|
Overpotential
In electrochemistry, overpotential is the potential difference (voltage) between a half-reaction's thermodynamically determined reduction potential and the potential at which the redox event is experimentally observed. The term is directly related to a cell's ''voltage efficiency''. In an electrolytic cell the existence of overpotential implies that the cell requires more energy than thermodynamically expected to drive a reaction. In a galvanic cell the existence of overpotential means less energy is recovered than thermodynamics predicts. In each case the extra/missing energy is lost as heat. The quantity of overpotential is specific to each cell design and varies across cells and operational conditions, even for the same reaction. Overpotential is experimentally determined by measuring the potential at which a given current density (typically small) is achieved. Thermodynamics The four possible polarities of overpotentials are listed below. * An electrolytic cell' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
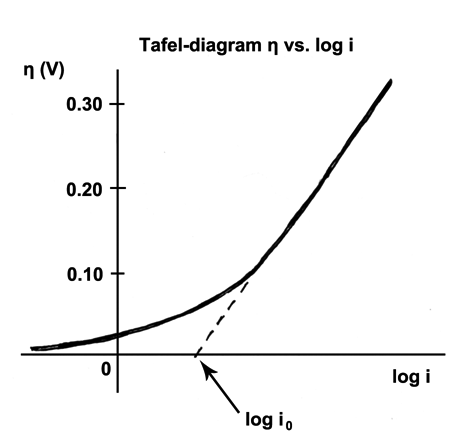
Tafel Equation
The Tafel equation is an equation in electrochemical kinetics relating the rate of an Electrochemistry, electrochemical reaction to the overpotential. The Tafel equation was first deduced experimentally and was later shown to have a theoretical justification. The equation is named after Swiss chemist Julius Tafel.It describes how the electrical current through an electrode depends on the voltage difference between the electrode and the bulk electrolyte for a simple, unimolecular redox reaction. : Ox + n e^- \leftrightarrows Red Where an electrochemical reaction occurs in two half reactions on separate electrodes, the Tafel equation is applied to each electrode separately. On a single electrode the Tafel equation can be stated as: where * the plus sign under the exponent refers to an anodic reaction, and a minus sign to a cathodic reaction, * \eta : overpotential, [V] * A : "Butler–Volmer equation#The limiting cases, Tafel slope", [V] * i : current density, [A/m2] * i_0 : "exch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature. It is an extremely reactive element and a strong oxidizing agent, oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Electronegativity#Pauling electronegativity, Pauling scale, behind only oxygen and fluorine. Chlorine played an important role in the experiments conducted by medieval Alchemy, alchemists, which commonly involved the heating of chloride Salt (chemistry), salts like ammonium chloride (sal ammoniac) and sodium chloride (common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and . However, the nature of fre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Faraday Efficiency
In electrochemistry, Faraday efficiency (also called faradaic efficiency, faradaic yield, coulombic efficiency, or current efficiency) describes the efficiency with which charge (electrons) is transferred in a system facilitating an electrochemical reaction. The word "Faraday" in this term has two interrelated aspects: first, the historic unit for charge is the faraday (F), but has since been replaced by the coulomb (C); and secondly, the related Faraday's constant () correlates charge with moles of matter and electrons (amount of substance). This phenomenon was originally understood through Michael Faraday's work and expressed in his laws of electrolysis. Sources of faradaic loss Faradaic losses are experienced by both electrolytic and galvanic cells when electrons or ions participate in unwanted side reactions. These losses appear as heat and/or chemical byproducts. An example can be found in the oxidation of water to oxygen at the positive electrode in electrolysis. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemical Reaction Mechanism
In electrochemistry, an electrochemical reaction mechanism is the step-by-step sequence of elementary steps, involving at least one outer-sphere electron transfer, by which an overall electrochemical reaction occurs. Overview Elementary steps like proton coupled electron transfer and the movement of electrons between an electrode and substrate are special to electrochemical processes. Electrochemical mechanisms are important to all redox chemistry including corrosion, redox active photochemistry including photosynthesis, other biological systems often involving electron transport chains and other forms of homogeneous and heterogeneous electron transfer. Such reactions are most often studied with standard three electrode techniques such as cyclic voltammetry(CV), chronoamperometry, and bulk electrolysis as well as more complex experiments involving rotating disk electrodes and rotating ring-disk electrodes. In the case of photoinduced electron transfer the use of ti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclic Voltammetry
In electrochemistry, cyclic voltammetry (CV) is a type of voltammetric measurement where the potential of the working electrode is ramped linearly versus time. Unlike in linear sweep voltammetry, after the set potential is reached in a CV experiment, the working electrode's potential is ramped in the opposite direction to return to the initial potential. These cycles in potential are repeated until the voltammetric trace reaches a cyclic steady state. The current at the working electrode is plotted versus the voltage at the working electrode to yield the cyclic voltammogram (see Figure 1). Cyclic voltammetry is generally used to study the electrochemical properties of an analyte in solution or of a molecule that is adsorbed onto the electrode. Experimental method In cyclic voltammetry (CV), the electrode potential is ramped linearly versus time in cyclical phases. The rate of voltage change over time during each of these phases is known as the scan rate (V/s). In a standar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Galvanic Cell
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. An example of a galvanic cell consists of two different metals, each immersed in separate beakers containing their respective metal ions in solution that are connected by a salt bridge or separated by a porous membrane. Volta was the inventor of the voltaic pile, the first electrical battery. Common usage of the word ''battery'' has evolved to include a single Galvanic cell, but the first batteries had many Galvanic cells. History In 1780, Luigi Galvani discovered that when two different metals (e.g., copper and zinc) are in contact and then both are touched at the same time to two different parts of a muscle of a frog leg, to close the circuit, the frog's leg contracts. He called this " animal electricity". The frog's leg, as well as being a detector ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemistry
Electrochemistry is the branch of physical chemistry concerned with the relationship between Electric potential, electrical potential difference and identifiable chemical change. These reactions involve Electron, electrons moving via an electronically conducting phase (typically an external electrical circuit, but not necessarily, as in Electroless nickel-phosphorus plating, electroless plating) between electrodes separated by an ionically conducting and electronically insulating electrolyte (or ionic chemical species, species in a Solution (chemistry), solution). When a chemical reaction is driven by an electrical Voltage, potential difference, as in electrolysis, or if a potential difference results from a chemical reaction as in an electric battery or fuel cell, it is called an ''electrochemical'' reaction. Unlike in other chemical reactions, in electrochemical reactions electrons are not transferred directly between atoms, ions, or molecules, but via the aforementioned electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), nonmetal, and a potent oxidizing agent that readily forms oxides with most elements as well as with other chemical compound, compounds. Oxygen is abundance of elements in Earth's crust, the most abundant element in Earth's crust, making up almost half of the Earth's crust in the form of various oxides such as water, carbon dioxide, iron oxides and silicates.Atkins, P.; Jones, L.; Laverman, L. (2016).''Chemical Principles'', 7th edition. Freeman. It is abundance of chemical elements, the third-most abundant element in the universe after hydrogen and helium. At standard temperature and pressure, two oxygen atoms will chemical bond, bind covalent bond, covalently to form dioxygen, a colorless and odorless diatomic gas with the chemical formula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver
Silver is a chemical element; it has Symbol (chemistry), symbol Ag () and atomic number 47. A soft, whitish-gray, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. Silver is found in the Earth's crust in the pure, free elemental form ("native metal, native silver"), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc Refining (metallurgy), refining. Silver has long been valued as a precious metal. Silver metal is used in many bullion coins, sometimes bimetallism, alongside gold: while it is more abundant than gold, it is much less abundant as a native metal. Its purity is typically measured on a per-mille basis; a 94%-pure alloy is described as "0.940 fine". As one of the seven metals of antiquity, silver has had an enduring role in most human cultures. Other than in currency and as an in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Second
The second (symbol: s) is a unit of time derived from the division of the day first into 24 hours, then to 60 minutes, and finally to 60 seconds each (24 × 60 × 60 = 86400). The current and formal definition in the International System of Units (SI) is more precise: The second ..is defined by taking the fixed numerical value of the caesium frequency, Δ''ν''Cs, the unperturbed ground-state hyperfine transition frequency of the caesium 133 atom, to be when expressed in the unit Hz, which is equal to s−1. This current definition was adopted in 1967 when it became feasible to define the second based on fundamental properties of nature with caesium clocks. As the speed of Earth's rotation varies and is slowing ever so slightly, a leap second is added at irregular intervals to civil time to keep clocks in sync with Earth's rotation. The definition that is based on of a rotation of the earth is still used by the Universal Time 1 (UT1) system. Etymology "Minute" ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has a great affinity towards oxygen, passivation (chemistry), forming a protective layer of aluminium oxide, oxide on the surface when exposed to air. It visually resembles silver, both in its color and in its great ability to reflect light. It is soft, magnetism, nonmagnetic, and ductility, ductile. It has one stable isotope, 27Al, which is highly abundant, making aluminium the abundance of the chemical elements, 12th-most abundant element in the universe. The radioactive decay, radioactivity of aluminium-26, 26Al leads to it being used in radiometric dating. Chemically, aluminium is a post-transition metal in the boron group; as is common for the group, aluminium forms compounds primarily in the +3 oxidation state. The aluminium cation Al3+ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |