organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clay ...
, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur ...
with the general formula , where R is an organic alkyl
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloal ...
or aryl
In organic chemistry, an aryl is any functional group or substituent derived from an aromatic ring, usually an aromatic hydrocarbon, such as phenyl and naphthyl. "Aryl" is used for the sake of abbreviation or generalization, and "Ar" is used as ...
group and the group a sulfonyl
In organosulfur chemistry, a sulfonyl group can refer either to a functional group found primarily in sulfones, or to a substituent obtained from a sulfonic acid by the removal of the hydroxyl group, similarly to acyl groups. Sulfonyl groups c ...
hydroxide. As a substituent, it is known as a sulfo group. A sulfonic acid can be thought of as sulfuric acid with one hydroxyl group replaced by an organic substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and '' functional group'', as well as '' ...
. The parent compound
In chemistry, a parent structure is the structure of an unadorned ion or molecule from which derivatives can be visualized. Parent structures underpin systematic nomenclature and facilitate classification. Fundamental parent structures have o ...
(with the organic substituent replaced by hydrogen) is the parent sulfonic acid, , a tautomer
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hyd ...
of sulfurous acid
Sulfurous acid (also sulfuric(IV) acid, sulphurous acid (UK), sulphuric(IV) acid (UK)) is the chemical compound with the formula . There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase. ...
, . Salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quanti ...
s or ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
s of sulfonic acids are called sulfonate
In organosulfur chemistry, a sulfonate is a salt or ester of a sulfonic acid. It contains the functional group , where R is an organic group. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non- ...
s.
Preparation
 Aryl sulfonic acids are produced by the process of
Aryl sulfonic acids are produced by the process of sulfonation Aromatic sulfonation is an organic reaction in which a hydrogen atom on an arene is replaced by a sulfonic acid functional group in an electrophilic aromatic substitution. Aryl sulfonic acids are used as detergents, dye, and drugs.
Stoichiometry ...
. Usually the sulfonating agent is sulfur trioxide. A large scale application of this method is the production of alkylbenzenesulfonic acid
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used synthet ...
s:
:electrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that ca ...
and the arene
Aromatic compounds, also known as "mono- and polycyclic aromatic hydrocarbons", are organic compounds containing one or more aromatic rings. The parent member of aromatic compounds is benzene. The word "aromatic" originates from the past grouping ...
is the nucleophile. The reaction is an example of electrophilic aromatic substitution
Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an aromatic system (usually hydrogen) is replaced by an electrophile. Some of the most important electrophilic aromatic substitutions are aromatic ni ...
.
Alkylsulfonic acids can be prepared by many methods. In in sulfoxidation
in chemistry, sulfoxidation refers to two distinct reactions.
In one meaning, sulfoxidation refers to the reaction of alkanes with a mixture of sulfur dioxide and oxygen. This reaction is employed industrially to produce alkyl sulfonic acids, ...
, alkanes are irradiated with a mixture of sulfur dioxide
Sulfur dioxide ( IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic ...
and oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as we ...
. This reaction is employed industrially to produce alkyl sulfonic acids, which are used as surfactant
Surfactants are chemical compounds that decrease the surface tension between two liquids, between a gas and a liquid, or interfacial tension between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, fo ...
s.
:methanesulfonic acid
Methanesulfonic acid (MsOH) or methanesulphonic acid (in British English) is an organosulfuric, colorless liquid with the chemical formula and structure . It is the simplest of the alkylsulfonic acids (). Salts and esters of methanesulfonic ac ...
to methanedisulfonic acid
Methanedisulfonic acid is the organosulfur compound with the formula CH2(SO3H)2. It is the di sulfonic acid of methane. It is prepared by treatment of methanesulfonic acid with oleum. Its acid strength (pKa) is comparable to that of sulfuri ...
.
Many alkane sulfonic acids can be obtained by the addition of bisulfite
The bisulfite ion (IUPAC-recommended nomenclature: hydrogensulfite) is the ion . Salts containing the ion are also known as "sulfite lyes". Sodium bisulfite is used interchangeably with sodium metabisulfite (Na2S2O5). Sodium metabisulfite diss ...
to terminal alkenes. Bisulfite can also be alkylated
Alkylation is the transfer of an alkyl group from one molecule to another. The alkyl group may be transferred as an alkyl carbocation, a free radical, a carbanion, or a carbene (or their equivalents). Alkylating agents are reagents for effecting a ...
by alkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
s:
:taurine
Taurine (), or 2-aminoethanesulfonic acid, is an organic compound that is widely distributed in animal tissues. It is a major constituent of bile and can be found in the large intestine, and accounts for up to 0.1% of total human body weight. It ...
.
Hydrolysis routes
Many sulfonic acids are prepared by hydrolysis ofsulfonyl halide
In inorganic chemistry, sulfonyl halide groups occur when a sulfonyl () functional group is singly bonded to a halogen atom. They have the general formula , where X is a halogen. The stability of sulfonyl halides decreases in the order fluoride ...
perfluorooctanesulfonic acid
Perfluorooctanesulfonic acid (PFOS) ( conjugate base perfluorooctanesulfonate) is a chemical compound having an eight-carbon fluorocarbon chain and a sulfonic acid functional group and thus a perfluorosulfonic acid. It is an anthropogenic (man ...
is prepared by hydrolysis of the sulfonyl fluoride, which in turn is generated by the electrofluorination Electrochemical fluorination (ECF), or electrofluorination, is a foundational organofluorine chemistry method for the preparation of fluorocarbon-based organofluorine compounds.G. Siegemund, W. Schwertfeger, A. Feiring, B. Smart, F. Behr, H. Vogel, ...
of octanesulfonic acid. Similarly the sulfonyl chloride derived from polyethylene is hydrolyzed to the sulfonic acid. These sulfonyl chlorides are produced by free-radical reactions of chlorine, sulfur dioxide, and the hydrocarbons using the Reed reaction The Reed reaction is a chemical reaction that utilizes photochemistry, light to redox, oxidize hydrocarbons to alkylsulfonyl chlorides. This reaction is employed in modifying polyethylene to give chlorosulfonated polyethylene (CSPE), which noted fo ...
.
Vinylsulfonic acid
Vinylsulfonic acid is the organosulfur compound with the chemical formula CH2=CHSO3H. It is the simplest unsaturated sulfonic acid. The C=C double bond is a site of high reactivity. Polymerization gives polyvinylsulfonic acid, especially when u ...
is derived by hydrolysis of carbyl sulfate, (), which in turn is obtained by the addition of sulfur trioxide to ethylene
Ethylene ( IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds).
Ethylene ...
.
Properties
Sulfonic acids are strong acids. They are commonly cited as being around a million times stronger than the correspondingcarboxyl
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxyl ...
ic acid. For example, ''p''-Toluenesulfonic acid and methanesulfonic acid
Methanesulfonic acid (MsOH) or methanesulphonic acid (in British English) is an organosulfuric, colorless liquid with the chemical formula and structure . It is the simplest of the alkylsulfonic acids (). Salts and esters of methanesulfonic ac ...
have p''K''a values of −2.8 and −1.9, respectively, while those of benzoic acid
Benzoic acid is a white (or colorless) solid organic compound with the formula , whose structure consists of a benzene ring () with a carboxyl () substituent. It is the simplest aromatic carboxylic acid. The name is derived from gum benzoin, w ...
and acetic acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main componen ...
are 4.20 and 4.76, respectively. However, as a consequence of their strong acidity, their p''K''a values cannot be measured directly, and values commonly quoted should be regarded as indirect estimates with significant uncertainties. For instance, various sources have reported the p''K''a of methanesulfonic acid to be as high as −0.6 or as low as −6.5. Sulfonic acids are known to react with solid sodium chloride (salt
Salt is a mineral composed primarily of sodium chloride (NaCl), a chemical compound belonging to the larger class of salts; salt in the form of a natural crystalline mineral is known as rock salt or halite. Salt is present in vast quanti ...
) to form the sodium sulfonate
In organosulfur chemistry, a sulfonate is a salt or ester of a sulfonic acid. It contains the functional group , where R is an organic group. Sulfonates are the conjugate bases of sulfonic acids. Sulfonates are generally stable in water, non- ...
and hydrogen chloride. This property implies an acidity within two or three orders of magnitude of that of HCl(g), whose p''K''a was recently accurately determined (p''K''aaq = −5.9).
Because of their polarity, sulfonic acids tend to be crystalline solids or viscous, high-boiling liquids. They are also usually colourless and nonoxidizing, which makes them suitable for use as acid catalysts in organic reactions. Their polarity, in conjunction with their high acidity, renders short-chain sulfonic acids water-soluble, while longer-chain ones exhibit detergent-like properties.
The structure of sulfonic acids is illustrated by the prototype, methanesulfonic acid
Methanesulfonic acid (MsOH) or methanesulphonic acid (in British English) is an organosulfuric, colorless liquid with the chemical formula and structure . It is the simplest of the alkylsulfonic acids (). Salts and esters of methanesulfonic ac ...
. The sulfonic acid group, RSO2OH features a tetrahedral sulfur centre, meaning that sulfur is at the center of four atoms: three oxygens and one carbon. The overall geometry of the sulfur centre is reminiscent of the shape of sulfuric acid.
Taurine
Taurine (), or 2-aminoethanesulfonic acid, is an organic compound that is widely distributed in animal tissues. It is a major constituent of bile and can be found in the large intestine, and accounts for up to 0.1% of total human body weight. It ...
, a bile acid
Bile acids are steroid acids found predominantly in the bile of mammals and other vertebrates. Diverse bile acids are synthesized in the liver. Bile acids are conjugated with taurine or glycine residues to give anions called bile salts.
Primary ...
, and one of the few naturally occurring sulfonic acids (shown in uncommon tautomer
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hyd ...
).
File:Perfluorooctanesulfonic acid structure.svg, PFOS
Perfluorooctanesulfonic acid (PFOS) ( conjugate base perfluorooctanesulfonate) is a chemical compound having an eight- carbon fluorocarbon chain and a sulfonic acid functional group and thus a perfluorosulfonic acid. It is an anthropogenic (man ...
, a surfactant and a controversial pollutant.
File:Tosic acid.png, ''p''-Toluenesulfonic acid, a widely used reagent in organic synthesis.
File:Nafion_structure.png, Nafion
Nafion is a brand name for a sulfonated tetrafluoroethylene based fluoropolymer- copolymer discovered in the late 1960s by Dr. Walther Grot of DuPont. Nafion is a brand of the Chemours company. It is the first of a class of synthetic polymers wi ...
, a polymeric sulfonic acid useful in fuel cell
A fuel cell is an electrochemical cell that converts the chemical energy of a fuel (often hydrogen fuel, hydrogen) and an oxidizing agent (often oxygen) into electricity through a pair of redox reactions. Fuel cells are different from most bat ...
s.
File:Sodium dodecylbenzenesulfonate skeletal.svg, Sodium dodecylbenzenesulfonate
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used syntheti ...
, an alkylbenzenesulfonate surfactant
Surfactants are chemical compounds that decrease the surface tension between two liquids, between a gas and a liquid, or interfacial tension between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, fo ...
used in laundry detergent
Laundry detergent is a type of detergent (cleaning agent) used for cleaning dirty laundry (clothes). Laundry detergent is manufactured in powder (washing powder) and liquid form.
While powdered and liquid detergents hold roughly equal share o ...
s.
File:Coenzyme M (CoM).svg, Coenzyme-M
Coenzyme M is a coenzyme required for methyl-transfer reactions in the metabolism of archaeal methanogens, and in the metabolism of other substrates in bacteria. It is also a necessary cofactor in the metabolic pathway of alkene-oxidizing bacteria. ...
, is a cofactor required for the biosynthesis of methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ear ...
, found in natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon ...
.
Applications
Although both alkyl and aryl sulfonic acids are known, most of the applications are associated with the aromatic derivatives.Detergents and surfactants
Detergent
A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are m ...
s and surfactant
Surfactants are chemical compounds that decrease the surface tension between two liquids, between a gas and a liquid, or interfacial tension between a liquid and a solid. Surfactants may act as detergents, wetting agents, emulsifiers, fo ...
s are molecules that combine highly nonpolar and highly polar groups. Traditionally, soap
Soap is a salt of a fatty acid used in a variety of cleansing and lubricating products. In a domestic setting, soaps are surfactants usually used for washing, bathing, and other types of housekeeping. In industrial settings, soaps are used ...
s are the popular surfactants, being derived from fatty acid
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, ...
s. Since the mid-20th century, the usage of sulfonic acids has surpassed soap in advanced societies. For example, an estimated 2 billion kilograms of alkylbenzenesulfonate
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used synthetic ...
s are produced annually for diverse purposes. Lignin sulfonates, produced by sulfonation of lignin
Lignin is a class of complex organic polymers that form key structural materials in the support tissues of most plants. Lignins are particularly important in the formation of cell walls, especially in wood and bark, because they lend rigidity ...
are components of drilling fluid
In geotechnical engineering, drilling fluid, also called drilling mud, is used to aid the drilling of boreholes into the earth. Often used while drilling oil and natural gas wells and on exploration drilling rigs, drilling fluids are also us ...
concrete
Concrete is a composite material composed of fine and coarse aggregate bonded together with a fluid cement (cement paste) that hardens (cures) over time. Concrete is the second-most-used substance in the world after water, and is the most ...
.
Dyes
Many if not most of theanthraquinone
Anthraquinone, also called anthracenedione or dioxoanthracene, is an aromatic organic compound with formula . Isomers include various quinone derivatives. The term anthraquinone however refers to the isomer, 9,10-anthraquinone (IUPAC: 9,10-dioxoan ...
dyes are produced or processed via sulfonation. Sulfonic acids tend to bind tightly to protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, respon ...
s and carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or ...
s. Most "washable" dye
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution and ...
s are sulfonic acids (or have the functional sulfonyl
In organosulfur chemistry, a sulfonyl group can refer either to a functional group found primarily in sulfones, or to a substituent obtained from a sulfonic acid by the removal of the hydroxyl group, similarly to acyl groups. Sulfonyl groups c ...
group in them) for this reason. p-Cresidinesulfonic acid is used to make food dyes.
Acid catalysts
Being strong acids, sulfonic acids are also used ascatalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
s. The simplest examples are methanesulfonic acid
Methanesulfonic acid (MsOH) or methanesulphonic acid (in British English) is an organosulfuric, colorless liquid with the chemical formula and structure . It is the simplest of the alkylsulfonic acids (). Salts and esters of methanesulfonic ac ...
, CH3SO2OH and ''p''-toluenesulfonic acid, which are regularly used in organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clay ...
as acids that are lipophilic (soluble in organic solvents). Polymeric sulfonic acids are also useful. Dowex resin are sulfonic acid derivatives of polystyrene
Polystyrene (PS) is a synthetic polymer made from monomers of the aromatic hydrocarbon styrene. Polystyrene can be solid or foamed. General-purpose polystyrene is clear, hard, and brittle. It is an inexpensive resin per unit weight. It is ...
and is used as catalysts and for ion exchange (water softening
Water softening is the removal of calcium, magnesium, and certain other metal cations in hard water. The resulting soft water requires less soap for the same cleaning effort, as soap is not wasted bonding with calcium ions. Soft water also extend ...
). Nafion
Nafion is a brand name for a sulfonated tetrafluoroethylene based fluoropolymer- copolymer discovered in the late 1960s by Dr. Walther Grot of DuPont. Nafion is a brand of the Chemours company. It is the first of a class of synthetic polymers wi ...
, a fluorinated polymeric sulfonic acid is a component of proton exchange membranes in fuel cell
A fuel cell is an electrochemical cell that converts the chemical energy of a fuel (often hydrogen fuel, hydrogen) and an oxidizing agent (often oxygen) into electricity through a pair of redox reactions. Fuel cells are different from most bat ...
s.
Drugs
Sulfa drugs, a class of antibacterials, are produced from sulfonic acids.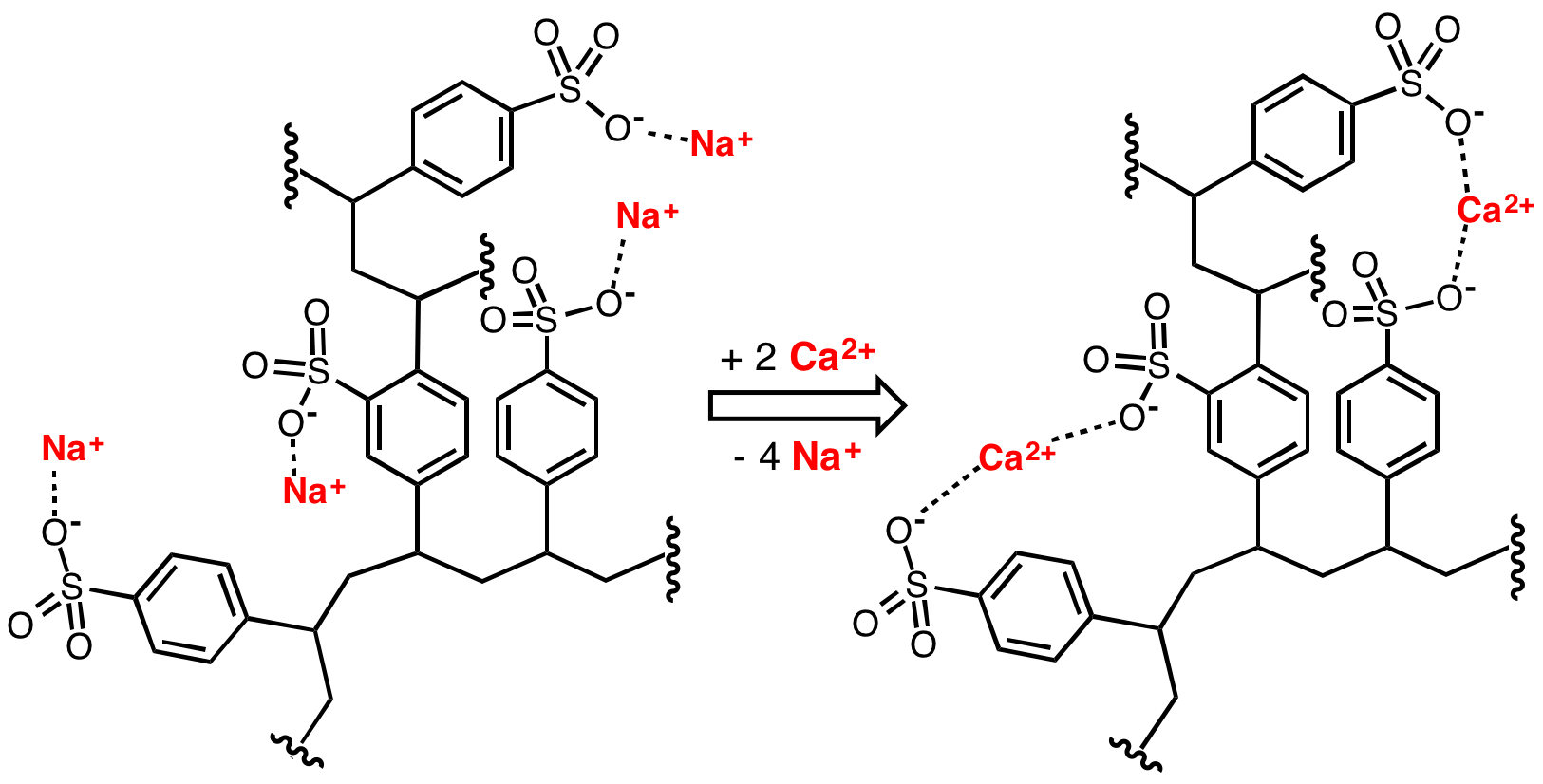
Lignosulfonates
In thesulfite process The sulfite process produces wood pulp that is almost pure cellulose fibers by treating wood chips with solutions of sulfite and bisulfite ions. These chemicals cleave the bonds between the cellulose and lignin components of the lignocellulose. A ...
for paper-making, lignin is removed from the lignocellulose by treating wood chips with solutions of sulfite and bisulfite ions. These reagents cleave the bonds between the cellulose and lignin components and especially within the lignin itself. The lignin is converted to lignosulfonates, useful ionomer
An ionomer () ('' iono-'' + '' -mer'') is a polymer composed of repeat units of both electrically neutral repeating units and ionized units covalently bonded to the polymer backbone as pendant group moieties. Usually no more than 15 mole perce ...
s, which are soluble and can be separated from the cellulose fibers.
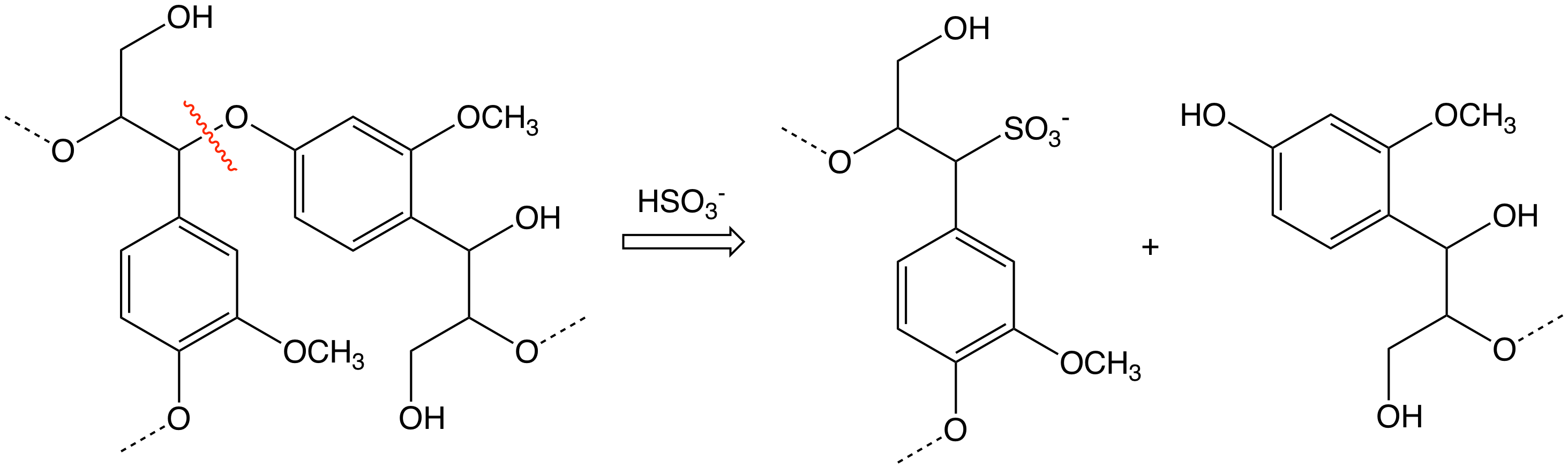
Reactions
Hydrolysis
Arylsulfonic acids are susceptible to hydrolysis, the reverse of the sulfonation reaction. Whereas benzenesulfonic acid hydrolyzes above 200 °C, most related derivatives are easier to hydrolyze. Thus, heating aryl sulfonic acids in aqueous acid produces the parent arene. This reaction is employed in several scenarios. In some cases the sulfonic acid serves as a water-solubilizing protecting group, as illustrated by the purification of para-xylene via its sulfonic acid derivative. In the synthesis of2,6-dichlorophenol
2,6-Dichlorophenol is a compound with formula C6H3Cl2OH. It is one of the six isomers of dichlorophenol. It is a colorless solid. Its pKa is 6.78, which is about 100x more acidic than 2-chlorophenol (8.52) and 1000x more acidic than phenol its ...
, phenol is converted to its 4-sulfonic acid derivative, which then selectively chlorinates at the positions flanking the phenol. Hydrolysis releases the sulfonic acid group.
Esterification
Sulfonic acids can be converted toester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
s. This class of organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon- hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. Th ...
s has the general formula R−SO2−OR. Sulfonic esters such as methyl triflate
Methyl trifluoromethanesulfonate, also commonly called methyl triflate and abbreviated MeOTf, is the organic compound with the formula CF3SO2OCH3. It is a colourless liquid which finds use in organic chemistry as a powerful methylating agent. ...
are considered good alkylating agent
Alkylation is the transfer of an alkyl group from one molecule to another. The alkyl group may be transferred as an alkyl carbocation, a free radical, a carbanion, or a carbene (or their equivalents). Alkylating agents are reagents for effecting ...
s in organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the intentional construction of organic compounds. Organic molecules are often more complex than inorganic compounds, and their synthesis has developed into one o ...
. Such sulfonate esters are often prepared by alcoholysis
In chemistry, solvolysis is a type of nucleophilic substitution (S1/S2) or elimination where the nucleophile is a solvent molecule. Characteristic of S1 reactions, solvolysis of a chiral reactant affords the racemate. Sometimes however, the ster ...
of the sulfonyl chlorides:
:RSO2Cl + R′OH → RSO2OR′ + HCl
Halogenation
Sulfonyl halide groups occur when a sulfonyl functional group is singly bonded to a halogen atom. They have the general formula R−SO2−X where X is a halide, almost invariably chloride. They are produced by chlorination of sulfonic acids usingthionyl chloride
Thionyl chloride is an inorganic compound with the chemical formula . It is a moderately volatile, colourless liquid with an unpleasant acrid odour. Thionyl chloride is primarily used as a chlorinating reagent, with approximately per year bein ...
and related reagents.
Displacement by hydroxide
Although strong, the (aryl)C−SO3− bond can be broken by nucleophilic reagents. Of historic and continuing significance is the α-sulfonation of anthroquinone followed by displacement of the sulfonate group by other nucleophiles, which cannot be installed directly. An early method for producingphenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it ...
involved the base hydrolysis of sodium benzenesulfonate, which can be generated readily from benzene.Manfred Weber, Markus Weber, Michael Kleine-Boymann "Phenol" in Ullmann's Encyclopedia of Industrial Chemistry 2004, Wiley-VCH. .
:C6H5SO3Na + NaOH → C6H5OH + Na2SO3
The conditions for this reaction are harsh, however, requiring 'fused alkali' or molten sodium hydroxide at 350 °C for benzenesulfonic acid itself. Unlike the mechanism for the fused alkali hydrolysis of chlorobenzene, which proceeds through elimination-addition ( benzyne mechanism), benzenesulfonic acid undergoes the analogous conversion by an SNAr mechanism, as revealed by a 14C labeling, despite the lack of stabilizing substituents. Sulfonic acids with electron-withdrawing groups (e.g., with NO2 or CN substituents) undergo this transformation much more readily.
Notes
References
{{Authority control Functional groups