Epistasis on:
[Wikipedia]
[Google]
[Amazon]
 Epistasis is a phenomenon in
Epistasis is a phenomenon in
 Terminology about epistasis can vary between scientific fields. Geneticists often refer to wild type and mutant
Terminology about epistasis can vary between scientific fields. Geneticists often refer to wild type and mutant
 In evolutionary genetics, the sign of epistasis is usually more significant than the magnitude of epistasis. This is because magnitude epistasis (positive and negative) simply affects how beneficial mutations are together, however sign epistasis affects whether mutation combinations are beneficial or deleterious.
A fitness landscape is a representation of the fitness where all genotypes are arranged in 2D space and the fitness of each genotype is represented by height on a surface. It is frequently used as a visual metaphor for understanding
In evolutionary genetics, the sign of epistasis is usually more significant than the magnitude of epistasis. This is because magnitude epistasis (positive and negative) simply affects how beneficial mutations are together, however sign epistasis affects whether mutation combinations are beneficial or deleterious.
A fitness landscape is a representation of the fitness where all genotypes are arranged in 2D space and the fitness of each genotype is represented by height on a surface. It is frequently used as a visual metaphor for understanding
INTERSNP
- a software for genome-wide interaction analysis (GWIA) of case-control and case-only SNP data, including analysis of quantitative traits.
High school (GCSE, Alevel) resource.
GeneticInteractions.orgEpistasis.org
{{Authority control Classical genetics Genetics concepts
 Epistasis is a phenomenon in
Epistasis is a phenomenon in genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinian friar work ...
in which the effect of a gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, m ...
is dependent on the presence or absence of mutations in one or more other genes, respectively termed modifier genes. In other words, the effect of the mutation is dependent on the genetic background in which it appears. Epistatic mutations therefore have different effects on their own than when they occur together. Originally, the term ''epistasis'' specifically meant that the effect of a gene variant is masked by that of a different gene.
The concept of ''epistasis'' originated in genetics in 1907 but is now used in biochemistry
Biochemistry or biological chemistry is the study of chemical processes within and relating to living organisms. A sub-discipline of both chemistry and biology, biochemistry may be divided into three fields: structural biology, enzymology and ...
, computational biology and evolutionary biology
Evolutionary biology is the subfield of biology that studies the evolutionary processes (natural selection, common descent, speciation) that produced the diversity of life on Earth. It is also defined as the study of the history of life ...
. The phenomenon arises due to interactions, either between genes (such as mutations also being needed in regulators of gene expression) or within them (multiple mutations being needed before the gene loses function), leading to non-linear effects. Epistasis has a great influence on the shape of evolutionary landscapes, which leads to profound consequences for evolution
Evolution is change in the heritable characteristics of biological populations over successive generations. These characteristics are the expressions of genes, which are passed on from parent to offspring during reproduction. Variation ...
and for the evolvability of phenotypic
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology or physical form and structure, its developmental processes, its biochemical and physiological pr ...
traits.
History
Understanding of epistasis has changed considerably through the history of genetics and so too has the use of the term. The term was first used by William Bateson and his collaborators Florence Durham and Muriel Wheldale Onslow. In early models ofnatural selection
Natural selection is the differential survival and reproduction of individuals due to differences in phenotype. It is a key mechanism of evolution, the change in the heritable traits characteristic of a population over generations. Cha ...
devised in the early 20th century, each gene was considered to make its own characteristic contribution to fitness, against an average background of other genes. Some introductory courses still teach population genetics
Population genetics is a subfield of genetics that deals with genetic differences within and between populations, and is a part of evolutionary biology. Studies in this branch of biology examine such phenomena as adaptation, speciation, and po ...
this way. Because of the way that the science of population genetics
Population genetics is a subfield of genetics that deals with genetic differences within and between populations, and is a part of evolutionary biology. Studies in this branch of biology examine such phenomena as adaptation, speciation, and po ...
was developed, evolutionary geneticists have tended to think of epistasis as the exception. However, in general, the expression of any one allele depends in a complicated way on many other alleles.
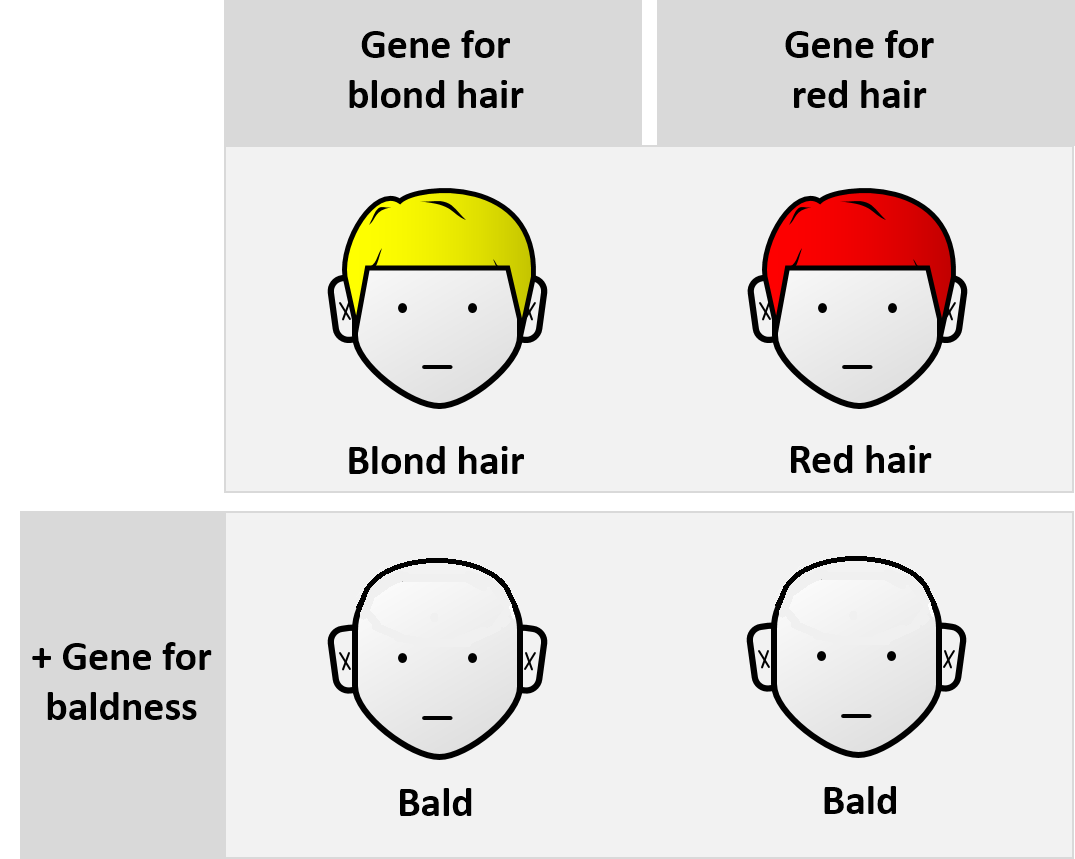
In classical genetics, if genes A and B are mutated, and each mutation by itself produces a unique phenotype but the two mutations together show the same phenotype as the gene A mutation, then gene A is epistatic and gene B is hypostatic. For example, the gene for total baldness is epistatic to the gene for brown hair
Brown hair, also referred to as brunet (male) or brunette (female), is the second most common human hair color, after black hair. It varies from light brown to a medium dark hair. It is characterized by higher levels of the dark pigment eumel ...
. In this sense, epistasis can be contrasted with genetic dominance, which is an interaction
Interaction is action that occurs between two or more objects, with broad use in philosophy and the sciences. It may refer to:
Science
* Interaction hypothesis, a theory of second language acquisition
* Interaction (statistics)
* Interaction ...
between alleles at the same gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
locus. As the study of genetics developed, and with the advent of molecular biology
Molecular biology is the branch of biology that seeks to understand the molecular basis of biological activity in and between cells, including biomolecular synthesis, modification, mechanisms, and interactions. The study of chemical and phys ...
, epistasis started to be studied in relation to quantitative trait loci (QTL) and polygenic inheritance
A quantitative trait locus (QTL) is a locus (section of DNA) that correlates with variation of a quantitative trait in the phenotype of a population of organisms. QTLs are mapped by identifying which molecular markers (such as SNPs or AFLPs ...
.
The effects of genes are now commonly quantifiable by assaying the magnitude of a phenotype (e.g. height
Height is measure of vertical distance, either vertical extent (how "tall" something or someone is) or vertical position (how "high" a point is).
For example, "The height of that building is 50 m" or "The height of an airplane in-flight is ab ...
, pigmentation or growth rate) or by biochemically assaying protein activity (e.g. binding or catalysis
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
). Increasingly sophisticated computational and evolutionary biology
Evolutionary biology is the subfield of biology that studies the evolutionary processes (natural selection, common descent, speciation) that produced the diversity of life on Earth. It is also defined as the study of the history of life ...
models aim to describe the effects of epistasis on a genome
In the fields of molecular biology and genetics, a genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding ...
-wide scale and the consequences of this for evolution
Evolution is change in the heritable characteristics of biological populations over successive generations. These characteristics are the expressions of genes, which are passed on from parent to offspring during reproduction. Variation ...
. Since identification of epistatic pairs is challenging both computationally and statistically, some studies try to prioritize epistatic pairs.
Classification
 Terminology about epistasis can vary between scientific fields. Geneticists often refer to wild type and mutant
Terminology about epistasis can vary between scientific fields. Geneticists often refer to wild type and mutant allele
An allele (, ; ; modern formation from Greek ἄλλος ''állos'', "other") is a variation of the same sequence of nucleotides at the same place on a long DNA molecule, as described in leading textbooks on genetics and evolution.
::"The chrom ...
s where the mutation is implicitly deleterious and may talk in terms of genetic enhancement, synthetic lethality and genetic suppressors. Conversely, a biochemist may more frequently focus on beneficial mutations and so explicitly state the effect of a mutation and use terms such as reciprocal sign epistasis and compensatory mutation. Additionally, there are differences when looking at epistasis within a single gene (biochemistry) and epistasis within a haploid or diploid
Ploidy () is the number of complete sets of chromosomes in a cell, and hence the number of possible alleles for autosomal and pseudoautosomal genes. Sets of chromosomes refer to the number of maternal and paternal chromosome copies, respectiv ...
genome (genetics). In general, epistasis is used to denote the departure from 'independence' of the effects of different genetic loci. Confusion often arises due to the varied interpretation of 'independence' among different branches of biology. The classifications below attempt to cover the various terms and how they relate to one another.
Additivity
Two mutations are considered to be purely additive if the effect of the double mutation is the sum of the effects of the single mutations. This occurs when genes do not interact with each other, for example by acting through differentmetabolic pathway
In biochemistry, a metabolic pathway is a linked series of chemical reactions occurring within a cell. The reactants, products, and intermediates of an enzymatic reaction are known as metabolites, which are modified by a sequence of chemical ...
s. Simply, additive traits were studied early on in the history of genetics, however they are relatively rare, with most genes exhibiting at least some level of epistatic interaction.
Magnitude epistasis
When the double mutation has a fitterphenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology (biology), morphology or physical form and structure, its Developmental biology, developmental proc ...
than expected from the effects of the two single mutations, it is referred to as positive epistasis. Positive epistasis between beneficial mutations generates greater improvements in function than expected. Positive epistasis between deleterious mutations protects against the negative effects to cause a less severe fitness drop.
Conversely, when two mutations together lead to a less fit phenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology (biology), morphology or physical form and structure, its Developmental biology, developmental proc ...
than expected from their effects when alone, it is called negative epistasis. Negative epistasis between beneficial mutations causes smaller than expected fitness improvements, whereas negative epistasis between deleterious mutations causes greater-than-additive fitness drops.
Independently, when the effect on fitness of two mutations is more radical than expected from their effects when alone, it is referred to as synergistic epistasis. The opposite situation, when the fitness difference of the double mutant from the wild type is smaller than expected from the effects of the two single mutations, it is called antagonistic epistasis. Therefore, for deleterious mutations, negative epistasis is also synergistic, while positive epistasis is antagonistic; conversely, for advantageous mutations, positive epistasis is synergistic, while negative epistasis is antagonistic.
The term genetic enhancement is sometimes used when a double (deleterious) mutant has a more severe phenotype than the additive effects of the single mutants. Strong positive epistasis is sometimes referred to by creationists as irreducible complexity (although most examples are misidentified).
Sign epistasis
Sign epistasis occurs when one mutation has the opposite effect when in the presence of another mutation. This occurs when a mutation that is deleterious on its own can enhance the effect of a particular beneficial mutation. For example, a large and complexbrain
A brain is an organ (biology), organ that serves as the center of the nervous system in all vertebrate and most invertebrate animals. It is located in the head, usually close to the sensory organs for senses such as Visual perception, vision. I ...
is a waste of energy without a range of sense organs, but sense organs are made more useful by a large and complex brain that can better process the information. If a fitness landscape has no sign epistasis then it is called ''smooth''.
At its most extreme, reciprocal sign epistasis occurs when two deleterious genes are beneficial when together. For example, producing a toxin
A toxin is a naturally occurring organic poison produced by metabolic activities of living cells or organisms. Toxins occur especially as a protein or conjugated protein. The term toxin was first used by organic chemist Ludwig Brieger (1849 ...
alone can kill a bacterium
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were am ...
, and producing a toxin exporter alone can waste energy, but producing both can improve fitness by killing competing organisms. If a fitness landscape has sign epistasis but no reciprocal sign epistasis then it is called ''semismooth''.
Reciprocal sign epistasis also leads to genetic suppression whereby two deleterious mutations are less harmful together than either one on its own, i.e. one compensates for the other. A clear example of genetic suppression was the demonstration that in the assembly of bacteriophage T4 two deleterious mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, m ...
s, each causing a deficiency in the level of a different morphogenetic
Morphogenesis (from the Greek ''morphê'' shape and ''genesis'' creation, literally "the generation of form") is the biological process that causes a cell, tissue or organism to develop its shape. It is one of three fundamental aspects of deve ...
protein, could interact positively. If a mutation causes a reduction in a particular structural component, this can bring about an imbalance in morphogenesis and loss of viable virus progeny, but production of viable progeny can be restored by a second (suppressor) mutation in another morphogenetic component that restores the balance of protein components.
The term genetic suppression can also apply to sign epistasis where the double mutant has a phenotype intermediate between those of the single mutants, in which case the more severe single mutant phenotype is suppressed by the other mutation or genetic condition. For example, in a diploid
Ploidy () is the number of complete sets of chromosomes in a cell, and hence the number of possible alleles for autosomal and pseudoautosomal genes. Sets of chromosomes refer to the number of maternal and paternal chromosome copies, respectiv ...
organism, a hypomorphic (or partial loss-of-function) mutant phenotype can be suppressed by knocking out one copy of a gene that acts oppositely in the same pathway. In this case, the second gene is described as a "dominant suppressor" of the hypomorphic mutant; "dominant" because the effect is seen when one wild-type copy of the suppressor gene is present (i.e. even in a heterozygote). For most genes, the phenotype of the heterozygous suppressor mutation by itself would be wild type (because most genes are not haplo-insufficient), so that the double mutant (suppressed) phenotype is intermediate between those of the single mutants.
In non reciprocal sign epistasis, fitness of the mutant lies in the middle of that of the extreme effects seen in reciprocal sign epistasis.
When two mutations are viable alone but lethal in combination, it is called Synthetic lethality or unlinked non-complementation.
Haploid organisms
In a haploid organism with genotypes (at two loci) ''ab'', ''Ab'', ''aB'' or ''AB'', we can think of different forms of epistasis as affecting the magnitude of a phenotype upon mutation individually (Ab and aB) or in combination (AB).Diploid organisms
Epistasis indiploid
Ploidy () is the number of complete sets of chromosomes in a cell, and hence the number of possible alleles for autosomal and pseudoautosomal genes. Sets of chromosomes refer to the number of maternal and paternal chromosome copies, respectiv ...
organisms is further complicated by the presence of two copies of each gene. Epistasis can occur between loci, but additionally, interactions can occur between the two copies of each locus in heterozygote
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
M ...
s. For a two locus, two allele
An allele (, ; ; modern formation from Greek ἄλλος ''állos'', "other") is a variation of the same sequence of nucleotides at the same place on a long DNA molecule, as described in leading textbooks on genetics and evolution.
::"The chrom ...
system, there are eight independent types of gene interaction.
Genetic and molecular causes
Additivity
This can be the case when multiple genes act in parallel to achieve the same effect. For example, when an organism is in need ofphosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ea ...
, multiple enzymes that break down different phosphorylated components from the environment
Environment most often refers to:
__NOTOC__
* Natural environment, all living and non-living things occurring naturally
* Biophysical environment, the physical and biological factors along with their chemical interactions that affect an organism or ...
may act additively to increase the amount of phosphorus available to the organism. However, there inevitably comes a point where phosphorus is no longer the limiting factor for growth and reproduction and so further improvements in phosphorus metabolism have smaller or no effect (negative epistasis). Some sets of mutations within genes have also been specifically found to be additive. It is now considered that strict additivity is the exception, rather than the rule, since most genes interact
Advocates for Informed Choice, doing business as, dba interACT or interACT Advocates for Intersex Youth, is a 501(c)(3) nonprofit organization using innovative strategies to advocate for the legal and human rights of children with intersex trai ...
with hundreds or thousands of other genes.
Epistasis between genes
Epistasis within the genomes of organisms occurs due to interactions between the genes within the genome. This interaction may be direct if the genes encode proteins that, for example, are separate components of a multi-component protein (such as the ribosome), inhibit each other's activity, or if the protein encoded by one gene modifies the other (such as byphosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
). Alternatively the interaction may be indirect, where the genes encode components of a metabolic pathway
In biochemistry, a metabolic pathway is a linked series of chemical reactions occurring within a cell. The reactants, products, and intermediates of an enzymatic reaction are known as metabolites, which are modified by a sequence of chemical ...
or network
Network, networking and networked may refer to:
Science and technology
* Network theory, the study of graphs as a representation of relations between discrete objects
* Network science, an academic field that studies complex networks
Mathematic ...
, developmental pathway, signalling pathway or transcription factor
In molecular biology, a transcription factor (TF) (or sequence-specific DNA-binding factor) is a protein that controls the rate of transcription of genetic information from DNA to messenger RNA, by binding to a specific DNA sequence. The f ...
network. For example, the gene encoding the enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
that synthesizes penicillin
Penicillins (P, PCN or PEN) are a group of β-lactam antibiotics originally obtained from '' Penicillium'' moulds, principally '' P. chrysogenum'' and '' P. rubens''. Most penicillins in clinical use are synthesised by P. chrysogenum usin ...
is of no use to a fungus
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately fr ...
without the enzymes that synthesize the necessary precursors in the metabolic pathway.
Epistasis within genes
Just as mutations in two separate genes can be non-additive if those genes interact, mutations in two codons within a gene can be non-additive. In genetics this is sometimes called intragenic suppression when one deleterious mutation can be compensated for by a second mutation within that gene. Analysis of bacteriophage T4mutant
In biology, and especially in genetics, a mutant is an organism or a new genetic character arising or resulting from an instance of mutation, which is generally an alteration of the DNA sequence of the genome or chromosome of an organism. It ...
s that were altered in the rIIB cistron (gene) revealed that certain pairwise combinations of mutations could mutually suppress each other; that is the double mutants had a more nearly wild-type phenotype
In genetics, the phenotype () is the set of observable characteristics or traits of an organism. The term covers the organism's morphology (biology), morphology or physical form and structure, its Developmental biology, developmental proc ...
than either mutant alone.Crick FH, Barnett L, Brenner S, Watts-Tobin RJ. General nature of the genetic code for proteins. Nature. 1961 Dec 30;192:1227-32. doi: 10.1038/1921227a0. PMID: 13882203 The linear map order of the mutants was established using genetic recombination
Genetic recombination (also known as genetic reshuffling) is the exchange of genetic material between different organisms which leads to production of offspring with combinations of traits that differ from those found in either parent. In eukaryo ...
data, From these sources of information, the triplet nature of the genetic code was logically deduced for the first time in 1961, and other key features of the code were also inferred.
Also intragenic suppression can occur when the amino acids within a protein interact. Due to the complexity of protein folding and activity, additive mutations are rare.
Proteins are held in their tertiary structure by a distributed, internal network of cooperative interactions (hydrophobic
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, ...
, polar
Polar may refer to:
Geography
Polar may refer to:
* Geographical pole, either of two fixed points on the surface of a rotating body or planet, at 90 degrees from the equator, based on the axis around which a body rotates
*Polar climate, the cli ...
and covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atom ...
). Epistatic interactions occur whenever one mutation alters the local environment of another residue (either by directly contacting it, or by inducing changes in the protein structure). For example, in a disulphide bridge, a single cysteine
Cysteine (symbol Cys or C; ) is a semiessential proteinogenic amino acid with the formula . The thiol side chain in cysteine often participates in enzymatic reactions as a nucleophile.
When present as a deprotonated catalytic residue, some ...
has no effect on protein stability
Protein folding is the physical process by which a protein chain is translated to its native three-dimensional structure, typically a "folded" conformation by which the protein becomes biologically functional. Via an expeditious and reproduc ...
until a second is present at the correct location at which point the two cysteines form a chemical bond which enhances the stability of the protein. This would be observed as positive epistasis where the double-cysteine variant had a much higher stability than either of the single-cysteine variants. Conversely, when deleterious mutations are introduced, proteins often exhibit mutational robustness whereby as stabilising interactions are destroyed the protein still functions until it reaches some stability threshold at which point further destabilising mutations have large, detrimental effects as the protein can no longer fold. This leads to negative epistasis whereby mutations that have little effect alone have a large, deleterious effect together.
In enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
s, the protein structure orients a few, key amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha ...
s into precise geometries to form an active site to perform chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a natural science that covers the elements that make up matter to the compounds made of atoms, molecules and ions: their composition, structure, proper ...
. Since these active site networks frequently require the cooperation of multiple components, mutating any one of these components massively compromises activity, and so mutating a second component has a relatively minor effect on the already inactivated enzyme. For example, removing any member of the catalytic triad of many enzymes will reduce activity to levels low enough that the organism is no longer viable.
Heterozygotic epistasis
Diploid
Ploidy () is the number of complete sets of chromosomes in a cell, and hence the number of possible alleles for autosomal and pseudoautosomal genes. Sets of chromosomes refer to the number of maternal and paternal chromosome copies, respectiv ...
organisms contain two copies of each gene. If these are different (heterozygous
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
...
/ heteroallelic), the two different copies of the allele may interact with each other to cause epistasis. This is sometimes called allelic complementation, or interallelic complementation. It may be caused by several mechanisms, for example transvection, where an enhancer from one allele acts in ''trans'' to activate transcription from the promoter of the second allele. Alternately, trans-splicing of two non-functional RNA molecules may produce a single, functional RNA.
Similarly, at the protein level, proteins that function as dimer
Dimer may refer to:
* Dimer (chemistry), a chemical structure formed from two similar sub-units
** Protein dimer, a protein quaternary structure
** d-dimer
* Dimer model, an item in statistical mechanics, based on ''domino tiling''
* Julius Dimer ( ...
s may form a heterodimer composed of one protein from each alternate gene and may display different properties to the homodimer of one or both variants. Two bacteriophage T4 mutants defective at different locations in the same gene can undergo allelic complementation during a mixed infection. That is, each mutant alone upon infection cannot produce viable progeny, but upon mixed infection with two complementing mutants, viable phage are formed. Intragenic complementation was demonstrated for several genes that encode structural proteins of the bacteriophageBernstein H, Edgar RS, Denhardt GH. Intragenic complementation among temperature sensitive mutants of bacteriaophage T4D. Genetics. 1965 Jun;51(6):987-1002. doi: 10.1093/genetics/51.6.987. PMID: 14337770; PMCID: PMC1210828 indicating that such proteins function as dimers or even higher order multimers.
Evolutionary consequences
Fitness landscapes and evolvability
 In evolutionary genetics, the sign of epistasis is usually more significant than the magnitude of epistasis. This is because magnitude epistasis (positive and negative) simply affects how beneficial mutations are together, however sign epistasis affects whether mutation combinations are beneficial or deleterious.
A fitness landscape is a representation of the fitness where all genotypes are arranged in 2D space and the fitness of each genotype is represented by height on a surface. It is frequently used as a visual metaphor for understanding
In evolutionary genetics, the sign of epistasis is usually more significant than the magnitude of epistasis. This is because magnitude epistasis (positive and negative) simply affects how beneficial mutations are together, however sign epistasis affects whether mutation combinations are beneficial or deleterious.
A fitness landscape is a representation of the fitness where all genotypes are arranged in 2D space and the fitness of each genotype is represented by height on a surface. It is frequently used as a visual metaphor for understanding evolution
Evolution is change in the heritable characteristics of biological populations over successive generations. These characteristics are the expressions of genes, which are passed on from parent to offspring during reproduction. Variation ...
as the process of moving uphill from one genotype to the next, nearby, fitter genotype.
