Polyethylene or polythene (abbreviated PE;
IUPAC name polyethene or poly(methylene)) is the most commonly produced
plastic.
It is a
polymer
A polymer (; Greek ''poly-'', "many" + '' -mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and ...
, primarily used for
packaging
Packaging is the science, art and technology of enclosing or protecting products for distribution, storage, sale, and use. Packaging also refers to the process of designing, evaluating, and producing packages. Packaging can be described as a ...
(
plastic bag
A plastic bag, poly bag, or pouch is a type of container made of thin, flexible, plastic film, nonwoven fabric, or plastic textile. Plastic bags are used for containing and transporting goods such as foods, produce, powders, ice, magazines, c ...
s,
plastic films,
geomembranes and containers including
bottle
A bottle is a narrow-necked container made of an impermeable material (such as glass, plastic or aluminium) in various shapes and sizes that stores and transports liquids. Its mouth, at the bottling line, can be sealed with an internal st ...
s, etc.). , over 100 million
tonnes of polyethylene
resins are being produced annually, accounting for 34% of the total plastics market.
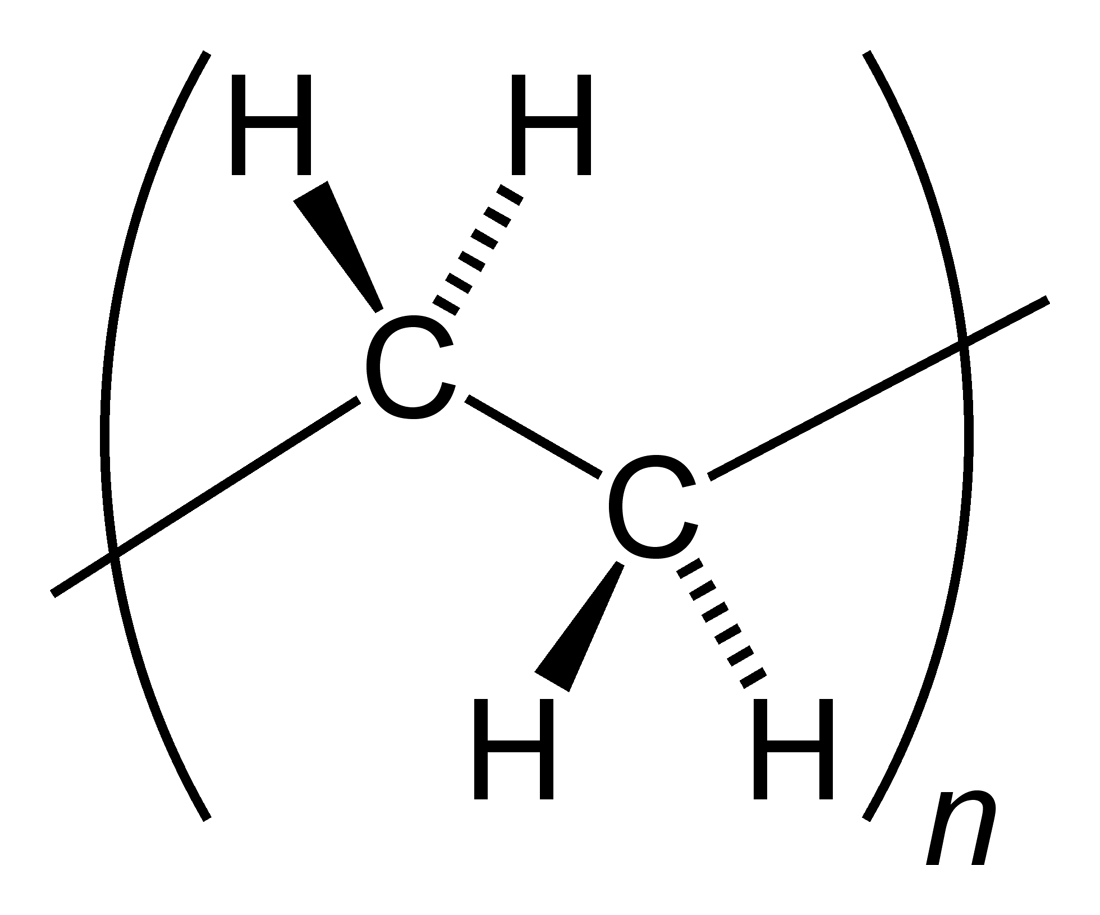
Many kinds of polyethylene are known, with most having the
chemical formula
In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, ...
(C
2H
4)
''n''. PE is usually a mixture of similar
polymer
A polymer (; Greek ''poly-'', "many" + '' -mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and ...
s of
ethylene, with various values of ''n''. It can be ''low-density'' or ''high-density'':
low-density polyethylene is extruded using high pressure () and high temperature (), while
high-density polyethylene is extruded using low pressure () and low temperature (). Polyethylene is usually
thermoplastic, but it can be modified to become
thermosetting instead, for example, in
cross-linked polyethylene.
History
Polyethylene was first synthesized by the German chemist
Hans von Pechmann, who prepared it by accident in 1898 while investigating
diazomethane.
When his colleagues
Eugen Bamberger and Friedrich Tschirner characterized the white, waxy substance that he had created, they recognized that it contained long −CH
2− chains and termed it ''polymethylene''.

The first industrially practical polyethylene synthesis (diazomethane is a notoriously unstable substance that is generally avoided in industrial application) was again accidentally discovered in 1933 by Eric Fawcett and Reginald Gibson at the
Imperial Chemical Industries
Imperial Chemical Industries (ICI) was a British chemical company. It was, for much of its history, the largest manufacturer in Britain.
It was formed by the merger of four leading British chemical companies in 1926.
Its headquarters were at M ...
(ICI) works in
Northwich
{{Infobox UK place
, static_image_name = Northwich - Town Bridge.jpg
, static_image_caption = Town Bridge, the River Weaver and the spire of Holy Trinity Church
, official_name = Northwich
, country ...
,
England
England is a country that is part of the United Kingdom. It shares land borders with Wales to its west and Scotland to its north. The Irish Sea lies northwest and the Celtic Sea to the southwest. It is separated from continental Europe ...
. Upon applying extremely high pressure (several hundred
atmospheres) to a mixture of ethylene and
benzaldehyde they again produced a white, waxy material. Because the reaction had been initiated by trace
oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as we ...
contamination in their apparatus, the experiment was difficult to reproduce at first. It was not until 1935 that another ICI chemist,
Michael Perrin, developed this accident into a reproducible high-pressure synthesis for polyethylene that became the basis for industrial low-density polyethylene (
LDPE) production beginning in 1939. Because polyethylene was found to have very low-loss properties at very high frequency radio waves, commercial distribution in Britain was suspended on the outbreak of World War II, secrecy imposed, and the new process was used to produce insulation for UHF and SHF
coaxial cable
Coaxial cable, or coax (pronounced ) is a type of electrical cable consisting of an inner conductor surrounded by a concentric conducting shield, with the two separated by a dielectric ( insulating material); many coaxial cables also have a ...
s of
radar
Radar is a detection system that uses radio waves to determine the distance ('' ranging''), angle, and radial velocity of objects relative to the site. It can be used to detect aircraft, ships, spacecraft, guided missiles, motor vehicles, w ...
sets. During World War II, further research was done on the ICI process and in 1944, Du Pont at Sabine River, Texas, and Bakelite Corporation at Charleston, West Virginia, began large-scale commercial production under license from ICI.
The landmark breakthrough in the commercial production of polyethylene began with the development of
catalysts that promoted the
polymerization at mild temperatures and pressures. The first of these was a catalyst based on
chromium trioxide discovered in 1951 by
Robert Banks and
J. Paul Hogan
John Paul Hogan (August 7, 1919 – February 19, 2012) was an American research chemist. Along with Robert Banks, he discovered methods of producing polypropylene and high-density polyethylene.
Hogan was born in Lowes, Kentucky to Charles Frankli ...
at
Phillips Petroleum
Phillips Petroleum Company was an American oil company incorporated in 1917 that expanded into petroleum refining, marketing and transportation, natural gas gathering and the chemicals sectors. It was Phillips Petroleum that first found oil in the ...
. In 1953 the German chemist
Karl Ziegler developed a catalytic system based on
titanium halides and organoaluminium compounds that worked at even milder conditions than the Phillips catalyst. The Phillips catalyst is less expensive and easier to work with, however, and both methods are heavily used industrially. By the end of the 1950s both the Phillips- and
Ziegler Ziegler is a common German-language surname meaning "brick-maker" and may refer to the following people:
Actors
* Ernst Ziegler (1894–1974), German actor
*Lulu Ziegler (1903–1973), Danish actress
* Matilda Ziegler (born 1964), English actress
* ...
-type catalysts were being used for high-density polyethylene (HDPE) production. In the 1970s, the Ziegler system was improved by the incorporation of
magnesium chloride. Catalytic systems based on soluble catalysts, the
metallocenes, were reported in 1976 by
Walter Kaminsky and
Hansjörg Sinn Hansjörg (or ''Hansjoerg'') may refer to:
Hansjörg
*Hansjörg Aschenwald (born 1965), Austrian Nordic combined skier
*Hansjörg Dittus, German physicist whose fields of expertise are gravitational physics, metrology, inertial sensors
*Hansjörg F ...
. The Ziegler- and metallocene-based catalysts families have proven to be very flexible at copolymerizing ethylene with other
olefin
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s and have become the basis for the wide range of polyethylene
resins available today, including
very-low-density polyethylene and
linear low-density polyethylene. Such resins, in the form of
UHMWPE fibers, have (as of 2005) begun to replace
aramids in many high-strength applications.
Properties
The properties of polyethylene can be divided into mechanical, chemical, electrical, optical, and thermal properties.
Mechanical
Polyethylene is of low strength,
hardness and rigidity, but has a high
ductility
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile str ...
and
impact strength as well as low friction. It shows strong
creep
Creep, Creeps or CREEP may refer to:
People
* Creep, a creepy person
Politics
* Committee for the Re-Election of the President (CRP), mockingly abbreviated as CREEP, an fundraising organization for Richard Nixon's 1972 re-election campaign
Art ...
under persistent force, which can be reduced by addition of short fibers. It feels waxy when touched.
Thermal
The commercial applicability of polyethylene is limited by its low melting point compared to other thermoplastics. For common commercial grades of medium- and high-density polyethylene the melting point is typically in the range . The melting point for average commercial low-density polyethylene is typically . These temperatures vary strongly with the type of polyethylene, but the theoretical upper limit of melting of polyethylene is reported to be . Combustion typically occurs above .
Chemical
Polyethylene consists of nonpolar, saturated, high-molecular-weight
hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ...
s. Therefore, its chemical behavior is similar to
paraffin. The individual macromolecules are not
covalently linked. Because of their symmetric molecular structure, they tend to crystallize; overall polyethylene is
partially crystalline. Higher
crystallinity increases
density and mechanical and chemical stability.
The standard method to test plastic density is ISO 1183 part 2 (gradient columns), alternatively ISO 1183 part 1
MVS2PRO density analyzer.
Most
LDPE,
MDPE, and
HDPE grades have excellent chemical resistance, meaning that they are not attacked by strong acids or strong bases and are resistant to gentle oxidants and reducing agents. Crystalline samples do not dissolve at room temperature. Polyethylene (other than cross-linked polyethylene) usually can be dissolved at elevated temperatures in
aromatic hydrocarbons such as
toluene or
xylene, or in chlorinated solvents such as
trichloroethane or
trichlorobenzene.
[Whiteley, Kenneth S.; Heggs, T. Geoffrey; Koch, Hartmut; Mawer, Ralph L. and Immel, Wolfgang (2005) "Polyolefins" in ''Ullmann's Encyclopedia of Industrial Chemistry''. Wiley-VCH, Weinheim. .]
Polyethylene absorbs almost no
water
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as ...
; the gas and water vapour permeability (only polar gases) is lower than for most plastics.
Oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as we ...
,
carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is t ...
and
flavorings, on the other hand, can pass it easily.
PE can become brittle when exposed to sunlight,
carbon black is usually used as a UV stabilizer.
Polyethylene burns slowly with a blue flame having a yellow tip and gives off an odour of paraffin (similar to
candle flame). The material continues burning on removal of the flame source and produces a drip.
Polyethylene cannot be imprinted or bonded with adhesives without pretreatment. High-strength joints are readily achieved with
plastic welding.
Electrical
Polyethylene is a good
electrical insulator. It offers good
electrical treeing resistance; however, it becomes easily
electrostatically charged (which can be reduced by additions of
graphite,
carbon black or
antistatic agents).
When pure, the dielectric constant is in the range 2.2 to 2.4 depending on the density and the loss tangent is very low, making it a good dielectric for a
capacitor
A capacitor is a device that stores electrical energy in an electric field by virtue of accumulating electric charges on two close surfaces insulated from each other. It is a passive electronic component with two terminals.
The effect of a ...
Optical
Depending on thermal history and film thickness, PE can vary between almost clear (
transparent), milky-opaque (
translucent) and
opaque. LDPE has the greatest, LLDPE slightly less, and HDPE the least transparency. Transparency is reduced by
crystallites if they are larger than the wavelength of visible light.
Manufacturing process
Monomer
The ingredient or
monomer is
ethylene (
IUPAC name ethene), a
gaseous
hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ...
with the formula C
2H
4, which can be viewed as a pair of
methylene groups (−−) connected to each other. Typical specifications for PE purity are <5 ppm for water, oxygen, and other
alkenes contents. Acceptable contaminants include N
2, ethane (common precursor to ethylene), and methane. Ethylene is usually produced from petrochemical sources, but also is generated by dehydration of ethanol.
Polymerization
Polymerization of ethylene to polyethylene is described by the following
chemical equation:
: (gas) → − (solid)
Δ/ =
Ethylene is a stable molecule that polymerizes only upon contact with catalysts. The conversion is highly
exothermic.
Coordination polymerization is the most pervasive technology, which means that metal chlorides or metal oxides are used. The most common catalysts consist of
titanium(III) chloride, the so-called
Ziegler–Natta catalysts. Another common catalyst is the
Phillips catalyst, prepared by depositing
chromium(VI) oxide
Chromium trioxide (also known as chromium(VI) oxide or chromic anhydride) is an inorganic compound with the formula CrO3. It is the acidic anhydride of chromic acid, and is sometimes marketed under the same name.
This compound is a dark-purple s ...
on silica.
Polyethylene can be produced through
radical polymerization, but this route has only limited utility and typically requires high-pressure apparatus.
Joining
Commonly used methods for joining polyethylene parts together include:
[Plastics Design Library (1997). ''Handbook of Plastics Joining: A Practical Guide''. Norwich, New York: Plastics Design Library. p. 326. .]
*
Welding
Welding is a fabrication process that joins materials, usually metals or thermoplastics, by using high heat to melt the parts together and allowing them to cool, causing fusion. Welding is distinct from lower temperature techniques such as br ...
**
Hot gas welding
**
Infrared welding
**
Laser welding
**
Ultrasonic welding
**
Heat sealing
**
Heat fusion
*
Fastening
*
Adhesives
**
Pressure-sensitive adhesive (PSAs)
*** Dispersion of solvent-type PSAs
**
Polyurethane
Polyurethane (; often abbreviated PUR and PU) refers to a class of polymers composed of organic chemistry, organic units joined by carbamate (urethane) links. In contrast to other common polymers such as polyethylene and polystyrene, polyurethan ...
contact adhesives
** Two-part polyurethane
**
Epoxy adhesives
**
Hot-melt adhesive
Hot-melt adhesive (HMA), also known as hot glue, is a form of thermoplastic adhesive that is commonly sold as solid cylindrical sticks of various diameters designed to be applied using a hot glue gun. The gun uses a continuous-duty heating eleme ...
s
**
Solvent bonding
Solvent bonding is one of several methods of adhesive bonding for joining plastic joining, plastics. Application of a solvent to a thermoplastic material softens the polymer, and with applied pressure this results in polymer chain interdiffusion a ...
—
Adhesives and
solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
s are rarely used as
solvent bonding
Solvent bonding is one of several methods of adhesive bonding for joining plastic joining, plastics. Application of a solvent to a thermoplastic material softens the polymer, and with applied pressure this results in polymer chain interdiffusion a ...
because polyethylene is
nonpolar and has a high resistance to solvents.
Pressure-sensitive adhesives (PSA) are feasible if the surface chemistry or charge is modified with
plasma activation,
flame treatment, or
corona treatment.
Classification
Polyethylene is classified by its
density and
branching. Its mechanical properties depend significantly on variables such as the extent and type of branching, the crystal structure, and the
molecular weight
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioch ...
. There are several types of polyethylene:
*
Ultra-high-molecular-weight polyethylene (UHMWPE)
*Ultra-low-molecular-weight polyethylene (ULMWPE or PE-WAX)
*High-molecular-weight polyethylene (HMWPE)
*
High-density polyethylene (HDPE)
*High-density cross-linked polyethylene (HDXLPE)
*
Cross-linked polyethylene (PEX or XLPE)
*
Medium-density polyethylene (MDPE)
*
Linear low-density polyethylene (LLDPE)
*
Low-density polyethylene (LDPE)
*Very-low-density polyethylene (VLDPE)
*
Chlorinated polyethylene (CPE)
With regard to sold volumes, the most important polyethylene grades are HDPE, LLDPE, and LDPE.
Ultra-high-molecular-weight (UHMWPE)

UHMWPE is polyethylene with a molecular weight numbering in the millions, usually between 3.5 and 7.5 million
amu. The high molecular weight makes it a very
tough
Tough may refer to:
* Toughness, the resistance to fracture of a material when stressed
* Machismo, prominently exhibited or excessive masculinity
* Psychological resilience
Tough may also refer to:
People
* Allen Tough (1936–2012), Canadian ...
material, but results in less efficient packing of the chains into the
crystal structure
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns t ...
as evidenced by densities of less than high-density polyethylene (for example, 0.930–0.935 g/cm
3). UHMWPE can be made through any catalyst technology, although Ziegler catalysts are most common. Because of its outstanding toughness and its cut, wear, and excellent chemical resistance, UHMWPE is used in a diverse range of applications. These include can- and
bottle
A bottle is a narrow-necked container made of an impermeable material (such as glass, plastic or aluminium) in various shapes and sizes that stores and transports liquids. Its mouth, at the bottling line, can be sealed with an internal st ...
-handling machine parts, moving parts on weaving machines, bearings, gears, artificial joints, edge protection on ice rinks, steel cable replacements on ships, and butchers' chopping boards. It is commonly used for the construction of articular portions of
implants
Implant can refer to:
Medicine
*Implant (medicine), or specifically:
**Brain implant
**Breast implant
**Buttock implant
**Cochlear implant
**Contraceptive implant
**Dental implant
**Fetal tissue implant
**Implantable cardioverter-defibrillator
** ...
used for
hip and
knee replacement
Knee replacement, also known as knee arthroplasty, is a surgical procedure to replace the weight-bearing surfaces of the knee joint to relieve pain and disability, most commonly offered when joint pain is not diminished by conservative sources a ...
s. As
fiber, it competes with
aramid in
bulletproof vests.
High-density (HDPE)

HDPE is defined by a density of greater or equal to 0.941 g/cm
3. HDPE has a low degree of branching. The mostly linear molecules pack together well, so intermolecular forces are stronger than in highly branched polymers. HDPE can be produced by
chromium
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal.
Chromium metal is valued for its high corrosion resistance and h ...
/silica catalysts,
Ziegler–Natta catalysts or
metallocene catalysts; by choosing catalysts and reaction conditions, the small amount of branching that does occur can be controlled. These catalysts prefer the formation of
free radicals at the ends of the growing polyethylene molecules. They cause new ethylene monomers to add to the ends of the molecules, rather than along the middle, causing the growth of a linear chain.
HDPE has high tensile strength. It is used in products and packaging such as milk jugs, detergent bottles, butter tubs, garbage containers, and
water pipes. One-third of all toys are manufactured from HDPE. In 2007, the global HDPE consumption reached a volume of more than 30 million tons.
Cross-linked (PEX or XLPE)
PEX is a medium- to high-density polyethylene containing
cross-link bonds introduced into the polymer structure, changing the thermoplastic into a
thermoset. The high-temperature properties of the polymer are improved, its flow is reduced, and its chemical resistance is enhanced. PEX is used in some potable-water plumbing systems because tubes made of the material can be expanded to fit over a metal nipple and it will slowly return to its original shape, forming a permanent, water-tight connection.
Medium-density (MDPE)
MDPE is defined by a density range of 0.926–0.940 g/cm
3. MDPE can be produced by chromium/silica catalysts, Ziegler–Natta catalysts, or metallocene catalysts. MDPE has good shock and drop resistance properties. It also is less notch-sensitive than HDPE; stress-cracking resistance is better than HDPE. MDPE is typically used in gas pipes and fittings, sacks, shrink film, packaging film, carrier bags, and screw closures.
Linear low-density (LLDPE)
LLDPE is defined by a density range of 0.915–0.925 g/cm
3. LLDPE is a substantially linear polymer with significant numbers of short branches, commonly made by
copolymerization of ethylene with short-chain
alpha-olefins (for example,
1-butene,
1-hexene, and
1-octene
1-Octene is an organic compound with a formula CH2CHC6H13. The alkene is classified as a higher olefin and alpha-olefin, meaning that the double bond is located at the alpha (primary) position, endowing this compound with higher reactivity and thu ...
). LLDPE has higher tensile strength than LDPE, and it exhibits higher impact and
puncture resistance
Puncture resistance denotes the relative ability of a material or object to inhibit the intrusion of a foreign object. This is defined by a test method, regulation, or technical specification. It can be measured in several ways ranging from a s ...
than LDPE. Lower-thickness (gauge) films can be blown, compared with LDPE, with better
environmental stress cracking resistance, but they are not as easy to process. LLDPE is used in packaging, particularly film for bags and sheets. Lower thickness may be used compared to LDPE. It is used for cable coverings, toys, lids, buckets, containers, and pipe. While other applications are available, LLDPE is used predominantly in film applications due to its toughness, flexibility, and relative transparency. Product examples range from agricultural films, Saran wrap, and bubble wrap to multilayer and composite films. In 2013, the world LLDPE market reached a volume of US$40 billion.
Low-density (LDPE)
LDPE is defined by a density range of 0.910–0.940 g/cm
3. LDPE has a high degree of short- and long-chain branching, which means that the chains do not pack into the
crystal structure
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions or molecules in a crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric patterns t ...
as well. It has, therefore, less strong intermolecular forces as the
instantaneous-dipole induced-dipole attraction is less. This results in a lower
tensile strength and increased
ductility
Ductility is a mechanical property commonly described as a material's amenability to drawing (e.g. into wire). In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile str ...
. LDPE is created by
free-radical polymerization
In polymer chemistry, free-radical polymerization (FRP) is a method of polymerization by which a polymer forms by the successive addition of free-radical building blocks (repeat units). Free radicals can be formed by a number of different mechanis ...
. The high degree of branching with long chains gives molten LDPE unique and desirable flow properties. LDPE is used for both rigid containers and plastic film applications such as plastic bags and film wrap. In 2013, the global LDPE market had a volume of almost US$33 billion.
The radical polymerization process used to make LDPE does not include a catalyst that "supervises" the radical sites on the growing PE chains. (In HDPE synthesis, the radical sites are at the ends of the PE chains, because the catalyst stabilizes their formation at the ends.) Secondary
radicals
Radical may refer to:
Politics and ideology Politics
*Radical politics, the political intent of fundamental societal change
*Radicalism (historical), the Radical Movement that began in late 18th century Britain and spread to continental Europe and ...
(in the middle of a chain) are more stable than primary radicals (at the end of the chain), and tertiary radicals (at a branch point) are more stable yet. Each time an ethylene monomer is added, it creates a primary radical, but often these will rearrange to form more stable secondary or tertiary radicals. Addition of ethylene monomers to the secondary or tertiary sites creates branching.
Very-low-density (VLDPE)
VLDPE is defined by a density range of 0.880–0.915 g/cm
3. VLDPE is a substantially linear polymer with high levels of short-chain branches, commonly made by copolymerization of ethylene with short-chain alpha-olefins (for example, 1-butene, 1-hexene and 1-octene). VLDPE is most commonly produced using metallocene catalysts due to the greater co-monomer incorporation exhibited by these catalysts. VLDPEs are used for hose and tubing, ice and frozen food bags, food packaging and stretch wrap as well as impact modifiers when blended with other polymers.
Recently, much research activity has focused on the nature and distribution of long chain branches in polyethylene. In HDPE, a relatively small number of these branches, perhaps one in 100 or 1,000 branches per backbone carbon, can significantly affect the
rheological properties of the polymer.
Copolymers
In addition to
copolymerization with alpha-olefins, ethylene can be copolymerized with a wide range of other monomers and ionic composition that creates ionized free radicals. Common examples include
vinyl acetate (the resulting product is
ethylene-vinyl acetate copolymer, or EVA, widely used in athletic-shoe sole foams) and a variety of
acrylates. Applications of
acrylic copolymer include packaging and sporting goods, and
superplasticizer, used in cement production.
Types of polyethylenes
The particular material properties of "polyethylene" depend on its molecular structure. Molecular weight and crystallinity are the most significant factors; crystallinity in turn depends on molecular weight and degree of branching. The less the polymer chains are branched, and the lower the molecular weight, the higher the crystallinity of polyethylene. Crystallinity ranges from 35% (PE-LD/PE-LLD) to 80% (PE-HD). Polyethylene has a density of 1.0 g/cm
3 in crystalline regions and 0.86 g/cm
3 in amorphous regions. An almost linear relationship exists between density and crystallinity.
The degree of branching of the different types of polyethylene can be schematically represented as follows:
The figure shows polyethylene backbones, short-chain branches and side-chain branches. The polymer chains are represented linearly.
Chain branches
The properties of polyethylene are highly dependent on type and number of chain branches. The chain branches in turn depend on the process used: either the high-pressure process (only PE-LD) or the low-pressure process (all other PE grades). Low-density polyethylene is produced by the high-pressure process by radical polymerization, thereby numerous short chain branches as well as long chain branches are formed. Short chain branches are formed by
intramolecular chain transfer reactions, they are always
butyl or
ethyl chain branches because the reaction proceeds after the following mechanism:
:

Environmental issues


Polyethylene is produced from ethylene, and although ethylene can be produced from
renewable resources, it is mainly obtained from
petroleum
Petroleum, also known as crude oil, or simply oil, is a naturally occurring yellowish-black liquid mixture of mainly hydrocarbons, and is found in geological formations. The name ''petroleum'' covers both naturally occurring unprocessed crude ...
or
natural gas
Natural gas (also called fossil gas or simply gas) is a naturally occurring mixture of gaseous hydrocarbons consisting primarily of methane in addition to various smaller amounts of other higher alkanes. Low levels of trace gases like carbon ...
.
Moreover, the widespread usage of polyethylene poses difficulties for
waste management if it is not recycled. Polyethylene, like other synthetic plastics, is not readily biodegradable, and thus accumulates in
landfills. Recycling is made easier if marked with a
recycling code
Recycling codes are used to identify the materials out of which the item is made, to facilitate easier recycling process. The presence on an item of a recycling code, a chasing arrows logo, or a resin code, is not an automatic indicator that a m ...
or
resin identification code. This can read "PE" or "02" ("plastic number 2") for PE-HD and "04" ("plastic number 4") for PE-LD.
In Japan, getting rid of plastics in an environmentally friendly way was the major problem discussed until the
Fukushima nuclear disaster in 2011 became a larger issue. It was listed as a US$90
billion market for solutions. Since 2008, Japan has rapidly increased the recycling of plastics, but still has a large amount of plastic wrapping which goes to waste.
In 2010, a Japanese researcher, Akinori Ito, released the prototype of a machine which creates oil from polyethylene using a small, self-contained vapor distillation process.
Biodegradability
Polyethylene, like other synthetic plastics, is not readily biodegradable, and thus accumulates in landfills. However, there are a number of species of bacteria and animals that are able to degrade polyethylene.
In May 2008, Daniel Burd, a 16-year-old Canadian, won the
Canada-Wide Science Fair
Started in 1962, the Canadian-Wide Science Fair (CWSF) is an annual science fair in Canada coordinated by Youth Science Canada. Finalists qualify from approximately 25,000 competitors at over 100 Youth Science Canada-affiliated regional science fa ...
in Ottawa after discovering that ''
Pseudomonas fluorescens
''Pseudomonas fluorescens'' is a common Gram-negative, rod-shaped bacterium. It belongs to the ''Pseudomonas'' genus; 16S rRNA analysis as well as phylogenomic analysis has placed ''P. fluorescens'' in the ''P. fluorescens'' group within the genu ...
'', with the help of ''
Sphingomonas'', can degrade over 40% of the weight of plastic bags within six weeks. He later guessed that it would be gone after six more weeks.
The thermophilic bacterium ''
Brevibacillus borstelensis
''Brevibacillus borstelensis'' is a Gram-positive, aerobic, rod-shaped, endospore-forming bacterium of the genus ''Brevibacillus
''Brevibacillus'' is a genus of Gram-positive bacteria in the family Paenibacillaceae
The Paenibacillaceae ar ...
'' (strain 707) was isolated from a soil sample and found to use low-density polyethylene as a sole carbon source when incubated together at 50 °C. Biodegradation increased with time exposed to
ultraviolet radiation.
''
Acinetobacter'' sp. 351 can degrade lower molecular-weight PE oligomers. When PE is subjected to thermo- and photo-oxidization, products including alkanes, alkenes, ketones, aldehydes, alcohols, carboxylic acid, keto-acids, dicarboxylic acids, lactones, and esters are released.
In 2014, a Chinese researcher named Jun Yang discovered that
Indian mealmoth larvae could metabolize polyethylene from observing that plastic bags at his home had small holes in them. Deducing that the hungry larvae must have digested the plastic somehow, he and his team analyzed their gut bacteria and found a few that could use plastic as their only carbon source. Not only could the bacteria from the guts of the ''Plodia interpunctella'' moth larvae metabolize polyethylene, they degraded it significantly, dropping its tensile strength by 50%, its mass by 10% and the molecular weights of its polymeric chains by 13%.
In 2017, researchers reported that the caterpillar of ''
Galleria mellonella'' eats
plastic garbage such as polyethylene.
Climate change
When exposed to ambient solar radiation the plastic produces two
greenhouse gases,
methane and
ethylene. Of particular concern is the plastic type which releases gases at the highest rate: low-density polyethylene (or
LDPE). Due to its low density properties it breaks down more easily over time, leading to higher surface areas. The production of these trace gases from virgin LDPE increase with surface area/time, with rates at the end of a 212-day incubation of 5.8 nmol g-1 d-1 of methane, 14.5 nmol g-1 d-1 of ethylene, 3.9 nmol g-1 d-1 of ethane and 9.7 nmol g-1 d-1 of propylene. When incubated in air, LDPE emits gases at rates ~2 times and ~76 times higher in comparison to water for methane and ethylene, respectively.
Chemically modified polyethylene
Polyethylene may either be modified in the polymerization by
polar or non-polar
comonomers or after polymerization through polymer-analogous reactions. Common polymer-analogous reactions are in case of polyethylene
crosslinking,
chlorination and
sulfochlorination.
Non-polar ethylene copolymers
α-olefins
In the low pressure process
α-olefins (e.g.
1-butene or
1-hexene) may be added, which are incorporated in the polymer chain during polymerization. These copolymers introduce short side chains, thus
crystallinity and
density are reduced. As explained above, mechanical and thermal properties are changed thereby. In particular, PE-LLD is produced this way.
Metallocene polyethylene (PE-MC)
Metallocene polyethylene (PE-M) is prepared by means of
metallocene catalysts, usually including copolymers (z. B. ethene / hexene). Metallocene polyethylene has a relatively narrow
molecular weight distribution, exceptionally high toughness, excellent optical properties and a uniform comonomer content. Because of the narrow molecular weight distribution it behaves less pseudoplastic (especially under larger shear rates). Metallocene polyethylene has a low proportion of low molecular weight (extractable) components and a low welding and sealing temperature. Thus, it is particularly suitable for the food industry.
Polyethylene with multimodal molecular weight distribution
Polyethylene with multimodal molecular weight distribution consists of several polymer fractions, which are homogeneously mixed. Such polyethylene types offer extremely high stiffness, toughness, strength, stress crack resistance and an increased crack propagation resistance. They consist of equal proportions higher and lower molecular polymer fractions. The lower molecular weight units crystallize easier and relax faster. The higher molecular weight fractions form linking molecules between crystallites, thereby increasing toughness and stress crack resistance. Polyethylene with multimodal molecular weight distribution can be prepared either in two-stage reactors, by catalysts with two active centers on a carrier or by blending in extruders.
Cyclic olefin copolymers (COC)
Cyclic olefin copolymer
Cyclic olefin copolymer (COC) is an amorphous polymer made by several polymer manufacturers. COC is a relatively new class of polymers as compared to commodities such as polypropylene and polyethylene. This newer material is used in a wide variet ...
s are prepared by copolymerization of ethene and
cycloolefins (usually
norbornene
Norbornene or norbornylene or norcamphene is a highly strained bridged cyclic hydrocarbon. It is a white solid with a pungent sour odor. The molecule consists of a cyclohexene ring with a methylene bridge between carbons 1 and 4. The molecule carr ...
) produced by using metallocene catalysts. The resulting polymers are amorphous polymers and particularly transparent and heat resistant.
Polar ethylene copolymers
The basic compounds used as polar comonomers are vinyl alcohol (
Ethenol, an unsaturated alcohol), acrylic acid (
propenoic acid, an unsaturated acid) and
ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
s containing one of the two compounds.
Ethylene copolymers with unsaturated alcohols
Ethylene/vinyl alcohol copolymer (EVOH) is (formally) a copolymer of PE and vinyl alcohol (ethenol), which is prepared by (partial) hydrolysis of ethylene-vinyl acetate copolymer (as vinyl alcohol itself is not stable). However, typically EVOH has a higher comonomer content than the VAC commonly used.
EVOH is used in multilayer films for packaging as a barrier layer (barrier plastic). As EVOH is hygroscopic (water-attracting), it absorbs water from the environment, whereby it loses its barrier effect. Therefore, it must be used as a core layer surrounded by other plastics (like LDPE, PP, PA or PET). EVOH is also used as a coating agent against corrosion at street lights, traffic light poles and noise protection walls.
Ethylene/acrylic acid copolymers (EAA)
Copolymer of ethylene and unsaturated carboxylic acids (such as acrylic acid) are characterized by good adhesion to diverse materials, by resistance to stress cracking and high flexibility. However, they are more sensitive to heat and oxidation than ethylene homopolymers. Ethylene/acrylic acid copolymers are used as
adhesion promoters.
If salts of an unsaturated carboxylic acid are present in the polymer, thermo-reversible ion networks are formed, they are called
ionomers. Ionomers are highly transparent thermoplastics which are characterized by high adhesion to metals, high abrasion resistance and high water absorption.
Ethylene copolymers with unsaturated esters
If unsaturated esters are copolymerized with ethylene, either the alcohol moiety may be in the polymer backbone (as it is the case in ethylene-vinyl acetate copolymer) or of the acid moiety (e. g. in ethylene-ethyl acrylate copolymer).
Ethylene-vinyl acetate copolymers are prepared similarly to LD-PE by high pressure polymerization. The proportion of comonomer has a decisive influence on the behaviour of the polymer.
The density decreases up to a comonomer share of 10% because of the disturbed crystal formation. With higher proportions it approaches to the one of
polyvinyl acetate (1.17 g/cm
3).
Due to decreasing crystallinity ethylene vinyl acetate copolymers are getting softer with increasing comonomer content. The polar side groups change the chemical properties significantly (compared to polyethylene):
weather resistance, adhesiveness and weldability rise with comonomer content, while the chemical resistance decreases. Also mechanical properties are changed: stress cracking resistance and toughness in the cold rise, whereas yield stress and heat resistance decrease. With a very high proportion of comonomers (about 50%) rubbery thermoplastics are produced (
thermoplastic elastomers).
Ethylene-ethyl acrylate copolymers behave similarly to ethylene-vinyl acetate copolymers.
Crosslinking
A basic distinction is made between peroxide crosslinking (PE-Xa), silane crosslinking (PE-Xb), electron beam crosslinking (PE-Xc) and azo crosslinking (PE-Xd).
[Vorschau auf kunststoffe.de]
/ref>
 Shown are the peroxide, the silane and irradiation crosslinking. In each method, a radical is generated in the polyethylene chain (top center), either by radiation (h·ν) or by peroxides (R-O-O-R). Then, two radical chains can either directly crosslink (bottom left) or indirectly by silane compounds (bottom right).
*Peroxide crosslinking (PE-Xa): The crosslinking of polyethylene using peroxides (e. g. dicumyl or di-tert-butyl peroxide) is still of major importance. In the so-called ''Engel process'', a mixture of HDPE and 2%
Shown are the peroxide, the silane and irradiation crosslinking. In each method, a radical is generated in the polyethylene chain (top center), either by radiation (h·ν) or by peroxides (R-O-O-R). Then, two radical chains can either directly crosslink (bottom left) or indirectly by silane compounds (bottom right).
*Peroxide crosslinking (PE-Xa): The crosslinking of polyethylene using peroxides (e. g. dicumyl or di-tert-butyl peroxide) is still of major importance. In the so-called ''Engel process'', a mixture of HDPE and 2%radicals
Radical may refer to:
Politics and ideology Politics
*Radical politics, the political intent of fundamental societal change
*Radicalism (historical), the Radical Movement that began in late 18th century Britain and spread to continental Europe and ...
. When these combine, a crosslinked network is formed.hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile.
Biological hydrolysis ...
, which condense then and crosslink the PE by the formation of Si-O-Si bridges. 6 Catalysts such as dibutyltin dilaurate may accelerate the reaction.isotopic radiator
Isotopic may refer to:
* In the physical sciences, to do with chemical isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong ...
). PE products are crosslinked below the crystalline melting point by splitting off hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
atoms. β-radiation possesses a penetration depth of 10 mm, ɣ-radiation 100 mm. Thereby the interior or specific areas can be excluded from the crosslinking.solid state
Solid state, or solid matter, is one of the four fundamental states of matter.
Solid state may also refer to:
Electronics
* Solid-state electronics, circuits built of solid materials
* Solid state ionics, study of ionic conductors and their u ...
. Thereby, the cross-linking takes place primarily in the amorphous regions, while the crystallinity remains largely intact.
Chlorination and sulfochlorination
Chlorinated Polyethylene (PE-C) is an inexpensive material having a chlorine content from 34 to 44%. It is used in blends with PVC because the soft, rubbery chloropolyethylene is embedded in the PVC matrix, thereby increasing the impact resistance. It also increases the weather resistance. Furthermore, it is used for softening PVC foils, without risking the migrate of plasticizers. Chlorinated polyethylene can be crosslinked peroxidically to form an elastomer which is used in cable and rubber industry.synthetic rubber
A synthetic rubber is an artificial elastomer. They are polymers synthesized from petroleum byproducts. About 32-million metric tons of rubbers are produced annually in the United States, and of that amount two thirds are synthetic. Synthetic rubbe ...
.
Bio-based polyethylene
Braskem and Toyota Tsusho Corporation
is a sōgō shōsha (trading company), a member of the Toyota Group. Toyota Tsusho has a worldwide presence through its many subsidiaries and operating divisions, including over 150 offices, and 900 subsidiaries and affiliates around the world. ...
started joint marketing activities to produce polyethylene from sugarcane
Sugarcane or sugar cane is a species of (often hybrid) tall, perennial grass (in the genus ''Saccharum'', tribe Andropogoneae) that is used for sugar production. The plants are 2–6 m (6–20 ft) tall with stout, jointed, fibrous stalks t ...
. Braskem will build a new facility at their existing industrial unit in Triunfo, Rio Grande do Sul, Brazil with an annual production capacity of , and will produce high-density and low-density polyethylene from bioethanol derived from sugarcane.
Polyethylene can also be made from other feedstocks, including wheat grain and sugar beet
A sugar beet is a plant whose root contains a high concentration of sucrose and which is grown commercially for sugar production. In plant breeding, it is known as the Altissima cultivar group of the common beet (''Beta vulgaris''). Together wi ...
. These developments are using renewable resources rather than fossil fuel, although the issue of plastic source is currently negligible in the wake of plastic waste and in particular polyethylene waste as shown above.
Nomenclature and general description of the process
The name polyethylene comes from the ingredient and not the resulting chemical compound, which contains no double bonds. The scientific name ''polyethene'' is systematically derived from the scientific name of the monomer.polystyrene
Polystyrene (PS) is a synthetic polymer made from monomers of the aromatic hydrocarbon styrene. Polystyrene can be solid or foamed. General-purpose polystyrene is clear, hard, and brittle. It is an inexpensive resin per unit weight. It is a ...
are shortened to PP and PS, respectively. In the United Kingdom and India the polymer is commonly called ''polythene'', from the ICI trade name
A trade name, trading name, or business name, is a pseudonym used by companies that do not operate under their registered company name. The term for this type of alternative name is a "fictitious" business name. Registering the fictitious name ...
, although this is not recognized scientifically.
Footnotes
References
Bibliography
*
*
External links
Polythene's story: The accidental birth of plastic bags
Kawawada, Karen, ''Waterloo Region Record'' (22 May 2008).
{{Authority control
Polyolefins
Dielectrics
Ethylene
Thermoplastics
Packaging materials
Transparent materials
Bioplastics
Commodity chemicals
German inventions
1898 in science
1898 in Germany
 Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a  UHMWPE is polyethylene with a molecular weight numbering in the millions, usually between 3.5 and 7.5 million amu. The high molecular weight makes it a very
UHMWPE is polyethylene with a molecular weight numbering in the millions, usually between 3.5 and 7.5 million amu. The high molecular weight makes it a very  HDPE is defined by a density of greater or equal to 0.941 g/cm3. HDPE has a low degree of branching. The mostly linear molecules pack together well, so intermolecular forces are stronger than in highly branched polymers. HDPE can be produced by
HDPE is defined by a density of greater or equal to 0.941 g/cm3. HDPE has a low degree of branching. The mostly linear molecules pack together well, so intermolecular forces are stronger than in highly branched polymers. HDPE can be produced by 
 Polyethylene is produced from ethylene, and although ethylene can be produced from renewable resources, it is mainly obtained from
Polyethylene is produced from ethylene, and although ethylene can be produced from renewable resources, it is mainly obtained from