PCl5 on:
[Wikipedia]
[Google]
[Amazon]
Phosphorus pentachloride is the
 In solutions of polar solvents, PCl5 undergoes self-
In solutions of polar solvents, PCl5 undergoes self-
 It also converts alcohols to alkyl chlorides.
It also converts alcohols to alkyl chlorides.
The period 3 chlorides
{{Authority control Phosphorus chlorides Hypervalent molecules Phosphorus(V) compounds
chemical compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
with the formula PCl5. It is one of the most important phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ear ...
chlorides, others being PCl3 and POCl3. PCl5 finds use as a chlorinating reagent. It is a colourless, water-sensitive and moisture-sensitive solid
Solid is one of the State of matter#Four fundamental states, four fundamental states of matter (the others being liquid, gas, and Plasma (physics), plasma). The molecules in a solid are closely packed together and contain the least amount o ...
, although commercial samples can be yellowish and contaminated with hydrogen chloride
The compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride ga ...
.
Structure
The structures for the phosphorus chlorides are invariably consistent withVSEPR theory
Valence shell electron pair repulsion (VSEPR) theory ( , ), is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm the ...
. The structure of PCl5 depends on its environment. Gaseous and molten PCl5 is a neutral molecule with trigonal bipyramidal
In chemistry, a trigonal bipyramid formation is a molecular geometry with one atom at the center and 5 more atoms at the corners of a triangular bipyramid. This is one geometry for which the bond angles surrounding the central atom are not ident ...
geometry and (''D''3h) symmetry
Symmetry (from grc, συμμετρία "agreement in dimensions, due proportion, arrangement") in everyday language refers to a sense of harmonious and beautiful proportion and balance. In mathematics, "symmetry" has a more precise definit ...
. The hypervalent
In chemistry, a hypervalent molecule (the phenomenon is sometimes colloquially known as expanded Octet rule, octet) is a molecule that contains one or more main group elements apparently bearing more than eight electrons in their valence shells. P ...
nature of this species (as well as of , see below) can be explained with the inclusion of non-bonding molecular orbital
In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of findin ...
s (molecular orbital theory
In chemistry, molecular orbital theory (MO theory or MOT) is a method for describing the electronic structure of molecules using quantum mechanics. It was proposed early in the 20th century.
In molecular orbital theory, electrons in a molecule ...
) or resonance
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied periodic force (or a Fourier component of it) is equal or close to a natural frequency of the system on which it acts. When an oscillatin ...
(valence bond theory
In chemistry, valence bond (VB) theory is one of the two basic theories, along with molecular orbital (MO) theory, that were developed to use the methods of quantum mechanics to explain chemical bonding. It focuses on how the atomic orbitals of ...
). This trigonal bipyramidal structure persists in nonpolar solvents, such as CS2 and CCl4. In the solid state PCl5 is an ionic compound, formulated .
 In solutions of polar solvents, PCl5 undergoes self-
In solutions of polar solvents, PCl5 undergoes self-ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule i ...
. Dilute solutions dissociate according to the following equilibrium:
:PCl5 + Cl−
At higher concentrations, a second equilibrium becomes more prevalent:
:2 PCl5 +
The cation and the anion are tetrahedral
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners. The tetrahedron is the simplest of all the ...
and octahedral
In geometry, an octahedron (plural: octahedra, octahedrons) is a polyhedron with eight faces. The term is most commonly used to refer to the regular octahedron, a Platonic solid composed of eight equilateral triangles, four of which meet a ...
, respectively. At one time, PCl5 in solution was thought to form a dimeric structure, P2Cl10, but this suggestion is not supported by Raman spectroscopic measurements.
Related pentachlorides
AsCl5 and SbCl5 also adopt trigonal bipyramidal structures. The relevant bond distances are 211 pm (As−Cleq), 221 pm (As−Clax), 227 pm (Sb−Cleq), and 233.3 pm (Sb−Clax). At low temperatures, SbCl5 converts to the dimer, dioctahedral Sb2Cl10, structurally related toniobium pentachloride
Niobium(V) chloride, also known as niobium pentachloride, is a yellow crystalline solid. It hydrolyzes in air, and samples are often contaminated with small amounts of NbOCl3. It is often used as a precursor to other compounds of niobium. NbC ...
.
Preparation
PCl5 is prepared by thechlorination Chlorination may refer to:
* Chlorination reaction
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transform ...
of PCl3. This reaction is used to produce around 10,000 tonnes of PCl5 per year (as of 2000).
:PCl3 + Cl2 PCl5 (Δ''H'' = −124 kJ/mol)
PCl5 exists in equilibrium with PCl3 and chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate betwee ...
, and at 180 °C the degree of dissociation is about 40%. Because of this equilibrium, samples of PCl5 often contain chlorine, which imparts a greenish coloration.
Reactions
Hydrolysis
In its most characteristic reaction, PCl5 reacts upon contact withwater
Water (chemical formula ) is an inorganic, transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a ...
to release hydrogen chloride
The compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride ga ...
and give phosphorus oxides. The first hydrolysis product is phosphorus oxychloride
Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula . It hydrolyses in moist air releasing phosphoric acid and fumes of hydrogen chloride. It is manufactured industrially on a large scale from pho ...
:
:PCl5 + H2O → POCl3 + 2 HCl
In hot water, hydrolysis proceeds completely to orthophosphoric acid
Phosphoric acid (orthophosphoric acid, monophosphoric acid or phosphoric(V) acid) is a colorless, odorless phosphorus-containing solid, and inorganic compound with the chemical formula . It is commonly encountered as an 85% aqueous solution, w ...
:
:PCl5 + 4 H2O → H3PO4 + 5 HCl
Lewis acidity
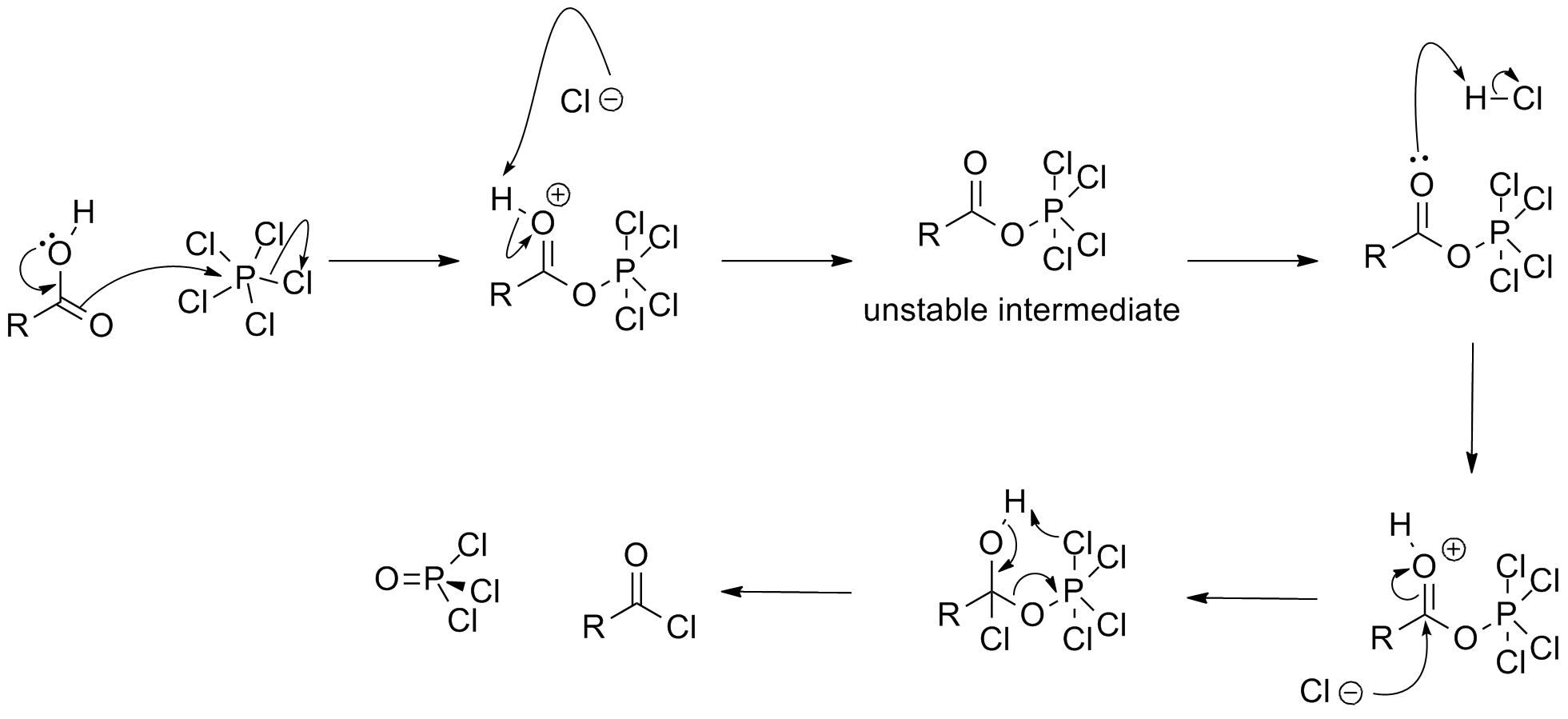
Phosphorus pentachloride is a Lewis acid. This property underpins many of its characteristic reactions, autoionization, chlorinations, hydrolysis. A well studied adduct is PCl5(pyridine).Chlorination of organic compounds
In synthetic chemistry, two classes of chlorination are usually of interest: oxidative chlorinations and substitutive chlorinations. Oxidative chlorinations entail the transfer of Cl2 from the reagent to the substrate. Substitutive chlorinations entail replacement of O or OH groups with chloride. PCl5 can be used for both processes. Upon treatment with PCl5,carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
s convert to the corresponding acyl chloride. The following mechanism has been proposed:
: It also converts alcohols to alkyl chlorides.
It also converts alcohols to alkyl chlorides. Thionyl chloride
Thionyl chloride is an inorganic compound with the chemical formula . It is a moderately volatile, colourless liquid with an unpleasant acrid odour. Thionyl chloride is primarily used as a chlorinating reagent, with approximately per year bein ...
is more commonly used in the laboratory because the resultant sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic activ ...
is more easily separated from the organic products than is POCl3.
PCl5 reacts with a tertiary amides, such as dimethylformamide
Dimethylformamide is an organic compound with the formula ( CH3)2NC(O)H. Commonly abbreviated as DMF (although this initialism is sometimes used for dimethylfuran, or dimethyl fumarate), this colourless liquid is miscible with water and the majo ...
(DMF), to give dimethylchloromethyleneammonium chloride, which is called the Vilsmeier reagent
The Vilsmeier reagent is an organic compound with the formula CH3)2NCHCll. It is a salt consisting of the N,N-dimethyliminium cation ( CH3)2N=CHClsup>+) and chloride anion. Depending on the particular reaction, the anion can vary. In typical POC ...
, CH3)2N=CClHl. More typically, a related salt is generated from the reaction of DMF and POCl3. Such reagents are useful in the preparation of derivatives of benzaldehyde by formylation and for the conversion of C−OH groups into C−Cl groups.
It is especially renowned for the conversion of C=O groups to CCl2 groups. For example, benzophenone
Benzophenone is the organic compound with the formula (C6H5)2CO, generally abbreviated Ph2CO. It is a white solid that is soluble in organic solvents. Benzophenone is a widely used building block in organic chemistry, being the parent diarylket ...
and phosphorus pentachloride react to give the diphenyldichloromethane
Diphenyldichloromethane is an organic compound with the formula (C6H5)2CCl2. It is a colorless solid that is used as a precursor to other organic compounds.
Synthesis
It is prepared from carbon tetrachloride and anhydrous aluminium chloride as c ...
:
:(C6H5)2CO + PCl5 → (C6H5)2CCl2 + POCl3
The electrophilic
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carri ...
character of PCl5 is highlighted by its reaction with styrene
Styrene () is an organic compound with the chemical formula C6H5CH=CH2. This derivative of benzene is a colorless oily liquid, although aged samples can appear yellowish. The compound evaporates easily and has a sweet smell, although high concen ...
to give, after hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water ...
, phosphonic acid derivatives.
Comparison with related reagents
Both PCl3 and PCl5 convert R3COH groups to the chloride R3CCl. The pentachloride is however a source of chlorine in many reactions. It chlorinates allylic andbenzylic
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure . Benzyl features a benzene ring () attached to a methylene group () group.
Nomenclature
In IUPAC nomenclature, the prefix benzyl refers to a subst ...
CH bonds. PCl5 bears a greater resemblance to SO2Cl2, also a source of Cl2. For oxidative chlorinations on the laboratory scale, sulfuryl chloride is often preferred over PCl5 since the gaseous SO2 by-product is readily separated.
Chlorination of inorganic compounds
As for the reactions with organic compounds, the use of PCl5 has been superseded by SO2Cl2. The reaction ofphosphorus pentoxide
Phosphorus pentoxide is a chemical compound with molecular formula P4 O10 (with its common name derived from its empirical formula, P2O5). This white crystalline solid is the anhydride of phosphoric acid. It is a powerful desiccant and dehydra ...
and PCl5 produces POCl3 :
:6 PCl5 + P4O10 → 10 POCl3
PCl5 chlorinates nitrogen dioxide
Nitrogen dioxide is a chemical compound with the formula . It is one of several nitrogen oxides. is an intermediate in the industrial synthesis of nitric acid, millions of tons of which are produced each year for use primarily in the producti ...
to form unstable nitryl chloride
Nitryl chloride is a volatile inorganic compound with formula ClNO2. At standard conditions it is a gas.
Formation
Nitryl chloride can be formed in the reaction of dinitrogen pentoxide with chlorides or hydrogen chloride:
:N2O5 + 2HCl → 2ClNO2 ...
:
:PCl5 + 2 NO2 → PCl3 + 2 NO2Cl
:2 NO2Cl → 2 NO2 + Cl2
PCl5 is a precursor for lithium hexafluorophosphate
Lithium hexafluorophosphate is an inorganic compound with the formula Li PF6. It is a white crystalline powder.
Production
LiPF6 is manufactured by reacting phosphorus pentachloride with hydrogen fluoride and lithium fluoride
:PCl5 + LiF + ...
, LiPF6. Lithium hexafluorophosphate is a commonly employed salt in electrolyte
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dis ...
s in lithium ion batteries
A lithium-ion or Li-ion battery is a type of rechargeable battery which uses the reversible reduction of lithium ions to store energy. It is the predominant battery type used in portable consumer electronics and electric vehicles. It also sees ...
. is produced by the reaction of with lithium fluoride
Lithium fluoride is an inorganic compound with the chemical formula LiF. It is a colorless solid, that transitions to white with decreasing crystal size. Although odorless, lithium fluoride has a bitter-saline taste. Its structure is analogous to ...
, with lithium chloride
Lithium chloride is a chemical compound with the formula Li Cl. The salt is a typical ionic compound (with certain covalent characteristics), although the small size of the Li+ ion gives rise to properties not seen for other alkali metal chlor ...
as a side product:
:PCl5 + 6 LiF → LiPF6 + 5 LiCl
Safety
PCl5 is a dangerous substance as it reacts violently with water. It is also corrosive when in contact with skin and can be fatal when inhaled.History
Phosphorus pentachloride was first prepared in 1808 by the English chemistHumphry Davy
Sir Humphry Davy, 1st Baronet, (17 December 177829 May 1829) was a British chemist and inventor who invented the Davy lamp and a very early form of arc lamp. He is also remembered for isolating, by using electricity, several elements for t ...
. Davy's analysis of phosphorus pentachloride was inaccurate; the first accurate analysis was provided in 1816 by the French chemist Pierre Louis Dulong
Pierre Louis Dulong FRS FRSE (; ; 12 February 1785 – 19 July 1838) was a French physicist and chemist. He is remembered today largely for the law of Dulong and Petit, although he was much-lauded by his contemporaries for his studies into ...
. On p. 148, Dulong presented the correct analysis of phosphorus pentachloride (which is 14.9% phosphorus and 85.1% chlorine by weight, vs. Dulong's values of 15.4% and 84.6%, respectively).
See also
*Phosphorus halides
In chemistry, there are three series of binary phosphorus halides, containing phosphorus in the oxidation states +5, +3 and +2. All compounds have been described, in varying degrees of detail, although serious doubts have been cast on the existenc ...
* Phosphorus trichloride
Phosphorus trichloride is an inorganic compound with the chemical formula PCl3. A colorless liquid when pure, it is an important industrial chemical, being used for the manufacture of phosphites and other organophosphorus compounds. It is toxi ...
* Phosphoryl chloride
Phosphoryl chloride (commonly called phosphorus oxychloride) is a colourless liquid with the formula . It hydrolyses in moist air releasing phosphoric acid and fumes of hydrogen chloride. It is manufactured industrially on a large scale from phosp ...
* Phosphorus trifluorodichloride
Phosphorus trifluorodichloride is a chemical compound with the chemical formula PF3Cl2. The covalent molecule trigonal bipyramidal molecular geometry. The central phosphorus atom has sp3d hybridization, and the molecule has an asymmetric charge d ...
References
External links
The period 3 chlorides
{{Authority control Phosphorus chlorides Hypervalent molecules Phosphorus(V) compounds