|
Upper Critical Solution Temperature
The upper critical solution temperature (UCST) or upper consolute temperature is the critical temperature above which the components of a mixture are miscible in all proportions. The word ''upper'' indicates that the UCST is an upper bound to a temperature range of partial miscibility, or miscibility for certain compositions only. For example, hexane-nitrobenzene mixtures have a UCST of , so that these two substances are miscible in all proportions above but not at lower temperatures. Examples at higher temperatures are the aniline-water system at (at pressures high enough for liquid water to exist at that temperature), and the lead-zinc system at (a temperature where both metals are liquid). A solid state example is the palladium-hydrogen system which has a solid solution phase (H2 in Pd) in equilibrium with a hydride phase (PdHn) below the UCST at 300 °C. Above this temperature there is a single solid solution phase. In the phase diagram of the mixture components, the UC ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Critical Temperature
Critical or Critically may refer to: *Critical, or critical but stable, medical states **Critical, or intensive care medicine * Critical juncture, a discontinuous change studied in the social sciences. * Critical Software, a company specializing in mission and business critical information systems * Critical theory, a school of thought that critiques society and culture by applying knowledge from the social sciences and the humanities * Critically endangered, a risk status for wild species *Criticality (status), the condition of sustaining a nuclear chain reaction Art, entertainment, and media * ''Critical'' (novel), a medical thriller written by Robin Cook * ''Critical'' (TV series), a Sky 1 TV series * "Critical" (''Person of Interest''), an episode of the American television drama series ''Person of Interest'' *"Critical", a 1999 single by Zion I People *Cr1TiKaL (born 1994), an American YouTuber and Twitch streamer See also *Critic *Criticality (other) *Critical Con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ACS Publications
The American Chemical Society (ACS) is a scientific society based in the United States that supports scientific inquiry in the field of chemistry. Founded in 1876 at New York University, the ACS currently has more than 155,000 members at all degree levels and in all fields of chemistry, chemical engineering, and related fields. It is one of the world's largest scientific societies by membership. The ACS is a 501(c)(3) non-profit organization and holds a congressional charter under Title 36 of the United States Code. Its headquarters are located in Washington, D.C., and it has a large concentration of staff in Columbus, Ohio. The ACS is a leading source of scientific information through its peer-reviewed scientific journals, national conferences, and the Chemical Abstracts Service. Its publications division produces over 60 scholarly journals including the prestigious ''Journal of the American Chemical Society'', as well as the weekly trade magazine ''Chemical & Engineering ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymers
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'', meani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flory–Huggins Solution Theory
Flory–Huggins solution theory is a lattice model of the thermodynamics of polymer solutions which takes account of the great dissimilarity in molecular sizes in adapting the usual expression for the entropy of mixing. The result is an equation for the Gibbs free energy change \Delta G_m for mixing a polymer with a solvent. Although it makes simplifying assumptions, it generates useful results for interpreting experiments. Theory The thermodynamic equation for the Gibbs energy change accompanying mixing at constant temperature and (external) pressure is :\Delta G_m = \Delta H_m - T\Delta S_m \, A change, denoted by \Delta, is the value of a variable for a solution or mixture minus the values for the pure components considered separately. The objective is to find explicit formulas for \Delta H_m and \Delta S_m, the enthalpy and entropy increments associated with the mixing process. The result obtained by Flory and Huggins is :\Delta G_m = RT ,n_1\ln\phi_1 + n_2\ln\phi_ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Statistical Mechanics
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. It does not assume or postulate any natural laws, but explains the macroscopic behavior of nature from the behavior of such ensembles. Statistical mechanics arose out of the development of classical thermodynamics, a field for which it was successful in explaining macroscopic physical properties—such as temperature, pressure, and heat capacity—in terms of microscopic parameters that fluctuate about average values and are characterized by probability distributions. This established the fields of statistical thermodynamics and statistical physics. The founding of the field of statistical mechanics is generally credited to three physicists: * Ludwig Boltzmann, who developed the fundamental interpretation of entropy in terms of a collection of microstates *James Clerk Maxwell, who developed models of probability di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dispersity
In chemistry, the dispersity is a measure of the heterogeneity of sizes of molecules or particles in a mixture. A collection of objects is called uniform if the objects have the same size, shape, or mass. A sample of objects that have an inconsistent size, shape and mass distribution is called non-uniform. The objects can be in any form of chemical dispersion, such as particles in a colloid, droplets in a cloud, crystals in a rock, or polymer macromolecules in a solution or a solid polymer mass. Polymers can be described by molecular mass distribution; a population of particles can be described by size, surface area, and/or mass distribution; and thin films can be described by film thickness distribution. IUPAC has deprecated the use of the term ''polydispersity index'', having replaced it with the term ''dispersity'', represented by the symbol Đ (pronounced D-strokeStepto, R. F. T.; Gilbert, R. G.; Hess, M.; Jenkins, A. D.; Jones, R. G.; Kratochvíl P. (2009).Dispersity in Po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
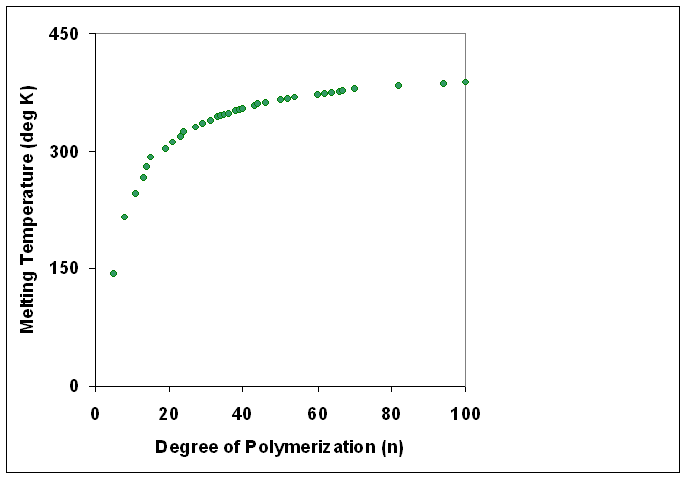
Degree Of Polymerization
The degree of polymerization, or DP, is the number of monomeric units in a macromolecule or polymer or oligomer molecule. For a homopolymer, there is only one type of monomeric unit and the ''number-average'' degree of polymerization is given by DP_n\equiv X_n=\frac, where Mn is the number-average molecular weight and M0 is the molecular weight of the monomer unit. For most industrial purposes, degrees of polymerization in the thousands or tens of thousands are desired. This number does not reflect the variation in molecule size of the polymer that typically occurs, it only represents the mean number of monomeric units. Some authors, however, define DP as the number of repeat units, where for copolymers the repeat unit may not be identical to the monomeric unit.Fried J.R. "Polymer Science and Technology" (Pearson Prentice-Hall, 2nd edn 2003), p.27 For example, in nylon-6,6, the repeat unit contains the two monomeric units —NH(CH2)6NH— and —OC(CH2)4CO—, so that a c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lower Critical Solution Temperature
The lower critical solution temperature (LCST) or lower consolute temperature is the critical temperature below which the components of a mixture are miscible in all proportions. The word ''lower'' indicates that the LCST is a lower bound to a temperature interval of partial miscibility, or miscibility for certain compositions only. The phase behavior of polymer solutions is an important property involved in the development and design of most polymer-related processes. Partially miscible polymer solutions often exhibit two solubility boundaries, the upper critical solution temperature (UCST) and the LCST, both of which depend on the molar mass and the pressure. At temperatures below LCST, the system is completely miscible in all proportions, whereas above LCST partial liquid miscibility occurs. In the phase diagram of the mixture components, the LCST is the shared minimum of the concave up spinodal and binodal (or coexistence) curves. It is in general pressure dependent, in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phase Separation
Phase separation is the creation of two distinct phases from a single homogeneous mixture. The most common type of phase separation is between two immiscible liquids, such as oil and water. Colloids are formed by phase separation, though not all phase separations forms colloids - for example oil and water can form separated layers under gravity rather than remaining as microscopic droplets in suspension. Phase separation in cold gases A mixture of two helium isotopes ( helium-3 and helium-4) in a certain range of temperatures and concentrations separates into parts. The initial mix of the two isotopes spontaneously separates into ^He-rich and ^3He-rich regions. Phase separation also exists in ultracold gas systems. It has been shown experimentally in a two-component ultracold Fermi gas case. The phase separation can compete with other phenomena as vortex lattice formation or an exotic Fulde-Ferrell-Larkin-Ovchinnikov phase. See also * Biomolecular condensate * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Miscible
Miscibility () is the property of two substances to mix in all proportions (that is, to fully dissolve in each other at any concentration), forming a homogeneous mixture (a solution). The term is most often applied to liquids but also applies to solids and gases. For example, water and ethanol are miscible because they mix in all proportions. By contrast, substances are said to be immiscible if there are certain proportions in which the mixture does not form a solution. For one example, oil is not soluble in water, so these two solvents are immiscible. As another example, butanone (methyl ethyl ketone) is significantly soluble in water, but these two solvents are also immiscible because in some proportions the mixture will separate into two phases. Organic compounds In organic compounds, the weight percent of hydrocarbon chain often determines the compound's miscibility with water. For example, among the alcohols, ethanol has two carbon atoms and is miscible with water, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |