Drosera on:
[Wikipedia]
[Google]
[Amazon]
''Drosera'', which is commonly known as the sundews, is one of the largest
 Sundews are
Sundews are
 * ''Petiolaris'' complex: A group of tropical Australian species, they live in constantly warm but sometimes wet conditions. Several of the 14 species that comprise this group have developed special strategies to cope with the alternately drier conditions. Many species, for example, have petioles densely covered in
* ''Petiolaris'' complex: A group of tropical Australian species, they live in constantly warm but sometimes wet conditions. Several of the 14 species that comprise this group have developed special strategies to cope with the alternately drier conditions. Many species, for example, have petioles densely covered in
 Sundews are characterised by the glandular tentacles, topped with sticky secretions, that cover their
Sundews are characterised by the glandular tentacles, topped with sticky secretions, that cover their 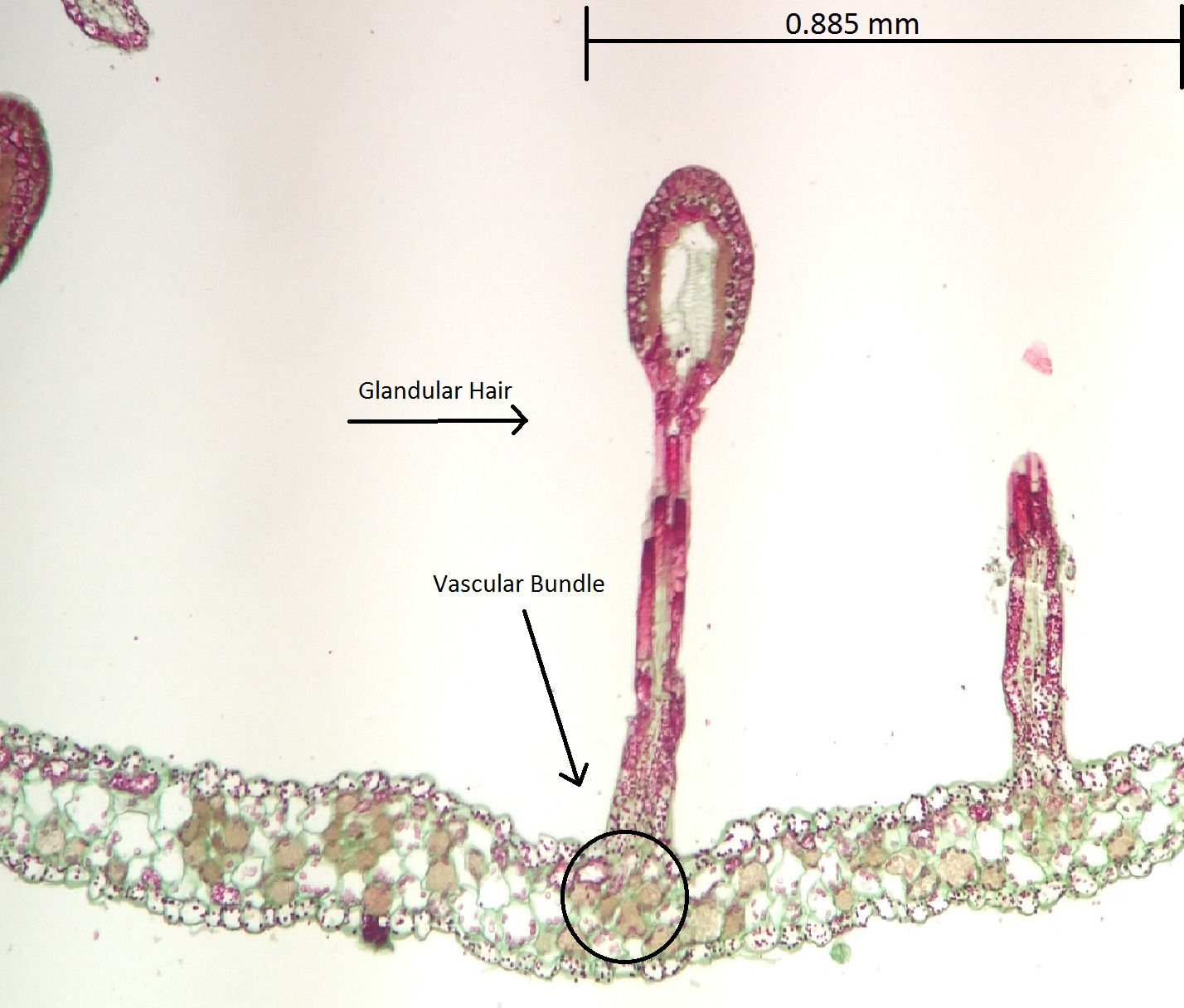 All species of sundew are able to move their tentacles in response to contact with edible prey. The tentacles are extremely sensitive and will bend toward the center of the leaf to bring the insect into contact with as many stalked glands as possible. According to
All species of sundew are able to move their tentacles in response to contact with edible prey. The tentacles are extremely sensitive and will bend toward the center of the leaf to bring the insect into contact with as many stalked glands as possible. According to  A further type of (mostly strong red and yellow) leaf coloration has recently been discovered in a few Australian species ('' D. hartmeyerorum'', '' D. indica''). Their function is not known yet, although they may help in attracting prey.
The leaf morphology of the species within the genus is extremely varied, ranging from the sessile
A further type of (mostly strong red and yellow) leaf coloration has recently been discovered in a few Australian species ('' D. hartmeyerorum'', '' D. indica''). Their function is not known yet, although they may help in attracting prey.
The leaf morphology of the species within the genus is extremely varied, ranging from the sessile
 The flowers of sundews, as with nearly all carnivorous plants, are held far above the leaves by a long stem. This physical isolation of the flower from the traps is commonly thought to be an adaptation meant to avoid trapping potential
The flowers of sundews, as with nearly all carnivorous plants, are held far above the leaves by a long stem. This physical isolation of the flower from the traps is commonly thought to be an adaptation meant to avoid trapping potential
 The root systems of most ''Drosera'' are often only weakly developed. Serving mainly to absorb water and to anchor the plant to the ground, the roots are relatively useless for nutrient uptake. A few South African species use their roots for water and food storage. Some species have wiry root systems that remain during frosts if the stem dies. Some species, such as ''D. adelae'' and ''D. hamiltonii'', use their roots for asexual propagation, by sprouting plantlets along their length. Some Australian species form underground
The root systems of most ''Drosera'' are often only weakly developed. Serving mainly to absorb water and to anchor the plant to the ground, the roots are relatively useless for nutrient uptake. A few South African species use their roots for water and food storage. Some species have wiry root systems that remain during frosts if the stem dies. Some species, such as ''D. adelae'' and ''D. hamiltonii'', use their roots for asexual propagation, by sprouting plantlets along their length. Some Australian species form underground
 The
The
 Sundews generally grow in seasonally moist or more rarely constantly wet habitats with acidic soils and high levels of sunlight. Common habitats include bogs,
Sundews generally grow in seasonally moist or more rarely constantly wet habitats with acidic soils and high levels of sunlight. Common habitats include bogs,
 None of the ''Drosera'' species in the United States are federally protected. Some are listed as
None of the ''Drosera'' species in the United States are federally protected. Some are listed as
File:Drosera. Tipulidae.jpg,
Artzneimittle, Tee, und Likör aus fleischfressenden Pflanzen
while another purple or yellow dye was traditionally prepared in the
genera
Genus ( plural genera ) is a taxonomic rank used in the biological classification of living and fossil organisms as well as viruses. In the hierarchy of biological classification, genus comes above species and below family. In binomial nomenclat ...
of carnivorous plant
Carnivorous plants are plants that derive some or most of their nutrients from trapping and consuming animals or protozoans
Protozoa (singular: protozoan or protozoon; alternative plural: protozoans) are a group of single-celled eukaryot ...
s, with at least 194 species
In biology, a species is the basic unit of classification and a taxonomic rank of an organism, as well as a unit of biodiversity. A species is often defined as the largest group of organisms in which any two individuals of the appropriate s ...
. 2 volumes. These members of the family Droseraceae
Droseraceae is a family (biology), family of carnivorous flowering plants, also known as the sundew family. It consists of approximately 180 species in three Extant taxon, extant genera. Representatives of the Droseraceae are found on all conti ...
lure, capture, and digest insects using stalked mucilaginous
Mucilage is a thick, gluey substance produced by nearly all plants and some microorganisms. These microorganisms include protists which use it for their locomotion. The direction of their movement is always opposite to that of the secretion of m ...
glands covering their leaf surfaces. The insects are used to supplement the poor mineral nutrition of the soil in which the plants grow. Various species, which vary greatly in size and form, are native to every continent except Antarctica
Antarctica () is Earth's southernmost and least-populated continent. Situated almost entirely south of the Antarctic Circle and surrounded by the Southern Ocean, it contains the geographic South Pole. Antarctica is the fifth-largest contine ...
.McPherson, S.R. 2008. ''Glistening Carnivores''. Redfern Natural
Nature, in the broadest sense, is the physical world or universe. "Nature" can refer to the phenomena of the physical world, and also to life in general. The study of nature is a large, if not the only, part of science. Although humans are p ...
History Productions Ltd., Poole.
Charles Darwin
Charles Robert Darwin ( ; 12 February 1809 – 19 April 1882) was an English naturalist, geologist, and biologist, widely known for his contributions to evolutionary biology. His proposition that all species of life have descended fr ...
performed much of the early research into ''Drosera'', engaging in a long series of experiments with Drosera rotundifolia
''Drosera rotundifolia'', the round-leaved sundew, roundleaf sundew, or common sundew, is a carnivorous species of flowering plant that grows in bogs, marshes and fens. One of the most widespread sundew species, it has a circumboreal distribu ...
which were the first to confirm carnivory in plants. In an 1860 letter, Darwin wrote, “…at the present moment, I care more about ''Drosera'' than the origin of all the species in the world.”
Both the botanical name (from the Greek
Greek may refer to:
Greece
Anything of, from, or related to Greece, a country in Southern Europe:
*Greeks, an ethnic group.
*Greek language, a branch of the Indo-European language family.
**Proto-Greek language, the assumed last common ancestor ...
δρόσος: ''drosos'' = "dew, dewdrops") and the English common name (sundew, derived from Latin
Latin (, or , ) is a classical language belonging to the Italic branch of the Indo-European languages. Latin was originally a dialect spoken in the lower Tiber area (then known as Latium) around present-day Rome, but through the power of the ...
''ros solis'', meaning "dew of the sun") refer to the glistening drops of mucilage
Mucilage is a thick, gluey substance produced by nearly all plants and some microorganisms. These microorganisms include protists which use it for their locomotion. The direction of their movement is always opposite to that of the secretion of m ...
at the tip of the glandular trichomes
Trichomes (); ) are fine outgrowths or appendages on plants, algae, lichens, and certain protists. They are of diverse structure and function. Examples are hairs, glandular hairs, scales, and papillae. A covering of any kind of hair on a plant ...
that resemble drops of morning dew
Dew is water in the form of droplets that appears on thin, exposed objects in the morning or evening due to condensation.
As the exposed surface cools by radiating its heat, atmospheric moisture condenses at a rate greater than that at wh ...
. The ''Principia Botanica'', published in 1787, states “Sun-dew (''Drosera'') derives its name from small drops of a liquor-like dew, hanging on its fringed leaves, and continuing in the hottest part of the day, exposed to the sun.”
Description
 Sundews are
Sundews are perennial
A perennial plant or simply perennial is a plant that lives more than two years. The term ('' per-'' + '' -ennial'', "through the years") is often used to differentiate a plant from shorter-lived annuals and biennials. The term is also wide ...
(or rarely annual
Annual may refer to:
*Annual publication, periodical publications appearing regularly once per year
** Yearbook
** Literary annual
*Annual plant
*Annual report
*Annual giving
*Annual, Morocco, a settlement in northeastern Morocco
*Annuals (band), ...
) herbaceous plant
Herbaceous plants are vascular plants that have no persistent woody stems above ground. This broad category of plants includes many perennials, and nearly all annuals and biennials.
Definitions of "herb" and "herbaceous"
The fourth edition of t ...
s, forming prostrate or upright rosettes between in height, depending on the species. Climbing species form scrambling stems which can reach much longer lengths, up to in the case of '' D. erythrogyne''. Sundews have been shown to be able to achieve a lifespan of 50 years. The genus is specialized for nutrient
A nutrient is a substance used by an organism to survive, grow, and reproduce. The requirement for dietary nutrient intake applies to animals, plants, fungi, and protists. Nutrients can be incorporated into cells for metabolic purposes or excret ...
uptake through its carnivorous behavior, for example the pygmy sundew is missing the enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. A ...
s (nitrate reductase
Nitrate reductases are molybdoenzymes that reduce nitrate (NO) to nitrite (NO). This reaction is critical for the production of protein in most crop plants, as nitrate is the predominant source of nitrogen in fertilized soils.
Types
Euka ...
, in particular) that plants normally use for the uptake of earth-bound nitrates.
Habit
The genus can be divided into severalhabits
A habit (or wont as a humorous and formal term) is a routine of behavior that is repeated regularly and tends to occur subconsciously.
, or growth forms:
* Temperate sundews: These species form a tight cluster of unfurled leaves called a hibernaculum in a winter dormancy period (= Hemicryptophyte). All of the North American and European species belong to this group. ''Drosera arcturi'' from Australia (including Tasmania) and New Zealand is another temperate species that dies back to a horn-shaped hibernaculum.
* Subtropical sundews: These species maintain vegetative growth year-round under uniform or nearly uniform climatic conditions.
* Pygmy sundews: A group of roughly 40 Australian species, they are distinguished by miniature growth, the formation of gemmae for asexual reproduction
Asexual reproduction is a type of reproduction that does not involve the fusion of gametes or change in the number of chromosomes. The offspring that arise by asexual reproduction from either unicellular or multicellular organisms inherit the fu ...
, and dense formation of hairs in the crown center. These hairs serve to protect the plants from Australia's intense summer sun. Pygmy sundews form the subgenus '' Bryastrum''.
* Tuberous sundews: These nearly 50 Australian species form an underground tuber
Tubers are a type of enlarged structure used as storage organs for nutrients in some plants. They are used for the plant's perennation (survival of the winter or dry months), to provide energy and nutrients for regrowth during the next growing ...
to survive the extremely dry summers of their habitat, re-emerging in the autumn. These so-called tuberous sundews can be further divided into two groups, those that form rosettes and those that form climbing or scrambling stems. Tuberous sundews comprise the subgenus '' Ergaleium''.
trichome
Trichomes (); ) are fine outgrowths or appendages on plants, algae, lichens, and certain protists. They are of diverse structure and function. Examples are hairs, glandular hairs, scales, and papillae. A covering of any kind of hair on a pla ...
s, which maintain a sufficiently humid environment and serve as an increased condensation
Condensation is the change of the state of matter from the gas phase into the liquid phase, and is the reverse of vaporization. The word most often refers to the water cycle. It can also be defined as the change in the state of water vapor to ...
surface for morning dew. The ''Petiolaris'' complex comprises the subgenus '' Lasiocephala''.
Although they do not form a single strictly defined growth form, a number of species are often put together in a further group:
* Queensland sundews: A small group of three species ('' D. adelae'', '' D. schizandra'' and '' D. prolifera''), all are native to highly humid habitats in the dim understories of the Australian rainforest.
Leaves and carnivory
leaves
A leaf (plural, : leaves) is any of the principal appendages of a vascular plant plant stem, stem, usually borne laterally aboveground and specialized for photosynthesis. Leaves are collectively called foliage, as in "autumn foliage", wh ...
. The trapping and digestion mechanism usually employs two types of glands: stalked glands that secrete sweet mucilage to attract and ensnare insects and enzymes to digest them, and sessile
Sessility, or sessile, may refer to:
* Sessility (motility), organisms which are not able to move about
* Sessility (botany), flowers or leaves that grow directly from the stem or peduncle of a plant
* Sessility (medicine), tumors and polyps that ...
gland
In animals, a gland is a group of cells in an animal's body that synthesizes substances (such as hormones) for release into the bloodstream (endocrine gland) or into cavities inside the body or its outer surface (exocrine gland).
Structure
De ...
s that absorb the resulting nutrient soup (the latter glands are missing in some species, such as '' D. erythrorhiza''). Small prey, mainly consisting of insects, are attracted by the sweet secretions of the peduncular glands. Upon touching these, the prey become entrapped by sticky mucilage which prevents their progress or escape. Eventually, the prey either succumb to death through exhaustion or through asphyxiation as the mucilage envelops them and clogs their spiracle Spiracle or spiraculum may refer to:
* Spiracle (arthropods), opening in the exoskeletons of some arthropods
* Spiracle (vertebrates), openings on the surface of some vertebrates
* Spiraculum, a genus of land snails in family Cyclophoridae
Cycl ...
s. Death usually occurs within 15 minutes. The plant meanwhile secretes esterase
An esterase is a hydrolase enzyme that splits esters into an acid and an alcohol in a chemical reaction with water called hydrolysis.
A wide range of different esterases exist that differ in their substrate specificity, their protein structure, ...
, peroxidase
Peroxidases or peroxide reductases ( EC numberbr>1.11.1.x are a large group of enzymes which play a role in various biological processes. They are named after the fact that they commonly break up peroxides.
Functionality
Peroxidases typically ca ...
, phosphatase
In biochemistry, a phosphatase is an enzyme that uses water to cleave a phosphoric acid Ester, monoester into a phosphate ion and an Alcohol (chemistry), alcohol. Because a phosphatase enzyme catalysis, catalyzes the hydrolysis of its Substrate ...
and protease
A protease (also called a peptidase, proteinase, or proteolytic enzyme) is an enzyme that catalyzes (increases reaction rate or "speeds up") proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the ...
enzymes
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecule ...
. These enzymes dissolve the insect and free the nutrients contained within it. This nutrient mixture is then absorbed through the leaf surfaces to be used by the rest of the plant.
 All species of sundew are able to move their tentacles in response to contact with edible prey. The tentacles are extremely sensitive and will bend toward the center of the leaf to bring the insect into contact with as many stalked glands as possible. According to
All species of sundew are able to move their tentacles in response to contact with edible prey. The tentacles are extremely sensitive and will bend toward the center of the leaf to bring the insect into contact with as many stalked glands as possible. According to Charles Darwin
Charles Robert Darwin ( ; 12 February 1809 – 19 April 1882) was an English naturalist, geologist, and biologist, widely known for his contributions to evolutionary biology. His proposition that all species of life have descended fr ...
, the contact of the legs of a small gnat with a single tentacle is enough to induce this response. This response to touch is known as thigmonasty
Thigmonasty or seismonasty is the nastic movement, nastic response of a plant or fungus to touch or vibration. Conspicuous examples of thigmonasty include many species in the Fabaceae, leguminous family (biology), subfamily Mimosoideae, active carn ...
, and is quite rapid in some species. The outer tentacles (recently coined as "snap-tentacles") of '' D. burmannii'' and '' D. sessilifolia'' can bend inwards toward prey in a matter of seconds after contact, while '' D. glanduligera'' is known to bend these tentacles in toward prey in tenths of a second. In addition to tentacle movement, some species are able to bend their leaves to various degrees to maximize contact with the prey. Of these, '' D. capensis'' exhibits what is probably the most dramatic movement, curling its leaf completely around prey in 30 minutes. Some species, such as '' D. filiformis'', are unable to bend their leaves in response to prey.
ovate
Ovate may refer to:
* Ovate (egg-shaped) leaves, tepals, or other botanical parts
*Ovate, a type of prehistoric stone hand axe
*Ovates, one of three ranks of membership in the Welsh Gorsedd
*Vates
In modern English, the nouns vates () and ova ...
leaves of ''D. erythrorhiza'' to the bipinnately divided acicular leaves of '' D. binata''.
While the exact physiological mechanism of the sundew's carnivorous response is not yet known, some studies have begun to shed light on how the plant is able to move in response to mechanical and chemical stimulation to envelop and digest prey. Individual tentacles, when mechanically stimulated, fire action potentials that terminate near the base of the tentacle, resulting in rapid movement of the tentacle towards the center of the leaf. This response is more prominent when marginal tentacles further away from the leaf center are stimulated. The tentacle movement response is achieved through auxin-mediated acid growth Acid growth refers to the ability of plant cells and plant cell walls to elongate or expand quickly at low (acidic) pH. The cell wall needs to be modified in order to maintain the turgor pressure. This modification is controlled by plant hormones l ...
. When action potentials reach their target cells, the plant hormone auxin
Auxins (plural of auxin ) are a class of plant hormones (or plant-growth regulators) with some morphogen-like characteristics. Auxins play a cardinal role in coordination of many growth and behavioral processes in plant life cycles and are essenti ...
causes protons (H+ ions) to be pumped out of the plasma membrane into the cell wall, thereby reducing the pH and making the cell wall more acidic. The resulting reduction in pH causes the relaxation of the cell wall protein, expansin, and allows for an increase in cell volume via osmosis and turgor. As a result of differential cell growth rates, the sundew tentacles are able to achieve movement towards prey and the leaf center through the bending caused by expanding cells. Among some drosera species, a second bending response occurs in which non-local, distant tentacles bend towards prey as well as the bending of the entire leaf blade to maximize contact with prey. While mechanical stimulation is sufficient to achieve a localized tentacle bend response, both mechanical and chemical stimuli are required for the secondary bending response to occur.
Flowers and fruit
 The flowers of sundews, as with nearly all carnivorous plants, are held far above the leaves by a long stem. This physical isolation of the flower from the traps is commonly thought to be an adaptation meant to avoid trapping potential
The flowers of sundews, as with nearly all carnivorous plants, are held far above the leaves by a long stem. This physical isolation of the flower from the traps is commonly thought to be an adaptation meant to avoid trapping potential pollinator
A pollinator is an animal that moves pollen from the male anther of a flower to the female stigma of a flower. This helps to bring about fertilization of the ovules in the flower by the male gametes from the pollen grains.
Insects are the maj ...
s. The mostly unforked inflorescence
An inflorescence is a group or cluster of flowers arranged on a stem that is composed of a main branch or a complicated arrangement of branches. Morphologically, it is the modified part of the shoot of seed plants where flowers are formed o ...
s are spikes
The SPIKES protocol is a method used in clinical medicine to break bad news to patients and families. As receiving bad news can cause distress and anxiety, clinicians need to deliver the news carefully. By using the SPIKES method for introducing a ...
, whose flowers open one at a time and usually only remain open for a short period. Flowers open in response to light intensity (often opening only in direct sunlight), and the entire inflorescence is also heliotropic
Heliotropism, a form of tropism, is the diurnal or seasonal motion of plant parts (flowers or leaves) in response to the direction of the Sun.
The habit of some plants to move in the direction of the Sun, a form of tropism, was already known by t ...
, moving in response to the sun's position in the sky.
The radially symmetrical
Symmetry in biology refers to the symmetry observed in organisms, including plants, animals, fungi, and bacteria. External symmetry can be easily seen by just looking at an organism. For example, take the face of a human being which has a pla ...
(actinomorphic
Floral symmetry describes whether, and how, a flower, in particular its perianth, can be divided into two or more identical or mirror-image parts.
Uncommonly, flowers may have no axis of symmetry at all, typically because their parts are spirall ...
) flowers are always perfect and have five parts (the exceptions to this rule are the four-petaled '' D. pygmaea'' and the eight to 12-petaled '' D. heterophylla''). Most of the species have small flowers (<1.5 cm or 0.6 in). A few species, however, such as '' D. regia'' and '' D. cistiflora'', have flowers or more in diameter. In general, the flowers are white or pink. Australian species display a wider range of colors, including orange ('' D. callistos''), red ('' D. adelae''), yellow ('' D. zigzagia'') or metallic violet ('' D. microphylla'').
The ovary
The ovary is an organ in the female reproductive system that produces an ovum. When released, this travels down the fallopian tube into the uterus, where it may become fertilized by a sperm. There is an ovary () found on each side of the body. ...
is superior and develops into a dehiscent seed capsule
In botany a capsule is a type of simple, dry, though rarely fleshy dehiscent fruit produced by many species of angiosperms (flowering plants).
Origins and structure
The capsule (Latin: ''capsula'', small box) is derived from a compound (multica ...
bearing numerous tiny seeds. The pollen
Pollen is a powdery substance produced by seed plants. It consists of pollen grains (highly reduced microgametophytes), which produce male gametes (sperm cells). Pollen grains have a hard coat made of sporopollenin that protects the gametophyt ...
grain type is compound, which means four microspore
Microspores are land plant spores that develop into male gametophytes, whereas megaspores develop into female gametophytes. The male gametophyte gives rise to sperm cells, which are used for fertilization of an egg cell to form a zygote. Megaspor ...
s (pollen grains) are stuck together with a protein called callose
Callose is a plant polysaccharide. Its production is due to the glucan synthase-like gene (GLS) in various places within a plant. It is produced to act as a temporary cell wall in response to stimuli such as stress or damage. Callose is composed ...
.
Roots
 The root systems of most ''Drosera'' are often only weakly developed. Serving mainly to absorb water and to anchor the plant to the ground, the roots are relatively useless for nutrient uptake. A few South African species use their roots for water and food storage. Some species have wiry root systems that remain during frosts if the stem dies. Some species, such as ''D. adelae'' and ''D. hamiltonii'', use their roots for asexual propagation, by sprouting plantlets along their length. Some Australian species form underground
The root systems of most ''Drosera'' are often only weakly developed. Serving mainly to absorb water and to anchor the plant to the ground, the roots are relatively useless for nutrient uptake. A few South African species use their roots for water and food storage. Some species have wiry root systems that remain during frosts if the stem dies. Some species, such as ''D. adelae'' and ''D. hamiltonii'', use their roots for asexual propagation, by sprouting plantlets along their length. Some Australian species form underground corm
A corm, bulbo-tuber, or bulbotuber is a short, vertical, swollen underground plant stem that serves as a storage organ that some plants use to survive winter or other adverse conditions such as summer drought and heat (perennation).
The word ' ...
s for this purpose, which also serve to allow the plants to survive dry summers. The roots of pygmy sundews are often extremely long in proportion to their size, with a 1-cm (0.4-in) plant extending roots over beneath the soil surface. Some pygmy sundews, such as '' D. lasiantha'' and '' D. scorpioides'', also form adventitious root Important structures in plant development are buds, shoots, roots, leaves, and flowers; plants produce these tissues and structures throughout their life from meristems located at the tips of organs, or between mature tissues. Thus, a living plant a ...
s as supports. ''D. intermedia'' and D. rotundifolia have been reported to form arbuscular mycorrhiza
An arbuscular mycorrhiza (AM) (plural ''mycorrhizae'', a.k.a. ''endomycorrhiza'') is a type of mycorrhiza in which the symbiont fungus (''AM fungi'', or AMF) penetrates the cortical cells of the roots of a vascular plant forming arbuscules. (N ...
e, which penetrate the plant's tissues.
Taxonomy and phylogenetics
The unrootedcladogram
A cladogram (from Greek ''clados'' "branch" and ''gramma'' "character") is a diagram used in cladistics to show relations among organisms. A cladogram is not, however, an evolutionary tree because it does not show how ancestors are related to d ...
to the right shows the relationship between various subgenera and classes as defined by the analysis of Rivadavia ''et al.'' The monotypic section Meristocaulis was not included in the study, so its place in this system is unclear. More recent studies have placed this group near section ''Bryastrum'', so it is placed there below. Also of note, the placement of the section ''Regiae'' in relation to ''Aldrovanda'' and ''Dionaea'' is uncertain. Since the section ''Drosera'' is polyphyletic
A polyphyletic group is an assemblage of organisms or other evolving elements that is of mixed evolutionary origin. The term is often applied to groups that share similar features known as homoplasies, which are explained as a result of converg ...
, it shows up multiple times in the cladogram (*).
This phylogenetic study has made the need for a revision of the genus even clearer.
Reproduction
Many species of sundews are self-fertile; their flowers will often self-pollinate upon closing. Often, numerous seeds are produced. The tiny black seeds germinate in response to moisture and light, while seeds of temperate species also require cold, damp,stratification
Stratification may refer to:
Mathematics
* Stratification (mathematics), any consistent assignment of numbers to predicate symbols
* Data stratification in statistics
Earth sciences
* Stable and unstable stratification
* Stratification, or st ...
to germinate. Seeds of the tuberous species require a hot, dry summer period followed by a cool, moist winter to germinate.
Vegetative reproduction
Vegetative reproduction (also known as vegetative propagation, vegetative multiplication or cloning) is any form of asexual reproduction occurring in plants in which a new plant grows from a fragment or cutting of the parent plant or spec ...
occurs naturally in some species that produce stolons
In biology, stolons (from Latin '' stolō'', genitive ''stolōnis'' – "branch"), also known as runners, are horizontal connections between organisms. They may be part of the organism, or of its skeleton; typically, animal stolons are external s ...
or when roots come close to the surface of the soil. Older leaves that touch the ground may sprout plantlets. Pygmy sundews reproduce asexually using specialized scale-like leaves called gemmae. Tuberous sundews can produce offsets from their corms.
In culture, sundews can often be propagated through leaf, crown, or root cuttings, as well as through seeds.
Distribution
range
Range may refer to:
Geography
* Range (geographic), a chain of hills or mountains; a somewhat linear, complex mountainous or hilly area (cordillera, sierra)
** Mountain range, a group of mountains bordered by lowlands
* Range, a term used to i ...
of the sundew genus stretches from Alaska in the north to New Zealand in the south. The centers of diversity
Diversity, diversify, or diverse may refer to:
Business
*Diversity (business), the inclusion of people of different identities (ethnicity, gender, age) in the workforce
*Diversity marketing, marketing communication targeting diverse customers
* ...
are Australia, with roughly 50% of all known species, and South America and southern Africa, each with more than 20 species. A few species are also found in large parts of Eurasia and North America. These areas, however, can be considered to form the outskirts of the generic range, as the ranges of sundews do not typically approach temperate or Arctic areas. Contrary to previous supposition, the evolutionary speciation
Speciation is the evolutionary process by which populations evolve to become distinct species. The biologist Orator F. Cook coined the term in 1906 for cladogenesis, the splitting of lineages, as opposed to anagenesis, phyletic evolution within ...
of this genus is no longer thought to have occurred with the breakup of Gondwana
Gondwana () was a large landmass, often referred to as a supercontinent, that formed during the late Neoproterozoic (about 550 million years ago) and began to break up during the Jurassic period (about 180 million years ago). The final stages ...
through continental drift
Continental drift is the hypothesis that the Earth's continents have moved over geologic time relative to each other, thus appearing to have "drifted" across the ocean bed. The idea of continental drift has been subsumed into the science of pla ...
. Rather, speciation is now thought to have occurred as a result of a subsequent wide dispersal of its range. The origins of the genus are thought to have been in Africa or Australia.
Europe is home to only three species: '' D. intermedia'', '' D. anglica'', and '' D. rotundifolia''. Where the ranges of the two latter species overlap, they sometimes hybridize to form the sterile '' D. × obovata''. In addition to the three species and the hybrid native to Europe, North America is also home to four additional species; '' D. brevifolia'' is a small annual
Annual may refer to:
*Annual publication, periodical publications appearing regularly once per year
** Yearbook
** Literary annual
*Annual plant
*Annual report
*Annual giving
*Annual, Morocco, a settlement in northeastern Morocco
*Annuals (band), ...
native to coastal states from Texas
Texas (, ; Spanish language, Spanish: ''Texas'', ''Tejas'') is a state in the South Central United States, South Central region of the United States. At 268,596 square miles (695,662 km2), and with more than 29.1 million residents in 2 ...
to Virginia
Virginia, officially the Commonwealth of Virginia, is a state in the Mid-Atlantic and Southeastern regions of the United States, between the Atlantic Coast and the Appalachian Mountains. The geography and climate of the Commonwealth ar ...
, while '' D. capillaris'', a slightly larger plant with a similar range, is also found in areas of the Caribbean. The third species, '' D. linearis'', is native to the northern United States and southern Canada. '' D. filiformis'' has two subspecies
In biological classification, subspecies is a rank below species, used for populations that live in different areas and vary in size, shape, or other physical characteristics (morphology), but that can successfully interbreed. Not all species ...
native to the East Coast of North America, the Gulf Coast
The Gulf Coast of the United States, also known as the Gulf South, is the coastline along the Southern United States where they meet the Gulf of Mexico. The coastal states that have a shoreline on the Gulf of Mexico are Texas, Louisiana, Mississ ...
, and the Florida panhandle
The Florida Panhandle (also West Florida and Northwest Florida) is the northwestern part of the U.S. state of Florida; it is a Salient (geography), salient roughly long and wide, lying between Alabama on the north and the west, Georgia (U. ...
.
This genus is often described as cosmopolitan
Cosmopolitan may refer to:
Food and drink
* Cosmopolitan (cocktail), also known as a "Cosmo"
History
* Rootless cosmopolitan, a Soviet derogatory epithet during Joseph Stalin's anti-Semitic campaign of 1949–1953
Hotels and resorts
* Cosmopoli ...
, meaning it has worldwide distribution. The botanist Ludwig Diels
Dr. Friedrich Ludwig Emil Diels (24 September 1874 – 30 November 1945) was a German botanist.
Diels was born in Hamburg, the son of the classical scholar Hermann Alexander Diels. From 1900 to 1902 he traveled together with Ernst Georg Prit ...
, author of the only monograph
A monograph is a specialist work of writing (in contrast to reference works) or exhibition on a single subject or an aspect of a subject, often by a single author or artist, and usually on a scholarly subject.
In library cataloging, ''monograph ...
of the family to date, called this description an "arrant misjudgment of this genus' highly unusual distributional circumstances (''arge Verkennung ihrer höchst eigentümlichen Verbreitungsverhältnisse'')", while admitting sundew species do "occupy a significant part of the Earth's surface (''einen beträchtlichen Teil der Erdoberfläche besetzt'')".
He particularly pointed to the absence of ''Drosera'' species from almost all arid
A region is arid when it severely lacks available water, to the extent of hindering or preventing the growth and development of plant and animal life. Regions with arid climates tend to lack vegetation and are called xeric or desertic. Most ar ...
climate zones, countless rainforest
Rainforests are characterized by a closed and continuous tree canopy, moisture-dependent vegetation, the presence of epiphytes and lianas and the absence of wildfire. Rainforest can be classified as tropical rainforest or temperate rainfores ...
s, the American Pacific Coast, Polynesia
Polynesia () "many" and νῆσος () "island"), to, Polinisia; mi, Porinihia; haw, Polenekia; fj, Polinisia; sm, Polenisia; rar, Porinetia; ty, Pōrīnetia; tvl, Polenisia; tkl, Polenihia (, ) is a subregion of Oceania, made up of ...
, the Mediterranean region, and North Africa, as well as the scarcity of species diversity in temperate zones, such as Europe and North America.
Habitat
 Sundews generally grow in seasonally moist or more rarely constantly wet habitats with acidic soils and high levels of sunlight. Common habitats include bogs,
Sundews generally grow in seasonally moist or more rarely constantly wet habitats with acidic soils and high levels of sunlight. Common habitats include bogs, fen
A fen is a type of peat-accumulating wetland fed by mineral-rich ground or surface water. It is one of the main types of wetlands along with marshes, swamps, and bogs. Bogs and fens, both peat-forming ecosystems, are also known as mires. T ...
s, swamp
A swamp is a forested wetland.Keddy, P.A. 2010. Wetland Ecology: Principles and Conservation (2nd edition). Cambridge University Press, Cambridge, UK. 497 p. Swamps are considered to be transition zones because both land and water play a role in ...
s, marsh
A marsh is a wetland that is dominated by herbaceous rather than woody plant species.Keddy, P.A. 2010. Wetland Ecology: Principles and Conservation (2nd edition). Cambridge University Press, Cambridge, UK. 497 p Marshes can often be found at ...
es, the tepui
A tepui , or tepuy (), is a table-top mountain or mesa found in South America, especially in Venezuela and western Guyana. The word tepui means "house of the gods" in the native tongue of the Pemon, the indigenous people who inhabit the Gran S ...
s of Venezuela, the wallum
Wallum, or wallum country, is an Australian ecosystem of coastal south-east Queensland, extending into north-eastern New South Wales. It is characterised by flora-rich shrubland and heathland on deep, nutrient-poor, acidic, sandy soils, and re ...
s of coastal Australia, the fynbos
Fynbos (; meaning fine plants) is a small belt of natural shrubland or heathland vegetation located in the Western Cape and Eastern Cape provinces of South Africa. This area is predominantly coastal and mountainous, with a Mediterranean clim ...
of South Africa, and moist streambanks. Many species grow in association with sphagnum moss
''Sphagnum'' is a genus of approximately 380 accepted species of mosses, commonly known as sphagnum moss, peat moss, also bog moss and quacker moss (although that term is also sometimes used for peat). Accumulations of ''Sphagnum'' can store wa ...
, which absorbs much of the soil's nutrient supply and also acidifies the soil, making nutrients less available to plant life. This allows sundews, which do not rely on soil-bound nutrients, to flourish where more dominating vegetation would usually outcompete them.
The genus, though, is very variable in terms of habitat. Individual sundew species have adapted to a wide variety of environments, including atypical habitats, such as rainforests, deserts ('' D. burmannii'' and '' D. indica''), and even highly shaded environments (Queensland sundews). The temperate species, which form hibernacula in the winter, are examples of such adaptation to habitats; in general, sundews tend to inhabit warm climates, and are only moderately frost-resistant.
Conservation status
 None of the ''Drosera'' species in the United States are federally protected. Some are listed as
None of the ''Drosera'' species in the United States are federally protected. Some are listed as threatened
Threatened species are any species (including animals, plants and fungi) which are vulnerable to endangerment in the near future. Species that are threatened are sometimes characterised by the population dynamics measure of ''critical depensat ...
or endangered
An endangered species is a species that is very likely to become extinct in the near future, either worldwide or in a particular political jurisdiction. Endangered species may be at risk due to factors such as habitat loss, poaching and inva ...
at the state level, but this gives little protection to lands under private ownership. Many of the remaining native populations are located on protected land, such as national park
A national park is a nature park, natural park in use for conservation (ethic), conservation purposes, created and protected by national governments. Often it is a reserve of natural, semi-natural, or developed land that a sovereign state dec ...
s or wildlife preserves.
''Drosera'' species are protected by law in many European countries, such as Germany, Austria, Switzerland, the Czech Republic, Finland, Hungary
Hungary ( hu, Magyarország ) is a landlocked country in Central Europe. Spanning of the Carpathian Basin, it is bordered by Slovakia to the north, Ukraine to the northeast, Romania to the east and southeast, Serbia to the south, Croatia a ...
, France
France (), officially the French Republic ( ), is a country primarily located in Western Europe. It also comprises of Overseas France, overseas regions and territories in the Americas and the Atlantic Ocean, Atlantic, Pacific Ocean, Pac ...
, and Bulgaria.
In Australia, they are listed as "threatened". In South America and the Caribbean, ''Drosera'' species in a number of areas are considered critical, endangered or vulnerable, while other areas have not been surveyed. At the same time that species are at risk in South Africa, new species continue to be discovered in the Western Cape
The Western Cape is a province of South Africa, situated on the south-western coast of the country. It is the fourth largest of the nine provinces with an area of , and the third most populous, with an estimated 7 million inhabitants in 2020 ...
and Madagascar
Madagascar (; mg, Madagasikara, ), officially the Republic of Madagascar ( mg, Repoblikan'i Madagasikara, links=no, ; french: République de Madagascar), is an island country in the Indian Ocean, approximately off the coast of East Africa ...
.
Worldwide, ''Drosera'' are at risk of extinction due to the destruction of natural habitat through urban and agricultural development. They are also threatened by the illegal collection of wild plants for the horticultural trade. An additional risk is environmental change Environmental change is a change or disturbance of the environment most often caused by human influences and natural ecological processes. Environmental changes include various factors, such as natural disasters, human interferences, or animal in ...
, because species are often specifically adapted to a precise location and set of conditions.
Currently, the largest threat in Europe and North America is loss of wetland habitat. Causes include urban development and the draining of bogs for agricultural uses and peat harvesting.
Such threats have led to the extirpation of some species from parts of their former range. Reintroduction of plants into such habitats is usually difficult or impossible, as the ecological needs of certain populations are closely tied to their geographical location. Increased legal protection of bogs and moors, and a concentrated effort to renaturalize such habitats, are possible ways to combat threats to ''Drosera'' plants' survival.
As part of the landscape, sundews are often overlooked or not recognized at all.
The relatively unimpressive image of these plants, as well as their small, low growth, makes them difficult to protect.
In South Africa and Australia, two of the three centers of species diversity
Species diversity is the number of different species that are represented in a given community (a dataset). The effective number of species refers to the number of equally abundant species needed to obtain the same mean proportional species abundan ...
, the natural habitats of these plants are undergoing a high degree of pressure from human activities. The African sundews '' D. insolita'' and '' D. katangensis'' are listed as critically endangered by the International Union for Conservation of Nature
The International Union for Conservation of Nature (IUCN; officially International Union for Conservation of Nature and Natural Resources) is an international organization working in the field of nature conservation and sustainable use of natu ...
(IUCN), while '' D. bequaertii'' is listed as vulnerable.
Expanding population centers such as Queensland
)
, nickname = Sunshine State
, image_map = Queensland in Australia.svg
, map_caption = Location of Queensland in Australia
, subdivision_type = Country
, subdivision_name = Australia
, established_title = Before federation
, established_ ...
, Perth
Perth is the capital and largest city of the Australian state of Western Australia. It is the fourth most populous city in Australia and Oceania, with a population of 2.1 million (80% of the state) living in Greater Perth in 2020. Perth is ...
, and Cape Town
Cape Town ( af, Kaapstad; , xh, iKapa) is one of South Africa's three capital cities, serving as the seat of the Parliament of South Africa. It is the legislative capital of the country, the oldest city in the country, and the second largest ...
, and the draining of moist areas for agriculture and forestry in rural areas threaten many such habitats. The droughts that have been sweeping Australia over the last 10 years also pose a threat to many species by drying up previously moist areas.
Those species endemic to a very limited area are often most threatened by the collection of plants from the wild. '' D. madagascariensis'' is considered endangered in Madagascar
Madagascar (; mg, Madagasikara, ), officially the Republic of Madagascar ( mg, Repoblikan'i Madagasikara, links=no, ; french: République de Madagascar), is an island country in the Indian Ocean, approximately off the coast of East Africa ...
because of the large-scale removal of plants from the wild for exportation; 10 - 200 million plants are harvested for commercial medicinal use annually.
Gallery of prey
Tipulidae
Crane fly is a common name referring to any member of the insect family (biology), family Tipulidae. Cylindrotominae, Limoniinae, and Pediciinae have been ranked as subfamilies of Tipulidae by most authors, though occasionally elevated to family ...
(crane fly) trapped by ''Drosera filiformis''
File:Drosera. Phalaenophana.jpg, Moth, ''Phalaenophana pyramusalis'' (Dark-banded Owlet) trapped by ''Drosera filiformis''
File:Drosera. Eusarca confusaria.jpg, ''Eusarca confusaria'' moth trapped by ''Drosera filiformis''
File:Drosera. Tabanus ventral.jpg, ''Tabanus'' fly trapped by ''Drosera filiformis''
File:Drosophila melanogaster ♀ Melgen, 1830, Drosera capensis Linnaeus, 1753 1100.1.2171.JPG, ''Drosophila melanogaster'' fly trapped by ''Drosera capensis''
Uses
As a medicinal plant
Sundews were used as medicinal herbs as early as the 12th century, when an Italian doctor from the School of Salerno,Matthaeus Platearius
Matthaeus Platearius was a physician from the medical school at Salerno, and is thought to have produced a twelfth-century Latin manuscript on medicinal herbs titled "''Circa Instans''" (also known as "''The Book of Simple Medicines''"), later tra ...
, described the plant as an herbal remedy for cough
A cough is a sudden expulsion of air through the large breathing passages that can help clear them of fluids, irritants, foreign particles and microbes. As a protective reflex, coughing can be repetitive with the cough reflex following three pha ...
s under the name ''herba sole''.
Culbreth's 1927 ''Materia Medica'' listed ''D. rotundifolia'', ''D. anglica'' and '' D. linearis'' as being used as stimulants
Stimulants (also often referred to as psychostimulants or colloquially as uppers) is an overarching term that covers many drugs including those that increase activity of the central nervous system and the body, drugs that are pleasurable and inv ...
and expectorant
Mucoactive agents are a class of chemical agents that aid in the clearance of mucus or sputum from the upper and lower airways, including the lungs, bronchi, and trachea. Mucoactive drugs include expectorants, mucolytics, mucoregulators, and muco ...
s, and "of doubtful efficacy" for treating bronchitis
Bronchitis is inflammation of the bronchi (large and medium-sized airways) in the lungs that causes coughing. Bronchitis usually begins as an infection in the nose, ears, throat, or sinuses. The infection then makes its way down to the bronchi. ...
, whooping cough
Whooping cough, also known as pertussis or the 100-day cough, is a highly contagious bacterial disease. Initial symptoms are usually similar to those of the common cold with a runny nose, fever, and mild cough, but these are followed by two or ...
, and tuberculosis
Tuberculosis (TB) is an infectious disease usually caused by '' Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, in ...
.
Sundew tea was recommended by herbalists for dry coughs, bronchitis
Bronchitis is inflammation of the bronchi (large and medium-sized airways) in the lungs that causes coughing. Bronchitis usually begins as an infection in the nose, ears, throat, or sinuses. The infection then makes its way down to the bronchi. ...
, whooping cough
Whooping cough, also known as pertussis or the 100-day cough, is a highly contagious bacterial disease. Initial symptoms are usually similar to those of the common cold with a runny nose, fever, and mild cough, but these are followed by two or ...
, asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, cou ...
and "bronchial cramps".
The ''French Pharmacopoeia'' of 1965 listed sundew for the treatment of inflammatory diseases such as asthma, chronic bronchitis
Bronchitis is inflammation of the bronchi (large and medium-sized airways) in the lungs that causes coughing. Bronchitis usually begins as an infection in the nose, ears, throat, or sinuses. The infection then makes its way down to the bronchi. ...
and whooping cough.
Modern studies have shown that ''Drosera'' exhibits antitussive
Cold medicines are a group of medications taken individually or in combination as a treatment for the symptoms of the common cold and similar conditions of the upper respiratory tract. The term encompasses a broad array of drugs, including ...
and anti-inflammatory
Anti-inflammatory is the property of a substance or treatment that reduces inflammation or swelling. Anti-inflammatory drugs, also called anti-inflammatories, make up about half of analgesics. These drugs remedy pain by reducing inflammation as o ...
properties.
It has been suggested that ''Drosera'' may stimulate the self-repair systems of bronchial epithelial cells to help resolve respiratory diseases.
''Drosera'' has been used commonly in cough preparations in Germany and elsewhere in Europe. ''Drosera'' is used to treat ailments such as asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, cou ...
, coughs, lung infections, and stomach ulcer
The stomach is a muscular, hollow organ in the gastrointestinal tract of humans and many other animals, including several invertebrates. The stomach has a dilated structure and functions as a vital organ in the digestive system. The stomach is ...
s. It may also have anti-fertility properties.
Medicinal preparations are primarily made using the roots, flowers, and fruit-like capsules. Since all native sundews species are protected in many parts of Europe and North America, extracts are usually prepared using cultivated fast-growing sundews (specifically ''D. rotundifolia'', '' D. intermedia'', ''D. anglica'', '' D. ramentacea'' and ''D. madagascariensis'') or from plants collected and imported from Madagascar, Spain, France, Finland and the Baltics
The Baltic states, et, Balti riigid or the Baltic countries is a geopolitical term, which currently is used to group three countries: Estonia, Latvia, and Lithuania. All three countries are members of NATO, the European Union, the Eurozone, ...
.
Sundews are historically mentioned as an aphrodisiac
An aphrodisiac is a substance that increases sexual desire, sexual attraction, sexual pleasure, or sexual behavior. Substances range from a variety of plants, spices, foods, and synthetic chemicals. Natural aphrodisiacs like cannabis or cocain ...
(hence the common name ''lustwort''). Other anecdotal uses include strengthening the heart and treating sunburn and toothache. They are mentioned as a folk remedy for treatment of warts, corns, and freckles.
As ornamental plants
Because of their carnivorous nature and the beauty of their glistening traps, sundews have become favoriteornamental plant
Ornamental plants or garden plants are plants that are primarily grown for their beauty but also for qualities such as scent or how they shape physical space. Many flowering plants and garden varieties tend to be specially bred cultivars that i ...
s; however, the environmental requirements of most species are relatively stringent and can be difficult to meet in cultivation. As a result, most species are unavailable commercially. A few of the hardiest varieties, however, have made their way into the mainstream nursery business and can often be found for sale next to Venus flytrap
The Venus flytrap (''Dionaea muscipula'') is a carnivorous plant native to subtropical wetlands on the East Coast of the United States in North Carolina and South Carolina. It catches its prey—chiefly insects and arachnids—with a trapping ...
s. These most often include '' D. capensis'', '' D. aliciae'', and '' D. spatulata''.Rice, Barry. 2006. ''Growing Carnivorous Plants''. Timber Press: Portland, Oregon.
Cultivation requirements vary greatly by species. In general, though, sundews require high environmental moisture content, usually in the form of a constantly moist or wet soil substrate. Most species also require this water to be pure, as nutrients, salts, or minerals in their soil can stunt their growth or even kill them. Commonly, plants are grown in a soil substrate containing some combination of dead or live sphagnum moss
''Sphagnum'' is a genus of approximately 380 accepted species of mosses, commonly known as sphagnum moss, peat moss, also bog moss and quacker moss (although that term is also sometimes used for peat). Accumulations of ''Sphagnum'' can store wa ...
, sphagnum
''Sphagnum'' is a genus of approximately 380 accepted species of mosses, commonly known as sphagnum moss, peat moss, also bog moss and quacker moss (although that term is also sometimes used for peat). Accumulations of ''Sphagnum'' can store wa ...
peat moss
''Sphagnum'' is a genus of approximately 380 accepted species of mosses, commonly known as sphagnum moss, peat moss, also bog moss and quacker moss (although that term is also sometimes used for peat). Accumulations of ''Sphagnum'' can store wa ...
, sand, and/or perlite
Perlite is an amorphous volcanic glass that has a relatively high water content, typically formed by the hydration of obsidian. It occurs naturally and has the unusual property of greatly expanding when heated sufficiently. It is an industrial m ...
, and are watered with distilled, reverse osmosis
Reverse osmosis (RO) is a water purification process that uses a partially permeable membrane to separate ions, unwanted molecules and larger particles from drinking water. In reverse osmosis, an applied pressure is used to overcome osmotic pre ...
, or rain water.
Nanobiotechnology
The mucilage produced by ''Drosera'' has remarkable elastic properties and has made this genus a very attractive subject in biomaterials research. In one recent study, the adhesive mucilages of three species (''D. binata'', ''D. capensis'', and ''D. spatulata'') were analyzed fornanofiber
Nanofibers are fibers with diameters in the nanometer range (typically, between 1 nm and 1 μm). Nanofibers can be generated from different polymers and hence have different physical properties and application potentials. Examples of natural polyme ...
and nanoparticle
A nanoparticle or ultrafine particle is usually defined as a particle of matter that is between 1 and 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 1 ...
content. Using atomic force microscopy
Atomic force microscopy (AFM) or scanning force microscopy (SFM) is a very-high-resolution type of scanning probe microscopy (SPM), with demonstrated resolution on the order of fractions of a nanometer, more than 1000 times better than the op ...
, transmission electron microscopy
Transmission electron microscopy (TEM) is a microscopy technique in which a beam of electrons is transmitted through a specimen to form an image. The specimen is most often an ultrathin section less than 100 nm thick or a suspension on a g ...
, and energy-dispersive X-ray spectroscopy
Energy-dispersive X-ray spectroscopy (EDS, EDX, EDXS or XEDS), sometimes called energy dispersive X-ray analysis (EDXA or EDAX) or energy dispersive X-ray microanalysis (EDXMA), is an analytical technique used for the elemental analysis or chemi ...
, researchers were able to observe networks of nanofibers and nanoparticles of various sizes within the mucilage residues. In addition, calcium
Calcium is a chemical element with the symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to ...
, magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
, and chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
– key components of biological salts - were identified. These nanoparticles are theorized to increase the viscosity and stickiness of the mucilage, in turn increasing the effectiveness of the trap.
More importantly for biomaterials research, however, is the fact that, when dried, the mucin provides a suitable substrate for the attachment of living cells. This has important implications for tissue engineering, especially because of the elastic qualities of the adhesive. Essentially, a coating of ''Drosera'' mucilage on a surgical implant, such as a replacement hip or an organ transplant, could drastically improve the rate of recovery and decrease the potential for rejection, because living tissue can effectively attach and grow on it. The authors also suggest a wide variety of applications for ''Drosera'' mucin, including wound treatment, regenerative medicine, or enhancing synthetic adhesives. Because this mucilage can stretch to nearly a million times its original size and is readily available for use, it can be an extremely cost-efficient source of biomaterial.
Other uses
Thecorm
A corm, bulbo-tuber, or bulbotuber is a short, vertical, swollen underground plant stem that serves as a storage organ that some plants use to survive winter or other adverse conditions such as summer drought and heat (perennation).
The word ' ...
s of the tuberous sundews native to Australia are considered a delicacy by the Indigenous Australians
Indigenous Australians or Australian First Nations are people with familial heritage from, and membership in, the ethnic groups that lived in Australia before British colonisation. They consist of two distinct groups: the Aboriginal peoples ...
. Some of these corms were also used to dye textiles,Plantarara (2001)Artzneimittle, Tee, und Likör aus fleischfressenden Pflanzen
while another purple or yellow dye was traditionally prepared in the
Scottish Highlands
The Highlands ( sco, the Hielands; gd, a’ Ghàidhealtachd , 'the place of the Gaels') is a historical region of Scotland. Culturally, the Highlands and the Lowlands diverged from the Late Middle Ages into the modern period, when Lowland Sco ...
using ''D. rotundifolia''. A sundew liqueur
A liqueur (; ; ) is an alcoholic drink composed of spirits (often rectified spirit) and additional flavorings such as sugar, fruits, herbs, and spices. Often served with or after dessert, they are typically heavily sweetened and un-aged beyond ...
is also still produced using a recipe from the 14th century. It is made using fresh leaves from mainly '' D. capensis'', '' D. spatulata'', and ''D. rotundifolia''.
Chemical constituents
Several chemical compounds with potential biological activities are found in sundews, includingflavonoid
Flavonoids (or bioflavonoids; from the Latin word ''flavus'', meaning yellow, their color in nature) are a class of polyphenolic secondary metabolites found in plants, and thus commonly consumed in the diets of humans.
Chemically, flavonoids ...
s (kaempferol
Kaempferol (3,4′,5,7-tetrahydroxyflavone) is a natural flavonol, a type of flavonoid, found in a variety of plants and plant-derived foods including kale, beans, tea, spinach, and broccoli. Kaempferol is a yellow crystalline solid with a meltin ...
, myricetin
Myricetin is a member of the flavonoid class of polyphenolic compounds, with antioxidant properties. Common dietary sources include vegetables (including tomatoes), fruits (including oranges), nuts, berries, tea, and red wine. Myricetin is structu ...
, quercetin
Quercetin is a plant flavonol from the flavonoid group of polyphenols. It is found in many fruits, vegetables, leaves, seeds, and grains; capers, red onions, and kale are common foods containing appreciable amounts of it. It has a bitter flavor a ...
and hyperoside
Hyperoside is a chemical compound. It is the 3-''O''-galactoside of quercetin.
Natural occurrences
Hyperoside has been isolated from ''Drosera rotundifolia'', from the Lamiaceae ''Stachys sp.'' and ''Prunella vulgaris'', from ''Rumex acetosella ...
), quinones
The quinones are a class of organic compounds that are formally "derived from aromatic compounds uch as benzene or naphthalene">benzene.html" ;"title="uch as benzene">uch as benzene or naphthalene] by conversion of an even number of –CH= group ...
(plumbagin
Plumbagin or 5-hydroxy-2-methyl-1,4-naphthoquinone is an organic compound with the chemical formula . It is regarded as a toxin and it is genotoxic and mutagenic.
Plumbagin is a yellow dye,hydroplumbagin glucoside and rossoliside (7–methyl–hydrojuglone–4–glucoside)), and other constituents such as
A key to ''Drosera'' species, with distribution maps and growing difficulty scale
* ttp://www.carnivorousplants.org/ International Carnivorous Plant Society
Carnivorous Plant FAQ
The Sundew Grow Guides
Sundew images from smugmug
Botanical Society of America, ''Drosera'' - the Sundews
{{good article Taxa named by Carl Linnaeus
carotenoid
Carotenoids (), also called tetraterpenoids, are yellow, orange, and red organic compound, organic pigments that are produced by plants and algae, as well as several bacteria, and Fungus, fungi. Carotenoids give the characteristic color to pumpki ...
s, plant acids (e.g. butyric acid
Butyric acid (; from grc, βούτῡρον, meaning "butter"), also known under the systematic name butanoic acid, is a straight-chain alkyl carboxylic acid with the chemical formula CH3CH2CH2CO2H. It is an oily, colorless liquid with an unple ...
, citric acid
Citric acid is an organic compound with the chemical formula HOC(CO2H)(CH2CO2H)2. It is a colorless weak organic acid. It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in t ...
, formic acid
Formic acid (), systematically named methanoic acid, is the simplest carboxylic acid, and has the chemical formula HCOOH and structure . It is an important intermediate in chemical synthesis and occurs naturally, most notably in some ants. Es ...
, gallic acid
Gallic acid (also known as 3,4,5-trihydroxybenzoic acid) is a trihydroxybenzoic acid with the formula C6 H2( OH)3CO2H. It is classified as a phenolic acid. It is found in gallnuts, sumac, witch hazel, tea leaves, oak bark, and other plants. It i ...
, malic acid
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms ...
, propionic acid
Propionic acid (, from the Greek words πρῶτος : ''prōtos'', meaning "first", and πίων : ''píōn'', meaning "fat"; also known as propanoic acid) is a naturally occurring carboxylic acid with chemical formula CH3CH2CO2H. It is a liq ...
), resin
In polymer chemistry and materials science, resin is a solid or highly viscous substance of plant or synthetic origin that is typically convertible into polymers. Resins are usually mixtures of organic compounds. This article focuses on natu ...
, tannins
Tannins (or tannoids) are a class of astringent, polyphenolic biomolecules that bind to and precipitate proteins and various other organic compounds including amino acids and alkaloids.
The term ''tannin'' (from Anglo-Norman ''tanner'', f ...
and ascorbic acid
Vitamin C (also known as ascorbic acid and ascorbate) is a water-soluble vitamin found in citrus and other fruits and vegetables, also sold as a dietary supplement and as a topical 'serum' ingredient to treat melasma (dark pigment spots) an ...
(vitamin C).
References
Further reading
Much of the content of this article comes from the equivalent German-language Wikipedia article (retrieved April 30, 2006). * * Correa A., Mireya D.; Silva, Tania Regina Dos Santos: ''Drosera (Droseraceae)'', in: Flora Neotropica, Monograph 96, New York, 2005 * Lowrie, Allen: ''Carnivorous Plants of Australia
''Carnivorous Plants of Australia'' is a three-volume work on carnivorous plants by Allen Lowrie. The three tomes were published in 1987, 1989, and 1998, by University of Western Australia Press.
An entirely updated three-volume work by Lowrie w ...
'', Vol. 1–3, English, Nedlands, Western Australia, 1987–1998
*
* Olberg, Günter: ''Sonnentau'', Natur und Volk, Bd. 78, Heft 1/3, pp. 32–37, Frankfurt, 1948
* Seine, Rüdiger; Barthlott, Wilhelm: ''Some proposals on the infrageneric classification of Drosera L.'', Taxon 43, 583 - 589, 1994
* Schlauer, Jan: ''A dichotomous key to the genus Drosera L. (Droseraceae)'', Carnivorous Plant Newsletter, Vol. 25 (1996)
External links
A key to ''Drosera'' species, with distribution maps and growing difficulty scale
* ttp://www.carnivorousplants.org/ International Carnivorous Plant Society
Carnivorous Plant FAQ
The Sundew Grow Guides
Sundew images from smugmug
Botanical Society of America, ''Drosera'' - the Sundews
{{good article Taxa named by Carl Linnaeus