|
Case Series
A case series (also known as a clinical series) is a type of medical research study that tracks subjects with a known exposure, such as patients who have received a similar treatment, or examines their medical records for exposure and outcome. Case series may be ''consecutive'' or ''non-consecutive'', depending on whether all cases presenting to the reporting authors over a period were included, or only a selection. When information on ''more than three patients'' is included, the case series is considered to be a systematic investigation designed to contribute to generalizable knowledge (i.e., ''research''), and therefore submission is required to an institutional review board (IRB). Case series usually contain demographic information about the patient(s), for example, age, gender, ethnic origin. etc. Case series have a descriptive study design; unlike studies that employ an analytic design (e.g. cohort studies, case-control studies or randomized controlled trials), case series ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Study Design
Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research (e.g., epidemiological) involving human beings. The goal of a clinical study is to assess the safety, efficacy, and / or the mechanism of action of an investigational medicinal product (IMP) or procedure, or new drug or device that is in development, but potentially not yet approved by a health authority (e.g. Food and Drug Administration). It can also be to investigate a drug, device or procedure that has already been approved but is still in need of further investigation, typically with respect to long-term effects or cost-effectiveness. Some of the considerations here are shared under the more general topic of design of experiments but there can be others, in particular related to patient confidentiality and ethics. Outline of types of designs for clinical studies Treatment studies * Randomized controlled trial ** Blind trial ** ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Placebo
A placebo ( ) is a substance or treatment which is designed to have no therapeutic value. Common placebos include inert tablets (like sugar pills), inert injections (like Saline (medicine), saline), sham surgery, and other procedures. In general, placebos can affect how patients perceive their condition and encourage the body's chemical processes for relieving pain and a few other symptoms, but have no impact on the disease itself. Improvements that patients experience after being treated with a placebo can also be due to unrelated factors, such as regression to the mean (a statistical effect where an unusually high or low measurement is likely to be followed by a less extreme one). The use of placebos in clinical medicine raises ethical concerns, especially if they are disguised as an active treatment, as this introduces dishonesty into the doctor–patient relationship and bypasses informed consent. While it was once assumed that this deception was necessary for placebos to have ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Consecutive Case Series
A consecutive case series is a clinical study that includes all eligible patients identified by the researchers Research is "creative and systematic work undertaken to increase the stock of knowledge". It involves the collection, organization and analysis of evidence to increase understanding of a topic, characterized by a particular attentiveness t ... during the study registration period. The patients are treated in the order in which they are identified. This type of study usually does not have a control group. For example, in Sugrue, et al. (2016), a consecutive case series design was used to determine trends in hand surgery research.Sugrue, C.M., Joyce, C.W., Sugrue, R.M. and Carroll, S.M. (2016)TRENDS IN THE LEVEL OF EVIDENCE IN CLINICAL HAND SURGERY RESEARCH Hand (New York, N.Y.) 1558944715627619, first published on February 26, 2016 doi:10.1177/1558944715627619 References External links Consecutive case seriesentry in the public domain NCI Dictionary of Cance ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Case Report
In medicine, a case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports may contain a demographic profile of the patient, but usually describe an unusual or novel occurrence. Some case reports also contain a literature review of other reported cases. Case reports are professional narratives that provide feedback on clinical practice guidelines and offer a framework for early signals of effectiveness, adverse events, and cost. They can be shared for medical, scientific, or educational purposes. Types Most case reports are on one of six topics: * An unexpected association between diseases or symptoms. * An unexpected event in the course of observing or treating a patient. * Findings that shed new light on the possible pathogenesis of a disease or an adverse effect. * Unique or rare features of a disease. * Unique therapeutic approaches. * A positional or quantitative variation of the anatomical structures. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Opinion Evidence
Opinion evidence refers to direct evidence outlining what the expert witness, believes, or infers in regard to facts, as distinguished from personal knowledge of the facts themselves. In common law jurisdictions the general rule is that a witness is supposed to testify as to what was observed and not to give an opinion on what was observed. However, there are two exceptions to this rule: expert evidence and non-expert opinion given by laymen which people in their daily lives reach without conscious ratiocination. General rule In general, witnesses should testify only as to the facts observed and should not give opinion. The main rationale for such a rule is that the admission of opinion evidence would not assist, or might even mislead, the court and in particular the jury. This is because opinion evidence is usually irrelevant. Moreover, admission of such evidence would usurp the functions of the jury, which alone should be the tribunal of fact and draw its own inferences. Exper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Case Report
In medicine, a case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports may contain a demographic profile of the patient, but usually describe an unusual or novel occurrence. Some case reports also contain a literature review of other reported cases. Case reports are professional narratives that provide feedback on clinical practice guidelines and offer a framework for early signals of effectiveness, adverse events, and cost. They can be shared for medical, scientific, or educational purposes. Types Most case reports are on one of six topics: * An unexpected association between diseases or symptoms. * An unexpected event in the course of observing or treating a patient. * Findings that shed new light on the possible pathogenesis of a disease or an adverse effect. * Unique or rare features of a disease. * Unique therapeutic approaches. * A positional or quantitative variation of the anatomical structures. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Observational Study
In fields such as epidemiology, social sciences, psychology and statistics, an observational study draws inferences from a sample (statistics), sample to a statistical population, population where the dependent and independent variables, independent variable is not under the Scientific control, control of the researcher because of ethical concerns or logistical constraints. One common observational study is about the possible effect of a treatment on subjects, where the assignment of subjects into a treated group versus a control group is outside the control of the investigator. This is in contrast with experiments, such as randomized controlled trials, where each subject is Random assignment, randomly assigned to a treated group or a control group. Observational studies, for lacking an assignment mechanism, naturally present difficulties for inferential analysis. Motivation The independent variable may be beyond the control of the investigator for a variety of reasons: * A ran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
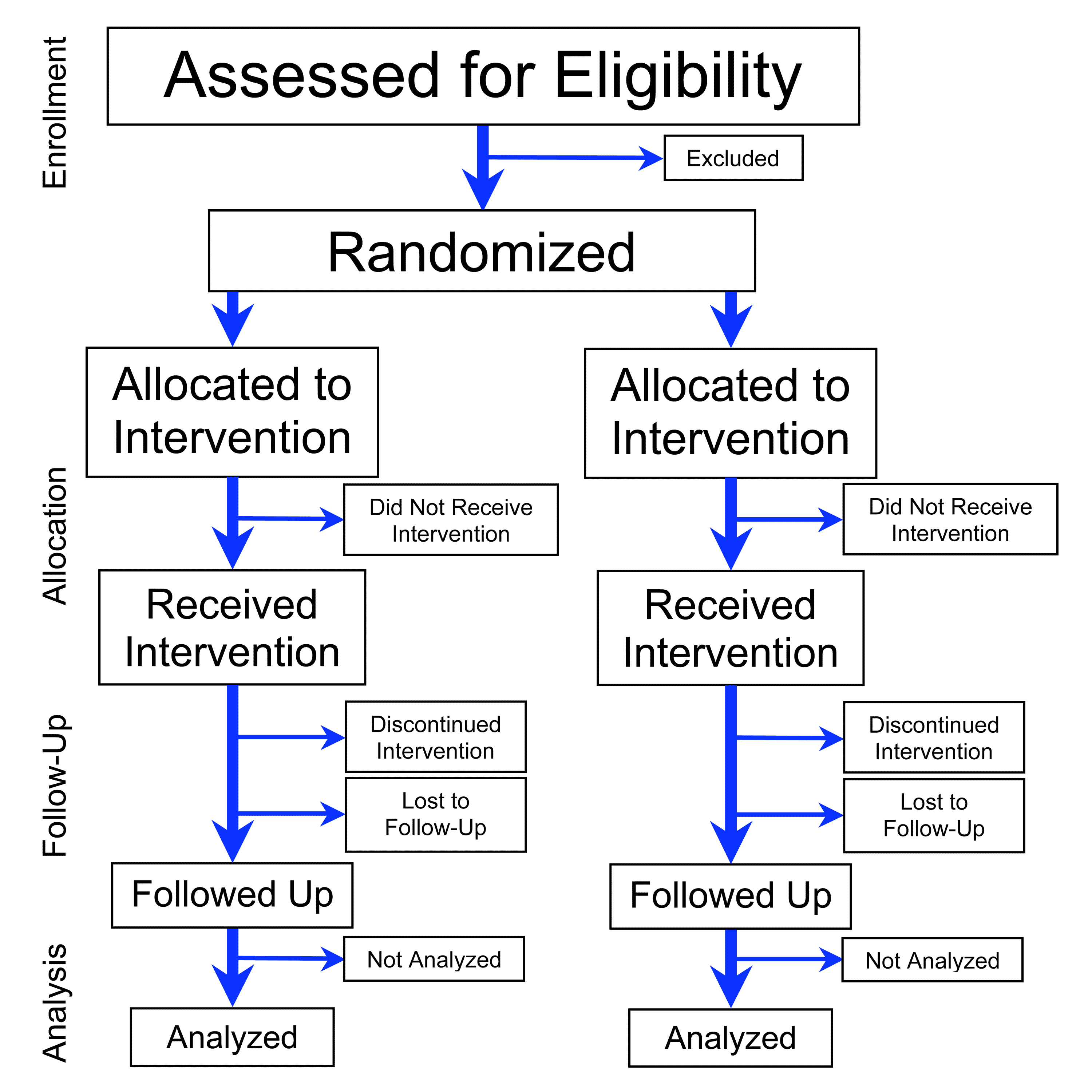
Randomized Controlled Trial
A randomized controlled trial (or randomized control trial; RCT) is a form of scientific experiment used to control factors not under direct experimental control. Examples of RCTs are clinical trials that compare the effects of drugs, surgical techniques, medical devices, diagnostic procedures or other medical treatments. Participants who enroll in RCTs differ from one another in known and unknown ways that can influence study outcomes, and yet cannot be directly controlled. By Random assignment, randomly allocating participants among compared treatments, an RCT enables ''statistical control'' over these influences. Provided it is designed well, conducted properly, and enrolls enough participants, an RCT may achieve sufficient control over these confounding factors to deliver a useful comparison of the treatments studied. Definition and examples An RCT in clinical research typically compares a proposed new treatment against an existing Standard of care#Medical standard of care, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Randomized Control Trial
A randomized controlled trial (or randomized control trial; RCT) is a form of scientific experiment used to control factors not under direct experimental control. Examples of RCTs are clinical trials that compare the effects of drugs, surgical techniques, medical devices, diagnostic procedures or other medical treatments. Participants who enroll in RCTs differ from one another in known and unknown ways that can influence study outcomes, and yet cannot be directly controlled. By randomly allocating participants among compared treatments, an RCT enables ''statistical control'' over these influences. Provided it is designed well, conducted properly, and enrolls enough participants, an RCT may achieve sufficient control over these confounding factors to deliver a useful comparison of the treatments studied. Definition and examples An RCT in clinical research typically compares a proposed new treatment against an existing standard of care; these are then termed the 'experimental' a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Systematic Review
A systematic review is a Literature review, scholarly synthesis of the evidence on a clearly presented topic using critical methods to identify, define and assess research on the topic. A systematic review extracts and interprets data from published studies on the topic, then analyzes, describes, and summarizes interpretations into a refined conclusion. For example, a systematic review of randomized controlled trials is a way of summarizing and implementing evidence-based medicine. While a systematic review may be applied in the Biomedical research, biomedical or health care context, it may also be used where an assessment of a precisely defined subject can advance understanding in a field of research. A systematic review may examine clinical tests, public health interventions, environmental interventions, social interventions, adverse effects, qualitative evidence syntheses, methodological reviews, policy reviews, and economic evaluations. An understanding of systematic review ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Rosenthal Effect
The Pygmalion effect, or Rosenthal effect, is a psychological phenomenon in which high expectations lead to improved performance in a given area. The effect is named for the Greek myth of Pygmalion, the sculptor who fell so much in love with the perfectly beautiful statue he created that the statue came to life. The psychologists Robert Rosenthal and Lenore Jacobson, in their book ''Pygmalion in the Classroom'', borrowed something of the myth by advancing the idea that teachers' expectations of their students affect the students' performance, a view that has been called into question as a result of later research findings. Rosenthal and Jacobson held that high expectations lead to better performance and low expectations lead to worse, both effects leading to self-fulfilling prophecy. According to the Pygmalion effect, the targets of the expectations internalize their positive labels, and those with positive labels succeed accordingly; a similar process works in the opposite direc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hawthorne Effect
The Hawthorne effect is a type of reactivity in which individuals modify an aspect of their behavior in response to their awareness of being observed. The effect was discovered in the context of research conducted at the Hawthorne Western Electric plant; however, some scholars feel the descriptions are apocryphal. The original research involved workers who made electrical relays at the Hawthorne Works, a Western Electric plant in Cicero, Illinois. Between 1924 and 1927, the lighting study was conducted. Workers experienced a series of lighting changes in which productivity was said to increase with almost any change in the lighting. This turned out ''not'' to be true. In the study that was associated with Elton Mayo, which ran from 1928 to 1932, a series of changes in work structure were implemented (e.g., changes in rest periods) in a group of five women. However, this was a methodologically poor, uncontrolled study that did not permit any firm conclusions to be drawn. One of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |