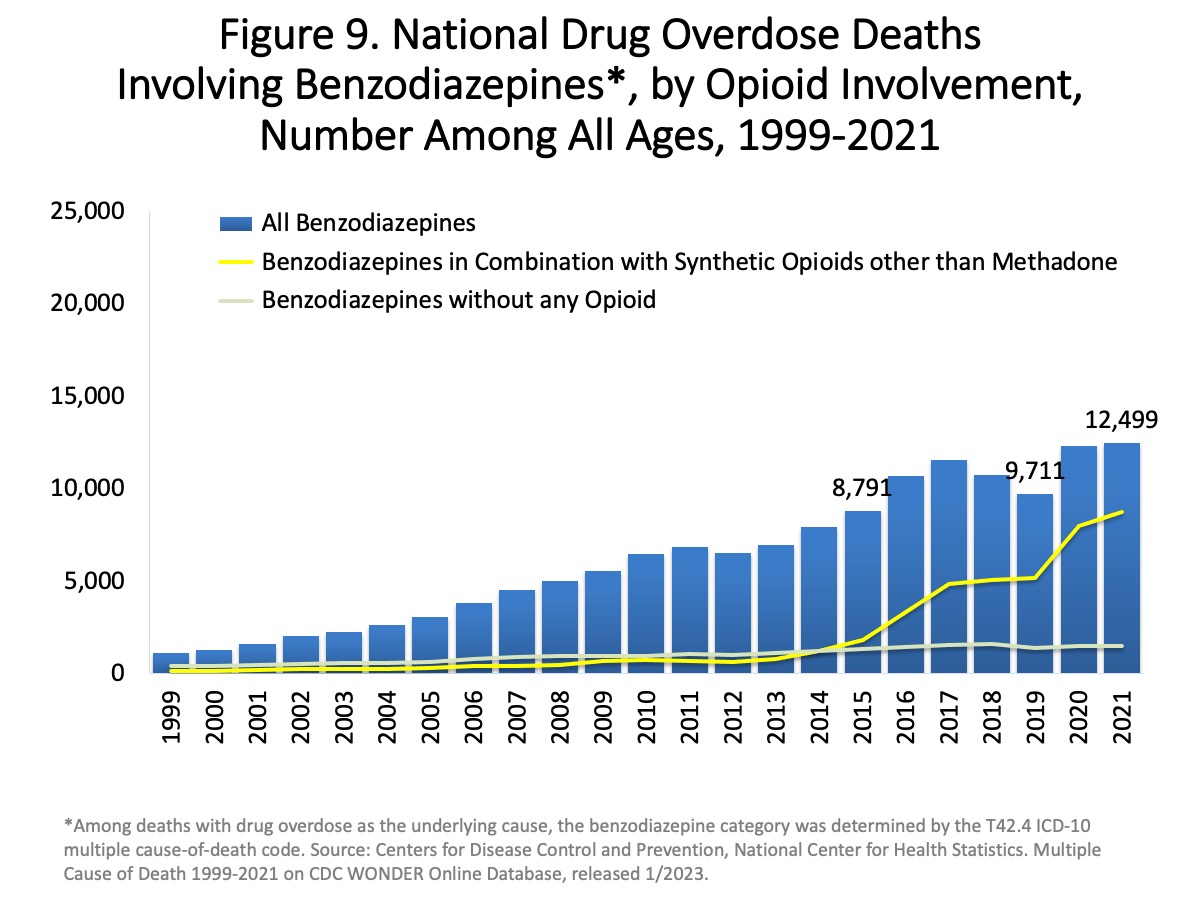
|
Thiambutene
The Thiambutenes are a family of opioid analgesic drugs developed at the British research laboratory of Burroughs-Wellcome in the late 1940s. The parent compound thiambutene has no analgesic effects, but several compounds from this group are analgesics with around the same potency as morphine. Notable compounds include dimethylthiambutene, diethylthiambutene, ethylmethylthiambutene, pyrrolidinylthiambutene and piperidylthiambutene. Of these, ethylmethylthiambutene is the most potent, with 1.3x the potency of morphine, pyrrolidinylthiambutene is the least potent at 0.7x, and the rest are all around the same potency as morphine. Diethylthiambutene has been the most widely used, mainly in veterinary medicine. All of these compounds produced anticholinergic and antihistamine side effects, except for two of the weaker compounds diallylthiambutene and morpholinylthiambutene. They also all have a chiral centre on the alpha carbon (where the R1 group is attached) and so have two stereois ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiambutene General
The Thiambutenes are a family of opioid analgesic drugs developed at the British research laboratory of Burroughs-Wellcome in the late 1940s. The parent compound thiambutene has no analgesic effects, but several compounds from this group are analgesics with around the same potency as morphine. Notable compounds include dimethylthiambutene, diethylthiambutene, ethylmethylthiambutene, pyrrolidinylthiambutene and piperidylthiambutene. Of these, ethylmethylthiambutene is the most potent, with 1.3x the potency of morphine, pyrrolidinylthiambutene is the least potent at 0.7x, and the rest are all around the same potency as morphine. Diethylthiambutene has been the most widely used, mainly in veterinary medicine. All of these compounds produced anticholinergic and antihistamine side effects, except for two of the weaker compounds diallylthiambutene and morpholinylthiambutene. They also all have a chiral centre on the alpha carbon (where the R1 group is attached) and so have two stereois ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Opioid
Opioids are substances that act on opioid receptors to produce morphine-like effects. Medically they are primarily used for pain relief, including anesthesia. Other medical uses include suppression of diarrhea, replacement therapy for opioid use disorder, reversing opioid overdose, and suppressing cough. Extremely potent opioids such as carfentanil are approved only for veterinary use. Opioids are also frequently used non-medically for their euphoric effects or to prevent withdrawal. Opioids can cause death and have been used for executions in the United States. Side effects of opioids may include itchiness, sedation, nausea, respiratory depression, constipation, and euphoria. Long-term use can cause tolerance, meaning that increased doses are required to achieve the same effect, and physical dependence, meaning that abruptly discontinuing the drug leads to unpleasant withdrawal symptoms. The euphoria attracts recreational use, and frequent, escalating recreational use of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Opioids
Opioids are substances that act on opioid receptors to produce morphine-like effects. Medically they are primarily used for pain relief, including anesthesia. Other medical uses include suppression of diarrhea, replacement therapy for opioid use disorder, reversing opioid overdose, and suppressing cough. Extremely potent opioids such as carfentanil are approved only for veterinary use. Opioids are also frequently used non-medically for their euphoric effects or to prevent withdrawal. Opioids can cause death and have been used for executions in the United States. Side effects of opioids may include itchiness, sedation, nausea, respiratory depression, constipation, and euphoria. Long-term use can cause tolerance, meaning that increased doses are required to achieve the same effect, and physical dependence, meaning that abruptly discontinuing the drug leads to unpleasant withdrawal symptoms. The euphoria attracts recreational use, and frequent, escalating recreational use of o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylthiambutene
Diethylthiambutene (Thiambutene, Themalon, Diethibutin, ''N'',''N''-Diethyl-1-methyl-3,3-di-2-thienylallylamine) is an opioid analgesic drug developed in the 1950s which was mainly used as an anesthetic in veterinary medicine and continues, along with the other two thiambutenes dimethylthiambutene and ethylmethylthiambutene Ethylmethylthiambutene (; Emethibutin) is an opioid analgesic drug from the thiambutene family, around 1.3x the potency of morphine Morphine is a strong opiate that is found naturally in opium, a dark brown resin in poppies ('' Papaver ... to be used for this purpose, particularly in Japan. It is now under international control under Schedule I of the UN Single Convention On Narcotic Drugs 1961, presumably due to high abuse potential, although little more information is available. It is listed under Schedule I of the US Controlled Substances Act as a Narcotic and has an ACSCN of 9616 with zero annual manufacturing quota as of 2013. Synthesis Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethylthiambutene
Diethylthiambutene (Thiambutene, Themalon, Diethibutin, ''N'',''N''-Diethyl-1-methyl-3,3-di-2-thienylallylamine) is an opioid analgesic drug developed in the 1950s which was mainly used as an anesthetic in veterinary medicine and continues, along with the other two thiambutenes dimethylthiambutene and ethylmethylthiambutene Ethylmethylthiambutene (; Emethibutin) is an opioid analgesic drug from the thiambutene family, around 1.3x the potency of morphine Morphine is a strong opiate that is found naturally in opium, a dark brown resin in poppies ('' Papaver ... to be used for this purpose, particularly in Japan. It is now under international control under Schedule I of the UN Single Convention On Narcotic Drugs 1961, presumably due to high abuse potential, although little more information is available. It is listed under Schedule I of the US Controlled Substances Act as a Narcotic and has an ACSCN of 9616 with zero annual manufacturing quota as of 2013. Synthesis Th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrrolidinylthiambutene
Pyrrolidinylthiambutene is an opioid analgesic drug from the thiambutene family with around 3/4 of the potency of morphine Morphine is a strong opiate that is found naturally in opium, a dark brown resin in poppies (''Papaver somniferum''). It is mainly used as a analgesic, pain medication, and is also commonly used recreational drug, recreationally, or to make .... It would be considered an illegal controlled substance analogue in some countries such as the US, Australia and New Zealand, but is legal in countries not possessing a controlled-substances-analog-act equivalent. References Opioids Thiophenes Pyrrolidines Mu-opioid receptor agonists {{analgesic-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Group
In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry's nomenclature of organic chemistry for a saturated two-carbon moiety in a molecule, while the prefix "''eth-''" is used to indicate the presence of two carbon atoms in the molecule. Ethylation Ethylation is the formation of a compound by introduction of the ethyl group. The most widely practiced example of this reaction is the ethylation of benzene with ethylene to yield ethylbenzene, a precursor to styrene, which is a precursor to polystyrene. Approximately 24.7 million tons of ethylbenzene were produced in 1999. :: Many ethyl-containing compounds are generated by electrophilic ethylation, i.e. treatment of nucleophiles with sources of Et+. Triethyloxonium tetrafluoroborate t3OF4 is such a reagent. For good nucleophiles, less electrophilic reagents are employed, such as ethyl h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Piperidylthiambutene
Piperidylthiambutene (Piperidinohton) is a synthetic opioid analgesic drug from the thiambutene family, which has around the same potency as morphine. Piperidylthiambutene is structurally distinct from fentanyl, its analogues, and other synthetic opioids previously reported. If sold or obtained for the purpose of human consumption it could be considered a controlled substance analogue in some countries such as the US, Australia and New Zealand. Piperidylthiambutene has been sold as a designer drug A designer drug is a structural or functional analog of a controlled substance that has been designed to mimic the pharmacological effects of the original drug, while avoiding classification as illegal and/or detection in standard drug tests. D ..., first appearing in late 2018. Synthesis The Grignard reaction between 3-Piperidinobutyric acid ethyl esterCID:10774378(1) and 2-Bromothiophene 003-09-4(2) gives 3. Dehydration in acid completes the synthesis. References Opioi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Piperidine
Piperidine is an organic compound with the molecular formula (CH2)5NH. This heterocyclic compound, heterocyclic amine consists of a six-membered ring containing five methylene bridges (–CH2–) and one amine bridge (–NH–). It is a colorless liquid with an odor described as objectionable, and typical of amines. The name comes from the genus name ''Piper (genus), Piper'', which is the Latin word for Black pepper, pepper. Although piperidine is a common organic compound, it is best known as a representative structure element within many pharmaceuticals and alkaloids, such as natural-occurring Solenopsin, solenopsins. Production Piperidine was first reported in 1850 by the Scottish chemist Thomas Anderson (chemist), Thomas Anderson and again, independently, in 1852 by the French chemist Auguste André Thomas Cahours, Auguste Cahours, who named it. Both of them obtained piperidine by reacting piperine with nitric acid. Industrially, piperidine is produced by the hydrogenation o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Racemic Mixture
In chemistry, a racemic mixture, or racemate (), is one that has equal amounts of left- and right-handed enantiomers of a chiral molecule or salt. Racemic mixtures are rare in nature, but many compounds are produced industrially as racemates. History The first known racemic mixture was racemic acid, which Louis Pasteur found to be a mixture of the two enantiomeric isomers of tartaric acid. He manually separated the crystals of a mixture by hand, starting from an aqueous solution of the sodium ammonium salt of racemate tartaric acid. Pasteur benefited from the fact that ammonium tartrate salt that gives enantiomeric crystals with distinct crystal forms (at 77 °F). Reasoning from the macroscopic scale down to the molecular, he reckoned that the molecules had to have non-superimposable mirror images. A sample with only a single enantiomer is an ''enantiomerically pure'' or ''enantiopure'' compound. Etymology From racemic acid found in grapes; from Latin ''racemus'', meani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrrolidine
Pyrrolidine, also known as tetrahydropyrrole, is an organic compound with the molecular formula (CH2)4NH. It is a cyclic secondary amine, also classified as a saturated heterocycle. It is a colourless liquid that is miscible with water and most organic solvents. It has a characteristic odor that has been described as "ammoniacal, fishy, shellfish-like". In addition to pyrrolidine itself, many substituted pyrrolidines are known. Production and synthesis Industrial production Pyrrolidine is prepared industrially by the reaction of 1,4-butanediol and ammonia at a temperature of 165–200 °C and a pressure of 17–21 MPa in the presence of a cobalt- and nickel oxide catalyst, which is supported on alumina. : The reaction is carried out in the liquid phase in a continuous tube- or tube bundle reactor, which is operated in the cycle gas method. The catalyst is arranged as a fixed-bed and the conversion is carried out in the downflow mode. The product is obtained after mu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
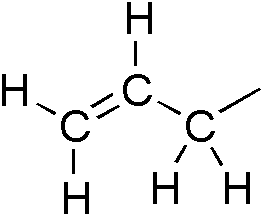
Allyl
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . In 1844, Theodor Wertheim isolated an allyl derivative from garlic oil and named it "". The term allyl applies to many compounds related to , some of which are of practical or of everyday importance, for example, allyl chloride. Allylation is any chemical reaction that adds an allyl group to a substrate. Nomenclature A site adjacent to the unsaturated carbon atom is called the allylic position or allylic site. A group attached at this site is sometimes described as allylic. Thus, "has an allylic hydroxyl group". Allylic C−H bonds are about 15% weaker than the C−H bonds in ordinary sp3 carbon centers and are thus more reactive. Benzylic and allylic are related in terms of structure, bond strength, and reactivity. Other re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |