|
Oxalic Acid
Oxalic acid is an organic acid with the systematic name ethanedioic acid and formula . It is the simplest dicarboxylic acid. It is a white crystalline solid that forms a colorless solution in water. Its name comes from the fact that early investigators isolated oxalic acid from flowering plants of the genus ''Oxalis'', commonly known as wood-sorrels. It occurs naturally in many foods. Excessive ingestion of oxalic acid or prolonged skin contact can be dangerous. Oxalic acid has much greater acid strength than acetic acid. It is a reducing agent and its conjugate base, known as oxalate (), is a chelating agent for metal cations. Typically, oxalic acid occurs as the dihydrate with the formula . History The preparation of salts of oxalic acid (crab acid) from plants had been known, at least since 1745, when the Dutch botanist and physician Herman Boerhaave isolated a salt from wood sorrel. By 1773, François Pierre Savary of Fribourg, Switzerland had isolated oxalic acid from i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society (professional association) in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 34,000 in the UK and a further 8,000 abroad. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxalate
Oxalate (IUPAC: ethanedioate) is an anion with the formula C2O42−. This dianion is colorless. It occurs naturally, including in some foods. It forms a variety of salts, for example sodium oxalate (Na2C2O4), and several esters such as dimethyl oxalate (C2O4(CH3)2). It is a conjugate base of oxalic acid. At neutral pH in aqueous solution, oxalic acid converts completely to oxalate. Relationship to oxalic acid The dissociation of protons from oxalic acid proceeds in a stepwise manner; as for other polyprotic acids, loss of a single proton results in the monovalent hydrogenoxalate anion . A salt with this anion is sometimes called an acid oxalate, monobasic oxalate, or hydrogen oxalate. The equilibrium constant ( ''K''a) for loss of the first proton is (p''K''a = 1.27). The loss of the second proton, which yields the oxalate ion, has an equilibrium constant of (p''K''a = 4.28). These values imply, in solutions with neutral pH, no oxalic acid and only trace am ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
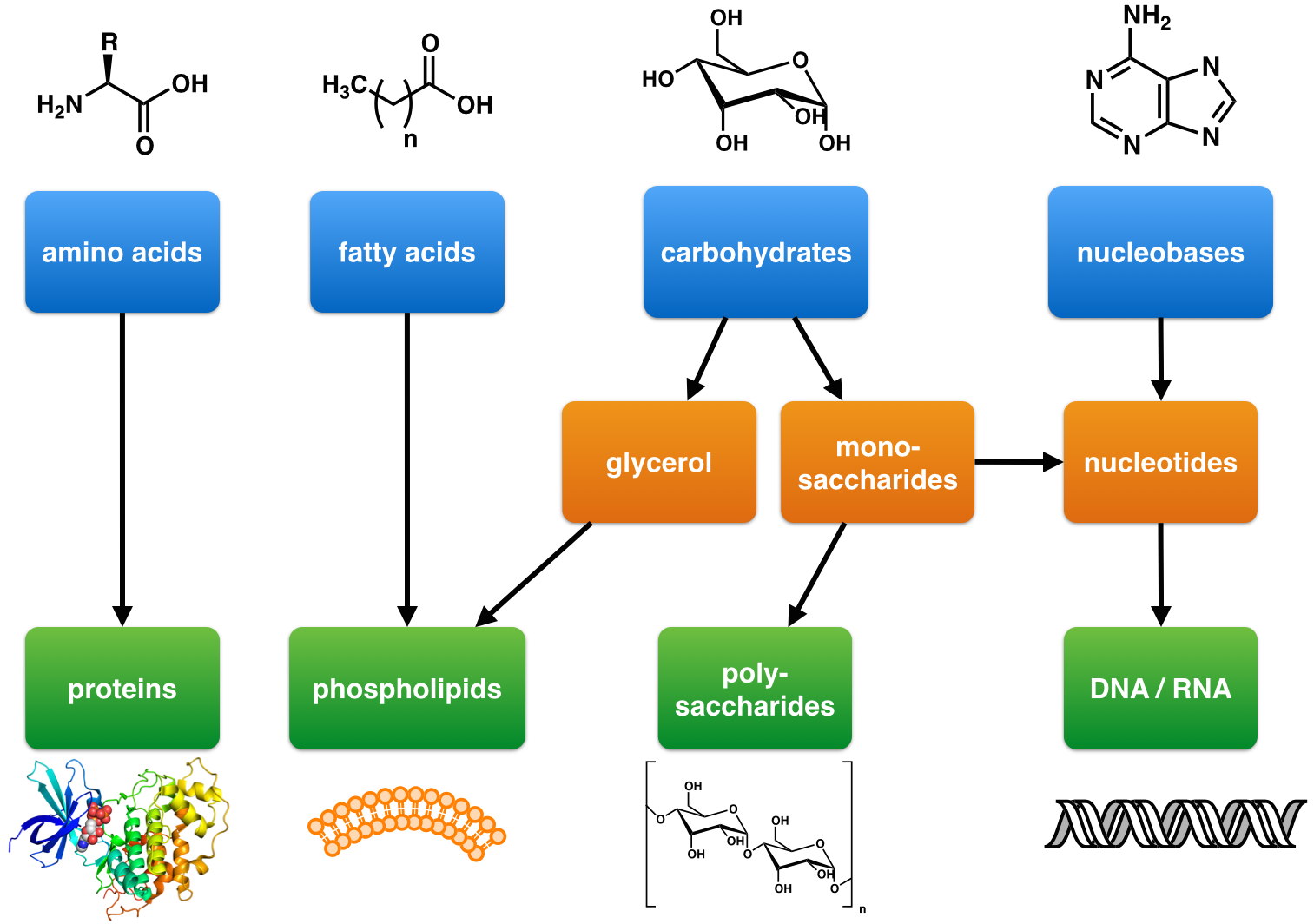
Glucose
Glucose is a simple sugar with the molecular formula . Glucose is overall the most abundant monosaccharide, a subcategory of carbohydrates. Glucose is mainly made by plants and most algae during photosynthesis from water and carbon dioxide, using energy from sunlight, where it is used to make cellulose in cell walls, the most abundant carbohydrate in the world. In energy metabolism, glucose is the most important source of energy in all organisms. Glucose for metabolism is stored as a polymer, in plants mainly as starch and amylopectin, and in animals as glycogen. Glucose circulates in the blood of animals as blood sugar. The naturally occurring form of glucose is -glucose, while -glucose is produced synthetically in comparatively small amounts and is less biologically active. Glucose is a monosaccharide containing six carbon atoms and an aldehyde group, and is therefore an aldohexose. The glucose molecule can exist in an open-chain (acyclic) as well as ring (cyclic) form. Gluco ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or may not be different from ''n''), which does not mean the H has covalent bonds with O (for example with , H has a covalent bond with C but not with O). However, not all carbohydrates conform to this precise stoichiometric definition (e.g., uronic acids, deoxy-sugars such as fucose), nor are all chemicals that do conform to this definition automatically classified as carbohydrates (e.g. formaldehyde and acetic acid). The term is most common in biochemistry, where it is a synonym of saccharide (), a group that includes sugars, starch, and cellulose. The saccharides are divided into four chemical groups: monosaccharides, disaccharides, oligosaccharides, and polysaccharides. Monosaccharides and disaccharides, the smallest (lower molecular wei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Natural Product
A natural product is a natural compound or substance produced by a living organism—that is, found in nature. In the broadest sense, natural products include any substance produced by life. Natural products can also be prepared by chemical synthesis (both semisynthesis and total synthesis) and have played a central role in the development of the field of organic chemistry by providing challenging synthetic targets. The term natural product has also been extended for commercial purposes to refer to cosmetics, dietary supplements, and foods produced from natural sources without added artificial ingredients. Within the field of organic chemistry, the definition of natural products is usually restricted to organic compounds isolated from natural sources that are produced by the pathways of primary or secondary metabolism. Within the field of medicinal chemistry, the definition is often further restricted to secondary metabolites. Secondary metabolites (or specialized metabolites ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
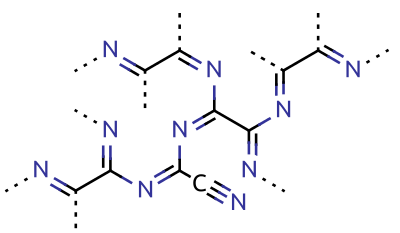
Cyanogen
Cyanogen is the chemical compound with the formula ( C N)2. It is a colorless and highly toxic gas with a pungent odor. The molecule is a pseudohalogen. Cyanogen molecules consist of two CN groups – analogous to diatomic halogen molecules, such as Cl2, but far less oxidizing. The two cyano groups are bonded together at their carbon atoms: N≡C‒ C≡N, although other isomers have been detected. The name is also used for the CN radical, and hence is used for compounds such as cyanogen bromide (NCBr) (but see also ''Cyano radical''.) Cyanogen is the anhydride of oxamide: :H2NC(O)C(O)NH2 → NCCN + 2 H2O although oxamide is manufactured from cyanogen by hydrolysis: :NCCN + 2 H2O → H2NC(O)C(O)NH2 Preparation Cyanogen is typically generated from cyanide compounds. One laboratory method entails thermal decomposition of mercuric cyanide: :2 Hg(CN)2 → (CN)2 + Hg2(CN)2 Alternatively, one can combine solutions of copper(II) salts (such as copper(II) sulfate) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Friedrich Wöhler
Friedrich Wöhler () FRS(For) HonFRSE (31 July 180023 September 1882) was a German chemist known for his work in inorganic chemistry, being the first to isolate the chemical elements beryllium and yttrium in pure metallic form. He was the first to prepare several inorganic compounds, including silane and silicon nitride. Wöhler is known for seminal contributions in organic chemistry, in particular, the Wöhler synthesis of urea. His synthesis of the organic compound urea in the laboratory from inorganic substances contradicted the belief that organic compounds could only be produced by living organisms due to a "life force". However, the exact extent of Wöhler's role in diminishing the belief in vitalism is considered by some to be questionable. Biography Friedrich Wöhler was born in Eschersheim, Germany, and was the son of a veterinarian. As a boy, he showed interest in mineral collecting, drawing, and science. His secondary education was at the Frankfurt Gymnasium. Durin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitric Acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitric acid has a concentration of 68% in water. When the solution contains more than 86% , it is referred to as ''fuming nitric acid''. Depending on the amount of nitrogen dioxide present, fuming nitric acid is further characterized as red fuming nitric acid at concentrations above 86%, or white fuming nitric acid at concentrations above 95%. Nitric acid is the primary reagent used for nitration – the addition of a nitro group, typically to an organic molecule. While some resulting nitro compounds are shock- and thermally-sensitive explosives, a few are stable enough to be used in munitions and demolition, while others are still more stable and used as pigments in inks and dyes. Nitric acid is also commonly used as a strong oxidizing agen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Torbern Bergman
Torbern Olaf (Olof) Bergman (''KVO'') (20 March 17358 July 1784) was a Swedish chemist and mineralogist noted for his 1775 ''Dissertation on Elective Attractions'', containing the largest chemical affinity tables ever published. Bergman was the first chemist to use the A, B, C, etc., system of notation for chemical species. Early life and education Torbern was born on 20 March 1735, the son of Barthold Bergman and Sara Hägg. He enrolled at the University of Uppsala at age 17. His father wished him to read either law or divinity, while he himself was anxious to study mathematics and natural science; in the effort to please both himself and his father, he overworked himself and harmed his health. During a period of enforced abstinence from study, he amused himself with field botany and entomology. He was able to send Linnaeus specimens of several new kinds of insects, and in 1756 he succeeded in proving that, contrary to the opinion of that naturalist, the so-called ''Coccus aqua ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish German pharmaceutical chemist. Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified molybdenum, tungsten, barium, hydrogen, and chlorine, among others. Scheele discovered organic acids tartaric, oxalic, uric, lactic, and citric, as well as hydrofluoric, hydrocyanic, and arsenic acids. He preferred speaking German to Swedish his whole life, as German was commonly spoken among Swedish pharmacists.Fors, Hjalmar 2008. Stepping through Science’s Door: C. W. Scheele, from Pharmacist's Apprentice to Man of Science. Ambix 55: 29–49 Biography Scheele was born in Stralsund, in western Pomerania, which at the time was a Swedish Dominion inside the Holy Roman Empire. Scheele's father, Joachim (or Johann) Christian Scheele, was a grain dealer and brewer from a respected Pomeranian family. His mother was Margaretha Eleanore Warnekros. Friends of Scheele's pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Hydrogenoxalate
Potassium hydrogenoxalate is a salt with formula KHC2O4 or K+·HO2C-CO2−. It is one of the most common salts of the hydrogenoxalate anion, and can be obtained by reacting potassium hydroxide with oxalic acid in 1:1 mole ratio. The salt is also known as: potassium hydrogen oxalate, potassium bioxalate, acid potassium oxalate, or monobasic potassium oxalate. In older literature, it was also called: Salt of sorrel, (retrieved via Internet Archive) sorrel salt, ''sel d'oseille'', Salt of Sorrel labelled “sel d'oseille”. Old dark-amber glass vial marked “sel d'oseille” with protective leaden cap. ''sal acetosella''; or, ''inaccurately, salt of lemon'' (due to the similar acidic ''“lemony”'' taste of the edible ''common sorrel'' or ''garden sorrel''.) “This fresh, “lemony” sourness has been highly prized in cuisines all over the world.” Potassium hydrogenoxalate occurs in some plants, notably sorrel. It is a commercial product used in photography, marble gr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxalis Acetosella
''Oxalis acetosella'', the wood sorrel or common wood sorrel, is a rhizomatous flowering plant in the family ''Oxalidaceae'', common in most of Europe and parts of Asia. The specific epithet ''acetosella'' refers to its sour taste. The common name wood sorrel is often used for other plants in the genus ''Oxalis''. In much of its range it is the only member of its genus and hence simply known as "the" wood sorrel. While common wood sorrel may be used to differentiate it from most other species of ''Oxalis'', in North America, ''Oxalis montana'' is also called common wood sorrel. It is also known as ''Alleluia'' because it blossoms between Easter and Pentecost, when the Psalms which end with Hallelujah are sung. Description The plant has trifoliate compound leaves, the leaflets heart-shaped and folded through the middle, that occur in groups of three on petioles up to long. It flowers from spring to midsummer with small white chasmogamous flowers with pink streaks. Red or viole ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |