Ethyl Glyoxylate on:
[Wikipedia]
[Google]
[Amazon]
Glyoxylic acid or oxoacetic acid is an
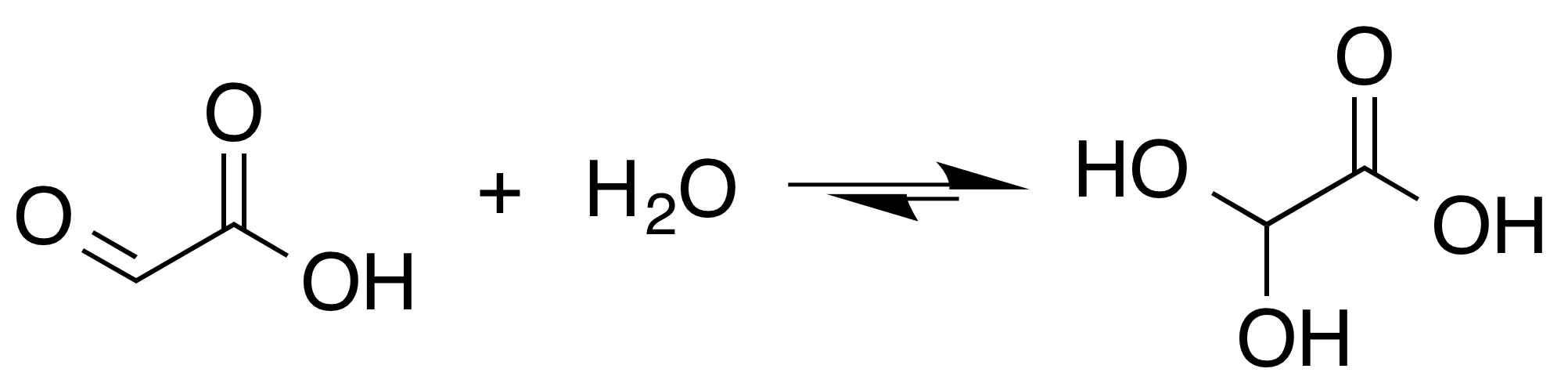 In solution, the monohydrate exists in equilibrium with a hemi
In solution, the monohydrate exists in equilibrium with a hemi In isolation, the aldehyde structure has as a major
In isolation, the aldehyde structure has as a major 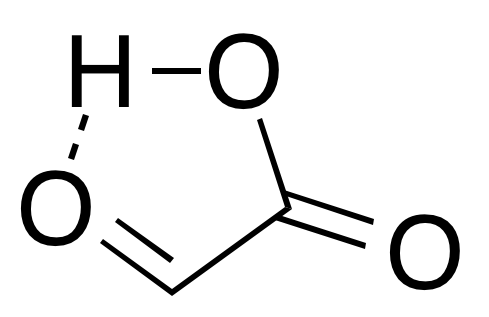 The
The


organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. T ...
. Together with acetic acid, glycolic acid
Glycolic acid (or hydroxyacetic acid; chemical formula HOCH2CO2H) is a colorless, odorless and hygroscopic crystalline solid, highly soluble in water. It is used in various skin-care products. Glycolic acid is widespread in nature. A glycolate (s ...
, and oxalic acid, glyoxylic acid is one of the C2 carboxylic acids. It is a colourless solid that occurs naturally and is useful industrially.
Structure and nomenclature
Although the structure of glyoxylic acid is described as having analdehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
, the aldehyde is only a minor component of the form most prevalent in some situations. Instead, it often exists as a hydrate or a cyclic dimer
Dimer may refer to:
* Dimer (chemistry), a chemical structure formed from two similar sub-units
** Protein dimer, a protein quaternary structure
** d-dimer
* Dimer model, an item in statistical mechanics, based on ''domino tiling''
* Julius Dimer ...
. For example, in the presence of water, the carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
rapidly converts to a geminal diol
In chemistry, the descriptor geminal () refers to the relationship between two atoms or functional groups that are attached to the same atom. A geminal diol, for example, is a diol (a molecule that has two alcohol functional groups) attached t ...
(described as the "monohydrate"). The equilibrium constant
The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical equilibrium, a state approached by a dynamic chemical system after sufficient time has elapsed at which its composition has no measurable tendency ...
(''K'') is 300 for the formation of dihydroxyacetic acid at room temperature:
: In solution, the monohydrate exists in equilibrium with a hemi
In solution, the monohydrate exists in equilibrium with a hemiacylal
Acylals in organic chemistry are a group of chemical compounds sharing a functional group with the general structure RCH(OOCR)2. Acylals are obtained by reaction of carbonyls with acetic anhydride or other acid anhydrides and a suitable catalyst, f ...
dimer form:Georges Mattioda and Yani Christidis “Glyoxylic Acid” Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim.
: In isolation, the aldehyde structure has as a major
In isolation, the aldehyde structure has as a major conformer
Conformer is a clear acrylic shell fitted after an enucleation if the final artificial eye is not available at the time of surgery to hold the shape of the eye socket and allow the eyelids to blink over the shell without rubbing the suture line.
...
a cyclic hydrogen-bonded structure with the aldehyde carbonyl in close proximity to the carboxyl
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
hydrogen:
: The
The Henry's law
In physical chemistry, Henry's law is a gas law that states that the amount of dissolved gas in a liquid is directly proportional to its partial pressure above the liquid. The proportionality factor is called Henry's law constant. It was formulate ...
constant of glyoxylic acid is KH = 1.09 × 104 × exp 40.0 × 103/R) × (1/T − 1/298)
Preparations
Theconjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid donates a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as in the reverse reaction it loses a ...
of glyoxylic acid is known as glyoxylate and is the form that the compound exists in solution at neutral pH. Glyoxylate is the byproduct of the amidation
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is p ...
process in biosynthesis of several amidated peptide
Peptides (, ) are short chains of amino acids linked by peptide bonds. Long chains of amino acids are called proteins. Chains of fewer than twenty amino acids are called oligopeptides, and include dipeptides, tripeptides, and tetrapeptides.
...
s.
For the historical record, glyoxylic acid was prepared from oxalic acid electrosynthetically: in organic synthesis, lead dioxide
Lead(IV) oxide is the inorganic compound with the formula PbO2. It is an oxide where lead is in an oxidation state of +4. It is a dark-brown solid which is insoluble in water. It exists in two crystalline forms. It has several important applicatio ...
cathodes were applied for preparing glyoxylic acid from oxalic acid in a sulfuric acid electrolyte.
: 380px
Hot nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
can oxidize
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a d ...
glyoxal
Glyoxal is an organic compound with the chemical formula OCHCHO. It is the smallest dialdehyde (a compound with two aldehyde groups). It is a crystalline solid, white at low temperatures and yellow near the melting point (15 °C). The liquid ...
to glyoxylic; however this reaction is highly exothermic and prone to thermal runaway. In addition, oxalic acid is the main side product.
Also, ozonolysis
In organic chemistry, ozonolysis is an organic reaction where the unsaturated bonds of alkenes (), alkynes (), or azo compounds () are cleaved with ozone (). Alkenes and alkynes form organic compounds in which the multiple carbon–carbon b ...
of maleic acid
Maleic acid or ''cis''-butenedioic acid is an organic compound that is a dicarboxylic acid, a molecule with two carboxyl groups. Its chemical formula is HO2CCH=CHCO2H. Maleic acid is the ''cis''-isomer of butenedioic acid, whereas fumaric ac ...
is effective.
Biological role
Glyoxylate is an intermediate of theglyoxylate cycle
The glyoxylate cycle, a variation of the tricarboxylic acid cycle, is an anabolic pathway occurring in plants, bacteria, protists, and fungi. The glyoxylate cycle centers on the conversion of acetyl-CoA to succinate for the synthesis of carbohydrat ...
, which enables organism
In biology, an organism () is any living system that functions as an individual entity. All organisms are composed of cells (cell theory). Organisms are classified by taxonomy into groups such as multicellular animals, plants, and ...
s, such as bacteria, fungi, and plants to convert fatty acid
In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, ...
s into carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or m ...
s. The glyoxylate cycle is also important for induction of plant defense mechanisms in response to fungi. The glyoxylate cycle is initiated through the activity of isocitrate lyase, which converts isocitrate into glyoxylate and succinate. Research is being done to co-opt the pathway for a variety of uses such as the biosynthesis of succinate.
In humans
Glyoxylate is produced via two pathways: through the oxidation of glycolate in peroxisomes or through the catabolism of hydroxyproline in mitochondria. In the peroxisomes, glyoxylate is converted into glycine by AGT1 or into oxalate by glycolate oxidase. In the mitochondria, glyoxylate is converted into glycine by AGT2 or into glycolate by glyoxylate reductase. A small amount of glyoxylate is converted into oxalate by cytoplasmic lactate dehydrogenase.
In plants
In addition to being an intermediate in theglyoxylate cycle
The glyoxylate cycle, a variation of the tricarboxylic acid cycle, is an anabolic pathway occurring in plants, bacteria, protists, and fungi. The glyoxylate cycle centers on the conversion of acetyl-CoA to succinate for the synthesis of carbohydrat ...
, glyoxylate is also an important intermediate in the photorespiration
Photorespiration (also known as the oxidative photosynthetic carbon cycle or C2 cycle) refers to a process in plant metabolism where the enzyme RuBisCO oxygenates RuBP, wasting some of the energy produced by photosynthesis. The desired reaction ...
pathway. Photorespiration is a result of the side reaction of RuBisCO with O2 instead of CO2. While at first considered a waste of energy and resources, photorespiration has been shown to be an important method of regenerating carbon and CO2, removing toxic phosphoglycolate, and initiating defense mechanisms. In photorespiration, glyoxylate is converted from glycolate through the activity of glycolate oxidase in the peroxisome. It is then converted into glycine through parallel actions by SGAT and GGAT, which is then transported into the mitochondria. It has also been reported that the pyruvate dehydrogenase complex may play a role in glycolate and glyoxylate metabolism.

Disease relevance
Diabetes
Glyoxylate is thought to be a potential early marker forType II diabetes
Type 2 diabetes, formerly known as adult-onset diabetes, is a form of diabetes mellitus that is characterized by high blood sugar, insulin resistance, and relative lack of insulin. Common symptoms include increased thirst, frequent urinatio ...
. One of the key conditions of diabetes pathology is the production of advanced glycation end-product
Advanced glycation end products (AGEs) are proteins or lipids that become glycated as a result of exposure to sugars. They are a bio-marker implicated in aging and the development, or worsening, of many degenerative diseases, such as diabetes, ath ...
s (AGEs) caused by the hyperglycemia. AGEs can lead to further complications of diabetes, such as tissue damage and cardiovascular disease. They are generally formed from reactive aldehydes, such as those present on reducing sugars and alpha-oxoaldehydes. In a study, glyoxylate levels were found to be significantly increased in patients who were later diagnosed with Type II diabetes. The elevated levels were found sometimes up to three years before the diagnosis, demonstrating the potential role for glyoxylate to be an early predictive marker.
Nephrolithiasis
Glyoxylate is involved in the development ofhyperoxaluria
Hyperoxaluria is an excessive urinary excretion of oxalate. Individuals with hyperoxaluria often have calcium oxalate kidney stones. It is sometimes called Bird's disease, after Golding Bird, who first described the condition.
Causes
Hyperoxalur ...
, a key cause of nephrolithiasis
Kidney stone disease, also known as nephrolithiasis or urolithiasis, is a crystallopathy where a solid piece of material (kidney stone) develops in the urinary tract. Kidney stones typically form in the kidney and leave the body in the urine s ...
(commonly known as kidney stones). Glyoxylate is both a substrate and inductor of sulfate anion transporter-1 (sat-1), a gene responsible for oxalate transportation, allowing it to increase sat-1 mRNA expression and as a result oxalate efflux from the cell. The increased oxalate release allows the buildup of calcium oxalate in the urine, and thus the eventual formation of kidney stones.
The disruption of glyoxylate metabolism provides an additional mechanism of hyperoxaluria development. Loss of function mutations in the HOGA1 gene leads to a loss of the 4-hydroxy-2-oxoglutarate aldolase, an enzyme in the hydroxyproline to glyoxylate pathway. The glyoxylate resulting from this pathway is normally stored away to prevent oxidation to oxalate in the cytosol. The disrupted pathway, however, causes a buildup of 4-hydroxy-2-oxoglutarate which can also be transported to the cytosol and converted into glyoxylate through a different aldolase. These glyoxylate molecules can be oxidized into oxalate increasing its concentration and causing hyperoxaluria.
Reactions and uses
Glyoxylic acid is about ten times stronger an acid than acetic acid, with an acid dissociation constant of 4.7 × 10−4 (p''K''a = 3.32): :OCHCO2H + H+ With base, glyoxylic acid disproportionates, forming hydroxyacetic acid and oxalic acid: :2 OCHCO2H + H2O → HOCH2CO2H + HO2CCO2H Glyoxylic acid gives heterocycles upon condensation withurea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
and 1,2-diaminobenzene.
Phenol derivatives
In general, glyoxylic acid undergoes anelectrophilic aromatic substitution
Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an aromatic system (usually hydrogen) is replaced by an electrophile. Some of the most important electrophilic aromatic substitutions are aromatic n ...
reaction with phenols
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of one or more hydroxyl groups (— O H) bonded directly to an aromatic hydrocarbon group. The simplest is phenol, . Phenolic compounds are ...
, a versatile step in the synthesis of several other compounds.
The immediate product with phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it ...
itself is 4-hydroxymandelic acid. This species reacts with ammonia to give hydroxyphenylglycine, a precursor to the drug amoxicillin
Amoxicillin is an antibiotic medication used to treat a number of bacterial infections. These include middle ear infection, strep throat, pneumonia, skin infections, and urinary tract infections among others. It is taken by mouth, or less c ...
. Reduction of the 4-hydroxymandelic acid gives 4-hydroxyphenylacetic acid
4-Hydroxyphenylacetic acid is a chemical compound found in olive oil and beer.
Synthesis
4-Hydroxyphenylacetic acid is obtained by reducing 4-hydroxymandelic acid with elemental phosphorus and iodine.
Uses
In industry, 4-hydroxyphenylacetic ac ...
, a precursor to the drug atenolol
Atenolol is a beta blocker medication primarily used to treat high blood pressure and heart-associated chest pain. Atenolol, however, does not seem to improve mortality in those with high blood pressure. Other uses include the prevention of mi ...
.
The sequence of reactions, in which glyoxylic acid reacts with guaiacol
Guaiacol () is an organic compound with the formula C6H4(OH)(OCH3). It is a phenolic compound containing an methoxy functional group. Guaiacol appears as a viscous colorless oil, although aged or impure samples are often yellowish. It occurs wid ...
the phenolic component followed by oxidation and decarboxylation, provides a route to vanillin
Vanillin is an organic compound with the molecular formula . It is a phenolic aldehyde. Its functional groups include aldehyde, hydroxyl, and ether. It is the primary component of the extract of the vanilla bean. Synthetic vanillin is now u ...
as a net formylation
In biochemistry, the addition of a formyl functional group is termed formylation. A formyl functional group consists of a carbonyl bonded to hydrogen. When attached to an R group, a formyl group is called an aldehyde.
Formylation has been ident ...
process.
Hopkins Cole reaction
Glyoxylic acid is a component of the Hopkins–Cole reaction, used to check for the presence oftryptophan
Tryptophan (symbol Trp or W)
is an α-amino acid that is used in the biosynthesis of proteins. Tryptophan contains an α-amino group, an α-carboxylic acid group, and a side chain indole, making it a polar molecule with a non-polar aromatic ...
in proteins.
Environmental chemistry
Glyoxylic acid is one of several ketone- and aldehyde-containing carboxylic acids that together are abundant in secondary organic aerosols. In the presence of water and sunlight, glyoxylic acid can undergophotochemical
Photochemistry is the branch of chemistry concerned with the chemical effects of light. Generally, this term is used to describe a chemical reaction caused by absorption of ultraviolet (wavelength from 100 to 400 nm), visible light (400–7 ...
oxidation. Several different reaction pathways can ensue, leading to various other carboxylic acid and aldehyde products.
Safety
The compound is not very toxic with an for rats of 2500 mg/kg.References