
Antimicrobial resistance (AMR) occurs when
microbe
A microorganism, or microbe,, ''mikros'', "small") and ''organism'' from the el, ὀργανισμός, ''organismós'', "organism"). It is usually written as a single word but is sometimes hyphenated (''micro-organism''), especially in olde ...
s evolve mechanisms that protect them from the effects of
antimicrobials
An antimicrobial is an agent that kills microorganisms or stops their growth. Antimicrobial medicines can be grouped according to the microorganisms they act primarily against. For example, antibiotics are used against bacteria, and antifungals a ...
.
All classes of microbes can evolve resistance.
Fungi
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately from ...
evolve
antifungal resistance.
Virus
A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea.
Since Dmitri Ivanovsky's 1 ...
es evolve
antiviral
Antiviral drugs are a class of medication used for treating viral infections. Most antivirals target specific viruses, while a broad-spectrum antiviral is effective against a wide range of viruses. Unlike most antibiotics, antiviral drugs do no ...
resistance.
Protozoa evolve
antiprotozoal
Antiprotozoal agents ( ATC code: ATC P01) is a class of pharmaceuticals used in treatment of protozoan infection.
A paraphyletic group, protozoans have little in common with each other. For example, ''Entamoeba histolytica'', a unikont eukaryot ...
resistance, and
bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
evolve
antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of ...
resistance. Those bacteria that are considered extensively drug resistant (XDR) or totally drug-resistant (TDR) are sometimes called "superbugs".
[A.-P. Magiorakos, A. Srinivasan, R. B. Carey, Y. Carmeli, M. E. Falagas, C. G. Giske, S. Harbarth, J. F. Hinndler ''et al'']
Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria...
Clinical Microbiology and Infection, Vol 8, Iss. 3 first published 27 July 2011 ia Wiley Online Library
IA, Ia, or ia may refer to:
Arts and entertainment
* ''Ia'', an 1892 novelette by Arthur Quiller-Couch
* "Iä", a fictional word in the works of H. P. Lovecraft
* International Alliance of Theatrical Stage Employees (IATSE), which also goes by ...
Retrieved 28 August 2020 Although antimicrobial resistance is a naturally-occurring process, it is often the result of improper usage of the drugs and management of the infections.
Antibiotic resistance is a major subset of AMR, that applies specifically to
bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
that become resistant to
antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of ...
s.
Resistance in bacteria can arise naturally by
genetic mutation
In biology, a mutation is an alteration in the nucleic acid sequence of the genome of an organism, virus, or extrachromosomal DNA. Viral genomes contain either DNA or RNA. Mutations result from errors during DNA or viral replication, ...
, or by one species acquiring resistance from another. Resistance can appear spontaneously because of random mutations. However, extended use of antimicrobials appears to encourage selection for mutations which can render antimicrobials ineffective.
Clinical conditions due to AMR cause millions of deaths each year. Infections caused by resistant microbes are more difficult to treat, requiring higher doses of antimicrobial drugs, or alternative
medication
A medication (also called medicament, medicine, pharmaceutical drug, medicinal drug or simply drug) is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy (pharmacotherapy) is an important part of the medical field and re ...
s which may prove
more toxic. These approaches may also be more expensive. Microbes resistant to multiple antimicrobials are called
multidrug resistant (MDR).
The prevention of
antibiotic misuse
Antibiotic misuse, sometimes called antibiotic abuse or antibiotic overuse, refers to the misuse or overuse of antibiotics, with potentially serious effects on health. It is a contributing factor to the development of antibiotic resistance, includ ...
, which can lead to antibiotic resistance, includes taking antibiotics only when prescribed.
Narrow-spectrum antibiotic
A narrow-spectrum antibiotic is an antibiotic that is only able to kill or inhibit limited species of bacteria. Examples of narrow-spectrum antibiotics include fidaxomicin and sarecycline.
Advantages
* Narrow-spectrum antibiotic allow to kill or ...
s are preferred over
broad-spectrum antibiotic
A broad-spectrum antibiotic is an antibiotic that acts on the two major bacterial groups, Gram-positive and Gram-negative, or any antibiotic that acts against a wide range of disease-causing bacteria. These medications are used when a bacterial ...
s when possible, as effectively and accurately targeting specific organisms is less likely to cause resistance, as well as side effects.
For people who take these medications at home, education about proper use is essential. Health care providers can minimize spread of resistant infections by use of proper
sanitation
Sanitation refers to public health conditions related to clean drinking water and treatment and disposal of human excreta and sewage. Preventing human contact with feces is part of sanitation, as is hand washing with soap. Sanitation systems ...
and
hygiene
Hygiene is a series of practices performed to preserve health.
According to the World Health Organization (WHO), "Hygiene refers to conditions and practices that help to maintain health and prevent the spread of diseases." Personal hygiene refer ...
, including
handwashing
Hand washing (or handwashing), also known as hand hygiene, is the act of cleaning one's hands with soap or handwash and water to remove viruses/bacteria/microorganisms, dirt, grease, or other harmful and unwanted substances stuck to the hands ...
and disinfecting between patients, and should encourage the same of the patient, visitors, and family members.
Rising drug resistance is caused mainly by use of antimicrobials in humans and other animals, and spread of resistant strains between the two.
Growing resistance has also been linked to releasing inadequately treated effluents from the pharmaceutical industry, especially in countries where bulk drugs are manufactured. Antibiotics increase
selective pressure
Any cause that reduces or increases reproductive success in a portion of a population potentially exerts evolutionary pressure, selective pressure or selection pressure, driving natural selection. It is a quantitative description of the amount of ...
in bacterial populations, causing vulnerable bacteria to die; this increases the percentage of resistant bacteria which continue growing. Even at very low levels of antibiotic, resistant bacteria can have a growth advantage and grow faster than vulnerable bacteria. As resistance to antibiotics becomes more common there is greater need for alternative treatments. Calls for new antibiotic therapies have been issued, but new drug development is becoming rarer.
Antimicrobial resistance is increasing globally due to increased prescription and dispensing of antibiotic drugs in
developing countries
A developing country is a sovereign state with a lesser developed industrial base and a lower Human Development Index (HDI) relative to other countries. However, this definition is not universally agreed upon. There is also no clear agreem ...
. Estimates are that 700,000 to several million deaths result per year and continues to pose a major public health threat worldwide.
Each year in the
United States
The United States of America (U.S.A. or USA), commonly known as the United States (U.S. or US) or America, is a country primarily located in North America. It consists of 50 states, a federal district, five major unincorporated territorie ...
, at least 2.8 million people become infected with bacteria that are resistant to antibiotics and at least 35,000 people die and US$55 billion in increased health care costs and lost productivity. According to
World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level of h ...
(WHO) estimates, 350 million deaths could be caused by AMR by 2050. By then, the yearly death toll will be 10 million, according to a
United Nations
The United Nations (UN) is an intergovernmental organization whose stated purposes are to maintain international peace and international security, security, develop friendly relations among nations, achieve international cooperation, and be ...
report.
There are public calls for global collective action to address the threat that include proposals for
international treaties
A treaty is a formal, legally binding written agreement between actors in international law. It is usually made by and between sovereign states, but can include international organizations, individuals, business entities, and other legal pers ...
on antimicrobial resistance.
Worldwide antibiotic resistance is not completely identified, but poorer countries with weaker healthcare systems are more affected.
During the
COVID-19 pandemic
The COVID-19 pandemic, also known as the coronavirus pandemic, is an ongoing global pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The novel virus was first identif ...
, action against antimicrobial resistance slowed due to scientists focusing more on
SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) is a strain of coronavirus that causes COVID-19 (coronavirus disease 2019), the respiratory illness responsible for the ongoing COVID-19 pandemic. The virus previously had a ...
research.
Definition
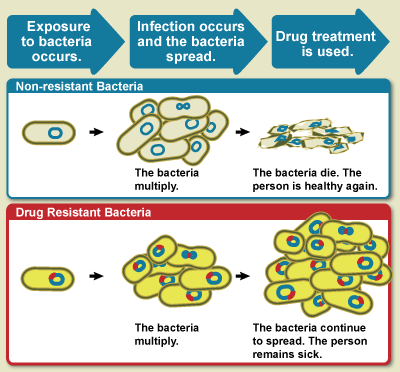
The WHO defines antimicrobial resistance as a microorganism's
resistance to an antimicrobial drug that was once able to treat an infection by that microorganism.
[ A person cannot become resistant to antibiotics. Resistance is a property of the microbe, not a person or other organism infected by a microbe. All types of microbes can develop drug resistance. Thus, there are antibiotic, antifungal, antiviral and antiparasitic resistance.]
Overview
WHO report released April 2014 stated, "this serious threat is no longer a prediction for the future, it is happening right now in every region of the world and has the potential to affect anyone, of any age, in any country. Antibiotic resistance—when bacteria change so antibiotics no longer work in people who need them to treat infections—is now a major threat to public health."["WHO's first global report on antibiotic resistance reveals serious, worldwide threat to public health"](_blank)
Retrieved 2 May 2014
Global deaths attributable to AMR numbered 1.27 million in 2019. That year, AMR may have contributed to 5 million deaths and one in five people who died due to AMR were children under five years old.food security
Food security speaks to the availability of food in a country (or geography) and the ability of individuals within that country (geography) to access, afford, and source adequate foodstuffs. According to the United Nations' Committee on World F ...
and development. Deaths attributable to AMR vary by area:
The European Centre for Disease Prevention and Control
The European Centre for Disease Prevention and Control (ECDC) is an agency of the European Union (EU) whose mission is to strengthen Europe's defences against infectious diseases. It covers a wide spectrum of activities, such as: surveillance, ...
calculated that in 2015 there were 671,689 infections in the EU and European Economic Area caused by antibiotic-resistant bacteria, resulting in 33,110 deaths. Most were acquired in healthcare settings.
Causes
Antimicrobial resistance is mainly caused by the overuse of antimicrobials. This leads to microbes either evolving a defense against drugs used to treat them, or certain strains of microbes that have a natural resistance to antimicrobials becoming much more prevalent than the ones that are easily defeated with medication. While antimicrobial resistance does occur naturally over time, the use of antimicrobial agents in a variety of settings both within the healthcare industry and outside of has led to antimicrobial resistance becoming increasingly more prevalent.
Natural occurrence
 Antimicrobial resistance can evolve naturally due to continued exposure to antimicrobials.
Antimicrobial resistance can evolve naturally due to continued exposure to antimicrobials. Natural selection
Natural selection is the differential survival and reproduction of individuals due to differences in phenotype. It is a key mechanism of evolution, the change in the heritable traits characteristic of a population over generations. Charle ...
means that organisms that are able to adapt to their environment, survive, and continue to produce offspring. As a result, the types of microorganisms that are able to survive over time with continued attack by certain antimicrobial agents will naturally become more prevalent in the environment, and those without this resistance will become obsolete.dermatophyte
Dermatophyte (from Greek '' derma'' "skin" ( GEN ''dermatos'') and ''phyton'' "plant") is a common label for a group of fungus of ''Arthrodermataceae'' that commonly causes skin disease in animals and humans. Traditionally, these anamorphic (a ...
that naturally produces antibiotics. Also, many soil fungi and bacteria are natural competitors and the original antibiotic Penicillin discovered by Alexander Flemming rapidly lost clinical effectiveness in treating humans and, furthermore, none of the other natural penicillins (F, K, N, X, O, U1 or U6) are currently in clinical use. https://en.wikipedia.org/wiki/Penicillin
Over time, most of the strains of bacteria and infections present will be the type resistant to the antimicrobial agent being used to treat them, making this agent now ineffective to defeat most microbes. With the increased use of antimicrobial agents, there is a speeding up of this natural process.
Self-medication
Self-medication
Self-medication is a human behavior in which an individual uses a substance or any exogenous influence to self-administer treatment for physical or psychological conditions: for example headaches or fatigue.
The substances most widely used in sel ...
by consumers is defined as "the taking of medicines on one's own initiative or on another person's suggestion, who is not a certified medical professional", and it has been identified as one of the primary reasons for the evolution of antimicrobial resistance.
In an effort to manage their own illness, patients take the advice of sometimes false media sources, friends, and family causing them to take antimicrobials unnecessarily or in excess. Many people resort to this out of necessity, when access to a physician is rationed or temporarily unavailable due to lockdowns or GP surgery closures, or when the patients have a limited amount of time or money to see a prescribing doctor, or, especially in many developing countries with growing populations but a poorly developed economy which lack cheap, convenient access to clinical professionals. In these situations, governments naturally allow the sale of antimicrobials as over the counter medications. This increased access makes it extremely easy to obtain antimicrobials without any targeting (which would enhance utility) or restriction via time and cost, and as a result many antimicrobials are taken incorrectly leading to resistant microbial strains. One major example of a place that faces these challenges is India, where in the state of
Punjab
Punjab (; Punjabi: پنجاب ; ਪੰਜਾਬ ; ; also romanised as ''Panjāb'' or ''Panj-Āb'') is a geopolitical, cultural, and historical region in South Asia, specifically in the northern part of the Indian subcontinent, comprising ...
73% of the population resorted to treating their minor health issues and chronic illnesses through self-medication.
Two significant issues with self-medication are the lack of knowledge of the public on, firstly, the dangerous effects of certain antimicrobials (for example Ciprofloxacin causing tendonitis and tendon rupture) and, secondly, broad microbial resistance. In order to determine the public's knowledge and preconceived notions on antibiotic resistance, a major type of antimicrobial resistance, a screening of 3537 articles published in Europe, Asia, and North America was done. Of the 55,225 total people surveyed, 70% had heard of antibiotic resistance previously, but 88% of those people thought it referred to some type of physical change in the body.
With so many people around the world with the ability to self-medicate using antibiotics, and a vast majority unaware of what antimicrobial resistance is, it makes the increase of antimicrobial resistance much more likely.
Clinical misuse
Clinical misuse by healthcare professionals is another cause leading to increased antimicrobial resistance. Studies done by the
CDC
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georgi ...
show that the indication for treatment of antibiotics, choice of the agent used, and the duration of therapy was incorrect in up to 50% of the cases studied. In another study done in an intensive care unit in a major hospital in France, it was shown that 30% to 60% of prescribed antibiotics were unnecessary.
These inappropriate uses of antimicrobial agents promote the evolution of antimicrobial resistance by supporting the bacteria in developing genetic alterations that lead to resistance.
In a study done by the ''
American Journal of Infection Control'' aimed to evaluate physicians' attitudes and knowledge on antimicrobial resistance in ambulatory settings, only 63% of those surveyed reported antibiotic resistance as a problem in their local practices, while 23% reported the aggressive prescription of antibiotics as necessary to avoid failing to provide adequate care. This demonstrates how a majority of doctors underestimate the impact that their own prescribing habits have on antimicrobial resistance as a whole. It also confirms that some physicians may be overly cautious and prescribe antibiotics for both medical or legal reasons, even when clinical indications for use for these medications are not always confirmed. This can lead to unnecessary antimicrobial use, a pattern which may have worsened during the
COVID-19
Coronavirus disease 2019 (COVID-19) is a contagious disease caused by a virus, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The first known case was COVID-19 pandemic in Hubei, identified in Wuhan, China, in December ...
pandemic.
Studies have shown that
common misconceptions
Each entry on this list of common misconceptions is worded as a correction; the misconceptions themselves are implied rather than stated. These entries are concise summaries of the main subject articles, which can be consulted for more detail.
...
about the effectiveness and necessity of antibiotics to treat common mild illnesses contribute to their overuse.
Pandemics, disinfectants and healthcare systems
Increased antibiotic use during the COVID-19 pandemic may exacerbate this
global health challenge. Moreover, pandemic burdens on some healthcare systems may contribute to antibiotic-resistant infections. On the other hand, a study suggests that "increased hand hygiene, decreased international travel, and decreased elective hospital procedures may reduce AMR pathogen selection and spread in the short term".
Disinfectant
A disinfectant is a chemical substance or compound used to inactivate or destroy microorganisms on inert surfaces. Disinfection does not necessarily kill all microorganisms, especially resistant bacterial spores; it is less effective than st ...
s such as in various forms of use of alcohol-based hand sanitizers, and antiseptic hand wash may also have the potential to increase antimicrobial resistance. According to a study, "Extensive disinfectant use leads to mutations that induce antimicrobial resistance".
Environmental pollution
Untreated effluents from pharmaceutical manufacturing industries, hospitals and clinics, and inappropriate disposal of unused or expired medication can expose microbes in the environment to antibiotics and trigger the evolution of resistance.
Food production
Livestock

The antimicrobial resistance crisis also extends to the food industry, specifically with food producing animals. Antibiotics are fed to livestock to act as growth supplements, and a preventative measure to decrease the likelihood of infections. This results in the transfer of resistant bacterial strains into the food that humans eat, causing potentially fatal transfer of disease. While this practice does result in better yields and
meat
Meat is animal flesh that is eaten as food. Humans have hunted, farmed, and scavenged animals for meat since prehistoric times. The establishment of settlements in the Neolithic Revolution allowed the domestication of animals such as chic ...
products, it is a major issue in terms of preventing antimicrobial resistance. Though the evidence linking antimicrobial usage in livestock to antimicrobial resistance is limited, the World Health Organization Advisory Group on Integrated Surveillance of Antimicrobial Resistance strongly recommended the reduction of use of medically important antimicrobials in livestock. Additionally, the Advisory Group stated that such antimicrobials should be expressly prohibited for both growth promotion and disease prevention.
In a study published by the
National Academy of Sciences
The National Academy of Sciences (NAS) is a United States nonprofit, non-governmental organization. NAS is part of the National Academies of Sciences, Engineering, and Medicine, along with the National Academy of Engineering (NAE) and the Nati ...
mapping antimicrobial consumption in livestock globally, it was predicted that in the 228 countries studied, there would be a total 67% increase in consumption of antibiotics by livestock by 2030. In some countries such as Brazil, Russia, India, China, and South Africa it is predicted that a 99% increase will occur.
Several countries have restricted the use of antibiotics in livestock, including Canada, China, Japan, and the US. These restrictions are sometimes associated with a reduction of the
prevalence
In epidemiology, prevalence is the proportion of a particular population found to be affected by a medical condition (typically a disease or a risk factor such as smoking or seatbelt use) at a specific time. It is derived by comparing the number o ...
of antimicrobial resistance in humans.
Pesticides
Most
pesticides protect crops against insects and plants, but in some cases antimicrobial pesticides are used to protect against various microorganisms such as bacteria, viruses, fungi, algae, and protozoa. The overuse of many pesticides in an effort to have a higher yield of crops has resulted in many of these microbes evolving a tolerance against these antimicrobial agents. Currently there are over 4000 antimicrobial pesticides registered with the
EPA
The Environmental Protection Agency (EPA) is an independent executive agency of the United States federal government tasked with environmental protection matters. President Richard Nixon proposed the establishment of EPA on July 9, 1970; it be ...
and sold to market, showing the widespread use of these agents. It is estimated that for every single meal a person consumes, 0.3 g of pesticides is used, as 90% of all pesticide use is used on agriculture. A majority of these products are used to help defend against the spread of infectious diseases, and hopefully protect public health. But out of the large amount of pesticides used, it is also estimated that less than 0.1% of those antimicrobial agents, actually reach their targets. That leaves over 99% of all pesticides used available to contaminate other resources. In soil, air, and water these antimicrobial agents are able to spread, coming in contact with more microorganisms and leading to these microbes evolving mechanisms to tolerate and further resist pesticides.
Prevention

There have been increasing public calls for global collective action to address the threat, including a proposal for international treaty on antimicrobial resistance. Further detail and attention is still needed in order to recognize and measure trends in resistance on the international level; the idea of a global tracking system has been suggested but implementation has yet to occur. A system of this nature would provide insight to areas of high resistance as well as information necessary for evaluating programs and other changes made to fight or reverse antibiotic resistance.
Duration of antibiotics
Antibiotic treatment duration should be based on the infection and other health problems a person may have.
[ For many infections once a person has improved there is little evidence that stopping treatment causes more resistance.][ Some, therefore, feel that stopping early may be reasonable in some cases.][ Other infections, however, do require long courses regardless of whether a person feels better.][
]
Monitoring and mapping
There are multiple national and international monitoring programs for drug-resistant threats, including methicillin-resistant ''Staphylococcus aureus'' (MRSA), vancomycin-resistant ''S. aureus'' (VRSA), extended spectrum beta-lactamase
Beta-lactamases, (β-lactamases) are enzymes () produced by bacteria that provide multi-resistance to beta-lactam antibiotics such as penicillins, cephalosporins, cephamycins, monobactams and carbapenems (ertapenem), although carbapenem ...
(ESBL), vancomycin-resistant ''Enterococcus'' (VRE), and multidrug-resistant ''Acinetobacter baumannii'' (MRAB).HealthMap HealthMap is a freely accessible, automated electronic information system for monitoring, organizing, and visualizing reports of global disease outbreaks according to geography, time, and infectious disease agent. In operation since September 2006, ...
which displays aggregated data on antimicrobial resistance from publicly available and user submitted data. The website can display data for a radius from a location. Users may submit data from antibiograms for individual hospitals or laboratories. European data is from the EARS-Net (European Antimicrobial Resistance Surveillance Network), part of the ECDC
The European Centre for Disease Prevention and Control (ECDC) is an agency of the European Union (EU) whose mission is to strengthen Europe's defences against infectious diseases. It covers a wide spectrum of activities, such as: surveillance, e ...
.
ResistanceMap is a website by the Center for Disease Dynamics, Economics & Policy and provides data on antimicrobial resistance on a global level.
Limiting antibiotic use
Antibiotic stewardship programmes appear useful in reducing rates of antibiotic resistance.
At the hospital level
Antimicrobial stewardship Antimicrobial stewardship is the systematic effort to educate and persuade prescribers of antimicrobials to follow evidence-based prescribing, in order to stem antimicrobial overuse, and thus antimicrobial resistance. AMS has been an organized eff ...
teams in hospitals are encouraging optimal use of antimicrobials. The goals of antimicrobial stewardship are to help practitioners pick the right drug at the right dose and duration of therapy while preventing misuse and minimizing the development of resistance. Stewardship may reduce the length of stay by an average of slightly over 1 day while not increasing the risk of death.
At the farming level
It is established that the use of antibiotics in animal husbandry can give rise to AMR resistances in bacteria found in food animals to the antibiotics being administered (through injections or medicated feeds). For this reason only antimicrobials that are deemed "not-clinically relevant" are used in these practices.
Recent studies have shown that the prophylactic use of "non-priority" or "non-clinically relevant" antimicrobials in feeds can potentially, under certain conditions, lead to co-selection of environmental AMR bacteria with resistance to medically important antibiotics.
At the level of GP
Given the volume of care provided in primary care (General Practice), recent strategies have focused on reducing unnecessary antibiotic prescribing in this setting. Simple interventions, such as written information explaining the futility of antibiotics for common infections such as upper respiratory tract infections, have been shown to reduce antibiotic prescribing.
The prescriber should closely adhere to the five rights of drug administration: the right patient, the right drug, the right dose, the right route, and the right time.
Cultures should be taken before treatment when indicated and treatment potentially changed based on the susceptibility report.
At the individual level
People can help tackle resistance by using antibiotics only when prescribed by a doctor; completing the full prescription, even if they feel better; never sharing antibiotics with others or using leftover prescriptions.
Country examples
* The Netherlands
)
, anthem = ( en, "William of Nassau")
, image_map =
, map_caption =
, subdivision_type = Sovereign state
, subdivision_name = Kingdom of the Netherlands
, established_title = Before independence
, established_date = Spanish Netherl ...
has the lowest rate of antibiotic prescribing in the OECD
The Organisation for Economic Co-operation and Development (OECD; french: Organisation de coopération et de développement économiques, ''OCDE'') is an intergovernmental organisation with 38 member countries, founded in 1961 to stimulate e ...
, at a rate of 11.4 defined daily doses (DDD) per 1,000 people per day in 2011.
* Germany
Germany,, officially the Federal Republic of Germany, is a country in Central Europe. It is the second most populous country in Europe after Russia, and the most populous member state of the European Union. Germany is situated betwe ...
and Sweden
Sweden, formally the Kingdom of Sweden,The United Nations Group of Experts on Geographical Names states that the country's formal name is the Kingdom of SwedenUNGEGN World Geographical Names, Sweden./ref> is a Nordic country located on ...
also have lower prescribing rates, with Sweden's rate having been declining since 2007.
* Greece
Greece,, or , romanized: ', officially the Hellenic Republic, is a country in Southeast Europe. It is situated on the southern tip of the Balkans, and is located at the crossroads of Europe, Asia, and Africa. Greece shares land borders with ...
, France
France (), officially the French Republic ( ), is a country primarily located in Western Europe. It also comprises of Overseas France, overseas regions and territories in the Americas and the Atlantic Ocean, Atlantic, Pacific Ocean, Pac ...
and Belgium
Belgium, ; french: Belgique ; german: Belgien officially the Kingdom of Belgium, is a country in Northwestern Europe. The country is bordered by the Netherlands to the north, Germany to the east, Luxembourg to the southeast, France to th ...
have high prescribing rates of more than 28 DDD.
Water, sanitation, hygiene
Infectious disease control through improved water, sanitation and hygiene (WASH) infrastructure needs to be included in the antimicrobial resistance (AMR) agenda. The "Interagency Coordination Group on Antimicrobial Resistance" stated in 2018 that "the spread of pathogens through unsafe water results in a high burden of gastrointestinal disease, increasing even further the need for antibiotic treatment."[IACG (2018]
Reduce unintentional exposure and the need for antimicrobials, and optimize their use IACG Discussion Paper
Interagency Coordination Group on Antimicrobial Resistance
public consultation process
at WHO, Geneva, Switzerland This is particularly a problem in developing countries
A developing country is a sovereign state with a lesser developed industrial base and a lower Human Development Index (HDI) relative to other countries. However, this definition is not universally agreed upon. There is also no clear agreem ...
where the spread of infectious diseases caused by inadequate WASH standards is a major driver of antibiotic demand.Sustainable Development Goals
The Sustainable Development Goals (SDGs) or Global Goals are a collection of 17 interlinked objectives designed to serve as a "shared blueprint for peace and prosperity for people and the planet, now and into the future".United Nations (2017) R ...
.
An increase in hand washing
Hand washing (or handwashing), also known as hand hygiene, is the act of cleaning one's hands with soap or handwash and water to remove viruses/bacteria/microorganisms, dirt, grease, or other harmful and unwanted substances stuck to the hands ...
compliance by hospital staff results in decreased rates of resistant organisms.
Water supply and sanitation infrastructure in health facilities offer significant co-benefits for combatting AMR, and investment should be increased.
Industrial wastewater treatment
Manufacturers of antimicrobials need to improve the treatment of their wastewater (by using industrial wastewater treatment processes) to reduce the release of residues into the environment.
Management in animal use
Europe
In 1997, European Union health ministers voted to ban avoparcin
Avoparcin is a glycopeptide antibiotic effective against Gram-positive bacteria. It has been used in agriculture as an additive to livestock feed to promote growth in chickens, pigs, and cattle. It is also used as an aid in the prevention of nec ...
and four additional antibiotics used to promote animal growth in 1999. In 2006 a ban on the use of antibiotics in European feed, with the exception of two antibiotics in poultry feeds, became effective. In Scandinavia, there is evidence that the ban has led to a lower prevalence of antibiotic resistance in (nonhazardous) animal bacterial populations. As of 2004, several European countries established a decline of antimicrobial resistance in humans through limiting the use of antimicrobials in agriculture and food industries without jeopardizing animal health or economic cost.
United States
The United States Department of Agriculture
The United States Department of Agriculture (USDA) is the United States federal executive departments, federal executive department responsible for developing and executing federal laws related to farming, forestry, rural economic development, ...
(USDA) and the Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respon ...
(FDA) collect data on antibiotic use in humans and in a more limited fashion in animals.fluoroquinolone
A quinolone antibiotic is a member of a large group of broad-spectrum bacteriocidals that share a bicyclic core structure related to the substance 4-quinolone. They are used in human and veterinary medicine to treat bacterial infections, as wel ...
use in poultry production because of substantial evidence linking it to the emergence of fluoroquinolone-resistant '' Campylobacter'' infections in humans. Legal challenges from the food animal and pharmaceutical industries delayed the final decision to do so until 2006.
Global action plans and awareness
The increasing interconnectedness of the world and the fact that new classes of antibiotics have not been developed and approved for more than 25 years highlight the extent to which antimicrobial resistance is a global health challenge. A global action plan to tackle the growing problem of resistance to antibiotics and other antimicrobial medicines was endorsed at the Sixty-eighth World Health Assembly
The World Health Assembly (WHA) is the forum through which the World Health Organization (WHO) is governed by its 194 member states. It is the world's highest health policy setting body and is composed of health ministers from member states.
Th ...
in May 2015.
Antibiotic Awareness Week
The World Health Organization has promoted the first World Antibiotic Awareness Week running from 16 to 22 November 2015. The aim of the week is to increase global awareness of antibiotic resistance. It also wants to promote the correct usage of antibiotics across all fields in order to prevent further instances of antibiotic resistance.
World Antibiotic Awareness Week has been held every November since 2015. For 2017, the Food and Agriculture Organization of the United Nations (FAO), the World Health Organization (WHO) and the World Organisation for Animal Health
The World Organisation for Animal Health (WOAH), formerly the (OIE), is an intergovernmental organization coordinating, supporting and promoting animal disease control.
Mission and status
The main objective of the WOAH is to control epizo ...
(OIE) are together calling for responsible use of antibiotics in humans and animals to reduce the emergence of antibiotic resistance.
United Nations
In 2016 the Secretary-General of the United Nations
The United Nations (UN) is an intergovernmental organization whose stated purposes are to maintain international peace and international security, security, develop friendly relations among nations, achieve international cooperation, and be ...
convened the Interagency Coordination Group (IACG) on Antimicrobial Resistance.
Mechanisms and organisms
Bacteria
 The five main mechanisms by which bacteria exhibit resistance to antibiotics are:
# Drug inactivation or modification: for example, enzymatic deactivation of
The five main mechanisms by which bacteria exhibit resistance to antibiotics are:
# Drug inactivation or modification: for example, enzymatic deactivation of penicillin G
Benzylpenicillin, also known as penicillin G (PenG) or BENPEN, and in military slang "Peanut Butter Shot" is an antibiotic used to treat a number of bacterial infections. This includes pneumonia, strep throat, syphilis, necrotizing enteroco ...
in some penicillin-resistant bacteria through the production of β-lactamases. Drugs may also be chemically modified through the addition of functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
s by transferase
A transferase is any one of a class of enzymes that catalyse the transfer of specific functional groups (e.g. a methyl or glycosyl group) from one molecule (called the donor) to another (called the acceptor). They are involved in hundreds of ...
enzymes; for example, acetylation, phosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
, or adenylation are common resistance mechanisms to aminoglycoside
Aminoglycoside is a medicinal and bacteriologic category of traditional Gram-negative antibacterial medications that inhibit protein synthesis and contain as a portion of the molecule an amino-modified glycoside (sugar). The term can also refer ...
s. Acetylation is the most widely used mechanism and can affect a number of drug class
A drug class is a set of medications and other compounds that have a similar chemical structures, the same mechanism of action (i.e. binding to the same biological target), a related mode of action, and/or are used to treat the same disease.
In ...
es.MRSA
Methicillin-resistant ''Staphylococcus aureus'' (MRSA) is a group of Gram-positive bacteria that are genetically distinct from other strains of ''Staphylococcus aureus''. MRSA is responsible for several difficult-to-treat infections in humans. ...
and other penicillin-resistant bacteria. Another protective mechanism found among bacterial species is ribosomal protection proteins. These proteins protect the bacterial cell from antibiotics that target the cell's ribosomes to inhibit protein synthesis. The mechanism involves the binding of the ribosomal protection proteins to the ribosomes of the bacterial cell, which in turn changes its conformational shape. This allows the ribosomes to continue synthesizing proteins essential to the cell while preventing antibiotics from binding to the ribosome to inhibit protein synthesis.
# Alteration of metabolic pathway: for example, some sulfonamide-resistant bacteria do not require para-aminobenzoic acid
4-Aminobenzoic acid (also known as ''para''-aminobenzoic acid or PABA because the two functional groups are attached to the benzene ring across from one another in the ''para'' position) is an organic compound with the formula H2NC6H4CO2H. PABA ...
(PABA), an important precursor for the synthesis of folic acid
Folate, also known as vitamin B9 and folacin, is one of the B vitamins. Manufactured folic acid, which is converted into folate by the body, is used as a dietary supplement and in food fortification as it is more stable during processing and ...
and nucleic acid
Nucleic acids are biopolymers, macromolecules, essential to all known forms of life. They are composed of nucleotides, which are the monomers made of three components: a 5-carbon sugar, a phosphate group and a nitrogenous base. The two main cl ...
s in bacteria inhibited by sulfonamides, instead, like mammalian cells, they turn to using preformed folic acid.
# Reduced drug accumulation: by decreasing drug permeability or increasing active efflux (pumping out) of the drugs across the cell surface These pumps within the cellular membrane of certain bacterial species are used to pump antibiotics out of the cell before they are able to do any damage. They are often activated by a specific substrate associated with an antibiotic, as in fluoroquinolone
A quinolone antibiotic is a member of a large group of broad-spectrum bacteriocidals that share a bicyclic core structure related to the substance 4-quinolone. They are used in human and veterinary medicine to treat bacterial infections, as wel ...
resistance.
# Ribosome splitting and recycling: for example, drug-mediated stalling of the ribosome by lincomycin
Lincomycin is a lincosamide antibiotic that comes from the actinomycete ''Streptomyces lincolnensis''. A related compound, clindamycin, is derived from lincomycin by using thionyl chloride to replace the 7-hydroxy group with a chlorine atom with ...
and erythromycin
Erythromycin is an antibiotic used for the treatment of a number of bacterial infections. This includes respiratory tract infections, skin infections, chlamydia infections, pelvic inflammatory disease, and syphilis. It may also be used duri ...
unstalled by a heat shock protein found in ''Listeria monocytogenes'', which is a homologue of HflX from other bacteria. Liberation of the ribosome from the drug allows further translation and consequent resistance to the drug.
 There are several different types of germs that have developed a resistance over time.
The six pathogens causing most deaths associated with resistance are ''Escherichia coli'', ''Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii'', and ''Pseudomonas aeruginosa''. They were responsible for 929,000 deaths attributable to resistance and 3.57 million deaths associated with resistance in 2019.
There are several different types of germs that have developed a resistance over time.
The six pathogens causing most deaths associated with resistance are ''Escherichia coli'', ''Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii'', and ''Pseudomonas aeruginosa''. They were responsible for 929,000 deaths attributable to resistance and 3.57 million deaths associated with resistance in 2019.DNA gyrase
DNA gyrase, or simply gyrase, is an enzyme within the class of topoisomerase and is a subclass of Type II topoisomerases that reduces topological strain in an ATP dependent manner while double-stranded DNA is being unwound by elongating RNA-poly ...
, protecting it from the action of quinolones. Finally, mutations at key sites in DNA gyrase or topoisomerase IV
Topoisomerase IV is one of two Type II topoisomerases in bacteria, the other being DNA gyrase. Like gyrase, topoisomerase IV is able to pass one double-strand of DNA through another double-strand of DNA, thereby changing the linking number of DNA b ...
can decrease their binding affinity to quinolones, decreasing the drug's effectiveness.
Some bacteria are naturally resistant to certain antibiotics; for example, gram-negative bacteria are resistant to most β-lactam antibiotic
β-lactam antibiotics (beta-lactam antibiotics) are antibiotics that contain a beta-lactam ring in their chemical
structure. This includes penicillin derivatives (penams), cephalosporins and cephamycins (cephems), monobactams, carbapenems and ...
s due to the presence of β-lactamase
Beta-lactamases, (β-lactamases) are enzymes () produced by bacteria that provide multi-resistance to beta-lactam antibiotics such as penicillins, cephalosporins, cephamycins, monobactams and carbapenems ( ertapenem), although carbapenems ...
. Antibiotic resistance can also be acquired as a result of either genetic mutation or horizontal gene transfer
Horizontal gene transfer (HGT) or lateral gene transfer (LGT) is the movement of genetic material between unicellular and/or multicellular organisms other than by the ("vertical") transmission of DNA from parent to offspring (reproduction). H ...
. Although mutations are rare, with spontaneous mutations in the pathogen
In biology, a pathogen ( el, πάθος, "suffering", "passion" and , "producer of") in the oldest and broadest sense, is any organism or agent that can produce disease. A pathogen may also be referred to as an infectious agent, or simply a germ ...
genome
In the fields of molecular biology and genetics, a genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding ge ...
occurring at a rate of about 1 in 105 to 1 in 108 per chromosomal replication, the fact that bacteria reproduce at a high rate allows for the effect to be significant. Given that lifespans and production of new generations can be on a timescale of mere hours, a new (de novo) mutation in a parent cell can quickly become an inherited mutation of widespread prevalence, resulting in the microevolution
Microevolution is the change in allele frequencies that occurs over time within a population. This change is due to four different processes: mutation, selection (natural and artificial), gene flow and genetic drift. This change happens over a ...
of a fully resistant colony. However, chromosomal mutations also confer a cost of fitness. For example, a ribosomal mutation may protect a bacterial cell by changing the binding site of an antibiotic but may result in slower growth rate. Moreover, some adaptive mutations can propagate not only through inheritance but also through horizontal gene transfer
Horizontal gene transfer (HGT) or lateral gene transfer (LGT) is the movement of genetic material between unicellular and/or multicellular organisms other than by the ("vertical") transmission of DNA from parent to offspring (reproduction). H ...
. The most common mechanism of horizontal gene transfer is the transferring of plasmids carrying antibiotic resistance genes between bacteria of the same or different species via conjugation
Conjugation or conjugate may refer to:
Linguistics
* Grammatical conjugation, the modification of a verb from its basic form
* Emotive conjugation or Russell's conjugation, the use of loaded language
Mathematics
* Complex conjugation, the chang ...
. However, bacteria can also acquire resistance through transformation
Transformation may refer to:
Science and mathematics
In biology and medicine
* Metamorphosis, the biological process of changing physical form after birth or hatching
* Malignant transformation, the process of cells becoming cancerous
* Tran ...
, as in ''Streptococcus pneumoniae'' uptaking of naked fragments of extracellular DNA that contain antibiotic resistance genes to streptomycin, through transduction, as in the bacteriophage-mediated transfer of tetracycline resistance genes between strains of ''S. pyogenes'', or through gene transfer agent
Gene transfer agents (GTAs) are DNA-containing virus-like particles that are produced by some bacteria and archaea and mediate horizontal gene transfer. Different GTA types have originated independently from viruses in several bacterial and archae ...
s, which are particles produced by the host cell that resemble bacteriophage structures and are capable of transferring DNA.
Antibiotic resistance can be introduced artificially into a microorganism through laboratory protocols, sometimes used as a selectable marker A selectable marker is a gene introduced into a cell, especially a bacterium or to cells in culture, that confers a trait suitable for artificial selection. They are a type of reporter gene used in laboratory microbiology, molecular biology, a ...
to examine the mechanisms of gene transfer or to identify individuals that absorbed a piece of DNA that included the resistance gene and another gene of interest.
Recent findings show no necessity of large populations of bacteria for the appearance of antibiotic resistance. Small populations of ''Escherichia coli
''Escherichia coli'' (),Wells, J. C. (2000) Longman Pronunciation Dictionary. Harlow ngland Pearson Education Ltd. also known as ''E. coli'' (), is a Gram-negative, facultative anaerobic, rod-shaped, coliform bacterium of the genus ''Escher ...
'' in an antibiotic gradient can become resistant. Any heterogeneous environment with respect to nutrient and antibiotic gradients may facilitate antibiotic resistance in small bacterial populations. Researchers hypothesize that the mechanism of resistance evolution is based on four SNP mutations in the genome of ''E. coli'' produced by the gradient of antibiotic.
In one study, which has implications for space microbiology, a non-pathogenic strain ''E. coli'' MG1655 was exposed to trace levels of the broad spectrum antibiotic chloramphenicol, under simulated microgravity (LSMMG, or Low Shear Modeled Microgravity) over 1000 generations. The adapted strain acquired resistance to not only chloramphenicol, but also cross-resistance to other antibiotics; this was in contrast to the observation on the same strain, which was adapted to over 1000 generations under LSMMG, but without any antibiotic exposure; the strain in this case did not acquire any such resistance. Thus, irrespective of where they are used, the use of an antibiotic would likely result in persistent resistance to that antibiotic, as well as cross-resistance to other antimicrobials.
In recent years, the emergence and spread of β-lactamases called carbapenemase
Beta-lactamases, (β-lactamases) are enzymes () produced by bacteria that provide multi-resistance to beta-lactam antibiotics such as penicillins, cephalosporins, cephamycins, monobactams and carbapenems ( ertapenem), although carbapenems ...
s has become a major health crisis. One such carbapenemase is New Delhi metallo-beta-lactamase 1
NDM-1 is an enzyme that makes bacteria resistant to a broad range of beta-lactam antibiotics. These include the antibiotics of the carbapenem family, which are a mainstay for the treatment of antibiotic-resistant bacterial infections. The gene f ...
(NDM-1),enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. A ...
that makes bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
resistant to a broad range of beta-lactam antibiotic
β-lactam antibiotics (beta-lactam antibiotics) are antibiotics that contain a beta-lactam ring in their chemical
structure. This includes penicillin derivatives (penams), cephalosporins and cephamycins (cephems), monobactams, carbapenems and ...
s. The most common bacteria that make this enzyme are gram-negative
Gram-negative bacteria are bacteria that do not retain the crystal violet stain used in the Gram staining method of bacterial differentiation. They are characterized by their cell envelopes, which are composed of a thin peptidoglycan cell wall ...
such as ''E. coli'' and ''Klebsiella pneumoniae
''Klebsiella pneumoniae'' is a Gram-negative, non-motile, encapsulated, lactose- fermenting, facultative anaerobic, rod-shaped bacterium. It appears as a mucoid lactose fermenter on MacConkey agar.
Although found in the normal flora of the mo ...
'', but the gene for NDM-1 can spread from one strain of bacteria to another by horizontal gene transfer
Horizontal gene transfer (HGT) or lateral gene transfer (LGT) is the movement of genetic material between unicellular and/or multicellular organisms other than by the ("vertical") transmission of DNA from parent to offspring (reproduction). H ...
.
Viruses
Specific antiviral drugs are used to treat some viral infections. These drugs prevent viruses from reproducing by inhibiting essential stages of the virus's replication cycle in infected cells. Antivirals are used to treat HIV
The human immunodeficiency viruses (HIV) are two species of ''Lentivirus'' (a subgroup of retrovirus) that infect humans. Over time, they cause acquired immunodeficiency syndrome (AIDS), a condition in which progressive failure of the immune ...
, hepatitis B
Hepatitis B is an infectious disease caused by the '' Hepatitis B virus'' (HBV) that affects the liver; it is a type of viral hepatitis. It can cause both acute and chronic infection.
Many people have no symptoms during an initial infection. ...
, hepatitis C, influenza, herpes viruses including varicella zoster virus
Varicella-zoster virus (VZV), also known as human herpesvirus 3 (HHV-3, HHV3) or ''Human alphaherpesvirus 3'' (taxonomically), is one of nine known herpes viruses that can infect humans. It causes chickenpox (varicella) commonly affecting chil ...
, cytomegalovirus and Epstein–Barr virus
The Epstein–Barr virus (EBV), formally called ''Human gammaherpesvirus 4'', is one of the nine known human herpesvirus types in the herpes family, and is one of the most common viruses in humans. EBV is a double-stranded DNA virus.
It is b ...
. With each virus, some strains have become resistant to the administered drugs.
Antiviral drugs typically target key components of viral reproduction; for example, oseltamivir
Oseltamivir, sold under the brand name Tamiflu, is an antiviral medication used to treat and prevent influenza A and influenza B, viruses that cause the flu. Many medical organizations recommend it in people who have complications or are at high ...
targets influenza neuraminidase
Exo-α-sialidase (EC 3.2.1.18, sialidase, neuraminidase; systematic name acetylneuraminyl hydrolase) is a glycoside hydrolase that cleaves the glycosidic linkages of neuraminic acids:
: Hydrolysis of α-(2→3)-, α-(2→6)-, α-(2→8)- glyc ...
, while guanosine analogs inhibit viral DNA polymerase. Resistance to antivirals is thus acquired through mutations in the genes that encode the protein targets of the drugs.
Resistance to HIV antivirals is problematic, and even multi-drug resistant strains have evolved. One source of resistance is that many current HIV drugs, including NRTIs and NNRTIs, target reverse transcriptase; however, HIV-1 reverse transcriptase is highly error prone and thus mutations conferring resistance arise rapidly. Resistant strains of the HIV virus emerge rapidly if only one antiviral drug is used. Using three or more drugs together, termed combination therapy
Combination therapy or polytherapy is therapy that uses more than one medication or modality. Typically, the term refers to using multiple therapies to treat a ''single'' disease, and often all the therapies are pharmaceutical (although it can also ...
, has helped to control this problem, but new drugs are needed because of the continuing emergence of drug-resistant HIV strains.
Fungi
Infections by fungi are a cause of high morbidity and mortality in immunocompromised
Immunodeficiency, also known as immunocompromisation, is a state in which the immune system's ability to fight infectious diseases and cancer is compromised or entirely absent. Most cases are acquired ("secondary") due to extrinsic factors that a ...
persons, such as those with HIV/AIDS, tuberculosis or receiving chemotherapy
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of cancer treatment that uses one or more anti-cancer drugs ( chemotherapeutic agents or alkylating agents) as part of a standardized chemotherapy regimen. Chemothe ...
. The fungi ''Candida'', ''Cryptococcus neoformans
''Cryptococcus neoformans'' is an encapsulated yeast belonging to the class Tremellomycetes and an obligate aerobe that can live in both plants and animals. Its teleomorph is a filamentous fungus, formerly referred to ''Filobasidiella neoforman ...
'' and '' Aspergillus fumigatus'' cause most of these infections and antifungal resistance occurs in all of them. Multidrug resistance in fungi is increasing because of the widespread use of antifungal drugs to treat infections in immunocompromised individuals.
Of particular note, Fluconazole
Fluconazole is an antifungal medication used for a number of fungal infections. This includes candidiasis, blastomycosis, coccidiodomycosis, cryptococcosis, histoplasmosis, dermatophytosis, and pityriasis versicolor. It is also used to pr ...
-resistant ''Candida'' species have been highlighted as a growing problem by the CDC.candidiasis
Candidiasis is a fungal infection due to any type of '' Candida'' (a type of yeast). When it affects the mouth, in some countries it is commonly called thrush. Signs and symptoms include white patches on the tongue or other areas of the mouth ...
infection, the most common of which is ''Candida albicans
''Candida albicans'' is an opportunistic pathogenic yeast that is a common member of the human gut flora. It can also survive outside the human body. It is detected in the gastrointestinal tract and mouth in 40–60% of healthy adults. It is usu ...
''. ''Candida'' yeasts normally inhabit the skin and mucous membranes without causing infection. However, overgrowth of ''Candida'' can lead to candidiasis. Some ''Candida'' strains are becoming resistant to first-line and second-line antifungal agents such as azoles
Azoles are a class of five-membered heterocyclic compounds containing a nitrogen atom and at least one other non-carbon atom (i.e. nitrogen, sulfur, or oxygen) as part of the ring.
Their names originate from the Hantzsch–Widman nomenclature. T ...
and echinocandin
Echinocandins are a class of antifungal drugs that inhibit the synthesis of β-glucan in the fungal cell wall via noncompetitive inhibition of the enzyme 1,3-β glucan synthase. The class has been termed the " penicillin of antifungals," along ...
s.
Parasites
The protozoan parasites that cause the diseases malaria
Malaria is a mosquito-borne infectious disease that affects humans and other animals. Malaria causes symptoms that typically include fever, tiredness, vomiting, and headaches. In severe cases, it can cause jaundice, seizures, coma, or death. S ...
, trypanosomiasis
Trypanosomiasis or trypanosomosis is the name of several diseases in vertebrates caused by parasitic protozoan trypanosomes of the genus ''Trypanosoma''. In humans this includes African trypanosomiasis and Chagas disease. A number of other diseas ...
, toxoplasmosis
Toxoplasmosis is a parasitic disease caused by '' Toxoplasma gondii'', an apicomplexan. Infections with toxoplasmosis are associated with a variety of neuropsychiatric and behavioral conditions. Occasionally, people may have a few weeks or mont ...
, cryptosporidiosis
Cryptosporidiosis, sometimes informally called crypto, is a parasitic disease caused by '' Cryptosporidium'', a genus of protozoan parasites in the phylum Apicomplexa. It affects the distal small intestine and can affect the respiratory tra ...
and leishmaniasis are important human pathogens.artemisinin
Artemisinin () and its semisynthetic derivatives are a group of drugs used in the treatment of malaria due to '' Plasmodium falciparum''. It was discovered in 1972 by Tu Youyou, who shared the 2015 Nobel Prize in Physiology or Medicine for her ...
has also been reported. The problem of drug resistance in malaria has driven efforts to develop vaccines.
Trypanosomes
Trypanosomatida is a group of kinetoplastid excavates distinguished by having only a single flagellum. The name is derived from the Greek ''trypano'' (borer) and ''soma'' (body) because of the corkscrew-like motion of some trypanosomatid species. ...
are parasitic protozoa that cause African trypanosomiasis and Chagas disease (American trypanosomiasis). There are no vaccines to prevent these infections so drugs such as pentamidine and suramin
Suramin is a medication used to treat African sleeping sickness and river blindness. It is the treatment of choice for sleeping sickness without central nervous system involvement. It is given by injection into a vein.
Suramin causes a fair ...
, benznidazole
Benznidazole is an antiparasitic medication used in the treatment of Chagas disease. While it is highly effective in early disease this decreases in those who have long-term infection. It is the first-line treatment given its moderate side effe ...
and nifurtimox
Nifurtimox, sold under the brand name Lampit, is a medication used to treat Chagas disease and sleeping sickness. For sleeping sickness it is used together with eflornithine in nifurtimox-eflornithine combination treatment. In Chagas disease it ...
are used to treat infections. These drugs are effective but infections caused by resistant parasites have been reported.
History
The 1950s to 1970s represented the golden age of antibiotic discovery, where countless new classes of antibiotics were discovered to treat previously incurable diseases such as tuberculosis and syphilis. However, since that time the discovery of new classes of antibiotics has been almost nonexistent, and represents a situation that is especially problematic considering the resiliency of bacteria shown over time and the continued misuse and overuse of antibiotics in treatment.Alexander Fleming
Sir Alexander Fleming (6 August 1881 – 11 March 1955) was a Scottish physician and microbiologist, best known for discovering the world's first broadly effective antibiotic substance, which he named penicillin. His discovery in 1928 of w ...
who said "The time may come when penicillin can be bought by anyone in the shops. Then there is the danger that the ignorant man may easily under-dose himself and by exposing his microbes to nonlethal quantities of the drug make them resistant." Without the creation of new and stronger antibiotics an era where common infections and minor injuries can kill, and where complex procedures such as surgery and chemotherapy become too risky, is a very real possibility. Antimicrobial resistance can lead to epidemics of enormous proportions if preventive actions are not taken. In this day and age current antimicrobial resistance leads to longer hospital stays, higher medical costs, and increased mortality.
Society and culture
Since the mid-1980s pharmaceutical companies have invested in medications for cancer or chronic disease that have greater potential to make money and have "de-emphasized or dropped development of antibiotics".World Economic Forum
The World Economic Forum (WEF) is an international non-governmental and lobbying organisation based in Cologny, canton of Geneva, Switzerland. It was founded on 24 January 1971 by German engineer and economist Klaus Schwab. The foundation, ...
in Davos, Switzerland
). Swiss law does not designate a ''capital'' as such, but the federal parliament and government are installed in Bern, while other federal institutions, such as the federal courts, are in other cities (Bellinzona, Lausanne, Luzern, Neuchâtel ...
, more than "80 pharmaceutical and diagnostic companies" from around the world called for "transformational commercial models" at a global level to spur research and development on antibiotics and on the "enhanced use of diagnostic tests that can rapidly identify the infecting organism".
Legal frameworks
Some global health scholars have argued that a global, legal framework is needed to prevent and control antimicrobial resistance.
United States
For the United States 2016 budget, U.S. president
The president of the United States (POTUS) is the head of state and head of government of the United States of America. The president directs the executive branch of the federal government and is the commander-in-chief of the United States ...
Barack Obama
Barack Hussein Obama II ( ; born August 4, 1961) is an American politician who served as the 44th president of the United States from 2009 to 2017. A member of the Democratic Party, Obama was the first African-American president of the U ...
proposed to nearly double the amount of federal funding to "combat and prevent" antibiotic resistance to more than $1.2 billion. Many international funding agencies like USAID, DFID, SIDA and Bill & Melinda Gates Foundation have pledged money for developing strategies to counter antimicrobial resistance.
On 27 March 2015, the White House
The White House is the official residence and workplace of the president of the United States. It is located at 1600 Pennsylvania Avenue NW in Washington, D.C., and has been the residence of every U.S. president since John Adams in 1800. ...
released a comprehensive plan to address the increasing need for agencies to combat the rise of antibiotic-resistant bacteria. The Task Force for Combating Antibiotic-Resistant Bacteria developed ''The National Action Plan for Combating Antibiotic-Resistant Bacteria'' with the intent of providing a roadmap to guide the US in the antibiotic resistance challenge and with hopes of saving many lives. This plan outlines steps taken by the Federal government over the next five years needed in order to prevent and contain outbreaks of antibiotic-resistant infections; maintain the efficacy of antibiotics already on the market; and to help to develop future diagnostics, antibiotics, and vaccines.
United Kingdom
Public Health England
Public Health England (PHE) was an executive agency of the Department of Health and Social Care in England which began operating on 1 April 2013 to protect and improve health and wellbeing and reduce health inequalities. Its formation came as a ...
reported that the total number of antibiotic resistant infections in England rose by 9% from 55,812 in 2017 to 60,788 in 2018, but antibiotic consumption had fallen by 9% from 20.0 to 18.2 defined daily doses per 1,000 inhabitants per day between 2014 and 2018.
Policies
According to World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level of h ...
, policymakers can help tackle resistance by strengthening resistance-tracking and laboratory capacity and by regulating and promoting the appropriate use of medicines.
Further research
Rapid viral testing
Clinical investigation to rule out bacterial infections are often done for patients with pediatric acute respiratory infections. Currently it is unclear if rapid viral testing affects antibiotic use in children.
Vaccines
Microorganisms usually do not develop resistance to vaccines because vaccines reduce the spread of the infection and target the pathogen in multiple ways in the same host and possibly in different ways between different hosts. Furthermore, if the use of vaccines increases, there is evidence that antibiotic resistant strains of pathogens will decrease; the need for antibiotics will naturally decrease as vaccines prevent infection before it occurs. However, there are well documented cases of vaccine resistance, although these are usually much less of a problem than antimicrobial resistance.
While theoretically promising, antistaphylococcal vaccines have shown limited efficacy, because of immunological variation between ''Staphylococcus'' species, and the limited duration of effectiveness of the antibodies produced. Development and testing of more effective vaccines is underway.
Two registrational trials have evaluated vaccine candidates in active immunization strategies against ''S. aureus'' infection. In a phase II trial, a bivalent vaccine of capsular proteins 5 & 8 was tested in 1804 hemodialysis patients with a primary fistula or synthetic graft vascular access. After 40 weeks following vaccination a protective effect was seen against ''S. aureus'' bacteremia, but not at 54 weeks following vaccination. Based on these results, a second trial was conducted which failed to show efficacy.
Alternating therapy
Alternating therapy is a proposed method in which two or three antibiotics are taken in a rotation versus taking just one antibiotic such that bacteria resistant to one antibiotic are killed when the next antibiotic is taken. Studies have found that this method reduces the rate at which antibiotic resistant bacteria emerge in vitro relative to a single drug for the entire duration.
Studies have found that bacteria that evolve antibiotic resistance towards one group of antibiotic may become more sensitive to others. This phenomenon can be used to select against resistant bacteria using an approach termed collateral sensitivity cycling, which has recently been found to be relevant in developing treatment strategies for chronic infections caused by ''Pseudomonas aeruginosa''. Despite its promise, large-scale clinical and experimental studies revealed limited evidence of susceptibility to antibiotic cycling across various pathogens.
Development of new drugs
Since the discovery of antibiotics, research and development (R&D) efforts have provided new drugs in time to treat bacteria that became resistant to older antibiotics, but in the 2000s there has been concern that development has slowed enough that seriously ill people may run out of treatment options. Another concern is that practitioners may become reluctant to perform routine surgeries because of the increased risk of harmful infection.aminoglycoside
Aminoglycoside is a medicinal and bacteriologic category of traditional Gram-negative antibacterial medications that inhibit protein synthesis and contain as a portion of the molecule an amino-modified glycoside (sugar). The term can also refer ...
s (such as amikacin
Amikacin is an antibiotic medication used for a number of bacterial infections. This includes joint infections, intra-abdominal infections, meningitis, pneumonia, sepsis, and urinary tract infections. It is also used for the treatment of mu ...
, gentamicin, kanamycin
Kanamycin A, often referred to simply as kanamycin, is an antibiotic used to treat severe bacterial infections and tuberculosis. It is not a first line treatment. It is used by mouth, injection into a vein, or injection into a muscle. Kanamycin ...
, streptomycin
Streptomycin is an antibiotic medication used to treat a number of bacterial infections, including tuberculosis, ''Mycobacterium avium'' complex, endocarditis, brucellosis, ''Burkholderia'' infection, plague, tularemia, and rat bite fever. F ...
, etc.) used for the treatment of drug-resistant tuberculosis
Tuberculosis management describes the techniques and procedures utilized for treating tuberculosis (TB).
The medical standard for active TB is a short course treatment involving a combination of isoniazid, rifampicin (also known as Rifampin), py ...
and cystic fibrosis can cause respiratory disorders, deafness and kidney failure. The potential crisis at hand is the result of a marked decrease in industry research and development.development
Development or developing may refer to:
Arts
*Development hell, when a project is stuck in development
*Filmmaking, development phase, including finance and budgeting
*Development (music), the process thematic material is reshaped
* Photograph ...
than for other pharmaceuticals. In 2011, Pfizer
Pfizer Inc. ( ) is an American multinational pharmaceutical and biotechnology corporation headquartered on 42nd Street in Manhattan, New York City. The company was established in 1849 in New York by two German entrepreneurs, Charles Pfizer ...
, one of the last major pharmaceutical companies developing new antibiotics, shut down its primary research effort, citing poor shareholder returns relative to drugs for chronic illnesses.high-throughput screening
High-throughput screening (HTS) is a method for scientific experimentation especially used in drug discovery and relevant to the fields of biology, materials science and chemistry. Using robotics, data processing/control software, liquid handling ...
manner to diversify novel lanthipeptides.
In the United States, drug companies and the administration of President Barack Obama
Barack Hussein Obama II ( ; born August 4, 1961) is an American politician who served as the 44th president of the United States from 2009 to 2017. A member of the Democratic Party, Obama was the first African-American president of the U ...
had been proposing changing the standards by which the FDA approves antibiotics targeted at resistant organisms.President's Council of Advisors on Science and Technology
The President's Council of Advisors on Science and Technology (PCAST) is a council, chartered (or re-chartered) in each administration with a broad mandate to advise the president of the United States on science and technology. The current PCAST w ...
(PCAST) which outlines strategies to stream-line clinical trials and speed up the R&D of new antibiotics. Among the proposals:
* Create a 'robust, standing national clinical trials network for antibiotic testing' which will promptly enroll patients once identified to have dangerous bacterial infections. The network will allow testing multiple new agents from different companies simultaneously for their safety and efficacy.
* Establish a 'Special Medical Use (SMU)' pathway for FDA to approve new antimicrobial agents for use in limited patient populations, shorten the approval timeline for new drug so patients with severe infections could benefit as quickly as possible.
* Provide economic incentives, especially for development of new classes of antibiotics, to offset the steep R&D costs which drive away the industry to develop antibiotics.
Scientists have started using advanced computational approaches with supercomputers for the development of new antibiotic derivatives to deal with antimicrobial resistance.
Biomaterials
Using antibiotic-free alternatives in bone infection treatment may help decrease the use of antibiotics and thus antimicrobial resistance.copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkis ...
and silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, whi ...
nanomaterials
*
Nanomaterials describe, in principle, materials of which a single unit is sized (in at least one dimension) between 1 and 100 nm (the usual definition of nanoscale).
Nanomaterials research takes a materials science-based approach to na ...
have demonstrated appealing features for the development of a new family of antimicrobial agents.
Rediscovery of ancient treatments
Similar to the situation in malaria therapy, where successful treatments based on ancient recipes have been found, there has already been some success in finding and testing ancient drugs and other treatments that are effective against AMR bacteria.
Rapid diagnostics
 Distinguishing infections requiring antibiotics from self-limiting ones is clinically challenging. In order to guide appropriate use of antibiotics and prevent the evolution and spread of antimicrobial resistance, diagnostic tests that provide clinicians with timely, actionable results are needed.
Acute febrile illness is a common reason for seeking medical care worldwide and a major cause of morbidity and mortality. In areas with decreasing malaria incidence, many febrile patients are inappropriately treated for malaria, and in the absence of a simple diagnostic test to identify alternative causes of fever, clinicians presume that a non-malarial febrile illness is most likely a bacterial infection, leading to inappropriate use of antibiotics. Multiple studies have shown that the use of malaria rapid diagnostic tests without reliable tools to distinguish other fever causes has resulted in increased antibiotic use.
Antimicrobial susceptibility testing (AST) can help practitioners avoid prescribing unnecessary antibiotics in the style of
Distinguishing infections requiring antibiotics from self-limiting ones is clinically challenging. In order to guide appropriate use of antibiotics and prevent the evolution and spread of antimicrobial resistance, diagnostic tests that provide clinicians with timely, actionable results are needed.
Acute febrile illness is a common reason for seeking medical care worldwide and a major cause of morbidity and mortality. In areas with decreasing malaria incidence, many febrile patients are inappropriately treated for malaria, and in the absence of a simple diagnostic test to identify alternative causes of fever, clinicians presume that a non-malarial febrile illness is most likely a bacterial infection, leading to inappropriate use of antibiotics. Multiple studies have shown that the use of malaria rapid diagnostic tests without reliable tools to distinguish other fever causes has resulted in increased antibiotic use.
Antimicrobial susceptibility testing (AST) can help practitioners avoid prescribing unnecessary antibiotics in the style of precision medicine
Precision, precise or precisely may refer to:
Science, and technology, and mathematics Mathematics and computing (general)
* Accuracy and precision, measurement deviation from true value and its scatter
* Significant figures, the number of digi ...
, and help them prescribe effective antibiotics, but with the traditional approach it could take 12 to 48 hours.molecular diagnostics
Molecular diagnostics is a collection of techniques used to analyze biological markers in the genome and proteome, and how their cells express their genes as proteins, applying molecular biology to medical testing. In medicine the technique is ...
innovations, is defined as "being feasible within an 8-h working shift".microfluidics
Microfluidics refers to the behavior, precise control, and manipulation of fluids that are geometrically constrained to a small scale (typically sub-millimeter) at which surface forces dominate volumetric forces. It is a multidisciplinary field tha ...
and cell tracking, to monitor bacterial response to antibiotics in less than 30 minutes overall manipulation time. Recently, this platform has been advanced by coupling microfluidic chip with optical tweezing in order to isolate bacteria with altered phenotype directly from the analytical matrix.
Phage therapy
Phage therapy
Phage therapy, viral phage therapy, or phagotherapy is the therapeutic use of bacteriophages for the treatment of pathogenic bacterial infections. This therapeutic approach emerged at the beginning of the 20th century but was progressively re ...
is the therapeutic
A therapy or medical treatment (often abbreviated tx, Tx, or Tx) is the attempted remediation of a health problem, usually following a medical diagnosis.
As a rule, each therapy has indications and contraindications. There are many different ...
use of bacteriophage
A bacteriophage (), also known informally as a ''phage'' (), is a duplodnaviria virus that infects and replicates within bacteria and archaea. The term was derived from "bacteria" and the Greek φαγεῖν ('), meaning "to devour". Bacteri ...
s to treat pathogenic bacterial infection
Pathogenic bacteria are bacteria that can cause disease. This article focuses on the bacteria that are pathogenic to humans. Most species of bacteria are harmless and are often beneficial but others can cause infectious diseases. The number ...
s.Georgia
Georgia most commonly refers to:
* Georgia (country), a country in the Caucasus region of Eurasia
* Georgia (U.S. state), a state in the Southeast United States
Georgia may also refer to:
Places
Historical states and entities
* Related to the ...
(George Eliava Institute
The George Eliava Institute of Bacteriophage, Microbiology and Virology (aka Tbilisi Institute) has been active since the 1930s in the field of phage therapy, which is used to combat microbial infection (cf. antibiotic-resistant strains).
Histor ...
) and in one institute in Wrocław
Wrocław (; german: Breslau, or . ; Silesian German: ''Brassel'') is a city in southwestern Poland and the largest city in the historical region of Silesia. It lies on the banks of the River Oder in the Silesian Lowlands of Central Europe, rou ...
, Poland. Bacteriophage cocktails are common drugs sold over the counter in pharmacies in eastern countries.
See also
References
Books
*
*
*
Journals
*
*
*
*
*
*
* 16-minute film about a post-antibiotic world. Review:
External links
*
*
Animation of Antibiotic Resistance
CDC Guideline "Management of Multidrug-Resistant Organisms in Healthcare Settings, 2006"
Antimicrobial Stewardship Project
at the Center for Infectious Disease Research and Policy
The Center for Infectious Disease Research and Policy (CIDRAP) is a center within the University of Minnesota that focuses on addressing public health preparedness and emerging infectious disease response. It was founded in 2001 by Dr. Michael Os ...
(CIDRAP), University of Minnesota
AMR Industry Alliance
"members from large R&D pharma, generic manufacturers, biotech, and diagnostic companies"
Why won't antibiotics cure us anymore? - prof. dr. Nathaniel Martin (Universiteit Leiden)
{{DEFAULTSORT:Antibiotic Resistance
Evolutionary biology
Health disasters
Pharmaceuticals policy
Veterinary medicine
Global issues
 Antimicrobial resistance (AMR) occurs when
Antimicrobial resistance (AMR) occurs when  The WHO defines antimicrobial resistance as a microorganism's resistance to an antimicrobial drug that was once able to treat an infection by that microorganism. A person cannot become resistant to antibiotics. Resistance is a property of the microbe, not a person or other organism infected by a microbe. All types of microbes can develop drug resistance. Thus, there are antibiotic, antifungal, antiviral and antiparasitic resistance.
Antibiotic resistance is a subset of antimicrobial resistance. This more specified resistance is linked to pathogenic bacteria and thus broken down into two further subsets, microbiological and clinical. Resistance linked microbiologically is the most common and occurs from genes, mutated or inherited, that allow the bacteria to resist the mechanism associated with certain antibiotics. Clinical resistance is shown through the failure of many therapeutic techniques where the bacteria that are normally susceptible to a treatment become resistant after surviving the outcome of the treatment. In both cases of acquired resistance, the bacteria can pass the genetic catalyst for resistance through conjugation, transduction, or transformation. This allows the resistance to spread across the same pathogen or even similar bacterial pathogens.
The WHO defines antimicrobial resistance as a microorganism's resistance to an antimicrobial drug that was once able to treat an infection by that microorganism. A person cannot become resistant to antibiotics. Resistance is a property of the microbe, not a person or other organism infected by a microbe. All types of microbes can develop drug resistance. Thus, there are antibiotic, antifungal, antiviral and antiparasitic resistance.
Antibiotic resistance is a subset of antimicrobial resistance. This more specified resistance is linked to pathogenic bacteria and thus broken down into two further subsets, microbiological and clinical. Resistance linked microbiologically is the most common and occurs from genes, mutated or inherited, that allow the bacteria to resist the mechanism associated with certain antibiotics. Clinical resistance is shown through the failure of many therapeutic techniques where the bacteria that are normally susceptible to a treatment become resistant after surviving the outcome of the treatment. In both cases of acquired resistance, the bacteria can pass the genetic catalyst for resistance through conjugation, transduction, or transformation. This allows the resistance to spread across the same pathogen or even similar bacterial pathogens.
 Antimicrobial resistance can evolve naturally due to continued exposure to antimicrobials.
Antimicrobial resistance can evolve naturally due to continued exposure to antimicrobials.  The antimicrobial resistance crisis also extends to the food industry, specifically with food producing animals. Antibiotics are fed to livestock to act as growth supplements, and a preventative measure to decrease the likelihood of infections. This results in the transfer of resistant bacterial strains into the food that humans eat, causing potentially fatal transfer of disease. While this practice does result in better yields and
The antimicrobial resistance crisis also extends to the food industry, specifically with food producing animals. Antibiotics are fed to livestock to act as growth supplements, and a preventative measure to decrease the likelihood of infections. This results in the transfer of resistant bacterial strains into the food that humans eat, causing potentially fatal transfer of disease. While this practice does result in better yields and  There have been increasing public calls for global collective action to address the threat, including a proposal for international treaty on antimicrobial resistance. Further detail and attention is still needed in order to recognize and measure trends in resistance on the international level; the idea of a global tracking system has been suggested but implementation has yet to occur. A system of this nature would provide insight to areas of high resistance as well as information necessary for evaluating programs and other changes made to fight or reverse antibiotic resistance.
There have been increasing public calls for global collective action to address the threat, including a proposal for international treaty on antimicrobial resistance. Further detail and attention is still needed in order to recognize and measure trends in resistance on the international level; the idea of a global tracking system has been suggested but implementation has yet to occur. A system of this nature would provide insight to areas of high resistance as well as information necessary for evaluating programs and other changes made to fight or reverse antibiotic resistance.
 There are several different types of germs that have developed a resistance over time.
The six pathogens causing most deaths associated with resistance are ''Escherichia coli'', ''Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii'', and ''Pseudomonas aeruginosa''. They were responsible for 929,000 deaths attributable to resistance and 3.57 million deaths associated with resistance in 2019.
Penicillinase-producing ''Neisseria gonorrhoeae'' developed a resistance to penicillin in 1976. Another example is Azithromycin-resistant ''Neisseria gonorrhoeae'', which developed a resistance to azithromycin in 2011.
In gram-negative bacteria, plasmid-mediated resistance genes produce proteins that can bind to
There are several different types of germs that have developed a resistance over time.
The six pathogens causing most deaths associated with resistance are ''Escherichia coli'', ''Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii'', and ''Pseudomonas aeruginosa''. They were responsible for 929,000 deaths attributable to resistance and 3.57 million deaths associated with resistance in 2019.
Penicillinase-producing ''Neisseria gonorrhoeae'' developed a resistance to penicillin in 1976. Another example is Azithromycin-resistant ''Neisseria gonorrhoeae'', which developed a resistance to azithromycin in 2011.
In gram-negative bacteria, plasmid-mediated resistance genes produce proteins that can bind to  Distinguishing infections requiring antibiotics from self-limiting ones is clinically challenging. In order to guide appropriate use of antibiotics and prevent the evolution and spread of antimicrobial resistance, diagnostic tests that provide clinicians with timely, actionable results are needed.
Acute febrile illness is a common reason for seeking medical care worldwide and a major cause of morbidity and mortality. In areas with decreasing malaria incidence, many febrile patients are inappropriately treated for malaria, and in the absence of a simple diagnostic test to identify alternative causes of fever, clinicians presume that a non-malarial febrile illness is most likely a bacterial infection, leading to inappropriate use of antibiotics. Multiple studies have shown that the use of malaria rapid diagnostic tests without reliable tools to distinguish other fever causes has resulted in increased antibiotic use.
Antimicrobial susceptibility testing (AST) can help practitioners avoid prescribing unnecessary antibiotics in the style of
Distinguishing infections requiring antibiotics from self-limiting ones is clinically challenging. In order to guide appropriate use of antibiotics and prevent the evolution and spread of antimicrobial resistance, diagnostic tests that provide clinicians with timely, actionable results are needed.
Acute febrile illness is a common reason for seeking medical care worldwide and a major cause of morbidity and mortality. In areas with decreasing malaria incidence, many febrile patients are inappropriately treated for malaria, and in the absence of a simple diagnostic test to identify alternative causes of fever, clinicians presume that a non-malarial febrile illness is most likely a bacterial infection, leading to inappropriate use of antibiotics. Multiple studies have shown that the use of malaria rapid diagnostic tests without reliable tools to distinguish other fever causes has resulted in increased antibiotic use.
Antimicrobial susceptibility testing (AST) can help practitioners avoid prescribing unnecessary antibiotics in the style of