|
Thiophene-3-acetic Acid
Thiophene-3-acetic acid is an organosulfur compound with the formula HO2CCH2C4H3S. It is a white solid. It is one of two isomers of thiophene acetic acid, the other being thiophene-2-acetic acid. Thiophene-3-acetic acid has attracted attention as a precursor to functionalized derivatives of polythiophene Polythiophenes (PTs) are polymerized thiophenes, a sulfur heterocyclic compound, heterocycle. The parent PT is an insoluble colored solid with the formula (C4H2S)n. The rings are linked through the 2- and 5-positions. Poly(alkylthiophene)s hav .... References Thiophenes Acetic acids {{organic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organosulfur Compound
Organosulfur compounds are organic compounds that contain sulfur. They are often associated with foul odors, but many of the sweetest compounds known are organosulfur derivatives, e.g., saccharin. Nature abounds with organosulfur compounds—sulfur is vital for life. Of the 20 common amino acids, two ( cysteine and methionine) are organosulfur compounds, and the antibiotics penicillin and sulfa drugs both contain sulfur. While sulfur-containing antibiotics save many lives, sulfur mustard is a deadly chemical warfare agent. Fossil fuels, coal, petroleum, and natural gas, which are derived from ancient organisms, necessarily contain organosulfur compounds, the removal of which is a major focus of oil refineries. Sulfur shares the chalcogen group with oxygen, selenium, and tellurium, and it is expected that organosulfur compounds have similarities with carbon–oxygen, carbon–selenium, and carbon–tellurium compounds. A classical chemical test for the detection of sulfur co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiophene-2-acetic Acid
Thiophene-2-acetic acid is the organosulfur compound with the formula HO2CCH2C4H3S. Together with thiophene-3-acetic acid, it is one of two isomeric thiophene acetic acids. Preparation and use It is prepared from 2-acetylthiophene. It is a precursor to the antibiotics cephaloridine and cephalothin Cefalotin (INN) or cephalothin (USAN) is a first-generation cephalosporin antibiotic. It was the first cephalosporin marketed (1964) and continues to be widely used. It is an intravenously administered agent with a similar antimicrobial spec ....{{cite book, first = Jonathan , last = Swanston , chapter = Thiophene , title = Ullmann's Encyclopedia of Industrial Chemistry , publisher = Wiley-VCH , location = Weinheim , date = 2006 , doi = 10.1002/14356007.a26_793.pub2, isbn = 3527306730 . References Thiophenes Carboxylic acids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polythiophene
Polythiophenes (PTs) are polymerized thiophenes, a sulfur heterocyclic compound, heterocycle. The parent PT is an insoluble colored solid with the formula (C4H2S)n. The rings are linked through the 2- and 5-positions. Poly(alkylthiophene)s have Alkyl, alkyl substituents at the 3- or 4-position(s). They are also colored solids, but tend to be soluble in organic solvents. PTs become Conductive polymer, conductive when oxidized. The electrical resistivity and conductivity, electrical conductivity results from the Delocalized electron, delocalization of electrons along the polymer backbone. Conductivity however is not the only interesting property resulting from electron delocalization. The optical properties of these materials respond to environmental stimuli, with dramatic color shifts in response to changes in solvent, temperature, Electric potential, applied potential, and binding to other molecules. Changes in both color and conductivity are induced by the same mechanism, tw ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
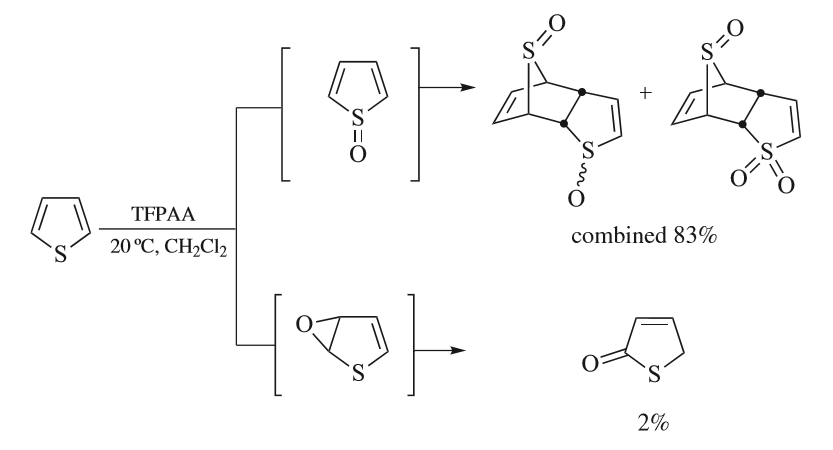
Thiophenes
Thiophene is a heterocyclic compound with the formula C4H4S. Consisting of a planar five-membered ring, it is aromatic as indicated by its extensive substitution reactions. It is a colorless liquid with a benzene-like odor. In most of its reactions, it resembles benzene. Compounds analogous to thiophene include furan (C4H4O), selenophene (C4H4Se) and pyrrole (C4H4NH), which each vary by the heteroatom in the ring. Isolation and occurrence Thiophene was discovered as a contaminant in benzene. It was observed that isatin (an indole) forms a blue dye if it is mixed with sulfuric acid and crude benzene. The formation of the blue indophenin had long been believed to be a reaction of benzene itself. Viktor Meyer was able to isolate thiophene as the actual substance responsible for this reaction. Thiophene and especially its derivatives occur in petroleum, sometimes in concentrations up to 1–3%. The thiophenic content of oil and coal is removed via the hydrodesulfurization (HDS) pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |