|
Trimethylamine
Trimethylamine (TMA) is an organic compound with the formula N(CH3)3. It is a colorless, hygroscopic, and flammable tertiary amine. It is a gas at room temperature but is usually sold as a 40% solution in water. (It is also sold in pressurized gas cylinders.) TMA is a nitrogenous base and can be readily protonated to give the trimethylammonium cation. Trimethylammonium chloride is a hygroscopic colorless solid prepared from hydrochloric acid. Trimethylamine is a good nucleophile, and this reaction is the basis of most of its applications. TMA is widely used in industry: it is used in the synthesis of choline, tetramethylammonium hydroxide, plant growth regulators or herbicides, strongly basic anion exchange resins, dye leveling agents, and a number of basic dyes. At higher concentrations it has an ammonia-like odor, and can cause necrosis of mucous membranes on contact. At lower concentrations, it has a "fishy" odor, the odor associated with rotting fish. In humans, ingesti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylamine
Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around 40%. An estimated 270,000 tons were produced in 2005. Structure and synthesis The molecule consists of a nitrogen atom with two methyl substituents and one proton. Dimethylamine is a weak base and the pKa of the ammonium CH3--CH3 is 10.73, a value above methylamine (10.64) and trimethylamine (9.79). Dimethylamine reacts with acids to form salts, such as dimethylamine hydrochloride, an odorless white solid with a melting point of 171.5 °C. Dimethylamine is produced by catalytic reaction of methanol and ammonia at elevated temperatures and high pressure: :2 CH3OH + NH3 → (CH3)2NH + 2 H2O Natural occurrence Dimethylamine is found quite widely distributed in animals and plants, and is present in many foods at the level of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tertiary Amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. Aromatic a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylenetriamine
Diethylenetriamine (abbreviated and also known as 2,2’-Iminodi(ethylamine)) is an organic compound with the formula HN(CH2CH2NH2)2. This colourless hygroscopic liquid is soluble in water and polar organic solvents, but not simple hydrocarbons. Diethylenetriamine is structural analogue of diethylene glycol. Its chemical properties resemble those for ethylene diamine, and it has similar uses. It is a weak base and its aqueous solution is alkaline. DETA is a byproduct of the production of ethylenediamine from ethylene dichloride. Reactions and uses Diethylenetriamine is a common curing agent for epoxy resins in epoxy adhesives and other thermosets. It is N-alkylated upon reaction with epoxide groups forming crosslinks. In coordination chemistry, it serves as a tridentate ligand forming complexes such as Co(dien)(NO2)3. Like some related amines, it is used in oil industry for the extraction of acid gas. Like ethylenediamine, DETA can also be used to sensitize nitromethane, ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
HN3 (nitrogen Mustard)
Tris(2-chloroethyl)amine is the organic compound with the formula N(CH2CH2Cl)3. Often abbreviated HN3 or HN-3, it is a powerful blister agent and a nitrogen mustard used for chemical warfare. HN3 was the last of the nitrogen mustard agents developed. It was designed as a military agent and is the only one of the nitrogen mustards that is still used for military purposes. It is the principal representative of the nitrogen mustards because its vesicant properties are almost equal to those of HD and thus the analogy between the two types of mustard is the strongest.NITROGEN MUSTARD HN-3 Emergency Response Safety and Health Database. |
Mechlorethamine
Chlormethine (INN, BAN), also known as mechlorethamine (USAN, USP), mustine, HN2, and (in post-Soviet states) embikhin (эмбихин), is a nitrogen mustard sold under the brand name Mustargen among others. It is the prototype of alkylating agents, a group of anticancer chemotherapeutic drugs. It works by binding to DNA, crosslinking two strands and preventing cell duplication. It binds to the N7 nitrogen on the DNA base guanine. As the chemical is a blister agent, its use is strongly restricted within the Chemical Weapons Convention where it is classified as a Schedule 1 substance. Mechlorethamine belongs to the group of nitrogen mustard alkylating agents. Uses It has been derivatized into the estrogen analogue estramustine phosphate, used to treat prostate cancer. It can also be used in chemical warfare where it has the code-name HN2. This chemical is a form of nitrogen mustard gas and a powerful vesicant. Historically, some uses of mechlorethamine have included lymphoid ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
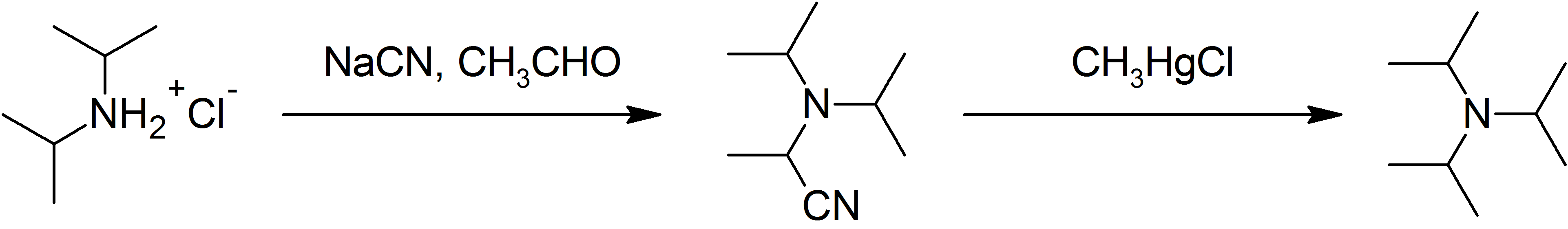
Triisopropylamine
Triisopropylamine is an organic chemical compound consisting of three isopropyl groups bound to a central nitrogen atom. As a hindered tertiary amine, it can be used as a non-nucleophilic base and as a stabilizer for polymers; however, its applications are limited by its relatively high cost and difficult synthesis. Structure Triisopropylamine is notable as being among the most sterically hindered amines currently known. The even more crowded tri-''tert''-butylamine (''t''Bu3N) has never been synthesized, although ''ab initio'' quantum chemical computations as well as the existence of the even more crowded 2,2,4,4-tetramethyl-3-''t''-butyl-pentane-3-ol (tri-''tert''-butylcarbinol, ''t''Bu3COH) implies that it should be a stable molecule if it could be prepared. To date, di-''tert''-butyl(isopropyl)amine (''t''Bu2iPrN) has been prepared in low yield, as have a handful of tri-''tert''-alkylamines in which two of the ''tert''-alkyl groups are tied together in a ring, but the auth ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tris(2-aminoethyl)amine
Tris(2-aminoethyl)amine is the organic compound with the chemical formula, formula N(CH2CH2NH2)3. This colourless liquid is soluble in water and is highly basic, consisting of a tertiary amine center and three pendant primary amine groups. Abbreviated tren or TREN it is a crosslinking agent in the synthesis of polyimine networks and a tripodal ligand in coordination chemistry. Tren is a C3-symmetric, tetradentate chelating ligand that forms stable complexes with transition metals, especially those in the 2+ and 3+ oxidation states. Tren complexes exist with relatively few isomers, reflecting the constrained connectivity of this tetramine. Thus, only a single achiral stereoisomer exists for [Co(tren)X2]+, where X is halide or pseudohalide. In contrast, for [Co(trien)X2]+ five diastereomers are possible, four of which are chiral. In a few cases, tren serves as a tridentate ligand with one of the primary amine groups non-coordinated. Tren is a common impurity in the more common trie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylaminopropylamine
Dimethylaminopropylamine (DMAPA) is a diamine used in the preparation of some surfactants, such as cocamidopropyl betaine which is an ingredient in many personal care products including soaps, shampoos, and cosmetics. BASF, a major producer, claims that DMAPA-derivatives do not sting the eyes and makes a fine-bubble foam, making it appropriate in shampoos. Preparation and reactions DMAPA is commonly produced commercially via the reaction between dimethylamine and acrylonitrile (a Michael reaction) to produce dimethylaminopropionitrile. A subsequent hydrogenation step yields DMAPA: : DMAPA is readily converted to the mustard dimethylaminopropyl-3-chloride, a powerful alkylating agent. Health effects Dimethylaminopropylamine is a known skin irritant and its presence as an impurity in cocamidopropyl betaine is thought to be the cause of irritation experienced by some individuals. See also * 1,1-Dimethylethylenediamine * 1,2-Dimethylethylenediamine References {{reflist Diamine ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triethylamine
Triethylamine is the chemical compound with the formula N(CH2CH3)3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine or tetraethylammonium, for which TEA is also a common abbreviation. It is a colourless volatile liquid with a strong fishy odor reminiscent of ammonia. Like diisopropylethylamine (Hünig's base), triethylamine is commonly employed in organic synthesis, usually as a base. Synthesis and properties Triethylamine is prepared by the alkylation of ammonia with ethanol: :NH3 + 3 C2H5OH → N(C2H5)3 + 3 H2O The pKa of protonated triethylamine is 10.75,David Evans Research Group and it can be used to prepare buffer solutions at that pH. The |
Diethylamine
Diethylamine is an organic compound with the formula (CH3CH2)2NH. It is a secondary amine. It is a flammable, weakly alkaline liquid that is miscible with most solvents. It is a colorless liquid, but commercial samples often appear brown due to impurities. It has a strong ammonia-like odor. Production and uses Diethylamine is made by the alumina-catalyzed reaction of ethanol and ammonia. It is obtained together with ethylamine and triethylamine. Annual production of three ethylamines was estimated in 2000 to be 80,000,000 kg. It is used in the production of corrosion inhibitor ''N'',''N''- diethylaminoethanol, by reaction with ethylene oxide. It is also a precursor to a wide variety of other commercial products. Diethylamine is also sometimes used in the illicit production of LSD. Supramolecular structure Diethylamine is the smallest and simplest molecule that features a supramolecular helix as its lowest energy aggregate. Other similarly sized hydrogen-bonding In c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetramethylammonium Hydroxide
Tetramethylammonium hydroxide (TMAH or TMAOH) is a quaternary ammonium salt with molecular formula N(CH3)4+ OH−. It is commonly encountered in form of concentrated solutions in water or methanol. TMAH in solid state and its aqueous solutions are all colorless, but may be yellowish if impure. Although TMAH has virtually no odor when pure, samples often have a strong fishy smell due to presence of trimethylamine which is a common impurity. TMAH has several diverse industrial and research applications. Chemical properties Structure TMAH is most commonly encountered as an aqueous solution, in concentrations from ~2–25%, and less frequently as solutions in methanol. These solutions are identified by CAS numbe75-59-2 Several hydrates such as N(CH3)4OH·xH2O. have been crystallized. These salts contain well separated Me4N+ cations and hydroxide anions ( Me is an abbreviation of methyl group). The hydroxide groups are linked by hydrogen bonds to the water of crystallization ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Plant Growth Regulator
Plant hormone (or phytohormones) are signal molecules, produced within plants, that occur in extremely low concentrations. Plant hormones control all aspects of plant growth and development, from embryogenesis, the regulation of organ size, pathogen defense, stress tolerance and through to reproductive development. Unlike in animals (in which hormone production is restricted to specialized glands) each plant cell is capable of producing hormones. Went and Thimann coined the term "phytohormone" and used it in the title of their 1937 book. Phytohormones occur across the plant kingdom, and even in algae, where they have similar functions to those seen in higher plants. Some phytohormones also occur in microorganisms, such as unicellular fungi and bacteria, however in these cases they do not play a hormonal role and can better be regarded as secondary metabolites. Characteristics The word hormone is derived from Greek, meaning ''set in motion''. Plant hormones affect gene expres ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.png)
