science and technologies of genomics.
Institute for Genome Sciences
Genomics research.
MIT OpenCourseWare HST.512 Genomic Medicine
A free, self-study course in genomic medicine. Resources include audio lectures and selected lecture notes.
ENCODE threads explorer
Machine learning approaches to genomics.
Nature (journal)
''Nature'' is a British weekly scientific journal founded and based in London, England. As a multidisciplinary publication, ''Nature'' features Peer review, peer-reviewed research from a variety of academic disciplines, mainly in science and t ...
Global map of genomics laboratories
Genomics: Scitable by nature education
Learn All About Genetics Online
{{Authority control