
In
quantum mechanics
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistr ...
, the uncertainty principle (also known as Heisenberg's uncertainty principle) is any of a variety of
mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physical quantities of a
particle
In the physical sciences, a particle (or corpuscule in older texts) is a small localized object which can be described by several physical or chemical properties, such as volume, density, or mass.
They vary greatly in size or quantity, from ...
, such as
position
Position often refers to:
* Position (geometry), the spatial location (rather than orientation) of an entity
* Position, a job or occupation
Position may also refer to:
Games and recreation
* Position (poker), location relative to the dealer
* ...
, ''x'', and
momentum, ''p'', can be predicted from
initial conditions.
Such variable pairs are known as
complementary variables
In physics, complementarity is a conceptual aspect of quantum mechanics that Niels Bohr regarded as an essential feature of the theory. The complementarity principle holds that objects have certain pairs of complementary properties which cannot al ...
or
canonically conjugate variables; and, depending on interpretation, the uncertainty principle limits to what extent such conjugate properties maintain their approximate meaning, as the mathematical framework of
quantum physics does not support the notion of simultaneously well-defined conjugate properties expressed by a single value. The uncertainty principle implies that it is in general not possible to predict the value of a quantity with arbitrary certainty, even if all initial conditions are specified.
Introduced first in 1927 by the German physicist
Werner Heisenberg
Werner Karl Heisenberg () (5 December 1901 – 1 February 1976) was a German theoretical physicist and one of the main pioneers of the theory of quantum mechanics. He published his work in 1925 in a breakthrough paper. In the subsequent serie ...
, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was
p
q ~ h using the full Planck constant.
[.
Annotated pre-publication proof sheet o]
Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik
March 21, 1927. The formal inequality relating the
standard deviation of position ''σ
x'' and the standard deviation of momentum ''σ
p'' was derived by
Earle Hesse Kennard later that year and by
Hermann Weyl in 1928:
where is the
reduced Planck constant
The Planck constant, or Planck's constant, is a fundamental physical constant of foundational importance in quantum mechanics. The constant gives the relationship between the energy of a photon and its frequency, and by the mass-energy equivalen ...
, ).
Historically, the uncertainty principle has been confused
with a related effect in
physics
Physics is the natural science that studies matter, its fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge which r ...
, called the
observer effect, which notes that measurements of certain systems cannot be made without affecting the system, that is, without changing something in a system. Heisenberg utilized such an observer effect at the quantum level (see below) as a physical "explanation" of quantum uncertainty. It has since become clearer, however, that the uncertainty principle is inherent in the properties of all
wave-like systems,
and that it arises in quantum mechanics simply due to the
matter wave
Matter waves are a central part of the theory of quantum mechanics, being an example of wave–particle duality. All matter exhibits wave-like behavior. For example, a beam of electrons can be diffracted just like a beam of light or a water wav ...
nature of all quantum objects. Thus, ''the uncertainty principle actually states a fundamental property of quantum systems and is not a statement about the observational success of current technology''.
Indeed the uncertainty principle has its roots in how we apply calculus to write the basic equations of mechanics. It must be emphasized that ''measurement'' does not mean only a process in which a physicist-observer takes part, but rather any interaction between classical and quantum objects regardless of any observer.
Since the uncertainty principle is such a basic result in quantum mechanics, typical experiments in quantum mechanics routinely observe aspects of it. Certain experiments, however, may deliberately test a particular form of the uncertainty principle as part of their main research program. These include, for example, tests of number–phase uncertainty relations in
superconducting
Superconductivity is a set of physical properties observed in certain materials where electrical resistance vanishes and magnetic flux fields are expelled from the material. Any material exhibiting these properties is a superconductor. Unlike ...
or
quantum optics
Quantum optics is a branch of atomic, molecular, and optical physics dealing with how individual quanta of light, known as photons, interact with atoms and molecules. It includes the study of the particle-like properties of photons. Photons have ...
systems. Applications dependent on the uncertainty principle for their operation include extremely low-noise technology such as that required in
gravitational wave interferometers.
Introduction
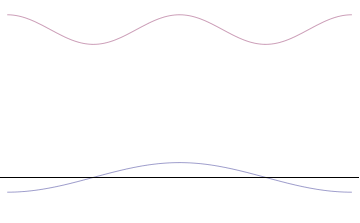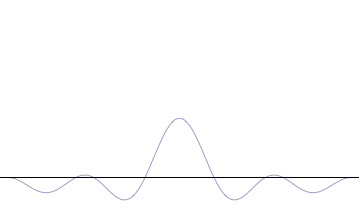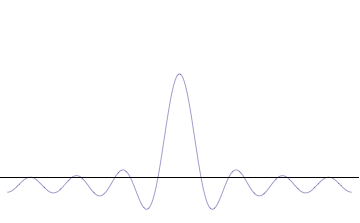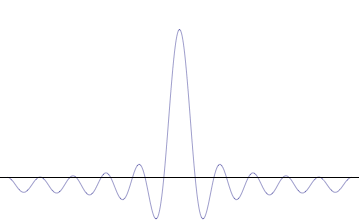
It is vital to illustrate how the principle applies to relatively intelligible physical situations since it is indiscernible on the macroscopic scales that humans experience. Two alternative frameworks for quantum physics offer different explanations for the uncertainty principle. The
wave mechanics Wave mechanics may refer to:
* the mechanics of waves
* the ''wave equation'' in quantum physics, see Schrödinger equation
See also
* Quantum mechanics
* Wave equation
The (two-way) wave equation is a second-order linear partial different ...
picture of the uncertainty principle is more visually intuitive, but the more abstract
matrix mechanics
Matrix mechanics is a formulation of quantum mechanics created by Werner Heisenberg, Max Born, and Pascual Jordan in 1925. It was the first conceptually autonomous and logically consistent formulation of quantum mechanics. Its account of quantum j ...
picture formulates it in a way that generalizes more easily.
Mathematically, in wave mechanics, the uncertainty relation between position and momentum arises because the expressions of the wavefunction in the two corresponding
orthonormal
In linear algebra, two vectors in an inner product space are orthonormal if they are orthogonal (or perpendicular along a line) unit vectors. A set of vectors form an orthonormal set if all vectors in the set are mutually orthogonal and all of un ...
bases in
Hilbert space are
Fourier transforms of one another (i.e., position and momentum are
conjugate variables). A nonzero function and its Fourier transform cannot both be sharply localized at the same time. A similar tradeoff between the variances of Fourier conjugates arises in all systems underlain by Fourier analysis, for example in sound waves: A pure tone is a
sharp spike at a single frequency, while its Fourier transform gives the shape of the sound wave in the time domain, which is a completely delocalized sine wave. In quantum mechanics, the two key points are that the position of the particle takes the form of a
matter wave
Matter waves are a central part of the theory of quantum mechanics, being an example of wave–particle duality. All matter exhibits wave-like behavior. For example, a beam of electrons can be diffracted just like a beam of light or a water wav ...
, and momentum is its Fourier conjugate, assured by the
de Broglie relation
Matter waves are a central part of the theory of quantum mechanics, being an example of wave–particle duality. All matter exhibits wave-like behavior. For example, a beam of electrons can be diffracted just like a beam of light or a water wave ...
, where is the
wavenumber
In the physical sciences, the wavenumber (also wave number or repetency) is the '' spatial frequency'' of a wave, measured in cycles per unit distance (ordinary wavenumber) or radians per unit distance (angular wavenumber). It is analogous to te ...
.
In
matrix mechanics
Matrix mechanics is a formulation of quantum mechanics created by Werner Heisenberg, Max Born, and Pascual Jordan in 1925. It was the first conceptually autonomous and logically consistent formulation of quantum mechanics. Its account of quantum j ...
, the
mathematical formulation of quantum mechanics
The mathematical formulations of quantum mechanics are those mathematical formalisms that permit a rigorous description of quantum mechanics. This mathematical formalism uses mainly a part of functional analysis, especially Hilbert spaces, which ...
, any pair of non-
commuting
Commuting is periodically recurring travel between one's place of residence and place of work or study, where the traveler, referred to as a commuter, leaves the boundary of their home community. By extension, it can sometimes be any regul ...
self-adjoint operator
In mathematics, a self-adjoint operator on an infinite-dimensional complex vector space ''V'' with inner product \langle\cdot,\cdot\rangle (equivalently, a Hermitian operator in the finite-dimensional case) is a linear map ''A'' (from ''V'' to its ...
s representing
observable
In physics, an observable is a physical quantity that can be measured. Examples include position and momentum. In systems governed by classical mechanics, it is a real-valued "function" on the set of all possible system states. In quantum phy ...
s are subject to similar uncertainty limits. An eigenstate of an observable represents the state of the wavefunction for a certain measurement value (the eigenvalue). For example, if a measurement of an observable is performed, then the system is in a particular eigenstate of that observable. However, the particular eigenstate of the observable need not be an eigenstate of another observable : If so, then it does not have a unique associated measurement for it, as the system is not in an eigenstate of that observable.
Wave mechanics interpretation
(Ref
[Online copy]
)
According to the
de Broglie hypothesis, every object in the universe is a
wave
In physics, mathematics, and related fields, a wave is a propagating dynamic disturbance (change from equilibrium) of one or more quantities. Waves can be periodic, in which case those quantities oscillate repeatedly about an equilibrium (re ...
, i.e., a situation which gives rise to this phenomenon. The position of the particle is described by a
wave function
A wave function in quantum physics is a mathematical description of the quantum state of an isolated quantum system. The wave function is a complex-valued probability amplitude, and the probabilities for the possible results of measurements ...
. The time-independent wave function of a single-moded plane wave of wavenumber ''k''
0 or momentum ''p''
0 is
The
Born rule
The Born rule (also called Born's rule) is a key postulate of quantum mechanics which gives the probability that a measurement of a quantum system will yield a given result. In its simplest form, it states that the probability density of findi ...
states that this should be interpreted as a
probability density amplitude function in the sense that the probability of finding the particle between ''a'' and ''b'' is
In the case of the single-moded plane wave,
is a
uniform distribution. In other words, the particle position is extremely uncertain in the sense that it could be essentially anywhere along the wave packet.
On the other hand, consider a wave function that is a
sum of many waves, which we may write as
where ''A''
''n'' represents the relative contribution of the mode ''p''
''n'' to the overall total. The figures to the right show how with the addition of many plane waves, the wave packet can become more localized. We may take this a step further to the
continuum limit
In mathematical physics and mathematics, the continuum limit or scaling limit of a lattice model refers to its behaviour in the limit as the lattice spacing goes to zero. It is often useful to use lattice models to approximate real-world processe ...
, where the wave function is an
integral
In mathematics, an integral assigns numbers to functions in a way that describes displacement, area, volume, and other concepts that arise by combining infinitesimal data. The process of finding integrals is called integration. Along wit ...
over all possible modes
with
representing the amplitude of these modes and is called the wave function in
momentum space
In physics and geometry, there are two closely related vector spaces, usually three-dimensional but in general of any finite dimension.
Position space (also real space or coordinate space) is the set of all ''position vectors'' r in space, and h ...
. In mathematical terms, we say that
is the ''
Fourier transform'' of
and that ''x'' and ''p'' are
conjugate variables. Adding together all of these plane waves comes at a cost, namely the momentum has become less precise, having become a mixture of waves of many different momenta.
One way to quantify the precision of the position and momentum is the
standard deviation ''σ''. Since
is a probability density function for position, we calculate its standard deviation.
The precision of the position is improved, i.e. reduced ''σ''
''x'', by using many plane waves, thereby weakening the precision of the momentum, i.e. increased ''σ''
''p''. Another way of stating this is that ''σ''
''x'' and ''σ''
''p'' have an
inverse relationship
In statistics, there is a negative relationship or inverse relationship between two variables if higher values of one variable tend to be associated with lower values of the other. A negative relationship between two variables usually implies that ...
or are at least bounded from below. This is the uncertainty principle, the exact limit of which is the Kennard bound. Click the ''show'' button below to see a semi-formal derivation of the Kennard inequality using wave mechanics.
Matrix mechanics interpretation
(Ref
)
In matrix mechanics, observables such as position and momentum are represented by
self-adjoint operator
In mathematics, a self-adjoint operator on an infinite-dimensional complex vector space ''V'' with inner product \langle\cdot,\cdot\rangle (equivalently, a Hermitian operator in the finite-dimensional case) is a linear map ''A'' (from ''V'' to its ...
s. When considering pairs of observables, an important quantity is the ''
commutator''. For a pair of operators and
, one defines their commutator as
In the case of position and momentum, the commutator is the
canonical commutation relation
In quantum mechanics, the canonical commutation relation is the fundamental relation between canonical conjugate quantities (quantities which are related by definition such that one is the Fourier transform of another). For example,
hat x,\hat p_ ...
The physical meaning of the non-commutativity can be understood by considering the effect of the commutator on position and momentum
eigenstates. Let
be a right eigenstate of position with a constant eigenvalue . By definition, this means that
Applying the commutator to
yields
where is the
identity operator
Identity may refer to:
* Identity document
* Identity (philosophy)
* Identity (social science)
* Identity (mathematics)
Arts and entertainment Film and television
* ''Identity'' (1987 film), an Iranian film
* ''Identity'' (2003 film) ...
.
Suppose, for the sake of
proof by contradiction
In logic and mathematics, proof by contradiction is a form of proof that establishes the truth or the validity of a proposition, by showing that assuming the proposition to be false leads to a contradiction. Proof by contradiction is also known ...
, that
is also a right eigenstate of momentum, with constant eigenvalue . If this were true, then one could write
On the other hand, the above canonical commutation relation requires that
This implies that no quantum state can simultaneously be both a position and a momentum eigenstate.
When a state is measured, it is projected onto an eigenstate in the basis of the relevant observable. For example, if a particle's position is measured, then the state amounts to a position eigenstate. This means that the state is ''not'' a momentum eigenstate, however, but rather it can be represented as a sum of multiple momentum basis eigenstates. In other words, the momentum must be less precise. This precision may be quantified by the
standard deviations,
As in the wave mechanics interpretation above, one sees a tradeoff between the respective precisions of the two, quantified by the uncertainty principle.
Heisenberg limit
In
quantum metrology
Quantum metrology is the study of making high-resolution and highly sensitive measurements of physical parameters using quantum theory to describe the physical systems, particularly exploiting quantum entanglement and quantum squeezing. This fie ...
, and especially
interferometry, the Heisenberg limit is the optimal rate at which the accuracy of a measurement can scale with the energy used in the measurement. Typically, this is the measurement of a phase (applied to one arm of a
beam-splitter
A beam splitter or ''beamsplitter'' is an optical device that splits a beam of light into a transmitted and a reflected beam. It is a crucial part of many optical experimental and measurement systems, such as interferometers, also finding wide ...
) and the energy is given by the number of photons used in an
interferometer. Although some claim to have broken the Heisenberg limit, this reflects disagreement on the definition of the scaling resource. Suitably defined, the Heisenberg limit is a consequence of the basic principles of quantum mechanics and cannot be beaten, although the weak Heisenberg limit can be beaten.
Robertson–Schrödinger uncertainty relations
The most common general form of the uncertainty principle is the ''
Robertson
Robertson may refer to:
People
* Robertson (surname) (includes a list of people with this name)
* Robertson (given name)
* Clan Robertson, a Scottish clan
* Robertson, stage name of Belgian magician Étienne-Gaspard Robert (1763–1837)
Places ...
uncertainty relation''.
For an arbitrary
Hermitian operator
In mathematics, a self-adjoint operator on an infinite-dimensional complex vector space ''V'' with inner product \langle\cdot,\cdot\rangle (equivalently, a Hermitian operator in the finite-dimensional case) is a linear map ''A'' (from ''V'' to it ...
we can associate a standard deviation
where the brackets
indicate an
expectation value
In probability theory, the expected value (also called expectation, expectancy, mathematical expectation, mean, average, or first moment) is a generalization of the weighted average. Informally, the expected value is the arithmetic mean of a l ...
. For a pair of operators
and
, we may define their ''
commutator'' as
In this notation, the Robertson uncertainty relation is given by
The Robertson uncertainty relation immediately
follows from a slightly stronger inequality, the ''Schrödinger uncertainty relation'',
where we have introduced the ''anticommutator'',
Mixed states
The Robertson–Schrödinger uncertainty relation may be generalized in a straightforward way to describe
mixed states.
The Maccone–Pati uncertainty relations
The Robertson–Schrödinger uncertainty relation can be trivial if the state of the system is chosen to be eigenstate of one of the observable. The stronger uncertainty relations proved by Maccone and Pati give non-trivial bounds on the sum of the variances for two incompatible observables. (Earlier works on uncertainty relations formulated as the sum of variances include, e.g., Ref. due to Huang.) For two non-commuting observables
and
the first stronger uncertainty relation is given by
where
,
,
is a normalized vector that is orthogonal to the state of the system
and one should choose the sign of
to make this real quantity a positive number.
The second stronger uncertainty relation is given by
where
is a state orthogonal to
.
The form of
implies that the right-hand side of the new uncertainty relation is nonzero unless
is an eigenstate of
. One may note that
can be an eigenstate of
without being an eigenstate of either
or
. However, when
is an eigenstate of one of the two observables the Heisenberg–Schrödinger uncertainty relation becomes trivial. But the lower bound in the new relation is nonzero unless
is an eigenstate of both.
Improving the Robertson–Schrödinger uncertainty relation based on decompositions of the density matrix
The Robertson–Schrödinger uncertainty can be improved noting that it must hold for all components
in any decomposition of the density matrix given as
Here, for the probailities
and
hold. Then, using the relation
for
,
it follows that
where the function in the bound is defined
The above relation very often has a bound larger than that of the original Robertson–Schrödinger uncertainty relation. Thus, we need to calculate the bound of the Robertson–Schrödinger uncertainty for the mixed components of the quantum state rather than for the quantum state, and compute an average of their square roots. The following expression is stronger than the Robertson–Schrödinger uncertainty relation
where on the right-hand side there is a concave roof over the decompositions of the density matrix.
The improved relation above is saturated by all single-qubit quantum states.
With similar arguments, one can derive a relation with a convex roof on the right-hand side
where
 In
In  It is vital to illustrate how the principle applies to relatively intelligible physical situations since it is indiscernible on the macroscopic scales that humans experience. Two alternative frameworks for quantum physics offer different explanations for the uncertainty principle. The
It is vital to illustrate how the principle applies to relatively intelligible physical situations since it is indiscernible on the macroscopic scales that humans experience. Two alternative frameworks for quantum physics offer different explanations for the uncertainty principle. The  In
In  It is vital to illustrate how the principle applies to relatively intelligible physical situations since it is indiscernible on the macroscopic scales that humans experience. Two alternative frameworks for quantum physics offer different explanations for the uncertainty principle. The
It is vital to illustrate how the principle applies to relatively intelligible physical situations since it is indiscernible on the macroscopic scales that humans experience. Two alternative frameworks for quantum physics offer different explanations for the uncertainty principle. The