Enthalpy on:
[Wikipedia]
[Google]
[Amazon]
Enthalpy () is the sum of a
 The enthalpy values of important substances can be obtained using commercial software. Practically all relevant material properties can be obtained either in tabular or in graphical form. There are many types of diagrams, such as diagrams, which give the specific enthalpy as function of temperature for various pressures, and diagrams, which give as function of for various . One of the most common diagrams is the temperature–specific entropy diagram ( diagram). It gives the melting curve and saturated liquid and vapor values together with isobars and isenthalps. These diagrams are powerful tools in the hands of the thermal engineer.
The enthalpy values of important substances can be obtained using commercial software. Practically all relevant material properties can be obtained either in tabular or in graphical form. There are many types of diagrams, such as diagrams, which give the specific enthalpy as function of temperature for various pressures, and diagrams, which give as function of for various . One of the most common diagrams is the temperature–specific entropy diagram ( diagram). It gives the melting curve and saturated liquid and vapor values together with isobars and isenthalps. These diagrams are powerful tools in the hands of the thermal engineer.
 One of the simple applications of the concept of enthalpy is the so-called throttling process, also known as Joule–Thomson expansion. It concerns a steady adiabatic flow of a fluid through a flow resistance (valve, porous plug, or any other type of flow resistance) as shown in the figure. This process is very important, since it is at the heart of domestic
One of the simple applications of the concept of enthalpy is the so-called throttling process, also known as Joule–Thomson expansion. It concerns a steady adiabatic flow of a fluid through a flow resistance (valve, porous plug, or any other type of flow resistance) as shown in the figure. This process is very important, since it is at the heart of domestic
 A power is applied e.g. as electrical power. If the compression is adiabatic, the gas temperature goes up. In the reversible case it would be at constant entropy, which corresponds with a vertical line in the diagram. For example, compressing nitrogen from 1 bar (point a) to 2 bar (point b) would result in a temperature increase from 300 K to 380 K. In order to let the compressed gas exit at ambient temperature , heat exchange, e.g. by cooling water, is necessary. In the ideal case the compression is isothermal. The average heat flow to the surroundings is . Since the system is in the steady state the first law gives
The minimal power needed for the compression is realized if the compression is reversible. In that case the
A power is applied e.g. as electrical power. If the compression is adiabatic, the gas temperature goes up. In the reversible case it would be at constant entropy, which corresponds with a vertical line in the diagram. For example, compressing nitrogen from 1 bar (point a) to 2 bar (point b) would result in a temperature increase from 300 K to 380 K. In order to let the compressed gas exit at ambient temperature , heat exchange, e.g. by cooling water, is necessary. In the ideal case the compression is isothermal. The average heat flow to the surroundings is . Since the system is in the steady state the first law gives
The minimal power needed for the compression is realized if the compression is reversible. In that case the
thermodynamic system
A thermodynamic system is a body of matter and/or radiation separate from its surroundings that can be studied using the laws of thermodynamics.
Thermodynamic systems can be passive and active according to internal processes. According to inter ...
's internal energy
The internal energy of a thermodynamic system is the energy of the system as a state function, measured as the quantity of energy necessary to bring the system from its standard internal state to its present internal state of interest, accoun ...
and the product of its pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
and volume
Volume is a measure of regions in three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch) ...
. It is a state function
In the thermodynamics of equilibrium, a state function, function of state, or point function for a thermodynamic system is a mathematical function relating several state variables or state quantities (that describe equilibrium states of a syste ...
in thermodynamics
Thermodynamics is a branch of physics that deals with heat, Work (thermodynamics), work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed b ...
used in many measurements in chemical, biological, and physical systems at a constant external pressure, which is conveniently provided by the large ambient atmosphere. The pressure–volume term expresses the work
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an ani ...
that was done against constant external pressure to establish the system's physical dimensions from to some final volume (as ), i.e. to make room for it by displacing its surroundings.
The pressure-volume term is very small for solids and liquids at common conditions, and fairly small for gases. Therefore, enthalpy is a stand-in for energy
Energy () is the physical quantity, quantitative physical property, property that is transferred to a physical body, body or to a physical system, recognizable in the performance of Work (thermodynamics), work and in the form of heat and l ...
in chemical systems; bond, lattice, solvation
Solvations describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the solute, includi ...
, and other chemical "energies" are actually enthalpy differences. As a state function, enthalpy depends only on the final configuration of internal energy, pressure, and volume, not on the path taken to achieve it.
In the International System of Units
The International System of Units, internationally known by the abbreviation SI (from French ), is the modern form of the metric system and the world's most widely used system of measurement. It is the only system of measurement with official s ...
(SI), the unit of measurement for enthalpy is the joule
The joule ( , or ; symbol: J) is the unit of energy in the International System of Units (SI). In terms of SI base units, one joule corresponds to one kilogram- metre squared per second squared One joule is equal to the amount of work d ...
. Other historical conventional units still in use include the calorie
The calorie is a unit of energy that originated from the caloric theory of heat. The large calorie, food calorie, dietary calorie, kilocalorie, or kilogram calorie is defined as the amount of heat needed to raise the temperature of one liter o ...
and the British thermal unit
The British thermal unit (Btu) is a measure of heat, which is a form of energy. It was originally defined as the amount of heat required to raise the temperature of one pound of water by one degree Fahrenheit. It is also part of the United Stat ...
(BTU).
The total enthalpy of a system cannot be measured directly because the internal energy contains components that are unknown, not easily accessible, or are not of interest for the thermodynamic problem at hand. In practice, a change in enthalpy is the preferred expression for measurements at constant pressure, because it simplifies the description of energy transfer
Energy Transfer LP is an American company engaged in the pipeline transportation, storage, and terminaling for natural gas, crude oil, NGLs, refined products and liquid natural gas. It is organized under Delaware state laws and headquartered i ...
. When transfer of matter into or out of the system is also prevented and no electrical or mechanical (stirring shaft or lift pumping) work is done, at constant pressure the enthalpy change equals the energy exchanged with the environment by heat
In thermodynamics, heat is energy in transfer between a thermodynamic system and its surroundings by such mechanisms as thermal conduction, electromagnetic radiation, and friction, which are microscopic in nature, involving sub-atomic, ato ...
.
In chemistry, the standard enthalpy of reaction
The standard enthalpy of reaction (denoted \Delta H_^\ominus) for a chemical reaction is the difference between total product and total reactant molar enthalpies, calculated for substances in their standard states. The value can be approximately i ...
is the enthalpy change when reactants in their standard state
The standard state of a material (pure substance, mixture or solution) is a reference point used to calculate its properties under different conditions. A degree sign (°) or a superscript ⦵ symbol (⦵) is used to designate a thermodynamic q ...
s ( usually ) change to products in their standard states.
This quantity is the standard heat of reaction at constant pressure and temperature, but it can be measured by calorimetric methods even if the temperature does vary during the measurement, provided that the initial and final pressure and temperature correspond to the standard state. The value does not depend on the path from initial to final state because enthalpy is a state function
In the thermodynamics of equilibrium, a state function, function of state, or point function for a thermodynamic system is a mathematical function relating several state variables or state quantities (that describe equilibrium states of a syste ...
.
Enthalpies of chemical substances are usually listed for pressure as a standard state. Enthalpies and enthalpy changes for reactions vary as a function of temperature,
but tables generally list the standard heats of formation of substances at . For endothermic
An endothermic process is a chemical or physical process that absorbs heat from its surroundings. In terms of thermodynamics, it is a thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, ...
(heat-absorbing) processes, the change is a positive value; for exothermic
In thermodynamics, an exothermic process () is a thermodynamic process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g. a spark, flame, or flash), electricity (e ...
(heat-releasing) processes it is negative.
The enthalpy of an ideal gas
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is ...
is independent of its pressure or volume, and depends only on its temperature, which correlates to its thermal energy. Real gases at common temperatures and pressures often closely approximate this behavior, which simplifies practical thermodynamic design and analysis.
The word "enthalpy" is derived from the Greek word ''enthalpein'', which means "to heat".
Definition
The enthalpy of a thermodynamic system is defined as the sum of its internal energy and the product of its pressure and volume: where is theinternal energy
The internal energy of a thermodynamic system is the energy of the system as a state function, measured as the quantity of energy necessary to bring the system from its standard internal state to its present internal state of interest, accoun ...
, is pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
, and is the volume
Volume is a measure of regions in three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch) ...
of the system; is sometimes referred to as the pressure energy .
Enthalpy is an extensive property
Physical or chemical properties of materials and systems can often be categorized as being either intensive or extensive, according to how the property changes when the size (or extent) of the system changes.
The terms "intensive and extensive ...
; it is proportional to the size of the system (for homogeneous systems). As intensive properties
Physical or chemical properties of materials and systems can often be categorized as being either intensive or extensive, according to how the property changes when the size (or extent) of the system changes.
The terms "intensive and extensive ...
, the ''specific enthalpy
Enthalpy () is the sum of a thermodynamic system's internal energy and the product of its pressure and volume. It is a state function in thermodynamics used in many measurements in chemical, biological, and physical systems at a constant extern ...
'' is referenced to a unit of mass
Mass is an Intrinsic and extrinsic properties, intrinsic property of a physical body, body. It was traditionally believed to be related to the physical quantity, quantity of matter in a body, until the discovery of the atom and particle physi ...
of the system, and the ''molar enthalpy'' where is the number of moles. For inhomogeneous systems the enthalpy is the sum of the enthalpies of the component subsystems:
where
: is the total enthalpy of all the subsystems,
: refers to the various subsystems,
: refers to the enthalpy of each subsystem.
A closed system may lie in thermodynamic equilibrium in a static gravitational field
In physics, a gravitational field or gravitational acceleration field is a vector field used to explain the influences that a body extends into the space around itself. A gravitational field is used to explain gravitational phenomena, such as ...
, so that its pressure varies continuously with altitude
Altitude is a distance measurement, usually in the vertical or "up" direction, between a reference datum (geodesy), datum and a point or object. The exact definition and reference datum varies according to the context (e.g., aviation, geometr ...
, while, because of the equilibrium requirement, its temperature is invariant with altitude. (Correspondingly, the system's gravitational potential energy
Gravitational energy or gravitational potential energy is the potential energy an object with mass has due to the gravitational potential of its position in a gravitational field. Mathematically, it is the minimum Work (physics), mechanical work t ...
density also varies with altitude.) Then the enthalpy summation becomes an integral
In mathematics, an integral is the continuous analog of a Summation, sum, which is used to calculate area, areas, volume, volumes, and their generalizations. Integration, the process of computing an integral, is one of the two fundamental oper ...
:
where
: ("rho
Rho (; uppercase Ρ, lowercase ρ or ; or ) is the seventeenth letter of the Greek alphabet. In the system of Greek numerals it has a value of 100. It is derived from Phoenician alphabet, Phoenician letter resh . Its uppercase form uses the same ...
") is density
Density (volumetric mass density or specific mass) is the ratio of a substance's mass to its volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' (or ''d'') can also be u ...
(mass per unit volume),
: is the specific enthalpy (enthalpy per unit mass),
: represents the enthalpy density (enthalpy per unit volume),
: denotes an infinitesimal
In mathematics, an infinitesimal number is a non-zero quantity that is closer to 0 than any non-zero real number is. The word ''infinitesimal'' comes from a 17th-century Modern Latin coinage ''infinitesimus'', which originally referred to the " ...
ly small element of volume within the system, for example, the volume of an infinitesimally thin horizontal layer.
The integral therefore represents the sum of the enthalpies of all the elements of the volume.
The enthalpy of a closed homogeneous system is its energy function with its entropy and its pressure as natural state variables which provide a differential relation for of the simplest form, derived as follows. We start from the first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy in the context of thermodynamic processes. For a thermodynamic process affecting a thermodynamic system without transfer of matter, the law distinguishes two ...
for closed systems for an infinitesimal process:
where
: is a small amount of heat added to the system,
: is a small amount of work performed by the system.
In a homogeneous system in which only reversible processes or pure heat transfer are considered, the second law of thermodynamics
The second law of thermodynamics is a physical law based on Universal (metaphysics), universal empirical observation concerning heat and Energy transformation, energy interconversions. A simple statement of the law is that heat always flows spont ...
gives with the absolute temperature
Thermodynamic temperature, also known as absolute temperature, is a physical quantity which measures temperature starting from absolute zero, the point at which particles have minimal thermal motion.
Thermodynamic temperature is typically expres ...
and the infinitesimal change in entropy
Entropy is a scientific concept, most commonly associated with states of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the micros ...
of the system. Furthermore, if only work is done, As a result,
Adding to both sides of this expression gives
or
So
and the coefficients of the natural variable differentials and are just the single variables and .
Other expressions
The above expression of in terms of entropy and pressure may be unfamiliar to some readers. There are also expressions in terms of more directly measurable variables such as temperature and pressure: where is theheat capacity
Heat capacity or thermal capacity is a physical property of matter, defined as the amount of heat to be supplied to an object to produce a unit change in its temperature. The SI unit of heat capacity is joule per kelvin (J/K).
Heat capacity is a ...
at ''constant pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
'', and is the coefficient of (cubic) thermal expansion:
With this expression one can, in principle, determine the enthalpy if and are known as functions of and . However the expression is more complicated than because is not a natural variable for the enthalpy .
At constant pressure, so that For an ideal gas
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is ...
, reduces to this form even if the process involves a pressure change, because
In a more general form, the first law describes the internal energy with additional terms involving the chemical potential
In thermodynamics, the chemical potential of a Chemical specie, species is the energy that can be absorbed or released due to a change of the particle number of the given species, e.g. in a chemical reaction or phase transition. The chemical potent ...
and the number of particles of various types. The differential statement for then becomes
where is the chemical potential per particle for a type particle, and is the number of such particles. The last term can also be written as (with the number of moles of component added to the system and, in this case, the molar chemical potential) or as (with the mass of component added to the system and, in this case, the specific chemical potential).
Characteristic functions and natural state variables
The enthalpy expresses the thermodynamics of a system in the ''energy representation''. As afunction of state
In the thermodynamics of equilibrium, a state function, function of state, or point function for a thermodynamic system is a mathematical function relating several state variables or state quantities (that describe equilibrium states of a syste ...
, its arguments include one intensive and several extensive state variable
A state variable is one of the set of Variable (mathematics), variables that are used to describe the mathematical "state" of a dynamical system. Intuitively, the state of a system describes enough about the system to determine its future behavi ...
s. The state variables , and are said to be the ''natural state variables'' in this representation. They are suitable for describing processes in which they are determined by factors in the surroundings. For example, when a virtual parcel of atmospheric air moves to a different altitude, the pressure surrounding it changes, and the process is often so rapid that there is too little time for heat transfer. This is the basis of the so-called adiabatic approximation that is used in meteorology
Meteorology is the scientific study of the Earth's atmosphere and short-term atmospheric phenomena (i.e. weather), with a focus on weather forecasting. It has applications in the military, aviation, energy production, transport, agricultur ...
.
Conjugate with the enthalpy, with these arguments, the other characteristic function of state of a thermodynamic system is its entropy, as a function of the same list of variables of state, except that the entropy is replaced in the list by the enthalpy . It expresses the ''entropy representation''. The state variables , , and are said to be the ''natural state variables'' in this representation. They are suitable for describing processes in which they are experimentally controlled. For example, and can be controlled by allowing heat transfer, and by varying only the external pressure on the piston that sets the volume of the system.
Physical interpretation
The term is the energy of the system, and the term can be interpreted as thework
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an ani ...
that would be required to "make room" for the system if the pressure of the environment remained constant. When a system, for example, moles of a gas of volume
Volume is a measure of regions in three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch) ...
at pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
and temperature
Temperature is a physical quantity that quantitatively expresses the attribute of hotness or coldness. Temperature is measurement, measured with a thermometer. It reflects the average kinetic energy of the vibrating and colliding atoms making ...
, is created or brought to its present state from absolute zero
Absolute zero is the lowest possible temperature, a state at which a system's internal energy, and in ideal cases entropy, reach their minimum values. The absolute zero is defined as 0 K on the Kelvin scale, equivalent to −273.15 ° ...
, energy must be supplied equal to its internal energy plus , where is the work
Work may refer to:
* Work (human activity), intentional activity people perform to support themselves, others, or the community
** Manual labour, physical work done by humans
** House work, housework, or homemaking
** Working animal, an ani ...
done in pushing against the ambient (atmospheric) pressure.
In physics
Physics is the scientific study of matter, its Elementary particle, fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge whi ...
and statistical mechanics
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. Sometimes called statistical physics or statistical thermodynamics, its applicati ...
it may be more interesting to study the internal properties of a constant-volume system and therefore the internal energy is used.
In chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules a ...
, experiments are often conducted at constant atmospheric pressure
Atmospheric pressure, also known as air pressure or barometric pressure (after the barometer), is the pressure within the atmosphere of Earth. The standard atmosphere (symbol: atm) is a unit of pressure defined as , which is equivalent to 1,013. ...
, and the pressure–volume work represents a small, well-defined energy exchange with the atmosphere, so that is the appropriate expression for the heat of reaction. For a heat engine
A heat engine is a system that transfers thermal energy to do mechanical or electrical work. While originally conceived in the context of mechanical energy, the concept of the heat engine has been applied to various other kinds of energy, pa ...
, the change in its enthalpy after a full cycle is equal to zero, since the final and initial state are equal.
Relationship to heat
In order to discuss the relation between the enthalpy increase and heat supply, we return to the first law for closed systems, with the physics sign convention: , where the heat is supplied by conduction, radiation,Joule heating
Joule heating (also known as resistive heating, resistance heating, or Ohmic heating) is the process by which the passage of an electric current through a conductor (material), conductor produces heat.
Joule's first law (also just Joule's law), ...
. We apply it to the special case with a constant pressure at the surface. In this case the work is given by (where is the pressure at the surface, is the increase of the volume of the system). Cases of long-range electromagnetic interaction require further state variables in their formulation and are not considered here. In this case the first law reads:
Now,
so
If the system is under constant pressure, and consequently, the increase in enthalpy of the system is equal to the heat
In thermodynamics, heat is energy in transfer between a thermodynamic system and its surroundings by such mechanisms as thermal conduction, electromagnetic radiation, and friction, which are microscopic in nature, involving sub-atomic, ato ...
added:
This is why the now-obsolete term ''heat content'' was used for enthalpy in the 19th century.
Applications
In thermodynamics, one can calculate enthalpy by determining the requirements for creating a system from "nothingness"; the mechanical work required, differs based upon the conditions that obtain during the creation of thethermodynamic system
A thermodynamic system is a body of matter and/or radiation separate from its surroundings that can be studied using the laws of thermodynamics.
Thermodynamic systems can be passive and active according to internal processes. According to inter ...
.
Energy
Energy () is the physical quantity, quantitative physical property, property that is transferred to a physical body, body or to a physical system, recognizable in the performance of Work (thermodynamics), work and in the form of heat and l ...
must be supplied to remove particles from the surroundings to make space for the creation of the system, assuming that the pressure remains constant; this is the term. The supplied energy must also provide the change in internal energy , which includes activation energies, ionization energies, mixing energies, vaporization energies, chemical bond energies, and so forth. Together, these constitute the change in the enthalpy For systems at constant pressure, with no external work done other than the work, the change in enthalpy is the heat received by the system.
For a simple system with a constant number of particles at constant pressure, the difference in enthalpy is the maximum amount of thermal energy derivable from an isobaric thermodynamic process.
Heat of reaction
The total enthalpy of a system cannot be measured directly; the ''enthalpy change'' of asystem
A system is a group of interacting or interrelated elements that act according to a set of rules to form a unified whole. A system, surrounded and influenced by its open system (systems theory), environment, is described by its boundaries, str ...
is measured instead. Enthalpy change is defined by the following equation:
where
: is the "enthalpy change",
: is the final enthalpy of the system (in a chemical reaction, the enthalpy of the products or the system at equilibrium),
: is the initial enthalpy of the system (in a chemical reaction, the enthalpy of the reactants).
For an exothermic reaction
In thermochemistry, an exothermic reaction is a "reaction for which the overall standard enthalpy change Δ''H''⚬ is negative." Exothermic reactions usually release heat. The term is often confused with exergonic reaction, which IUPAC define ...
at constant pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
, the system's change in enthalpy, , is negative due to the products of the reaction having a smaller enthalpy than the reactants, and equals the heat released in the reaction if no electrical or mechanical work is done. In other words, the overall decrease in enthalpy is achieved by the generation of heat.
Conversely, for a constant-pressure endothermic
An endothermic process is a chemical or physical process that absorbs heat from its surroundings. In terms of thermodynamics, it is a thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, ...
reaction, is positive and equal to the heat ''absorbed'' in the reaction.
From the definition of enthalpy as the enthalpy change at constant pressure is However, for most chemical reactions, the work term is much smaller than the internal energy change , which is approximately equal to . As an example, for the combustion of carbon monoxide and
Since the differences are so small, reaction enthalpies are often described as reaction energies and analyzed in terms of bond energies
In chemistry, bond energy (''BE'') is one measure of the strength of a chemical bond. It is sometimes called the mean bond, bond enthalpy, average bond enthalpy, or bond strength. IUPAC defines bond energy as the average value of the gas-phase bo ...
.
Specific enthalpy
The ''specific enthalpy'' of a uniform system is defined as , where is the mass of the system. ItsSI unit
The International System of Units, internationally known by the abbreviation SI (from French ), is the modern form of the metric system and the world's most widely used system of units of measurement, system of measurement. It is the only system ...
is joule per kilogram. It can be expressed in other specific quantities by where is the specific internal energy
The internal energy of a thermodynamic system is the energy of the system as a state function, measured as the quantity of energy necessary to bring the system from its standard internal state to its present internal state of interest, accoun ...
, is the pressure, and is specific volume
In thermodynamics, the specific volume of a substance (symbol: , nu) is the quotient of the substance's volume () to its mass ():
:\nu = \frac
It is a mass-specific intrinsic property of the substance. It is the reciprocal of density (rho) ...
, which is equal to , where is the density
Density (volumetric mass density or specific mass) is the ratio of a substance's mass to its volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' (or ''d'') can also be u ...
.
Enthalpy changes
An enthalpy change describes the change in enthalpy observed in the constituents of a thermodynamic system when undergoing a transformation or chemical reaction. It is the difference between the enthalpy after the process has completed, i.e. the enthalpy of theproducts
Product may refer to:
Business
* Product (business), an item that can be offered to a market to satisfy the desire or need of a customer.
* Product (project management), a deliverable or set of deliverables that contribute to a business solution
...
assuming that the reaction goes to completion, and the initial enthalpy of the system, namely the reactants. These processes are specified solely by their initial and final states, so that the enthalpy change for the reverse is the negative of that for the forward process.
A common standard enthalpy change is the enthalpy of formation, which has been determined for a large number of substances. Enthalpy changes are routinely measured and compiled in chemical and physical reference works, such as the CRC Handbook of Chemistry and Physics
The ''CRC Handbook of Chemistry and Physics'' is a comprehensive one-volume reference resource for science research. First published in 1914, it is currently () in its 105th edition, published in 2024. It is known colloquially among chemists as ...
. The following is a selection of enthalpy changes commonly recognized in thermodynamics.
When used in these recognized terms the qualifier ''change'' is usually dropped and the property is simply termed ''enthalpy of "process"''. Since these properties are often used as reference values, it is very common to quote them for a standardized set of environmental parameters, or standard conditions, including:
* A pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
of one atmosphere (1 atm = 1013.25 hPa) or 1 bar
* A temperature
Temperature is a physical quantity that quantitatively expresses the attribute of hotness or coldness. Temperature is measurement, measured with a thermometer. It reflects the average kinetic energy of the vibrating and colliding atoms making ...
of 25 °C = 298.15 K
* A concentration
In chemistry, concentration is the abundance of a constituent divided by the total volume of a mixture. Several types of mathematical description can be distinguished: '' mass concentration'', '' molar concentration'', '' number concentration'', ...
of 1.0 M when the element or compound is present in solution
* Elements or compounds in their normal physical states, i.e. standard state
The standard state of a material (pure substance, mixture or solution) is a reference point used to calculate its properties under different conditions. A degree sign (°) or a superscript ⦵ symbol (⦵) is used to designate a thermodynamic q ...
For such standardized values the name of the enthalpy is commonly prefixed with the term ''standard'', e.g. ''standard enthalpy of formation''.
Chemical properties
Enthalpy of reaction
The standard enthalpy of reaction (denoted \Delta H_^\ominus) for a chemical reaction is the difference between total product and total reactant molar enthalpies, calculated for substances in their standard states. The value can be approximately i ...
is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of substance reacts completely.
Enthalpy of formation is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of a compound is formed from its elementary antecedents.
Enthalpy of combustion
The heating value (or energy value or calorific value) of a substance, usually a fuel or food (see food energy), is the amount of heat released during the combustion of a specified amount of it.
The ''calorific value'' is the total energy release ...
is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of a substance burns completely with oxygen.
Enthalpy of hydrogenation is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of an unsaturated compound reacts completely with an excess of hydrogen to form a saturated compound.
Enthalpy of atomization is defined as the enthalpy change required to separate one mole of a substance into its constituent atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
s completely.
Enthalpy of neutralization is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of water is formed when an acid and a base react.
Standard enthalpy of solution is defined as the enthalpy change observed in a constituent of a thermodynamic system when one mole of a solute is dissolved completely in an excess of solvent, so that the solution is at infinite dilution.
Standard enthalpy of denaturation is defined as the enthalpy change required to denature one mole of compound.
Enthalpy of hydration is defined as the enthalpy change observed when one mole of gaseous ions is completely dissolved in water forming one mole of aqueous ions.
Physical properties
Enthalpy of fusion
In thermodynamics, the enthalpy of fusion of a substance, also known as (latent) heat of fusion, is the change in its enthalpy resulting from providing energy, typically heat, to a specific quantity of the substance to change its state from a ...
is defined as the enthalpy change required to completely change the state of one mole of substance from solid to liquid.
Enthalpy of vaporization
In thermodynamics, the enthalpy of vaporization (symbol ), also known as the (latent) heat of vaporization or heat of evaporation, is the amount of energy (enthalpy) that must be added to a liquid substance to transform a quantity of that sub ...
is defined as the enthalpy change required to completely change the state of one mole of substance from liquid to gas.
Enthalpy of sublimation
In thermodynamics, the enthalpy of sublimation, or heat of sublimation, is the heat required to sublimate (change from solid to gas) one mole of a substance at a given combination of temperature and pressure, usually standard temperature and p ...
is defined as the enthalpy change required to completely change the state of one mole of substance from solid to gas.
Lattice enthalpy is defined as the energy required to separate one mole of an ionic compound into separated gaseous ions to an infinite distance apart (meaning no force of attraction).
Enthalpy of mixing
In thermodynamics, the enthalpy of mixing (also heat of mixing and excess enthalpy) is the enthalpy liberated or absorbed from a substance upon mixing. When a substance or compound is combined with any other substance or compound, the enthalpy ...
is defined as the enthalpy change upon mixing of two (non-reacting) chemical substances.
Open systems
Inthermodynamic
Thermodynamics is a branch of physics that deals with heat, work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed by the four laws of th ...
open systems, mass (of substances) may flow in and out of the system boundaries. The first law of thermodynamics for open systems states: The increase in the internal energy of a system is equal to the amount of energy added to the system by mass flowing in and by heating, minus the amount lost by mass flowing out and in the form of work done by the system:
where is the average internal energy entering the system, and is the average internal energy leaving the system.
The region of space enclosed by the boundaries of the open system is usually called a control volume
In continuum mechanics and thermodynamics, a control volume (CV) is a mathematical abstraction employed in the process of creating mathematical models of physical processes. In an inertial frame of reference, it is a fictitious region of a given v ...
, and it may or may not correspond to physical walls. If we choose the shape of the control volume such that all flow in or out occurs perpendicular to its surface, then the flow of mass into the system performs work as if it were a piston of fluid pushing mass into the system, and the system performs work on the flow of mass out as if it were driving a piston of fluid. There are then two types of work performed: ''flow work'' described above, which is performed on the fluid (this is also often called '' work''), and ''mechanical work'' (''shaft work''), which may be performed on some mechanical device such as a turbine or pump.
These two types of work are expressed in the equation
Substitution into the equation above for the control volume (cv) yields
The definition of enthalpy permits us to use this thermodynamic potential
Thermodynamics is a branch of physics that deals with heat, Work (thermodynamics), work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed b ...
to account for both internal energy and work in fluids for open systems:
If we allow also the system boundary to move (e.g. due to moving pistons), we get a rather general form of the first law for open systems.
In terms of time derivatives, using Newton's dot notation for time derivatives, it reads:
with sums over the various places where heat is supplied, mass flows into the system, and boundaries are moving. The terms represent enthalpy flows, which can be written as
with the mass flow and the molar flow at position respectively. The term represents the rate of change of the system volume at position that results in power done by the system. The parameter represents all other forms of power done by the system such as shaft power, but it can also be, say, electric power produced by an electrical power plant.
Note that the previous expression holds true only if the kinetic energy flow rate is conserved between system inlet and outlet. Otherwise, it has to be included in the enthalpy balance. During steady-state
In systems theory, a system or a process is in a steady state if the variables (called state variables) which define the behavior of the system or the process are unchanging in time. In continuous time, this means that for those properties ''p'' ...
operation of a device (see Turbine
A turbine ( or ) (from the Greek , ''tyrbē'', or Latin ''turbo'', meaning vortex) is a rotary mechanical device that extracts energy from a fluid flow and converts it into useful work. The work produced can be used for generating electrical ...
, Pump
A pump is a device that moves fluids (liquids or gases), or sometimes Slurry, slurries, by mechanical action, typically converted from electrical energy into hydraulic or pneumatic energy.
Mechanical pumps serve in a wide range of application ...
, and Engine
An engine or motor is a machine designed to convert one or more forms of energy into mechanical energy.
Available energy sources include potential energy (e.g. energy of the Earth's gravitational field as exploited in hydroelectric power ge ...
), the average may be set equal to zero. This yields a useful expression for the average power
Power may refer to:
Common meanings
* Power (physics), meaning "rate of doing work"
** Engine power, the power put out by an engine
** Electric power, a type of energy
* Power (social and political), the ability to influence people or events
Math ...
generation for these devices in the absence of chemical reactions:
where the angle bracket
A bracket is either of two tall fore- or back-facing punctuation marks commonly used to isolate a segment of text or data from its surroundings. They come in four main pairs of shapes, as given in the box to the right, which also gives their n ...
s denote time averages. The technical importance of the enthalpy is directly related to its presence in the first law for open systems, as formulated above.
Diagrams
 The enthalpy values of important substances can be obtained using commercial software. Practically all relevant material properties can be obtained either in tabular or in graphical form. There are many types of diagrams, such as diagrams, which give the specific enthalpy as function of temperature for various pressures, and diagrams, which give as function of for various . One of the most common diagrams is the temperature–specific entropy diagram ( diagram). It gives the melting curve and saturated liquid and vapor values together with isobars and isenthalps. These diagrams are powerful tools in the hands of the thermal engineer.
The enthalpy values of important substances can be obtained using commercial software. Practically all relevant material properties can be obtained either in tabular or in graphical form. There are many types of diagrams, such as diagrams, which give the specific enthalpy as function of temperature for various pressures, and diagrams, which give as function of for various . One of the most common diagrams is the temperature–specific entropy diagram ( diagram). It gives the melting curve and saturated liquid and vapor values together with isobars and isenthalps. These diagrams are powerful tools in the hands of the thermal engineer.
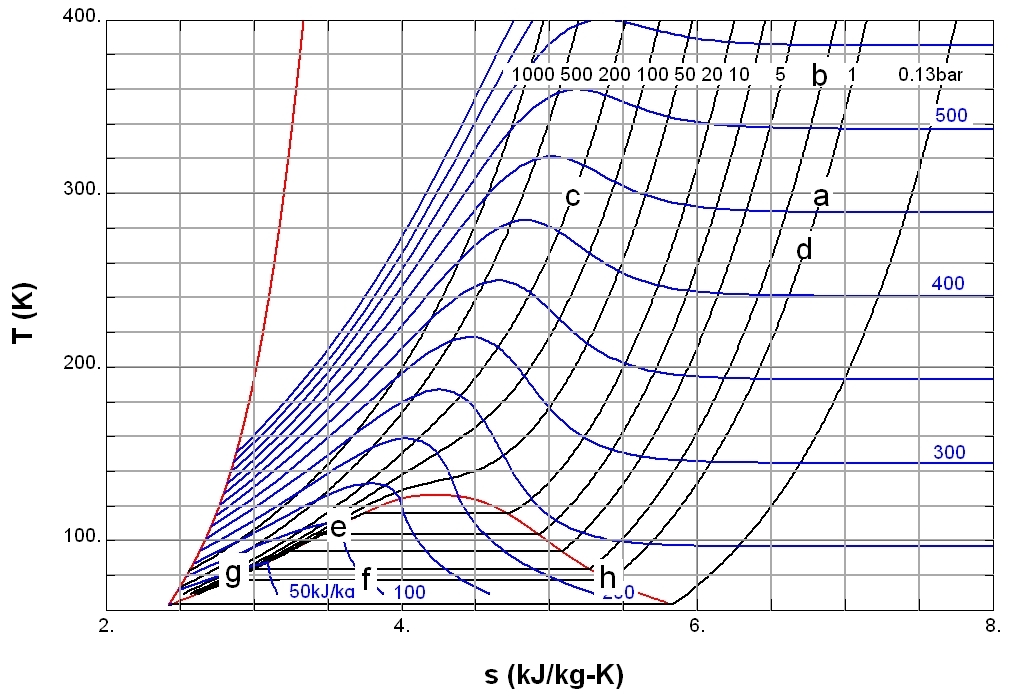
Some basic applications
The points through in the figure play a role in the discussion in this section. : Points and are saturated liquids, and point is a saturated gas.Throttling
 One of the simple applications of the concept of enthalpy is the so-called throttling process, also known as Joule–Thomson expansion. It concerns a steady adiabatic flow of a fluid through a flow resistance (valve, porous plug, or any other type of flow resistance) as shown in the figure. This process is very important, since it is at the heart of domestic
One of the simple applications of the concept of enthalpy is the so-called throttling process, also known as Joule–Thomson expansion. It concerns a steady adiabatic flow of a fluid through a flow resistance (valve, porous plug, or any other type of flow resistance) as shown in the figure. This process is very important, since it is at the heart of domestic refrigerator
A refrigerator, commonly shortened to fridge, is a commercial and home appliance consisting of a thermal insulation, thermally insulated compartment and a heat pump (mechanical, electronic or chemical) that transfers heat from its inside to ...
s, where it is responsible for the temperature drop between ambient temperature and the interior of the refrigerator. It is also the final stage in many types of liquefiers.
For a steady state flow regime, the enthalpy of the system (dotted rectangle) has to be constant. Hence
Since the mass flow is constant, the specific enthalpies at the two sides of the flow resistance are the same:
that is, the enthalpy per unit mass does not change during the throttling. The consequences of this relation can be demonstrated using the diagram above.
Example 1
Point c is at 200 bar and room temperature (300 K). A Joule–Thomson expansion from 200 bar to 1 bar follows a curve of constant enthalpy of roughly 425 (not shown in the diagram) lying between the 400 and 450 isenthalps and ends in point d, which is at a temperature of about 270 K . Hence the expansion from 200 bar to 1 bar cools nitrogen from 300 K to 270 K . In the valve, there is a lot of friction, and a lot of entropy is produced, but still the final temperature is below the starting value.Example 2
Point e is chosen so that it is on the saturated liquid line with It corresponds roughly with and Throttling from this point to a pressure of 1 bar ends in the two-phase region (point f). This means that a mixture of gas and liquid leaves the throttling valve. Since the enthalpy is an extensive parameter, the enthalpy in is equal to the enthalpy in multiplied by the liquid fraction in plus the enthalpy in multiplied by the gas fraction in So With numbers: : so This means that the mass fraction of the liquid in the liquid–gas mixture that leaves the throttling valve is 64%.Compressors
 A power is applied e.g. as electrical power. If the compression is adiabatic, the gas temperature goes up. In the reversible case it would be at constant entropy, which corresponds with a vertical line in the diagram. For example, compressing nitrogen from 1 bar (point a) to 2 bar (point b) would result in a temperature increase from 300 K to 380 K. In order to let the compressed gas exit at ambient temperature , heat exchange, e.g. by cooling water, is necessary. In the ideal case the compression is isothermal. The average heat flow to the surroundings is . Since the system is in the steady state the first law gives
The minimal power needed for the compression is realized if the compression is reversible. In that case the
A power is applied e.g. as electrical power. If the compression is adiabatic, the gas temperature goes up. In the reversible case it would be at constant entropy, which corresponds with a vertical line in the diagram. For example, compressing nitrogen from 1 bar (point a) to 2 bar (point b) would result in a temperature increase from 300 K to 380 K. In order to let the compressed gas exit at ambient temperature , heat exchange, e.g. by cooling water, is necessary. In the ideal case the compression is isothermal. The average heat flow to the surroundings is . Since the system is in the steady state the first law gives
The minimal power needed for the compression is realized if the compression is reversible. In that case the second law of thermodynamics
The second law of thermodynamics is a physical law based on Universal (metaphysics), universal empirical observation concerning heat and Energy transformation, energy interconversions. A simple statement of the law is that heat always flows spont ...
for open systems gives
Eliminating gives for the minimal power
For example, compressing 1 kg of nitrogen from 1 bar to 200 bar costs at least :
With the data, obtained with the diagram, we find a value of
The relation for the power can be further simplified by writing it as
With
:
this results in the final relation
History and etymology
The term ''enthalpy'' was coined relatively late in the history of thermodynamics, in the early 20th century.Energy
Energy () is the physical quantity, quantitative physical property, property that is transferred to a physical body, body or to a physical system, recognizable in the performance of Work (thermodynamics), work and in the form of heat and l ...
was introduced in a modern sense by Thomas Young in 1802, while entropy
Entropy is a scientific concept, most commonly associated with states of disorder, randomness, or uncertainty. The term and the concept are used in diverse fields, from classical thermodynamics, where it was first recognized, to the micros ...
by Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
in 1865. ''Energy'' uses the root of the Greek
Greek may refer to:
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group
*Greek language, a branch of the Indo-European language family
**Proto-Greek language, the assumed last common ancestor of all kno ...
word (''ergon''), meaning "work", to express the idea of capacity to perform work. ''Entropy'' uses the Greek word (''tropē'') meaning ''transformation'' or ''turning''. ''Enthalpy'' uses the root of the Greek word (''thalpos'') "warmth, heat".
The term expresses the obsolete concept of ''heat content'', as refers to the amount of heat gained in a process at constant pressure only, but not in the general case when pressure is variable. J. W. Gibbs used the term "a heat function for constant pressure" for clarity.
Introduction of the concept of "heat content" is associated with Benoît Paul Émile Clapeyron
Benoît () is a French male given name. It is less frequently spelled Benoist. The name comes from the Latin word , which means "blessed", equivalent in meaning to Bénédicte or the English name Benedict. A female derivative of the name is Ben ...
and Rudolf Clausius
Rudolf Julius Emanuel Clausius (; 2 January 1822 – 24 August 1888) was a German physicist and mathematician and is considered one of the central founding fathers of the science of thermodynamics. By his restatement of Sadi Carnot's principle ...
(Clausius–Clapeyron relation
The Clausius–Clapeyron relation, in chemical thermodynamics, specifies the temperature dependence of pressure, most importantly vapor pressure, at a discontinuous phase transition between two phases of matter of a single constituent. It is nam ...
, 1850).
The term ''enthalpy'' first appeared in print in 1909. It is attributed to Heike Kamerlingh Onnes
Heike Kamerlingh Onnes (; 21 September 1853 – 21 February 1926) was a Dutch Experimental physics, experimental physicist. After studying in Groningen and Heidelberg, he became Professor of Experimental Physics at Leiden University, where he tau ...
, who most likely introduced it orally the year before, at the first meeting of the Institute of Refrigeration in Paris. It gained currency only in the 1920s, notably with the '' Mollier Steam Tables and Diagrams'', published in 1927.
Until the 1920s, the symbol was used, somewhat inconsistently, for "heat" in general. The definition of as strictly limited to enthalpy or "heat content at constant pressure" was formally proposed by A. W. Porter in 1922.
.
Notes
See also
*Calorimetry
In chemistry and thermodynamics, calorimetry () is the science or act of measuring changes in '' state variables'' of a body for the purpose of deriving the heat transfer associated with changes of its state due, for example, to chemical reac ...
* Calorimeter
A calorimeter is a device used for calorimetry, or the process of measuring the heat of chemical reactions or physical changes as well as heat capacity. Differential scanning calorimeters, isothermal micro calorimeters, titration calorimeters ...
* Departure function
In thermodynamics, a departure function is defined for any thermodynamic property as the difference between the property as computed for an ideal gas and the property of the species as it exists in the real world, for a specified temperature ''T'' ...
* Hess's law
* Isenthalpic process
An isenthalpic process or isoenthalpic process is a process that proceeds without any change in enthalpy, ''H''; or specific enthalpy, ''h''.
Overview
If a steady-state, steady-flow process is analysed using a control volume, everything outside ...
* Laws of thermodynamics
The laws of thermodynamics are a set of scientific laws which define a group of physical quantities, such as temperature, energy, and entropy, that characterize thermodynamic systems in thermodynamic equilibrium. The laws also use various param ...
* Stagnation enthalpy
* Standard enthalpy of formation
In chemistry and thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of enthalpy during the formation of 1 mole of the substance from its constituent elements in their reference state, w ...
* Thermodynamic databases
* Thermodynamics
Thermodynamics is a branch of physics that deals with heat, Work (thermodynamics), work, and temperature, and their relation to energy, entropy, and the physical properties of matter and radiation. The behavior of these quantities is governed b ...
References
Bibliography
* * * * * * * *External links
* * * {{Authority control State functions Energy (physics) Physical quantities