If all mutations are additive, they can be acquired in any order and still give a continuous uphill trajectory. The landscape is perfectly smooth, with only one peak (global maximum
In mathematical analysis, the maxima and minima (the respective plurals of maximum and minimum) of a function, known collectively as extrema (the plural of extremum), are the largest and smallest value of the function, either within a given ran ...
) and all sequences can evolve uphill to it by the accumulation of beneficial mutations ''in any order''. Conversely, if mutations interact with one another by epistasis, the fitness landscape becomes rugged as the effect of a mutation depends on the genetic background of other mutations. At its most extreme, interactions are so complex that the fitness is ‘uncorrelated’ with gene sequence and the topology of the landscape is random. This is referred to as a rugged fitness landscape and has profound implications for the evolutionary optimisation of organisms. If mutations are deleterious in one combination but beneficial in another, the fittest genotypes can only be accessed by accumulating mutations ''in one specific order''. This makes it more likely that organisms will get stuck at local maxima
In mathematical analysis, the maxima and minima (the respective plurals of maximum and minimum) of a function, known collectively as extrema (the plural of extremum), are the largest and smallest value of the function, either within a given ran ...
in the fitness landscape having acquired mutations in the 'wrong' order. For example, a variant of TEM1 β-lactamase with 5 mutations is able to cleave cefotaxime
Cefotaxime is an antibiotic used to treat a number of bacterial infections in human, other animals and plant tissue culture. Specifically in humans it is used to treat joint infections, pelvic inflammatory disease, meningitis, pneumonia, urinar ...
(a third generation antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention ...
). However, of the 120 possible pathways to this 5-mutant variant, only 7% are accessible to evolution as the remainder passed through fitness valleys where the combination of mutations reduces activity. In contrast, changes in environment (and therefore the shape of the fitness landscape) have been shown to provide escape from local maxima. In this example, selection in changing antibiotic environments resulted in a "gateway mutation" which epistatically interacted in a positive manner with other mutations along an evolutionary pathway, effectively crossing a fitness valley. This gateway mutation alleviated the negative epistatic interactions of other individually beneficial mutations, allowing them to better function in concert. Complex environments or selections may therefore bypass local maxima found in models assuming simple positive selection.
High epistasis is usually considered a constraining factor on evolution, and improvements in a highly epistatic trait are considered to have lower evolvability. This is because, in any given genetic background, very few mutations will be beneficial, even though many mutations may need to occur to eventually improve the trait. The lack of a smooth landscape makes it harder for evolution to access fitness peaks. In highly rugged landscapes, fitness valleys block access to some genes, and even if ridges exist that allow access, these may be rare or prohibitively long. Moreover, adaptation can move proteins into more precarious or rugged regions of the fitness landscape. These shifting "fitness territories" may act to decelerate evolution and could represent tradeoffs for adaptive traits.
The frustration of adaptive evolution by rugged fitness landscapes was recognized as a potential force for the evolution of evolvability. Michael Conrad in 1972 was the first to propose a mechanism for the evolution of evolvability by noting that a mutation which smoothed the fitness landscape at other loci could facilitate the production of advantageous mutations and hitchhike along with them. Rupert Riedl in 1975 proposed that new genes which produced the same phenotypic effects with a single mutation as other loci with reciprocal sign epistasis would be a new means to attain a phenotype otherwise too unlikely to occur by mutation.
Rugged, epistatic fitness landscapes also affect the trajectories of evolution. When a mutation has a large number of epistatic effects, each accumulated mutation drastically changes the set of available beneficial mutations. Therefore, the evolutionary trajectory followed depends highly on which early mutations were accepted. Thus, repeats of evolution from the same starting point tend to diverge to different local maxima rather than converge on a single global maximum as they would in a smooth, additive landscape.
Evolution of sex
Negative epistasis and sex are thought to be intimately correlated. Experimentally, this idea has been tested in using digital simulations of asexual and sexual populations. Over time, sexual populations move towards more negative epistasis, or the lowering of fitness by two interacting alleles. It is thought that negative epistasis allows individuals carrying the interacting deleterious mutations to be removed from the populations efficiently. This removes those alleles from the population, resulting in an overall more fit population. This hypothesis was proposed by Alexey Kondrashov, and is sometimes known as the ''deterministic mutation hypothesis'' and has also been tested using artificialgene networks
A gene (or genetic) regulatory network (GRN) is a collection of molecular regulators that interact with each other and with other substances in the cell to govern the gene expression levels of mRNA and proteins which, in turn, determine the fun ...
.
However, the evidence for this hypothesis has not always been straightforward and the model proposed by Kondrashov has been criticized for assuming mutation parameters far from real world observations. In addition, in those tests which used artificial gene networks, negative epistasis is only found in more densely connected networks, whereas empirical evidence indicates that natural gene networks are sparsely connected, and theory shows that selection for robustness will favor more sparsely connected and minimally complex networks.
Methods and model systems
Regression analysis
Quantitative genetics focuses ongenetic variance
Genetic variance is a concept outlined by the English biologist and statistician Ronald Fisher in his fundamental theorem of natural selection. In his 1930 book ''The Genetical Theory of Natural Selection'', Fisher postulates that the rate of cha ...
due to genetic interactions. Any two locus interactions at a particular gene frequency can be decomposed into eight independent genetic effects using a weighted regression. In this regression, the observed two locus genetic effects are treated as dependent variables and the "pure" genetic effects are used as the independent variables. Because the regression is weighted, the partitioning among the variance components will change as a function of gene frequency. By analogy it is possible to expand this system to three or more loci, or to cytonuclear interactions
Double mutant cycles
When assaying epistasis within a gene, site-directed mutagenesis can be used to generate the different genes, and their protein products can be assayed (e.g. for stability or catalytic activity). This is sometimes called a double mutant cycle and involves producing and assaying the wild type protein, the two single mutants and the double mutant. Epistasis is measured as the difference between the effects of the mutations together versus the sum of their individual effects. This can be expressed as a free energy of interaction. The same methodology can be used to investigate the interactions between larger sets of mutations but all combinations have to be produced and assayed. For example, there are 120 different combinations of 5 mutations, some or all of which may show epistasis...Computational prediction
Numerous computational methods have been developed for the detection and characterization of epistasis. Many of these rely onmachine learning
Machine learning (ML) is a field of inquiry devoted to understanding and building methods that 'learn', that is, methods that leverage data to improve performance on some set of tasks. It is seen as a part of artificial intelligence.
Machine ...
to detect non-additive effects that might be missed by statistical approaches such as linear regression.
For example, multifactor dimensionality reduction (MDR) was designed specifically for nonparametric and model-free detection of combinations of genetic variants that are predictive of a phenotype such as disease status in human populations. Several of these approaches have been broadly reviewed in the literature. Even more recently, methods that utilize insights from theoretical computer science (the Hadamard transform and compressed sensing) or maximum-likelihood inference were shown to distinguish epistatic effects from overall non-linearity in genotype–phenotype map structure, while others used patient survival analysis to identify non-linearity.
See also
* Co-adaptation *Epistasis and functional genomics
Epistasis refers to genetic interactions in which the mutation of one gene masks the phenotypic effects of a mutation at another locus. Systematic analysis of these epistatic interactions can provide insight into the structure and function of gene ...
*Mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, m ...
*Synthetic viability Synthetic rescue (or synthetic recovery or synthetic viability when a lethal phenotype is rescued ) refers to a genetic interaction in which a cell that is nonviable, sensitive to a specific drug, or otherwise impaired due to the presence of a genet ...
* Synthetic lethality
*Quantitative trait locus
A quantitative trait locus (QTL) is a locus (section of DNA) that correlates with variation of a quantitative trait in the phenotype of a population of organisms. QTLs are mapped by identifying which molecular markers (such as SNPs or AFLPs ...
* Interactome (Genetic interaction network)
* Fitness landscape
* Evolvability
*Pleiotropy
Pleiotropy (from Greek , 'more', and , 'way') occurs when one gene influences two or more seemingly unrelated phenotypic traits. Such a gene that exhibits multiple phenotypic expression is called a pleiotropic gene. Mutation in a pleiotropic g ...
* Evolution of sexual reproduction
References
External links
INTERSNP
- a software for genome-wide interaction analysis (GWIA) of case-control and case-only SNP data, including analysis of quantitative traits.
High school (GCSE, Alevel) resource.
GeneticInteractions.org
{{Authority control Classical genetics Genetics concepts