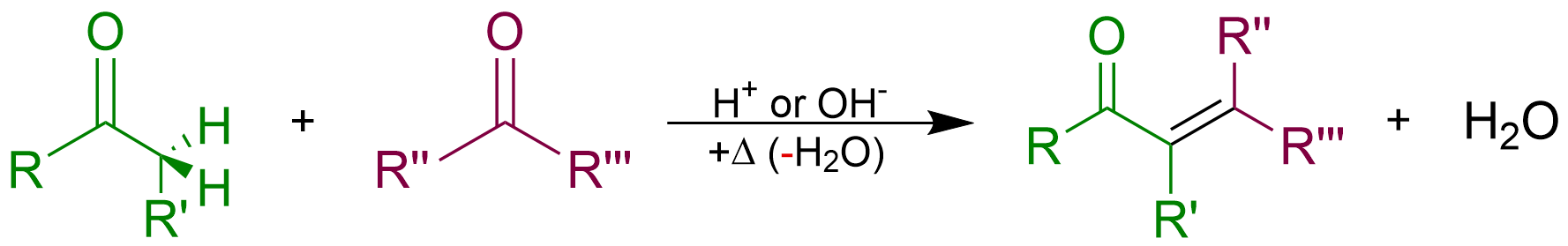|
Triacetone Amine
Triacetonamine is an organic compound with the formula OC(CH2CMe2)2NH (where Me = CH3). It is a colorless or white solid that melts near room temperature. The compound is an intermediate in the preparation of 2,2,6,6-tetramethylpiperidine, a sterically hindered base and precursor to the reagent called TEMPO. Triacetonamine is formed by the poly- aldol condensation of acetone in the presence of ammonia and calcium chloride: :3 (CH3)2CO + NH3 → OC(CH2CMe2)2NH + 2 H2O Reductive amination of triacetonamine gives 4-amino-2,2,6,6-tetramethylpiperidine. It is primarily used as a stabilizer for plastics, typically via its conversion to number of hindered amine light stabilizers, but also finds use as a chemical feedstock. It is used to prepare the hindered amine 2,2,6,6-tetramethylpiperidine, CH2 H2C(CH3)2sub>2NH, as well as the radical oxidizer 4-Hydroxy-TEMPO 4-Hydroxy-TEMPO or TEMPOL, formally 4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl, is a heterocyclic compoun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic compound, heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless liquid with an odor described as objectionable, and typical of amines. The name comes from the genus name ''Piper (genus), Piper'', which is the Latin word for Black pepper, pepper. Although piperidine is a common organic compound, it is best known as a representative structure element within many pharmaceuticals and alkaloids, such as natural-occurring Solenopsin, solenopsins. Production Piperidine was first reported in 1850 by the Scottish chemist Thomas Anderson (chemist), Thomas Anderson and again, independently, in 1852 by the French chemist Auguste André Thomas Cahours, Auguste Cahours, who named it. Both of them obtained piperidine by reacting piperine with nitric acid. Industrially, piperidine is produced by the hydrogenation o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon- hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. The study of the properties, reactions, and syntheses of organic compounds comprise the discipline known as organic chemistry. For historical reasons, a few classes of carbon-containing compounds (e.g., carbonate salts and cyanide salts), along with a few other exceptions (e.g., carbon dioxide, hydrogen cyanide), are not classified as organic compounds and are considered inorganic. Other than those just named, little consensus exists among chemists on precisely which carbon-containing compounds are excluded, making any rigorous definition of an organic compound elusive. Although organic compounds make up only a small percentage of Earth's crust, they are of central importance because all known life is based on organic compounds. Livin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TEMPO
In musical terminology, tempo ( Italian, 'time'; plural ''tempos'', or ''tempi'' from the Italian plural) is the speed or pace of a given piece. In classical music, tempo is typically indicated with an instruction at the start of a piece (often using conventional Italian terms) and is usually measured in beats per minute The minute is a unit of time usually equal to (the first sexagesimal fraction) of an hour, or 60 seconds. In the UTC time standard, a minute on rare occasions has 61 seconds, a consequence of leap seconds (there is a provision to insert a nega ... (or bpm). In modern classical compositions, a " metronome mark" in beats per minute may supplement or replace the normal tempo marking, while in modern genres like electronic dance music, tempo will typically simply be stated in BPM. Tempo may be separated from articulation and meter (music), meter, or these aspects may be indicated along with tempo, all contributing to the overall texture (music), texture. W ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldol Condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to give a conjugated enone. The overall reaction is as follows (where the Rs can be H): Aldol condensations are important in organic synthesis and biochemistry as ways to form carbon–carbon bonds. In its usual form, it involves the nucleophilic addition of a ketone enolate to an aldehyde to form a β-hydroxy ketone, or "aldol" (aldehyde + alcohol), a structural unit found in many naturally occurring molecules and pharmaceuticals. The term ''aldol condensation'' is also commonly used, especially in biochemistry, to refer to just the first (addition) stage of the process—the aldol reaction itself—as catalyzed by aldolases. However, this is formally an addition reaction rather than a condensation reaction because it does n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium Chloride
Calcium chloride is an inorganic compound, a salt with the chemical formula . It is a white crystalline solid at room temperature, and it is highly soluble in water. It can be created by neutralising hydrochloric acid with calcium hydroxide. Calcium chloride is commonly encountered as a hydrated solid with generic formula , where ''n'' = 0, 1, 2, 4, and 6. These compounds are mainly used for de-icing and dust control. Because the anhydrous salt is hydroscopic and deliquescent, it is used as a desiccant.Robert Kemp, Suzanne E. Keegan "Calcium Chloride" in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH, Weinheim. Uses De-icing and freezing-point depression By depressing the freezing point of water, calcium chloride is used to prevent ice formation and is used to de-ice. This application consumes the greatest amount of calcium chloride. Calcium chloride is relatively harmless to plants and soil. As a deicing agent, it is much more effective at lower temperat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
James M
James is a common English language surname and given name: *James (name), the typically masculine first name James * James (surname), various people with the last name James James or James City may also refer to: People * King James (other), various kings named James * Saint James (other) * James (musician) * James, brother of Jesus Places Canada * James Bay, a large body of water * James, Ontario United Kingdom * James College, a college of the University of York United States * James, Georgia, an unincorporated community * James, Iowa, an unincorporated community * James City, North Carolina * James City County, Virginia ** James City (Virginia Company) ** James City Shire * James City, Pennsylvania * St. James City, Florida Arts, entertainment, and media * ''James'' (2005 film), a Bollywood film * ''James'' (2008 film), an Irish short film * ''James'' (2022 film), an Indian Kannada-language film * James the Red Engine, a character in ''Tho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reductive Amination
Reductive amination (also known as reductive alkylation) is a form of amination that involves the conversion of a carbonyl group to an amine via an intermediate imine. The carbonyl group is most commonly a ketone or an aldehyde. It is considered the most important way to make amines, and a majority of amines made in the pharmaceutical industry are made this way. Reaction process In this organic reaction, the amine first reacts with the carbonyl group to form a hemiaminal species, which subsequently loses one molecule of water in a reversible manner by alkylimino-de-oxo-bisubstitution, to form the imine. The equilibrium between aldehyde/ketone and imine can be shifted toward imine formation by removal of the formed water through physical or chemical means. This intermediate imine can then be isolated and reduced with a suitable reducing agent (e.g., sodium borohydride). This method is sometimes called indirect reductive amination. In a separate approach, imine formation and redu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-Amino-2,2,6,6-tetramethylpiperidine
4-Amino-2,2,6,6-tetramethyl-4-piperidine is an organic compound with the formula H2NCH(CH2CMe2)2NH (where Me = CH3). Classified as a diamine, it is a colorless oily liquid. The compound is an intermediate in the preparation of Bobbitt's salt, an oxidant used in organic synthesis. It is prepared by the reductive amination of the corresponding ketone: :OC(CH2CMe2)2NH + NH3 + H2 → H2NCH(CH2CMe2)2NH + H2O Compound Properties Boiling point is 188.5 °C Melting point is 17 °C. Density is 0.8966 g/cm3 @ Temp: 20 °C Toxicity A study by Truda et al, has reported the median lethal dose LD(50) as 906mg/kg in rats. . Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(5), Pg. 53, 1984/ref> References Related compounds *Pempidine Pempidine is a ganglion-blocking drug, first reported in 1958 by two research groups working independently, and introduced as an oral treatment for hypertension. Pharmacology Reports on th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hindered Amine Light Stabilizers
Hindered amine light stabilizers (HALS) are chemical compounds containing an amine functional group that are used as stabilizers in plastics and polymers. These compounds are typically derivatives of tetramethylpiperidine and are primarily used to protect the polymers from the effects of photo-oxidation; as opposed to other forms of polymer degradation such as ozonolysis. They are also increasingly being used as thermal stabilizers, particularly for low and moderate level of heat, however during the high temperature processing of polymers (e.g. injection moulding) they remain less effective than traditional phenolic antioxidants. Mechanism of action HALS do not absorb UV radiation, but act to inhibit degradation of the polymer by continuously and cyclically removing free radicals that are produced by photo-oxidation of the polymer. The overall process is sometimes referred to as the Denisov cycle, after Evguenii T. Denisov and is exceedingly complex. Broadly, HALS react with t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Steric Hindrance
Steric effects arise from the spatial arrangement of atoms. When atoms come close together there is a rise in the energy of the molecule. Steric effects are nonbonding interactions that influence the shape ( conformation) and reactivity of ions and molecules. Steric effects complement electronic effects, which dictate the shape and reactivity of molecules. Steric repulsive forces between overlapping electron clouds result in structured groupings of molecules stabilized by the way that opposites attract and like charges repel. Steric hindrance Steric hindrance is a consequence of steric effects. Steric hindrance is the slowing of chemical reactions due to steric bulk. It is usually manifested in ''intermolecular reactions'', whereas discussion of steric effects often focus on ''intramolecular interactions''. Steric hindrance is often exploited to control selectivity, such as slowing unwanted side-reactions. Steric hindrance between adjacent groups can also affect torsional ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-Hydroxy-TEMPO
4-Hydroxy-TEMPO or TEMPOL, formally 4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl, is a heterocyclic compound. Like the related TEMPO, it is used as a catalyst and chemical oxidant by virtue of being a stable aminoxyl radical. Its major appeal over TEMPO is that is less expensive, being produced from triacetone amine, which is itself made via the condensation of acetone and ammonia. This makes it economically viable on an industrial scale. In biochemical research, 4-hydroxy-TEMPO has been investigated as an agent for limiting reactive oxygen species. It catalyzes the disproportionation of superoxide, facilitates hydrogen peroxide Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ... metabolism, and inhibits Fenton chemistry. 4-Hydroxy-TEMPO, along with related nitroxides, are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
.jpg)