|
Hypervalent
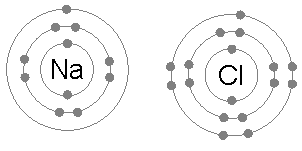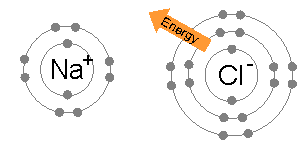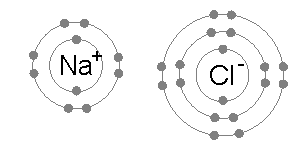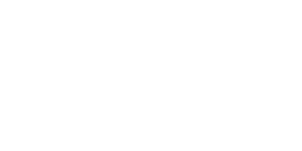
In chemistry, a hypervalent molecule (the phenomenon is sometimes colloquially known as expanded Octet rule, octet) is a molecule that contains one or more main group elements apparently bearing more than eight electrons in their valence shells. Phosphorus pentachloride (), sulfur hexafluoride (), chlorine trifluoride (), the chlorite () ion, and the triiodide () ion are examples of hypervalent molecules. Definitions and nomenclature Hypervalent molecules were first formally defined by Jeremy I. Musher in 1969 as molecules having central atoms of group 15–18 in any valence (chemistry), valence other than the lowest (i.e. 3, 2, 1, 0 for Groups 15, 16, 17, 18 respectively, based on the octet rule). Several specific classes of hypervalent molecules exist: * Hypervalent iodine compounds are useful reagents in organic chemistry (e.g. Dess–Martin periodinane) * Tetra-, penta- and hexavalent phosphorus, silicon, and sulfur compounds (e.g. PCl5, PF5, SF6, sulfuranes and persulfuranes) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hypervalent Iodine
Iodane generally refers to any organic derivative of iodine. Without modifier, ''iodane'' is the systematic name for the parent hydride of iodine, HI. Thus, any organoiodine compound with general formula RI (e.g., iodomethane , or iodobenzene ) is a substituted iodane. However, as used in the context of organic synthesis, the term ''iodane'' more specifically refers to organoiodine compounds with nonstandard bond order of bonds between iodine and other atoms, i.e., bond order of iodine greater than 1, making this term a synonym for hypervalent iodine. These iodine compounds are hypervalent because the iodine atom formally contains more than the 8 electrons in the valence shell required for the octet rule. When iodine is ligated to an organic residue and electronegative ligands (e.g. halides or carboxylates), hypervalent iodine occurs in a +3 oxidation state as iodine(III) or λ3-iodane, or in a +5 oxidation state as iodine(V) or λ5-iodane, or in a +7 oxidation state as iodine(V ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Three-center Four-electron Bond
The 3-center 4-electron (3c–4e) bond is a model used to explain bonding in certain hypervalent molecules such as tetratomic and hexatomic interhalogen compounds, sulfur tetrafluoride, the xenon fluorides, and the bifluoride ion. It is also known as the Pimentel–Rundle three-center model after the work published by George C. Pimentel in 1951,Pimentel, G. C. The Bonding of Trihalide and Bifluoride Ions by the Molecular Orbital Method. ''J. Chem. Phys.'' 1951, ''19'', 446-448. which built on concepts developed earlier by Robert E. Rundle for electron-deficient bonding.Rundle, R. E. Electron Deficient Compounds. II. Relative Energies of "Half-Bonds". ''J. Chem. Phys.'' 1949, ''17'', 671–675. An extended version of this model is used to describe the whole class of hypervalent molecules such as phosphorus pentafluoride and sulfur hexafluoride as well as multi-center π-bonding such as ozone and sulfur trioxide. There are also molecules such as diborane (B2H6) and dialane (Al2H6) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anthony Joseph Arduengo III
Anthony Joseph Arduengo III is Professor of the Practice at the Georgia Institute of Technology, Saxon Professor Emeritus of Chemistry at the University of Alabama, adjunct professor at the Institute for Inorganic Chemistry of Braunschweig University of Technology in Germany, and co-founder of the ''StanCE'' coalition for sustainable chemistry based on woody biomassXylochemistry. He is notable for his work on chemical compounds with unusual valency, especially in the field of stable carbene research. Early life Anthony "Bo" Arduengo was born in 1952 in Tampa, Florida. He grew up in the Atlanta, Georgia area. His father was a pressman and mechanic with the Atlanta Journal-Constitution and instilled his son with an interest and skill for all things mechanical and scientific. By the age of 16, he and his father had built his first car from miscellaneous parts. The car was registered as street-legal and road-worthy. With some re-engineering, the car was later fitted to run on al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octet Rule
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens; although more generally the rule is applicable for the s-block and p-block of the periodic table. Other rules exist for other elements, such as the duplet rule for hydrogen and helium, or the 18-electron rule for transition metals. The valence electrons can be counted using a Lewis electron dot diagram as shown at the right for carbon dioxide. The electrons shared by the two atoms in a covalent bond are counted twice, once for each atom. In carbon dioxide each oxygen shares four electrons with the central carbon, two (shown in red) from the oxygen itself and two (shown in black) from the carbon. All four of these electrons are counted in both the carbon octet and the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octet Rule
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens; although more generally the rule is applicable for the s-block and p-block of the periodic table. Other rules exist for other elements, such as the duplet rule for hydrogen and helium, or the 18-electron rule for transition metals. The valence electrons can be counted using a Lewis electron dot diagram as shown at the right for carbon dioxide. The electrons shared by the two atoms in a covalent bond are counted twice, once for each atom. In carbon dioxide each oxygen shares four electrons with the central carbon, two (shown in red) from the oxygen itself and two (shown in black) from the carbon. All four of these electrons are counted in both the carbon octet and the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Valence (chemistry)
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules. Description The combining capacity, or affinity of an atom of a given element is determined by the number of hydrogen atoms that it combines with. In methane, carbon has a valence of 4; in ammonia, nitrogen has a valence of 3; in water, oxygen has a valence of 2; and in hydrogen chloride, chlorine has a valence of 1. Chlorine, as it has a valence of one, can be substituted for hydrogen. Phosphorus has a valence of 5 in phosphorus pentachloride, . Valence diagrams of a compound represent the connectivity of the elements, with lines drawn between two elements, sometimes called bonds, representing a saturated valency for each element. The two tables below show some examples of different compounds, their valence diagrams, and the valences for each element of the compound. Modern definitions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Pentachloride
Phosphorus pentachloride is the chemical compound with the formula PCl5. It is one of the most important phosphorus chlorides, others being PCl3 and POCl3. PCl5 finds use as a chlorinating reagent. It is a colourless, water-sensitive and moisture-sensitive solid, although commercial samples can be yellowish and contaminated with hydrogen chloride. Structure The structures for the phosphorus chlorides are invariably consistent with VSEPR theory. The structure of PCl5 depends on its environment. Gaseous and molten PCl5 is a neutral molecule with trigonal bipyramidal geometry and (''D''3h) symmetry. The hypervalent nature of this species (as well as of , see below) can be explained with the inclusion of non-bonding molecular orbitals (molecular orbital theory) or resonance (valence bond theory). This trigonal bipyramidal structure persists in nonpolar solvents, such as CS2 and CCl4. In the solid state PCl5 is an ionic compound, formulated . In solutions of polar solvents, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
James Cullen Martin
James Cullen Martin (January 14, 1928 – April 20, 1999) was an American chemist. Known in the field as "J.C.", he specialized in physical organic chemistry with an emphasis on main group element chemistry. Martin received his undergraduate and master's degree at Vanderbilt University. His PhD work was conducted with Paul Bartlett at Harvard. Most of his professional career was at the University of Illinois at Urbana-Champaign, where he was a colleague of Roger Adams, Speed Marvel, David Y. Curtin, Nelson J. Leonard, and Reynold C. Fuson. Late in his career, he moved back to Vanderbilt, but soon succumbed to poor health. Professor Martin is best known for his work on bonding of main group elements. He is responsible for the hexafluorocumyl alcohol derived "Martin" bidentate ligand and a tridentate analog. With his doctoral student Daniel Benjamin Dess, he invented the Dess–Martin periodinane that is used for selective oxidation of alcohols. He is also known for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Persulfuranes
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two ( cysteine and methionine) are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries. Sulfur shares the chalcogen group with oxygen, selenium, and tellurium, and it is expected that organosulfur compounds have similarities with carbon–oxygen, carbon–selenium, and carbon–tellurium compounds. A classical chemical test for the detection of sulfur compou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfuranes
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two (cysteine and methionine) are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries. Sulfur shares the chalcogen group with oxygen, selenium, and tellurium, and it is expected that organosulfur compounds have similarities with carbon–oxygen, carbon–selenium, and carbon–tellurium compounds. A classical chemical test for the detection of sulfur compound ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triiodide
In chemistry, triiodide usually refers to the triiodide ion, . This anion, one of the polyhalogen ions, is composed of three iodine atoms. It is formed by combining aqueous solutions of iodide salts and iodine. Some salts of the anion have been isolated, including thallium(I) triiodide (Tl+ 3sup>−) and ammonium triiodide ( H4sup>+ 3sup>−). Triiodide is observed to be a red colour in solution. Nomenclature Other chemical compounds with "triiodide" in their name may contain three iodide centers that are not bonded to each other as the triiodide ion, but exist instead as separate iodine atoms or iodide ions. Examples include nitrogen triiodide (NI3) and phosphorus triiodide (PI3), where individual iodine atoms are covalently bonded to a central atom. As some cations have the theoretical possibility to form compounds with both triiodide and iodide ions, such as ammonium, compounds containing iodide anions in a 3:1 stoichiometric ratio should only be referred to as triiodides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Trifluoride
Chlorine trifluoride is an interhalogen compound with the formula ClF3. This colorless, poisonous, corrosive, and extremely reactive gas condenses to a pale-greenish yellow liquid, the form in which it is most often sold (pressurized at room temperature). The compound is primarily of interest in plasmaless cleaning and etching operations in the semiconductor industry, in nuclear reactor fuel processing, as a component in rocket fuels, and other industrial operations. Preparation, structure, and properties It was first reported in 1930 by Ruff and Krug who prepared it by fluorination of chlorine; this also produced ClF (chlorine monofluoride) and the mixture was separated by distillation. :3 F2 + Cl2 → 2 ClF3 The molecular geometry of ClF3 is approximately T-shaped, with one short bond (1.598 Å) and two long bonds (1.698 Å). This structure agrees with the prediction of VSEPR theory, which predicts lone pairs of electrons as occupying two equatorial positions o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |