|
Shishijimicin A
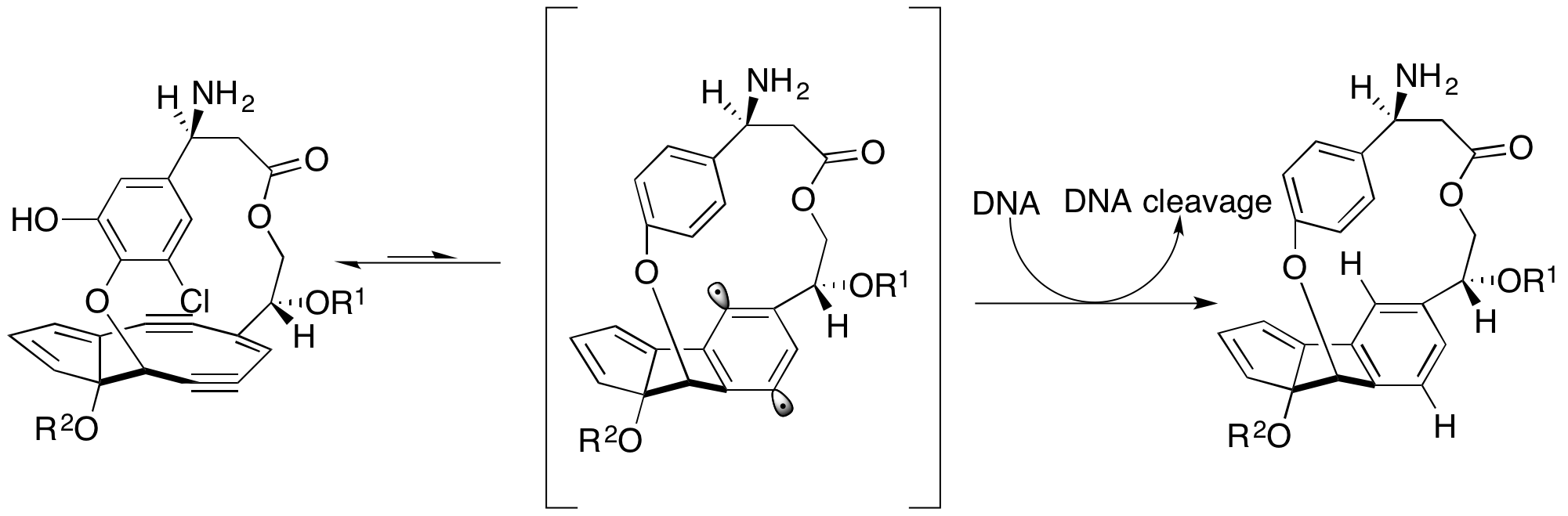
Shishijimicin A is an enediyne antibiotic isolated from '' Didemnum proliferum.'' Isolated in 2003 it is part of the family of 10 member ringed enediyne antitumor antibiotic agents, which includes: namenamicin, esperamicin and, calicheamicin. Due to its high potency from cytotoxicity, Shishjimicin A is currently undergoing testing as a possiblAntibody-antibiotic Conjugate (ADCs)cancer treatment. Laboratory tests indicate it to be “more than 1,000 times as toxic to cancer cells as the anticancer drug taxol”, also known as Paclitaxel, a prevalent chemotherapy medication. As such, theoretically, only an administration of a minuscule dose of the molecule would be necessary per each treatment. As shishjimicin A supply is scarce and the full extent of its side effects is not yet established, there is still a need for further biological and clinical studies. Total synthesis The total synthesis of shishjimicin A was published by scientists at Rice University in 2015, led by K. C. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enediyne
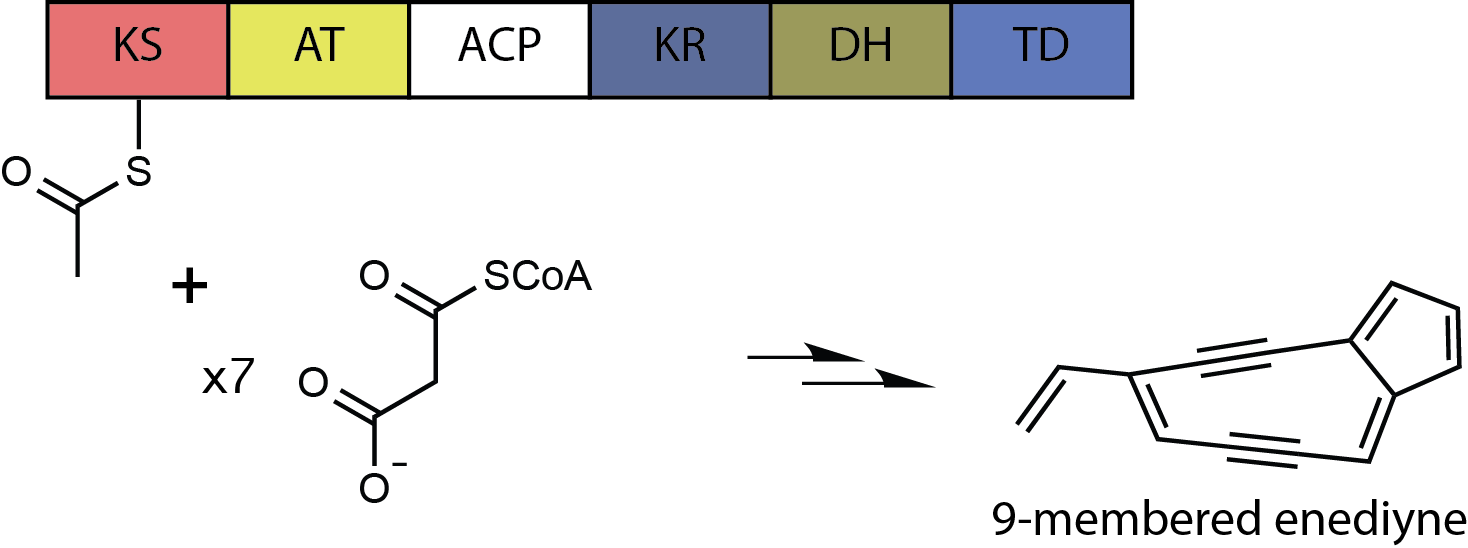
In organic chemistry, enediynes are organic compounds containing two triple bonds and one double bond. Enediynes are most notable for their limited use as antitumor antibiotics (known as enediyne Chemotherapy, anticancer antibiotics). They are efficient at inducing apoptosis in Cell biology, cells, but cannot differentiate Cancer, cancerous cells from healthy cells. Consequently, research is being conducted to increase the specificity of enediyne toxicity. Structure and reactivity A nine- or ten-membered ring containing a double bond between two triple bonds is termed the warhead of the enediyne. In this state, the warhead is inactive. Enediynes are triggered into a chemically active state via Bergman cyclization, Bergman or Myers-Saito cyclization. The triggering mechanism can be attributed to an intramolecular nucleophilic attack initiated by one of the variable regions of the molecule. Triggering can also occur via attack by an external nucleophile. Bergman cyclization re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deprotection
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis. In many preparations of delicate organic compounds, some specific parts of their molecules cannot survive the required reagents or chemical environments. Then, these parts, or groups, must be protected. For example, lithium aluminium hydride is a highly reactive but useful reagent capable of reducing esters to alcohols. It will always react with carbonyl groups, and this cannot be discouraged by any means. When a reduction of an ester is required in the presence of a carbonyl, the attack of the hydride on the carbonyl has to be prevented. For example, the carbonyl is converted into an acetal, which does not react with hydrides. The acetal is then called a protecting group for the carbonyl. After the step involving the hydride is complete, the acet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enediynes
In organic chemistry, enediynes are organic compounds containing two triple bonds and one double bond. Enediynes are most notable for their limited use as antitumor antibiotics (known as enediyne anticancer antibiotics). They are efficient at inducing apoptosis in cells, but cannot differentiate cancerous cells from healthy cells. Consequently, research is being conducted to increase the specificity of enediyne toxicity. Structure and reactivity A nine- or ten-membered ring containing a double bond between two triple bonds is termed the warhead of the enediyne. In this state, the warhead is inactive. Enediynes are triggered into a chemically active state via Bergman or Myers-Saito cyclization. The triggering mechanism can be attributed to an intramolecular nucleophilic attack initiated by one of the variable regions of the molecule. Triggering can also occur via attack by an external nucleophile. Bergman cyclization restructures the enediyne ring into two smaller rings. O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antibiotics
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention of such infections. They may either kill or inhibit the growth of bacteria. A limited number of antibiotics also possess antiprotozoal activity. Antibiotics are not effective against viruses such as the common cold or influenza; drugs which inhibit viruses are termed antiviral drugs or antivirals rather than antibiotics. Sometimes, the term ''antibiotic''—literally "opposing life", from the Greek roots ἀντι ''anti'', "against" and βίος ''bios'', "life"—is broadly used to refer to any substance used against microbes, but in the usual medical usage, antibiotics (such as penicillin) are those produced naturally (by one microorganism fighting another), whereas non-antibiotic antibacterials (such as sulfonamides and antisep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lidamycin
C-1027 or Lidamycin is an antitumor antibiotic consisting of a complex of an enediyne chromophore and an apoprotein. It shows antibiotic activity against most Gram-positive bacteria. It is one of the most potent cytotoxic molecules known, due to its induction of a higher ratio of DNA double-strand breaks than single-strand breaks. C-1027's chromophore contains a nine-membered enediyne that is responsible for most of the molecule's biological activity. Unlike other enediynes, this molecule contains no triggering mechanism. It is already primed to undergo the cycloaromatization reaction without external activation to produce the toxic 1,4-benzenoid diradical species. C-1027 can induce oxygen-independent interstrand DNA crosslinks in addition to the oxygen-dependent single- and double-stranded DNA breaks typically generated by other enediynes. This unique oxygen-independent mechanism suggests that C-1027 may be effective against hypoxic tumor cells. C-1027 shows promise as an a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kedarcidin
Kedarcidin is a chromoprotein antitumor antibiotic first isolated from an Actinomycete in 1992, comprising an ansa-bridged enediyne chromophore (shown) as well as an apoprotein that serves to stabilize the toxin in the Actinomycete. Like other members of the enediyne class of drugs—so named for the nine-or-ten-membered core structure bearing an alkene directly attached to two alkynyl appendages—kedarcidin was likely evolved to kill bacteria that compete with the producing organism. Because it achieves this by causing DNA damage, however, kedarcidin is capable of harming tumor cells, as well. Kedarcidin is thus the subject of scientific research, both for its structural complexity as well as its anticancer properties. Discovery and structure elucidation Kedarcidin was first discovered in 1992 when bioassays conducted at Bristol-Myers Squibb indicated the presence of a DNA-damaging chromoprotein in the fermentation broth of an Actinomycete strain. The involvement of a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Esperamicin
The esperamicins are chromoprotein enediyne antitumor antibiotics of bacterial origin. Esperamicin A1 is the most well studied compound in this class. Esperamcin A1 and the related enediyne calicheamicin The calicheamicins are a class of enediyne antitumor antibiotics derived from the bacterium ''Micromonospora echinospora'', with calicheamicin γ1 being the most notable. It was isolated originally in the mid-1980s from the chalky soil, or "calich ... are the two most potent antitumor agents known. The esperamicins are extremely toxic DNA splicing compounds. Oxygen and active oxygen-radical scavengers have no significant influence upon DNA strand breakage by esperamicin, but the cleavage of DNA by esperamicin is greatly accelerated in the presence of thiol compounds. The preferential cutting sites of esperamicin are at thymidylate residues, and the frequency of nucleobase attacked (T greater than C greater than A greater than G) is different from that of calicheamicin (C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calicheamicin
The calicheamicins are a class of enediyne antitumor antibiotics derived from the bacterium ''Micromonospora echinospora'', with calicheamicin γ1 being the most notable. It was isolated originally in the mid-1980s from the chalky soil, or "caliche pits", located in Kerrville, Texas. The sample was collected by a scientist working for Lederle Labs. It is extremely toxic to all cells and, in 2000, a CD33 antigen-targeted immunoconjugate N-acetyl dimethyl hydrazide calicheamicin was developed and marketed as targeted therapy against the non-solid tumor cancer acute myeloid leukemia (AML). A second calicheamicin-linked monoclonal antibody, inotuzumab ozogamicin (marketed as Besponsa) an anti-CD22-directed antibody-drug conjugate, was approved by the U.S. Food and Drug Administration on August 17, 2017, for use in the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia. Calicheamicin γ1 and the related enediyne esperamicin are the two of the m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IC50
The half maximal inhibitory concentration (IC50) is a measure of the potency of a substance in inhibiting a specific biological or biochemical function. IC50 is a quantitative measure that indicates how much of a particular inhibitory substance (e.g. drug) is needed to inhibit, ''in vitro'', a given biological process or biological component by 50%. The biological component could be an enzyme, cell, cell receptor or microorganism. IC50 values are typically expressed as molar concentration. IC50 is commonly used as a measure of antagonist drug potency in pharmacological research. IC50 is comparable to other measures of potency, such as EC50 for excitatory drugs. EC50 represents the dose or plasma concentration required for obtaining 50% of a maximum effect ''in vivo''. IC50 can be determined with functional assays or with competition binding assays. Sometimes, IC50 values are converted to the pIC50 scale. :\ce = -\log_ \ce Due to the minus sign, higher values of pIC50 indica ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hela Cells
HeLa (; also Hela or hela) is an immortalized cell line used in scientific research. It is the oldest and most commonly used human cell line. The line is derived from cervical cancer cells taken on February 8, 1951, named after Henrietta Lacks, a 31-year-old African-American mother of five, who died of cancer on October 4, 1951. The cell line was found to be remarkably durable and prolific, which allows it to be used extensively in scientific study. The cells from Lacks's cancerous cervical tumor were taken without her knowledge or consent, which was common practice in the United States at the time. Cell biologist George Otto Gey found that they could be kept alive, and developed a cell line. Previously, cells cultured from other human cells would only survive for a few days. Cells from Lacks's tumor behaved differently. History Origin In 1951, a patient named Henrietta Lacks was admitted to the Johns Hopkins Hospital with symptoms of irregular vaginal bleeding, and was s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Histone
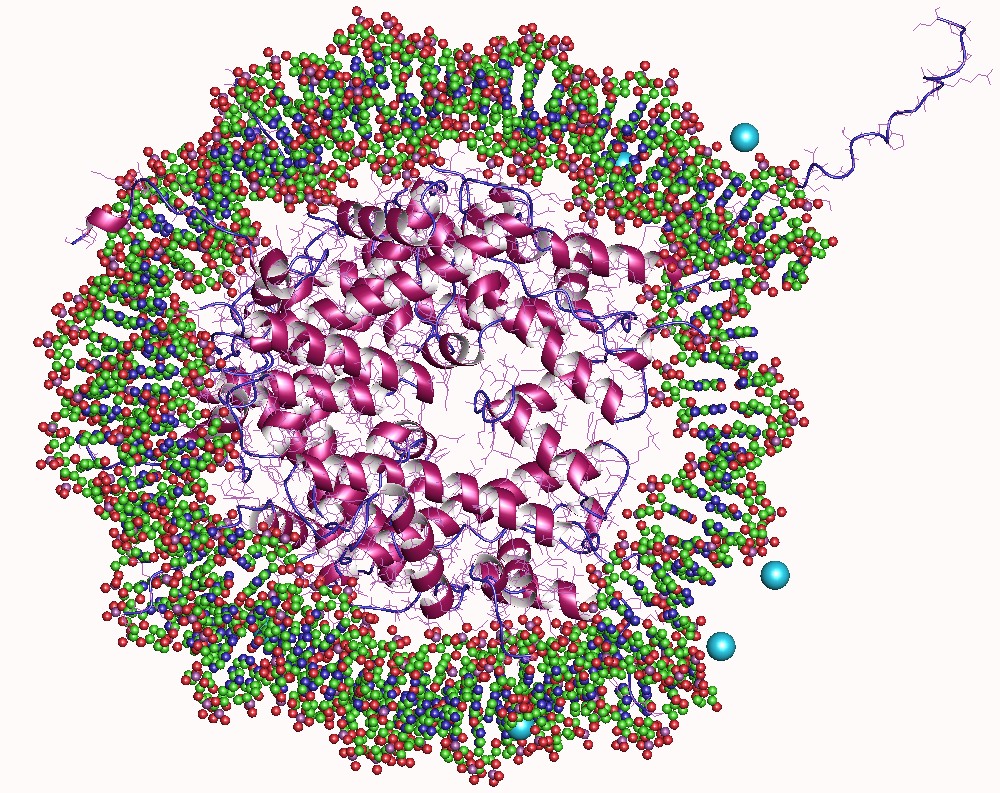
In biology, histones are highly basic proteins abundant in lysine and arginine residues that are found in eukaryotic cell nuclei. They act as spools around which DNA winds to create structural units called nucleosomes. Nucleosomes in turn are wrapped into 30-nanometer fibers that form tightly packed chromatin. Histones prevent DNA from becoming tangled and protect it from DNA damage. In addition, histones play important roles in gene regulation and DNA replication. Without histones, unwound DNA in chromosomes would be very long. For example, each human cell has about 1.8 meters of DNA if completely stretched out; however, when wound about histones, this length is reduced to about 90 micrometers (0.09 mm) of 30 nm diameter chromatin fibers. There are five families of histones which are designated H1/H5 (linker histones), H2, H3, and H4 (core histones). The nucleosome core is formed of two H2A-H2B dimers and a H3-H4 tetramer. The tight wrapping of DNA around histones ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |