Phosphoenolpyruvate Carboxylase on:
[Wikipedia]
[Google]
[Amazon]
Phosphoenolpyruvate carboxylase (also known as PEP carboxylase, PEPCase, or PEPC; , PDB ID: 3ZGE) is an
 # The bicarbonate acts as a nucleophile to attack the
# The bicarbonate acts as a nucleophile to attack the
 PEP carboxylase is mainly subject to two levels of regulation:
PEP carboxylase is mainly subject to two levels of regulation:
enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. A ...
in the family of carboxy-lyases
Carboxy-lyases, also known as decarboxylases, are carbon–carbon lyases that add or remove a carboxyl group from organic compounds. These enzymes catalyze the decarboxylation of amino acids, beta-keto acids and alpha-keto acids.
Classification ...
found in plants and some bacteria that catalyzes the addition of bicarbonate
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogencarbonate) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula .
Bicarbonate serves a crucial biochem ...
(HCO3−) to phosphoenolpyruvate
Phosphoenolpyruvate (2-phosphoenolpyruvate, PEP) is the ester derived from the enol of pyruvate and phosphate. It exists as an anion. PEP is an important intermediate in biochemistry. It has the highest-energy phosphate bond found (−61.9 kJ/ ...
(PEP) to form the four-carbon compound oxaloacetate
Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes ...
and inorganic phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phospho ...
:
:PEP + HCO3− → oxaloacetate + Pi
This reaction is used for carbon fixation in CAM
Calmodulin (CaM) (an abbreviation for calcium-modulated protein) is a multifunctional intermediate calcium-binding messenger protein expressed in all eukaryotic cells. It is an intracellular target of the secondary messenger Ca2+, and the bin ...
(crassulacean acid metabolism) and organisms, as well as to regulate flux
Flux describes any effect that appears to pass or travel (whether it actually moves or not) through a surface or substance. Flux is a concept in applied mathematics and vector calculus which has many applications to physics. For transport ph ...
through the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protein ...
(also known as Krebs or TCA cycle) in bacteria and plants. The enzyme structure and its two step catalytic, irreversible mechanism have been well studied. PEP carboxylase is highly regulated, both by phosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
and allostery
In biochemistry, allosteric regulation (or allosteric control) is the regulation of an enzyme by binding an effector molecule at a site other than the enzyme's active site.
The site to which the effector binds is termed the ''allosteric sit ...
.
Enzyme structure
The PEP carboxylase enzyme is present in plants and some types of bacteria, but not in fungi or animals (including humans). The genes vary between organisms, but are strictly conserved around theactive
Active may refer to:
Music
* ''Active'' (album), a 1992 album by Casiopea
* Active Records, a record label
Ships
* ''Active'' (ship), several commercial ships by that name
* HMS ''Active'', the name of various ships of the British Royal ...
and allosteric sites discussed in the mechanism and regulation sections. Tertiary structure
Protein tertiary structure is the three dimensional shape of a protein. The tertiary structure will have a single polypeptide chain "backbone" with one or more protein secondary structures, the protein domains. Amino acid side chains may int ...
of the enzyme is also conserved.
The crystal structure of PEP carboxylase in multiple organisms, including ''Zea mays'' (maize), and ''Escherichia coli
''Escherichia coli'' (),Wells, J. C. (2000) Longman Pronunciation Dictionary. Harlow ngland Pearson Education Ltd. also known as ''E. coli'' (), is a Gram-negative, facultative anaerobic, rod-shaped, coliform bacterium of the genus ''Escher ...
'' has been determined. The overall enzyme exists as a dimer-of-dimers: two identical subunits closely interact to form a dimer through salt bridges between arginine (R438 - exact positions may vary depending on the origin of the gene) and glutamic acid (E433) residues. This dimer assembles (more loosely) with another of its kind to form the four subunit complex. The monomer subunits are mainly composed of alpha helices
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues ear ...
(65%), and have a mass of 106kDa each. The sequence length is about 966 amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha am ...
s.;
The enzyme active site is not completely characterized. It includes a conserved aspartic acid (D564) and a glutamic acid (E566) residue that non-covalently bind a divalent metal cofactor ion through the carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
functional groups. This metal ion can be magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ta ...
, manganese
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard, brittle, silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy use ...
or cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, pr ...
depending on the organism, and its role is to coordinate the phosphoenolpyruvate molecule as well as the reaction intermediates. A histidine
Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the d ...
(H138) residue at the active site is believed to facilitate proton transfer during the catalytic mechanism.
Enzyme mechanism
The mechanism of PEP carboxylase has been well studied. The enzymatic mechanism of formingoxaloacetate
Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes ...
is very exothermic and thereby irreversible; the biological Gibbs free energy
In thermodynamics, the Gibbs free energy (or Gibbs energy; symbol G) is a thermodynamic potential that can be used to calculate the maximum amount of work that may be performed by a thermodynamically closed system at constant temperature and ...
change (△G°’) is -30kJmol−1. The substrates and cofactor bind in the following order: metal cofactor (either Co2+, Mg2+, or Mn2+), PEP, bicarbonate
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogencarbonate) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula .
Bicarbonate serves a crucial biochem ...
(HCO3−). The mechanism proceeds in two major steps, as described below and shown in figure 2:
 # The bicarbonate acts as a nucleophile to attack the
# The bicarbonate acts as a nucleophile to attack the phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phospho ...
group in PEP. This results in the splitting of PEP into a carboxyphosphate and the (very reactive) enol
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The t ...
ate form of pyruvate.
# Proton transfer takes place at the carboxyphosphate. This is most likely modulated by a histidine
Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the d ...
(H138) residue that first deprotonates the carboxy side, and then, as an acid, protonates the phosphate part. The carboxyphosphate then exothermically decomposes into carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
and inorganic phosphate, at this point making this an irreversible reaction. Finally, after the decomposition, the carbon dioxide is attacked by the enolate to form oxaloacetate.
The metal cofactor is necessary to coordinate the enolate and carbon dioxide intermediates; the CO2 molecule is only lost 3% of the time. The active site is hydrophobic
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, t ...
to exclude water
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ...
, since the carboxyphosphate intermediate is susceptible to hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water ...
.
Function
The three most important roles that PEP carboxylase plays in plants and bacteria metabolism are in the cycle, the CAM cycle, and thecitric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protein ...
biosynthesis flux.
The primary mechanism of carbon dioxide assimilation in plants is through the enzyme ribulose-1,5-bisphosphate carboxylase/oxygenase (also known as RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
), that adds CO2 to ribulose-1,5-bisphosphate
Ribulose 1,5-bisphosphate (RuBP) is an organic substance that is involved in photosynthesis, notably as the principal acceptor in plants. It is a colourless anion, a double phosphate ester of the ketopentose (ketone-containing sugar with five car ...
(a 5 carbon sugar), to form two molecules of 3-phosphoglycerate (2x3 carbon sugars). However, at higher temperatures and lower CO2 concentrations, RuBisCO adds oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
instead of carbon dioxide, to form the unusable product glycolate
Glycolic acid (or hydroxyacetic acid; chemical formula HOCH2CO2H) is a colorless, odorless and hygroscopic crystalline solid, highly soluble in water. It is used in various skin-care products. Glycolic acid is widespread in nature. A glycolate (so ...
in a process called photorespiration
Photorespiration (also known as the oxidative photosynthetic carbon cycle or C2 cycle) refers to a process in plant metabolism where the enzyme RuBisCO oxygenates RuBP, wasting some of the energy produced by photosynthesis. The desired reaction ...
. To prevent this wasteful process, plants increase the local CO2 concentration in a process called the cycle. PEP carboxylase plays the key role of binding CO2 in the form of bicarbonate
In inorganic chemistry, bicarbonate (IUPAC-recommended nomenclature: hydrogencarbonate) is an intermediate form in the deprotonation of carbonic acid. It is a polyatomic anion with the chemical formula .
Bicarbonate serves a crucial biochem ...
with PEP to create oxaloacetate in the mesophyll tissue
A leaf ( : leaves) is any of the principal appendages of a vascular plant stem, usually borne laterally aboveground and specialized for photosynthesis. Leaves are collectively called foliage, as in "autumn foliage", while the leaves, ste ...
. This is then converted back to pyruvate (through a malate
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L ...
intermediate), to release the CO2 in the deeper layer of bundle sheath cells for carbon fixation by RuBisCO
Ribulose-1,5-bisphosphate carboxylase-oxygenase, commonly known by the abbreviations RuBisCo, rubisco, RuBPCase, or RuBPco, is an enzyme () involved in the first major step of carbon fixation, a process by which atmospheric carbon dioxide is con ...
and the Calvin cycle
The Calvin cycle, light-independent reactions, bio synthetic phase, dark reactions, or photosynthetic carbon reduction (PCR) cycle of photosynthesis is a series of chemical reactions that convert carbon dioxide and hydrogen-carrier compounds into ...
. Pyruvate is converted back to PEP in the mesophyll cells, and the cycle begins again, thus actively pumping CO2.
The second important and very similar biological significance of PEP carboxylase is in the CAM cycle. This cycle is common in organisms living in arid habitats. Plants cannot afford to open stomata during the day to take in CO2, as they would lose too much water by transpiration
Transpiration is the process of water movement through a plant and its evaporation from aerial parts, such as leaves, stems and flowers. Water is necessary for plants but only a small amount of water taken up by the roots is used for growth ...
. Instead, stomata open at night, when water evaporation is minimal, and take in CO2 by fixing with PEP to form oxaloacetate
Oxaloacetic acid (also known as oxalacetic acid or OAA) is a crystalline organic compound with the chemical formula HO2CC(O)CH2CO2H. Oxaloacetic acid, in the form of its conjugate base oxaloacetate, is a metabolic intermediate in many processes ...
though PEP carboxylase. Oxaloacetate is converted to malate
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L ...
by malate dehydrogenase
Malate dehydrogenase () (MDH) is an enzyme that reversibly catalyzes the oxidation of malate to oxaloacetate using the reduction of NAD+ to NADH. This reaction is part of many metabolic pathways, including the citric acid cycle. Other malate ...
, and stored for use during the day when the light dependent reaction generates energy (mainly in the form of ATP) and reducing equivalents
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
such as NADPH to run the Calvin cycle
The Calvin cycle, light-independent reactions, bio synthetic phase, dark reactions, or photosynthetic carbon reduction (PCR) cycle of photosynthesis is a series of chemical reactions that convert carbon dioxide and hydrogen-carrier compounds into ...
.
Third, PEP carboxylase is significant in non-photosynthetic metabolic pathways. Figure 3 shows this metabolic flow (and its regulation). Similar to pyruvate carboxylase
Pyruvate carboxylase (PC) encoded by the gene PC is an enzyme () of the ligase class that catalyzes (depending on the species) the physiologically irreversible carboxylation of pyruvate to form oxaloacetate (OAA).
Image:Pyruvic-acid-2D-sk ...
, PEP carboxylase replenishes oxaloacetate in the citric acid cycle. At the end of glycolysis, PEP is converted to pyruvate, which is converted to acetyl-coenzyme-A ( acetyl-CoA), which enters the citric acid cycle by reacting with oxaloacetate to form citrate
Citric acid is an organic compound with the chemical formula HOC(CO2H)(CH2CO2H)2. It is a colorless weak organic acid. It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the ...
. To increase flux through the cycle, some of the PEP is converted to oxaloacetate by PEP carboxylase. Since the citric acid cycle intermediates provide a hub for metabolism, increasing flux is important for the biosynthesis of many molecules, such as for example amino acids
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha am ...
.
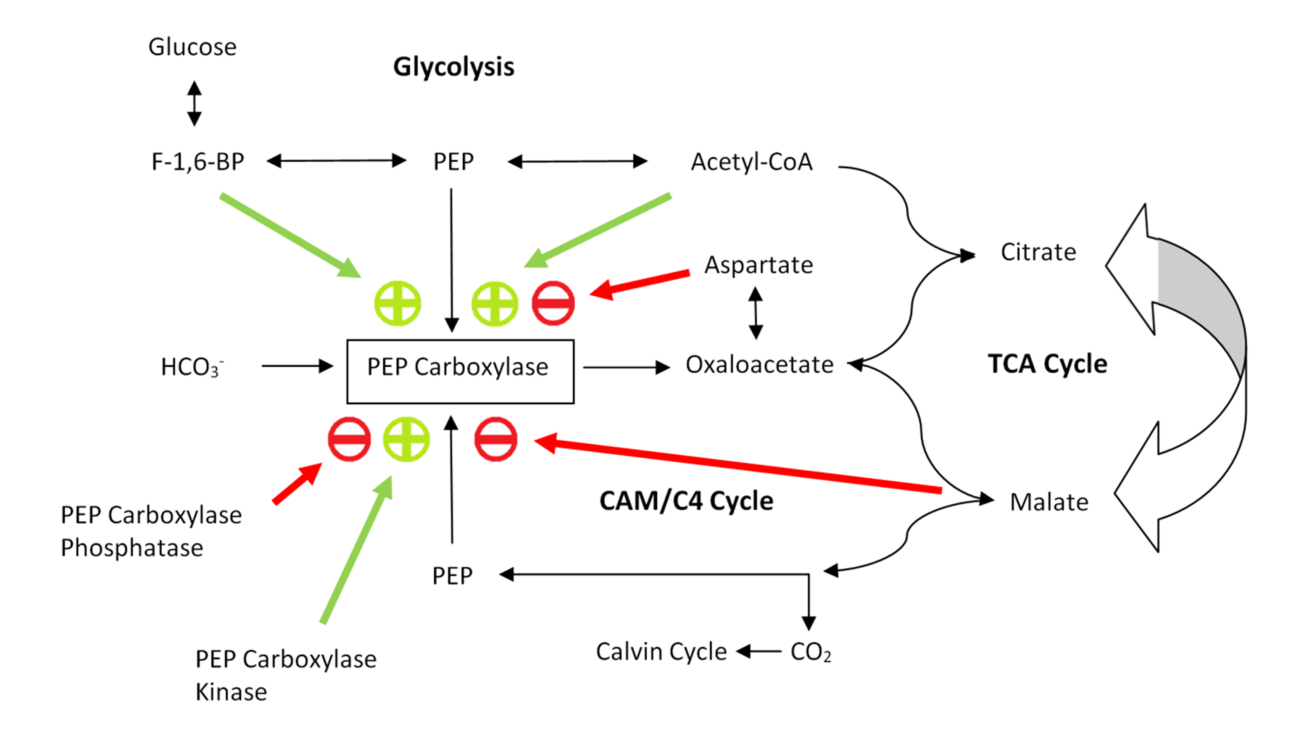
Regulation
 PEP carboxylase is mainly subject to two levels of regulation:
PEP carboxylase is mainly subject to two levels of regulation: phosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
and allostery
In biochemistry, allosteric regulation (or allosteric control) is the regulation of an enzyme by binding an effector molecule at a site other than the enzyme's active site.
The site to which the effector binds is termed the ''allosteric sit ...
. Figure 3 shows a schematic of the regulatory mechanism.
Phosphorylation
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, wh ...
by phosphoenolpyruvate carboxylase kinase turns the enzyme on, whereas phosphoenolpyruvate carboxylase phosphatase turns it back off. Both kinase and phosphate are regulated by transcription
Transcription refers to the process of converting sounds (voice, music etc.) into letters or musical notes, or producing a copy of something in another medium, including:
Genetics
* Transcription (biology), the copying of DNA into RNA, the fir ...
. It is further believed that malate
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L ...
acts as a feedback inhibitor of kinase expression levels, and as an activator for phosphatase expression (transcription). Since oxaloacetate is converted to malate in CAM and organisms, high concentrations of malate activate phosphatase expression - the phosphatase subsequently de-phosphorylates and thus de-actives PEP carboxylase, leading to no further accumulation of oxaloacetate and thus no further conversion of oxaloacetate to malate. Hence malate production is down-regulated.
The main allosteric inhibitors of PEP carboxylase are the carboxylic acids
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
malate
Malic acid is an organic compound with the molecular formula . It is a dicarboxylic acid that is made by all living organisms, contributes to the sour taste of fruits, and is used as a food additive. Malic acid has two stereoisomeric forms (L ...
(weak) and aspartate (strong). Since malate is formed in the next step of the CAM and cycles after PEP carboxylase catalyses the condensation of CO2 and PEP to oxaloacetate, this works as a feedback inhibition pathway. Oxaloacetate and aspartate are easily inter-convertible through a transaminase
Transaminases or aminotransferases are enzymes that catalyze a transamination reaction between an amino acid and an α- keto acid. They are important in the synthesis of amino acids, which form proteins.
Function and mechanism
An amino acid ...
mechanism; thus high concentrations of aspartate are also a pathway of feedback inhibition of PEP carboxylase.
The main allosteric activators of PEP carboxylase are acetyl-CoA and fructose-1,6-bisphosphate
Fructose 1,6-bisphosphate, also known as Harden-Young ester, is fructose sugar phosphorylated on carbons 1 and 6 (i.e., is a fructosephosphate). The β-D-form of this compound is common in cells. Upon entering the cell, most glucose and fructos ...
(F-1,6-BP). Both molecules are indicators of increased glycolysis levels, and thus positive feed-forward effectors of PEP carboxylase. They signal the need to produce oxaloacetate to allow more flux through the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protein ...
. Additionally, increased glycolysis means a higher supply of PEP is available, and thus more storage capacity for binding CO2 in transport to the Calvin cycle
The Calvin cycle, light-independent reactions, bio synthetic phase, dark reactions, or photosynthetic carbon reduction (PCR) cycle of photosynthesis is a series of chemical reactions that convert carbon dioxide and hydrogen-carrier compounds into ...
. It is also noteworthy that the negative effectors aspartate competes with the positive effector acetyl-CoA, suggesting that they share an allosteric binding site.
Studies have shown that energy equivalents such as AMP #REDIRECT Amp
{{Redirect category shell, {{R from other capitalisation{{R from ambiguous page ...
, ADP and ATP have no significant effect on PEP carboxylase.
The magnitudes of the allosteric effects of these different molecules on PEP carboxylase activity depend on individual organisms.
References
{{Portal bar, Biology, border=no EC 4.1.1 Photosynthesis