|
Thrombin
Thrombin (, ''fibrinogenase'', ''thrombase'', ''thrombofort'', ''topical'', ''thrombin-C'', ''tropostasin'', ''activated blood-coagulation factor II'', ''blood-coagulation factor IIa'', ''factor IIa'', ''E thrombin'', ''beta-thrombin'', ''gamma-thrombin'') is a serine protease, an enzyme that, in humans, is encoded by the ''F2'' gene. Prothrombin (coagulation factor II) is proteolytically cleaved to form thrombin in the clotting process. Thrombin in turn acts as a serine protease that converts soluble fibrinogen into insoluble strands of fibrin, as well as catalyzing many other coagulation-related reactions. History After the description of fibrinogen and fibrin, Alexander Schmidt hypothesised the existence of an enzyme that converts fibrinogen into fibrin in 1872. Prothrombin was discovered by Pekelharing in 1894. Physiology Synthesis Thrombin is produced by the enzymatic cleavage of two sites on prothrombin by activated Factor X (Xa). The activity of factor Xa is greatly ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antithrombin
Antithrombin (AT) is a small glycoprotein that inactivates several enzymes of the coagulation system. It is a 432-amino-acid protein produced by the liver. It contains three disulfide bonds and a total of four possible glycosylation sites. α-Antithrombin is the dominant form of antithrombin found in blood plasma and has an oligosaccharide occupying each of its four glycosylation sites. A single glycosylation site remains consistently un-occupied in the minor form of antithrombin, β-antithrombin. Its activity is increased manyfold by the anticoagulant drug heparin, which enhances the binding of antithrombin to factor IIa (prothrombin) and factor Xa. Nomenclature Antithrombin is also termed antithrombin III (AT III). The designations antithrombin I through to antithrombin IV originate in early studies carried out in the 1950s by Seegers, Johnson and Fell. Antithrombin I (AT I) refers to the absorption of thrombin onto fibrin after thrombin has activated fibrinogen. Antithro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Prothrombinase
The prothrombinase complex consists of the serine protease, Factor Xa, and the protein cofactor, Factor Va. The complex assembles on negatively charged phospholipid membranes in the presence of calcium ions. The prothrombinase complex catalyzes the conversion of prothrombin (Factor II), an inactive zymogen, to thrombin (Factor IIa), an active serine protease. The activation of thrombin is a critical reaction in the coagulation cascade, which functions to regulate hemostasis in the body. To produce thrombin, the prothrombinase complex cleaves two peptide bonds in prothrombin, one after Arg271 and the other after Arg320. Although it has been shown that Factor Xa can activate prothrombin when unassociated with the prothrombinase complex, the rate of thrombin formation is severely decreased under such circumstances. The prothrombinase complex can catalyze the activation of prothrombin at a rate 3 x 105-fold faster than can Factor Xa alone. Thus, the prothrombinase complex is required ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
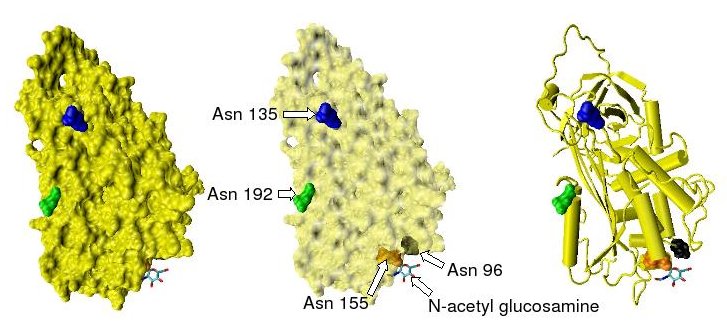
Prothrombin
Thrombin (, ''fibrinogenase'', ''thrombase'', ''thrombofort'', ''topical'', ''thrombin-C'', ''tropostasin'', ''activated blood-coagulation factor II'', ''blood-coagulation factor IIa'', ''factor IIa'', ''E thrombin'', ''beta-thrombin'', ''gamma-thrombin'') is a serine protease, an enzyme that, in humans, is encoded by the ''F2'' gene. Prothrombin (coagulation factor II) is proteolytically cleaved to form thrombin in the clotting process. Thrombin in turn acts as a serine protease that converts soluble fibrinogen into insoluble strands of fibrin, as well as catalyzing many other coagulation-related reactions. History After the description of fibrinogen and fibrin, Alexander Schmidt hypothesised the existence of an enzyme that converts fibrinogen into fibrin in 1872. Prothrombin was discovered by Pekelharing in 1894. Physiology Synthesis Thrombin is produced by the enzymatic cleavage of two sites on prothrombin by activated Factor X (Xa). The activity of factor Xa is greatl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coagulation Full
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechanism of coagulation involves activation, adhesion and aggregation of platelets, as well as deposition and maturation of fibrin. Coagulation begins almost instantly after an injury to the endothelium lining a blood vessel. Exposure of blood to the subendothelial space initiates two processes: changes in platelets, and the exposure of subendothelial tissue factor to plasma factor VII, which ultimately leads to cross-linked fibrin formation. Platelets immediately form a plug at the site of injury; this is called ''primary hemostasis. Secondary hemostasis'' occurs simultaneously: additional coagulation (clotting) factors beyond factor VII ( listed below) respond in a cascade to form fibrin strands, which strengthen the platelet plug. Disorders of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coagulation
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechanism of coagulation involves activation, adhesion and aggregation of platelets, as well as deposition and maturation of fibrin. Coagulation begins almost instantly after an injury to the endothelium lining a blood vessel. Exposure of blood to the subendothelial space initiates two processes: changes in platelets, and the exposure of subendothelial tissue factor to plasma factor VII, which ultimately leads to cross-linked fibrin formation. Platelets immediately form a plug at the site of injury; this is called ''primary hemostasis. Secondary hemostasis'' occurs simultaneously: additional coagulation (clotting) factors beyond factor VII ( listed below) respond in a cascade to form fibrin strands, which strengthen the platelet plug. Disorders of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Factor X
Factor X, also known by the eponym Stuart–Prower factor, is an enzyme () of the coagulation cascade. It is a serine endopeptidase (protease group S1, PA clan). Factor X is synthesized in the liver and requires vitamin K for its synthesis. Factor X is activated, by hydrolysis, into factor Xa by both factor IX (with its cofactor, factor VIII in a complex known as ''intrinsic tenase'') and factor VII with its cofactor, tissue factor (a complex known as ''extrinsic tenase''). It is therefore the first member of the ''final common pathway'' or ''thrombin pathway''. It acts by cleaving prothrombin in two places (an arg- thr and then an arg-ile bond), which yields the active thrombin. This process is optimized when factor Xa is complexed with activated co-factor V in the prothrombinase complex. Factor Xa is inactivated by protein Z-dependent protease inhibitor (ZPI), a serine protease inhibitor (serpin). The affinity of this protein for factor Xa is increased 1000-fold by the pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Factor V
Factor V (pronounced factor five) is a protein of the coagulation system, rarely referred to as proaccelerin or labile factor. In contrast to most other coagulation factors, it is not enzymatically active but functions as a cofactor. Deficiency leads to predisposition for hemorrhage, while some mutations (most notably factor V Leiden) predispose for thrombosis. Genetics The gene for factor V is located on the first chromosome (1q24). It is genomically related to the family of multicopper oxidases, and is homologous to coagulation factor VIII. The gene spans 70 kb, consists of 25 exons, and the resulting protein has a relative molecular mass of approximately 330kDa. Structure Factor V protein consists of six domains: A1-A2-B-A3-C1-C2. The A domains are homologous to the A domains of the copper-binding protein ceruloplasmin, and form a triangular as in that protein. A copper ion is bound in the A1-A3 interface, and A3 interacts with the plasma. The C domains belong to the p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Factor V
Factor V (pronounced factor five) is a protein of the coagulation system, rarely referred to as proaccelerin or labile factor. In contrast to most other coagulation factors, it is not enzymatically active but functions as a cofactor. Deficiency leads to predisposition for hemorrhage, while some mutations (most notably factor V Leiden) predispose for thrombosis. Genetics The gene for factor V is located on the first chromosome (1q24). It is genomically related to the family of multicopper oxidases, and is homologous to coagulation factor VIII. The gene spans 70 kb, consists of 25 exons, and the resulting protein has a relative molecular mass of approximately 330kDa. Structure Factor V protein consists of six domains: A1-A2-B-A3-C1-C2. The A domains are homologous to the A domains of the copper-binding protein ceruloplasmin, and form a triangular as in that protein. A copper ion is bound in the A1-A3 interface, and A3 interacts with the plasma. The C domains belong to the p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thrombomodulin
Thrombomodulin (TM), CD141 or BDCA-3 is an integral membrane protein expressed on the surface of endothelial cells and serves as a cofactor for thrombin. It reduces blood coagulation by converting thrombin to an anticoagulant enzyme from a procoagulant enzyme. Thrombomodulin is also expressed on human mesothelial cell, monocyte and a dendritic cell subset. Genetics and structure In humans, thrombomodulin is encoded by the gene. The protein has a molecular mass of 74k Da, and consists of a single chain with six tandemly repeated EGF-like domains, a Serine/Threonine-rich spacer and a transmembrane domain. It is a member of the C-type lectin domain (CTLD) group 14 family. Function Thrombomodulin functions as a cofactor in the thrombin-induced activation of protein C in the anticoagulant pathway by forming a 1:1 stoichiometric complex with thrombin. This raises the speed of protein C activation thousandfold. Thrombomodulin-bound thrombin has procoagulant effect at the same ti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fibrinogen
Fibrinogen (factor I) is a glycoprotein complex, produced in the liver, that circulates in the blood of all vertebrates. During tissue and vascular injury, it is converted enzymatically by thrombin to fibrin and then to a fibrin-based blood clot. Fibrin clots function primarily to occlude blood vessels to stop bleeding. Fibrin also binds and reduces the activity of thrombin. This activity, sometimes referred to as antithrombin I, limits clotting. Fibrin also mediates blood platelet and endothelial cell spreading, tissue fibroblast proliferation, capillary tube formation, and angiogenesis and thereby promotes revascularization and wound healing. Reduced and/or dysfunctional fibrinogens occur in various congenital and acquired human fibrinogen-related disorders. These disorders represent a group of rare conditions in which individuals may present with severe episodes of pathological bleeding and thrombosis; these conditions are treated by supplementing blood fibrinogen levels an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Warfarin
Warfarin, sold under the brand name Coumadin among others, is a medication that is used as an anticoagulant (blood thinner). It is commonly used to prevent blood clots such as deep vein thrombosis and pulmonary embolism, and to prevent stroke in people who have atrial fibrillation, valvular heart disease, or artificial heart valves. Less commonly, it is used following ST-segment elevation myocardial infarction and orthopedic surgery. It is generally taken by mouth, but may also be used intravenously. The common side effect is bleeding. Less common side effects may include areas of tissue damage and purple toes syndrome. Use is not recommended during pregnancy. The effects of warfarin typically should be monitored by checking prothrombin time (INR) every one to four weeks. Many other medications and dietary factors can interact with warfarin, either increasing or decreasing its effectiveness. The effects of warfarin may be reversed with phytomenadione (vitamin K1), fresh f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vitamin K
Vitamin K refers to structurally similar, fat-soluble vitamers found in foods and marketed as dietary supplements. The human body requires vitamin K for post-synthesis modification of certain proteins that are required for blood coagulation (K from ''Koagulation'', German for "coagulation") or for controlling binding of calcium in bones and other tissues. The complete synthesis involves final modification of these so-called "Gla proteins" by the enzyme gamma-glutamyl carboxylase that uses vitamin K as a cofactor. Vitamin K is used in the liver as the intermediate VKH2 to deprotonate a glutamate residue and then is reprocessed into vitamin K through a vitamin K oxide intermediate. The presence of uncarboxylated proteins indicates a vitamin K deficiency. Carboxylation allows them to bind (chelate) calcium ions, which they cannot do otherwise. Without vitamin K, blood coagulation is seriously impaired, and uncontrolled bleeding occurs. Research suggests that deficiency of v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |