|
Triazabicyclodecene
Triazabicyclodecene (1,5,7-triazabicyclo .4.0ec-5-ene or TBD) is an organic compound consisting of a bicyclic guanidine. For a charge-neutral compound, it is a relatively strong base that is effectively for a variety of organic transformations. TBD is colorless solid that is soluble in a variety of solvents. Reactivity As a strong base, TBD fully deprotonates most phenols, carboxylic acids, and some C-acids. It catalyzes a variety of reactions including Michael reactions, Henry reactions ( nitroaldol reactions), transesterification reactions, and Knoevenagel condensations. Deprotonation at the 7-position gives a particularly electron-rich ligand In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's elec ... as manifested in the redox properties of ditungsten tetra(hpp). See also * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Guanidine
Guanidine is the compound with the formula HNC(NH2)2. It is a colourless solid that dissolves in polar solvents. It is a strong base that is used in the production of plastics and explosives. It is found in urine predominantly in patients experiencing renal failure. A guanidine moiety also appears in larger organic molecules, including on the side chain of arginine. Structure Guanidine can be thought of as a nitrogenous analogue of carbonic acid. That is, the C=O group in carbonic acid is replaced by a C=NH group, and each OH is replaced by a group. Isobutene can be seen as the carbon analogue in much the same way. A detailed crystallographic analysis of guanidine was elucidated 148 years after its first synthesis, despite the simplicity of the molecule. In 2013, the positions of the hydrogen atoms and their displacement parameters were accurately determined using single-crystal neutron diffraction. Production Guanidine can be obtained from natural sources, being first isolat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Guanidines
Guanidine is the compound with the formula HNC(NH2)2. It is a colourless solid that dissolves in polar solvents. It is a strong base that is used in the production of plastics and explosives. It is found in urine predominantly in patients experiencing renal failure. A guanidine moiety also appears in larger organic molecules, including on the side chain of arginine. Structure Guanidine can be thought of as a nitrogenous analogue of carbonic acid. That is, the C=O group in carbonic acid is replaced by a C=NH group, and each OH is replaced by a group. Isobutene can be seen as the carbon analogue in much the same way. A detailed crystallographic analysis of guanidine was elucidated 148 years after its first synthesis, despite the simplicity of the molecule. In 2013, the positions of the hydrogen atoms and their displacement parameters were accurately determined using single-crystal neutron diffraction. Production Guanidine can be obtained from natural sources, being first isolat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sigma-Aldrich
Sigma-Aldrich (formally MilliporeSigma) is an American chemical, life science, and biotechnology company that is owned by the German chemical conglomerate Merck Group. Sigma-Aldrich was created in 1975 by the merger of Sigma Chemical Company and Aldrich Chemical Company. It grew through various acquisitions until it had over 9,600 employees and was listed on the Fortune 1000. The company is headquartered in St. Louis and has operations in approximately 40 countries. In 2015, the German chemical conglomerate Merck Group acquired Sigma-Aldrich for $17 billion. The company is currently a part of Merck's life science business and in combination with Merck's earlier acquired Millipore Corporation, Millipore, operates as MilliporeSigma. History Sigma Chemical Company of St. Louis and Aldrich Chemical Company of Milwaukee were both American specialty chemical companies when they merged in August 1975. The company grew throughout the 1980s and 1990s, with significant expansion in fac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
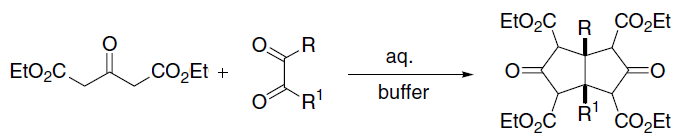
Knoevenagel Condensation
In organic chemistry, the Knoevenagel condensation () reaction is a type of chemical reaction named after German chemist Emil Knoevenagel. It is a modification of the aldol condensation. A Knoevenagel condensation is a nucleophilic addition of an active hydrogen compound to a carbonyl group followed by a dehydration reaction in which a molecule of water is eliminated (hence '' condensation''). The product is often an α,β-unsaturated ketone (a conjugated enone). In this reaction the carbonyl group is an aldehyde or a ketone. The catalyst is usually a weakly basic amine. The active hydrogen component has the form * or for instance diethyl malonate, Meldrum's acid, ethyl acetoacetate or malonic acid, or cyanoacetic acid. * , for instance nitromethane. where Z is an electron withdrawing group. Z must be powerful enough to facilitate deprotonation to the enolate ion even with a mild base. Using a strong base in this reaction would induce self-condensation of the alde ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amines
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. Aro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalysts
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usually gaseous or liquid) as the reactant, or heterogeneous, whose components are not in the same phase. Enzymes and other biocatalysts are often considered as a third category. Catalysis is ubiquitous in chemical industry of all kinds. Estimates are that 90% of all commercially produced chemical products involve catalysts at some stag ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
7-Methyl-TBD
7-Methyl-1,5,7-triazabicyclo .4.0ec-5-ene (mTBD) is a bicyclic strong guanidine base (p''K''a = 25.43 in CH3CN and p''K''a = 17.9 in THF). mTBD, like 1,5,7-triazabicyclo .4.0ec-5-ene and other guanidine super bases, can be used as a catalyst in a variety of chemical reactions. It also reacts with CO2, which could make it useful for carbon capture and storage. When brought into contact with some acids, mTBD reacts to form an ionic liquid. Some of these ionic liquids can dissolve cellulose Cellulose is an organic compound with the formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units. Cellulose is an important structural component of the primary cell w .... References {{DEFAULTSORT:Methyl-1,5,7-triazabicyclo(4.4.0)dec-5-ene, 7- Catalysts Guanidines Pyrimidopyrimidines Amines Reagents for organic chemistry Superbases ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ditungsten Tetra(hpp)
Tetrakis(hexahydropyrimidinopyrimidine)ditungsten(II), known as ditungsten tetra(hpp), is the name of the coordination compound with the formula W2(hpp)4. This material consists of a pair of tungsten centers linked by the conjugate base of four hexahydropyrimidopyrimidine (hpp) ligands. It adopts a structure sometimes called a Chinese lantern structure or paddlewheel compound, the prototype being copper(II) acetate. The molecule is of research interest because it has the lowest ionization energy (3.51 eV) of all stable chemical elements or chemical compounds as of the year 2005. This value is even lower than of caesium with 3.89 eV (or 375 kJ/mol) located at the extreme left lower corner of the periodic table (although francium is at a lower position in the periodic table compared to caesium, it has a higher ionization energy and is radioactive) or known metallocene reducing agents such as decamethylcobaltocene with 4.71 eV. Preparation This coordinat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand
In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environment ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron-rich
In chemistry, electron-rich is jargon that is used in multiple related meanings with either or both kinetic and thermodynamic implications: *with regards to electron-transfer, electron-rich species have low ionization energy and/or are reducing agents. Tetrakis(dimethylamino)ethylene is an electron-rich alkene because, unlike ethylene, it forms isolable radical cation. In contrast, electron-poor alkene tetracyanoethylene is an electron acceptor, forming isolable anions. *with regards to acid-base reactions, electron-rich species have high pKa's and react with weak Lewis acids. *with regards to nucleophilic substitution reactions, electron-rich species are relatively strong nucleophiles, as judged by rates of attack by electrophiles. For example, compared to benzene, pyrrole is more rapidly attacked by electrophiles. Pyrrole is therefore considered to be an electron-rich aromatic ring. Similarly, benzene derivatives with electron-donating groups (EDGs) are attacked by ele ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-3D-balls.png)