|
Deoxycytidine
Deoxycytidine is a deoxyribonucleoside, a component of deoxyribonucleic acid. It is similar to the ribonucleoside cytidine, but with one hydroxyl group removed from the C2' position. Deoxycytidine can be phosphorylated at C5' of the deoxyribose by deoxycytidine kinase, converting it to deoxycytidine monophosphate (dCMP), a DNA precursor. dCMP can be converted to dUMP and dTMP. It can also be used as a precursor for 5-aza-2′-deoxycytidine, a treatment for MDS patients. This compound slows the cell cycle by interfering with the methylation of the P15/INK4B gene, increasing the expression of P15/INK4B protein which subdues the transformation of MDS to leukemia Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or ' .... References Nucleosides Pyrimidones Hydroxymethyl compounds< ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
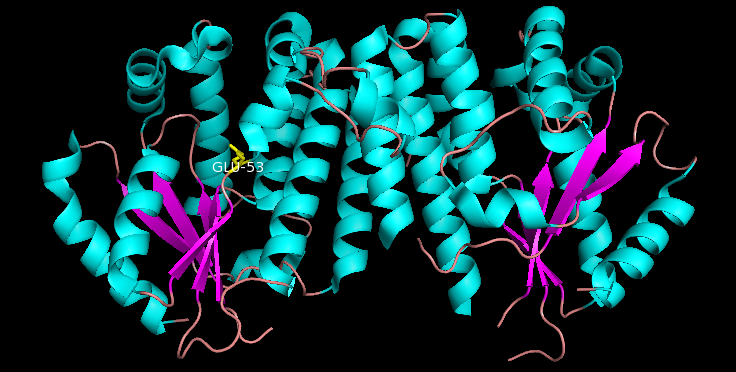
Deoxycytidine Kinase
Deoxycytidine kinase (dCK) is an enzyme which is encoded by the ''DCK'' gene in humans. dCK predominantly phosphorylates deoxycytidine (dC) and converts dC into deoxycytidine monophosphate. dCK catalyzes one of the initial steps in the nucleoside salvage pathway and has the potential to phosphorylate other preformed nucleosides, specifically deoxyadenosine (dA) and deoxyguanosine (dG), and convert them into their monophosphate forms. There has been recent biomedical research interest in investigating dCK's potential as a therapeutic target for different types of cancer. Structure dCK is a homodimer where each monomer subunit consists of multiple alpha helices surrounding a beta sheet core. Each subunit includes a nucleotide donor binding site, nucleoside acceptor binding site, nucleotide base sensing loop (240-254 residues), insert region (12-15 residues) that connects helices 2 and 3. dCK has several different protein conformations but its conformation depends on the nucleos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-aza-2′-deoxycytidine
Decitabine, sold under the brand name Dacogen among others, acts as a nucleic acid synthesis inhibitor. It is a medication for the treatment of myelodysplastic syndromes, a class of conditions where certain blood cells are dysfunctional, and for acute myeloid leukemia (AML). Chemically, it is a cytidine analog. Medical uses Decitabine is used to treat myelodysplastic syndromes (MDS) including previously treated and untreated, de novo and secondary MDS of all French-American-British subtypes ( refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia) and Intermediate-1, Intermediate-2, and High-Risk International Prognostic Scoring System groups. In patients with chronic kidney disease, Batty and colleagues reported the first case series on the feasibility of therapy with hypomethylating agents in patients with chronic kidney disease. It also h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deoxycytidine Monophosphate
Deoxycytidine monophosphate (dCMP), also known as deoxycytidylic acid or deoxycytidylate in its conjugate acid and conjugate base forms, respectively, is a deoxynucleotide, and one of the four monomers that make up DNA. In a DNA double helix, it will base pair with deoxyguanosine monophosphate. See also * Cytidine monophosphate Cytidine monophosphate, also known as 5'-cytidylic acid or simply cytidylate, and abbreviated CMP, is a nucleotide that is used as a monomer in RNA. It is an ester of phosphoric acid with the nucleoside cytidine. CMP consists of the phosphate grou ... Nucleotides {{biochem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Myelodysplastic Syndrome
A myelodysplastic syndrome (MDS) is one of a group of cancers in which immature blood cells in the bone marrow do not mature, and as a result, do not develop into healthy blood cells. Early on, no symptoms typically are seen. Later, symptoms may include feeling tired, shortness of breath, bleeding disorders, anemia, or frequent infections. Some types may develop into acute myeloid leukemia. Risk factors include previous chemotherapy or radiation therapy, exposure to certain chemicals such as tobacco smoke, pesticides, and benzene, and exposure to heavy metals such as mercury or lead. Problems with blood cell formation result in some combination of low red blood cell, platelet, and white blood cell counts. Some types have an increase in immature blood cells, called blasts, in the bone marrow or blood. The types of MDS are based on specific changes in the blood cells and bone marrow. Treatments may include supportive care, drug therapy, and hematopoietic stem cell transplantati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
DNA Methylation
DNA methylation is a biological process by which methyl groups are added to the DNA molecule. Methylation can change the activity of a DNA segment without changing the sequence. When located in a gene promoter, DNA methylation typically acts to repress gene transcription. In mammals, DNA methylation is essential for normal development and is associated with a number of key processes including genomic imprinting, X-chromosome inactivation, repression of transposable elements, aging, and carcinogenesis. As of 2016, two nucleobases have been found on which natural, enzymatic DNA methylation takes place: adenine and cytosine. The modified bases are N6-methyladenineD. B. Dunn, J. D. Smith: ''The occurrence of 6-methylaminopurine in deoxyribonucleic acids.'' In: ''Biochem J.'' 68(4), Apr 1958, S. 627–636. PMID 13522672. ., 5-methylcytosineB. F. Vanyushin, S. G. Tkacheva, A. N. Belozersky: ''Rare bases in animal DNA.'' In: ''Nature.'' 225, 1970, S. 948–949. PMID 4391887. and N4- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleosides
Nucleosides are glycosylamines that can be thought of as nucleotides without a phosphate group. A nucleoside consists simply of a nucleobase (also termed a nitrogenous base) and a five-carbon sugar (ribose or 2'-deoxyribose) whereas a nucleotide is composed of a nucleobase, a five-carbon sugar, and one or more phosphate groups. In a nucleoside, the anomeric carbon is linked through a glycosidic bond to the N9 of a purine or the N1 of a pyrimidine. Nucleotides are the molecular building-blocks of DNA and RNA. List of nucleosides and corresponding nucleobases The reason for 2 symbols, shorter and longer, is that the shorter ones are better for contexts where explicit disambiguation is superfluous (because context disambiguates) and the longer ones are for contexts where explicit disambiguation is judged to be needed or wise. For example, when discussing long nucleobase sequences in genomes, the CATG symbol system is much preferable to the Cyt-Ade-Thy-Gua symbol system (see '' N ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deoxyribonucleoside
A deoxyribonucleotide is a nucleotide that contains deoxyribose. They are the monomeric units of the informational biopolymer, deoxyribonucleic acid ( DNA). Each deoxyribonucleotide comprises three parts: a deoxyribose sugar (monosaccharide), a nitrogenous base, and one phosphoryl group. The nitrogenous bases are either purines or pyrimidines, heterocycles whose structures support the specific base-pairing interactions that allow nucleic acids to carry information. The base is always bonded to the 1'-carbon of the deoxyribose, an analog of ribose in which the hydroxyl group of the 2'-carbon is replaced with a hydrogen atom. The third component, the phosphoryl group, attaches to the deoxyribose monomer via the hydroxyl group on the 5'-carbon of the sugar. When deoxyribonucleotides polymerize to form DNA, the phosphate group from one nucleotide will bond to the 3' carbon on another nucleotide, forming a phosphodiester bond via dehydration synthesis. New nucleotides are always added ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ribonucleoside
A ribonucleoside is a type of nucleoside including ribose as a component. One example of a ribonucleoside is cytidine Cytidine (symbol C or Cyd) is a nucleoside molecule that is formed when cytosine is attached to a ribose ring (also known as a ribofuranose) via a β-N1- glycosidic bond. Cytidine is a component of RNA. It is a white water-soluble solid. which .... References {{Chem-stub * Ribosides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cytidine
Cytidine (symbol C or Cyd) is a nucleoside molecule that is formed when cytosine is attached to a ribose ring (also known as a ribofuranose) via a β-N1- glycosidic bond. Cytidine is a component of RNA. It is a white water-soluble solid. which is only slightly soluble in ethanol. Dietary sources Dietary sources of cytidine include foods with high RNA (ribonucleic acid) content, such as organ meats, brewer's yeast, as well as pyrimidine-rich foods such as beer. During digestion, RNA-rich foods are broken-down into ribosyl pyrimidines (cytidine and uridine), which are absorbed intact. In humans, dietary cytidine is converted into uridine, which is probably the compound behind cytidine's metabolic effects. Cytidine analogues A variety of cytidine analogues are known, some with potentially useful pharmacology. For example, KP-1461 is an anti-HIV agent that works as a viral mutagen, and zebularine exists in '' E. coli'' and is being examined for chemotherapy. Low doses of azacit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy groups. Both the negatively charged anion , called hydroxide, and the neutral radical , known as the hydroxyl radical, consist of an unbonded hydroxy group. According to IUPAC definitions, the term ''hydroxyl'' refers to the hydroxyl radical () only, while the functional group is called a ''hydroxy group''. Properties Water, alcohols, carboxylic acids, and many other hydroxy-containing compounds can be readily deprotonated due to a large difference between the electronegativity of oxygen (3.5) and that of hydrogen (2.1). Hydroxy-containing compounds engage in intermolecular hydrogen bonding increasing the electrostatic attraction between molecules and thus to higher boiling and melting points than found for compounds that lack this f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deoxyribose
Deoxyribose, or more precisely 2-deoxyribose, is a monosaccharide with idealized formula H−(C=O)−(CH2)−(CHOH)3−H. Its name indicates that it is a deoxy sugar, meaning that it is derived from the sugar ribose by loss of a hydroxy group. Discovered in 1929 by Phoebus Levene, deoxyribose is most notable for its presence in DNA. Since the pentose sugars arabinose and ribose only differ by the stereochemistry at C2′, 2-deoxyribose and 2-deoxyarabinose are equivalent, although the latter term is rarely used because ribose, not arabinose, is the precursor to deoxyribose. Structure Several isomers exist with the formula H−(C=O)−(CH2)−(CHOH)3−H, but in deoxyribose all the hydroxyl groups are on the same side in the Fischer projection. The term "2-deoxyribose" may refer to either of two enantiomers: the biologically important -2-deoxyribose and to the rarely encountered mirror image -2-deoxyribose.C Bernelot-Moens and B Demple (1989), ''Multiple DNA repair activities f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |