|
Radioactivity In The Life Sciences
Radioactivity is generally used in life sciences for highly sensitive and direct measurements of biological phenomena, and for visualizing the location of biomolecules radiolabelled with a radioisotope. All atoms exist as stable or unstable isotopes and the latter decay at a given half-life ranging from attoseconds to billions of years; radioisotopes useful to biological and experimental systems have half-lives ranging from minutes to months. In the case of the hydrogen isotope tritium (half-life = 12.3 years) and carbon-14 (half-life = 5,730 years), these isotopes derive their importance from all organic life containing hydrogen and carbon and therefore can be used to study countless living processes, reactions, and phenomena. Most short lived isotopes are produced in cyclotrons, linear particle accelerators, or nuclear reactors and their relatively short half-lives give them high maximum theoretical specific activities which is useful for detection in biological systems. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biomolecule
A biomolecule or biological molecule is a loosely used term for molecules present in organisms that are essential to one or more typically biological processes, such as cell division, morphogenesis, or developmental biology, development. Biomolecules include large macromolecules (or polyelectrolytes) such as proteins, carbohydrates, lipids, and nucleic acids, as well as small molecules such as primary metabolites, secondary metabolites and natural products. A more general name for this class of material is biological materials. Biomolecules are an important element of living organisms, those biomolecules are often endogeny (biology), endogenous, produced within the organism but organisms usually need exogeny, exogenous biomolecules, for example certain nutrients, to survive. Biology and its subfields of biochemistry and molecular biology study biomolecules and their organic reaction, reactions. Most biomolecules are organic compounds, and just four chemical element, elements ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Iodine
There are 37 known isotopes of iodine (53I) from 108I to 144I; all undergo radioactive decay except 127I, which is stable. Iodine is thus a monoisotopic element. Its longest-lived radioactive isotope, 129I, has a half-life of 15.7 million years, which is far too short for it to exist as a primordial nuclide. Cosmogenic sources of 129I produce very tiny quantities of it that are too small to affect atomic weight measurements; iodine is thus also a mononuclidic element—one that is found in nature only as a single nuclide. Most 129I derived radioactivity on Earth is man-made, an unwanted long-lived byproduct of early nuclear tests and nuclear fission accidents. All other iodine radioisotopes have half-lives less than 60 days, and four of these are used as tracers and therapeutic agents in medicine. These are 123I, 124I, 125I, and 131I. All industrial production of radioactive iodine isotopes involves these four useful radionuclides. The isotope 135I has a half-life less than s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
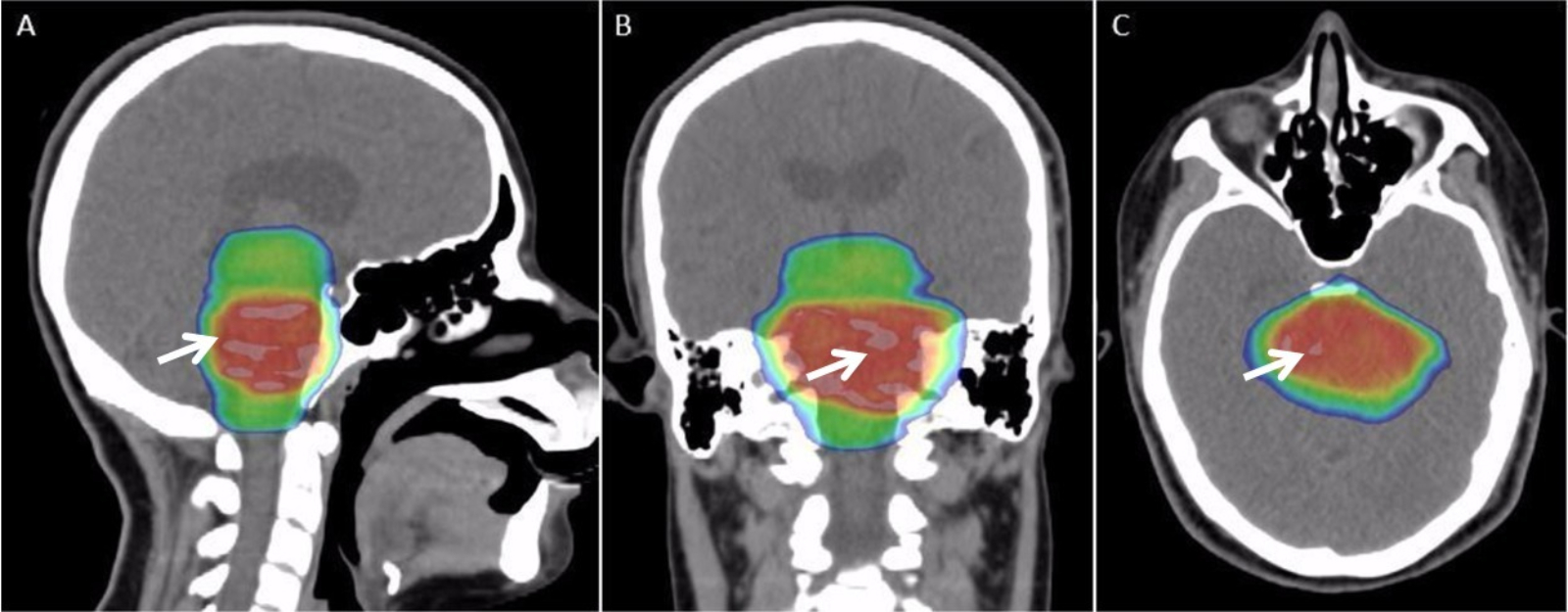
Radiation Therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Radiation therapy may be curative in a number of types of cancer if they are localized to one area of the body. It may also be used as part of adjuvant therapy, to prevent tumor recurrence after surgery to remove a primary malignant tumor (for example, early stages of breast cancer). Radiation therapy is synergistic with chemotherapy, and has been used before, during, and after chemotherapy in susceptible cancers. The subspecialty of oncology concerned with radiotherapy is called radiation oncology. A physician who practices in this subspecialty is a radiation oncologist. Radiation therapy is commonly applied to the cancerous tumor because of its ability to control cell growth. Ionizing radiation works by damaging the DNA of cancerous tissue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiobiology
Radiobiology (also known as radiation biology, and uncommonly as actinobiology) is a field of clinical and basic medical sciences that involves the study of the action of ionizing radiation on living things, especially health effects of radiation. Ionizing radiation is generally harmful and potentially lethal to living things but can have health benefits in radiation therapy for the treatment of cancer and thyrotoxicosis. Its most common impact is the induction of cancer with a latent period of years or decades after exposure. High doses can cause visually dramatic radiation burns, and/or rapid fatality through acute radiation syndrome. Controlled doses are used for medical imaging and radiotherapy. Health effects In general, ionizing radiation is harmful and potentially lethal to living beings but can have health benefits in radiation therapy for the treatment of cancer and thyrotoxicosis. Most adverse health effects of radiation exposure may be grouped in two general categor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
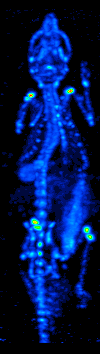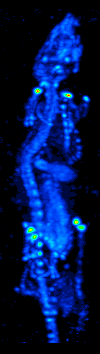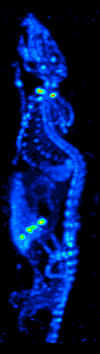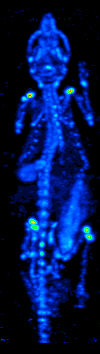
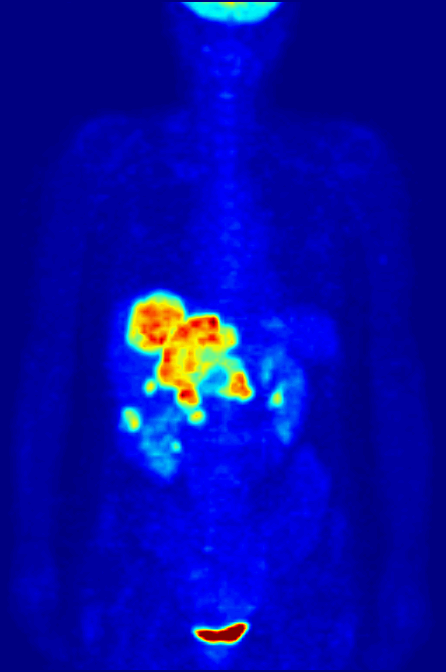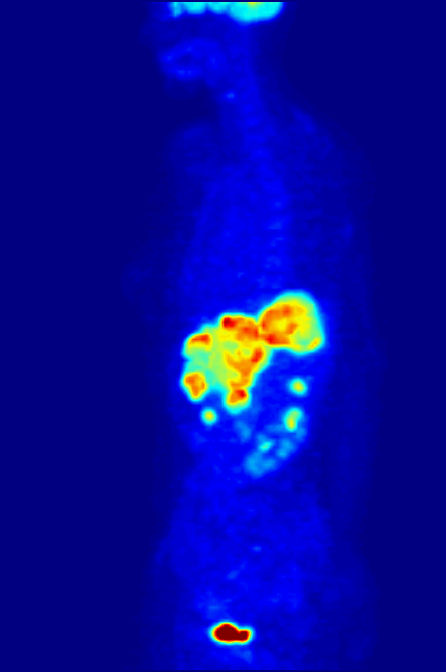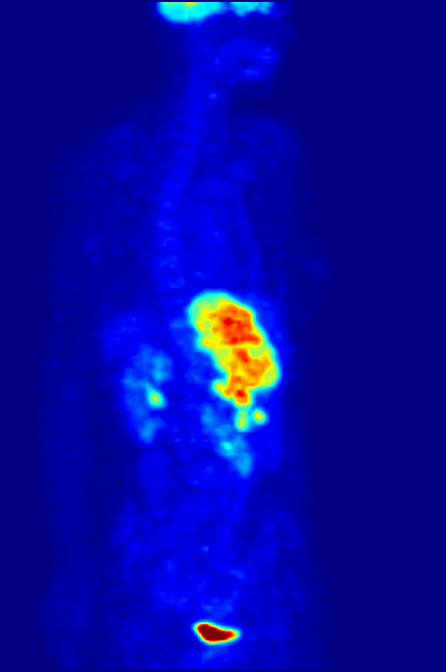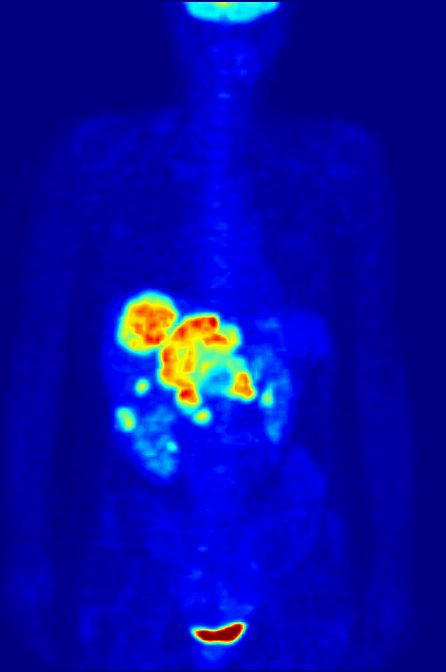
Single-photon Emission Computed Tomography
Single-photon emission computed tomography (SPECT, or less commonly, SPET) is a nuclear medicine tomographic imaging technique using gamma rays. It is very similar to conventional nuclear medicine planar imaging using a gamma camera (that is, scintigraphy), but is able to provide true 3D information. This information is typically presented as cross-sectional slices through the patient, but can be freely reformatted or manipulated as required. The technique needs delivery of a gamma-emitting radioisotope (a radionuclide) into the patient, normally through injection into the bloodstream. On occasion, the radioisotope is a simple soluble dissolved ion, such as an isotope of gallium(III). Most of the time, though, a marker radioisotope is attached to a specific ligand to create a radioligand, whose properties bind it to certain types of tissues. This marriage allows the combination of ligand and radiopharmaceutical to be carried and bound to a place of interest in the body, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Positron Emission Tomography
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as radiotracers to visualize and measure changes in metabolic processes, and in other physiological activities including blood flow, regional chemical composition, and absorption. Different tracers are used for various imaging purposes, depending on the target process within the body. For example, -FDG is commonly used to detect cancer, NaF is widely used for detecting bone formation, and oxygen-15 is sometimes used to measure blood flow. PET is a common imaging technique, a medical scintillography technique used in nuclear medicine. A radiopharmaceutical — a radioisotope attached to a drug — is injected into the body as a tracer. When the radiopharmaceutical undergoes beta plus decay, a positron is emitted, and when the positron collides with an ordinary electron, the two particles annihilate and gamma rays are emitted. These gamma rays are detecte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Imaging
Molecular imaging is a field of medical imaging that focuses on imaging molecules of medical interest within living patients. This is in contrast to conventional methods for obtaining molecular information from preserved tissue samples, such as histology. Molecules of interest may be either ones produced naturally by the body, or synthetic molecules produced in a laboratory and injected into a patient by a doctor. The most common example of molecular imaging used clinically today is to inject a contrast agent (e.g., a microbubble, metal ion, or radioactive isotope) into a patient's bloodstream and to use an imaging modality (e.g., ultrasound, MRI, CT, PET) to track its movement in the body. Molecular imaging originated from the field of radiology from a need to better understand fundamental molecular processes inside organisms in a noninvasive manner. The ultimate goal of molecular imaging is to be able to noninvasively monitor all of the biochemical processes occurring inside ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radioiodine
There are 37 known isotopes of iodine (53I) from 108I to 144I; all undergo radioactive decay except 127I, which is stable. Iodine is thus a monoisotopic element. Its longest-lived radioactive isotope, 129I, has a half-life of 15.7 million years, which is far too short for it to exist as a primordial nuclide. Cosmogenic sources of 129I produce very tiny quantities of it that are too small to affect atomic weight measurements; iodine is thus also a mononuclidic element—one that is found in nature only as a single nuclide. Most 129I derived radioactivity on Earth is man-made, an unwanted long-lived byproduct of early nuclear tests and nuclear fission accidents. All other iodine radioisotopes have half-lives less than 60 days, and four of these are used as tracers and therapeutic agents in medicine. These are 123I, 124I, 125I, and 131I. All industrial production of radioactive iodine isotopes involves these four useful radionuclides. The isotope 135I has a half-life less than se ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
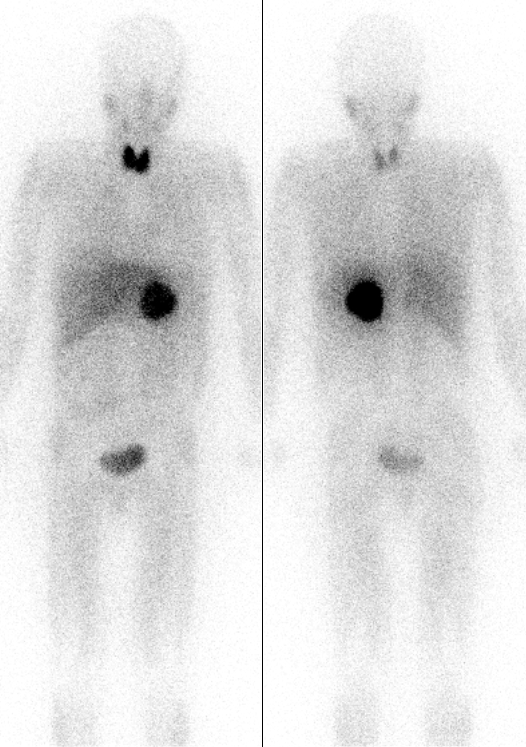
Gallium-68
Natural gallium (31Ga) consists of a mixture of two stable isotopes: gallium-69 and gallium-71. The most commercially important radioisotopes are gallium-67 and gallium-68. Gallium-67 (half-life 3.3 days) is a gamma-emitting isotope (the gamma ray emitted immediately after electron capture) used in standard nuclear medical imaging, in procedures usually referred to as gallium scans. It is usually used as the free ion, Ga3+. It is the longest-lived radioisotope of gallium. The shorter-lived gallium-68 (half-life 68 minutes) is a positron-emitting isotope generated in very small quantities from germanium-68 in gallium-68 generators or in much greater quantities by proton bombardment of 68Zn in low-energy medical cyclotrons, for use in a small minority of diagnostic PET scans. For this use, it is usually attached as a tracer to a carrier molecule (for example the somatostatin analogue DOTATOC), which gives the resulting radiopharmaceutical a different tissue-uptake specificity fro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium-67
Natural gallium (31Ga) consists of a mixture of two stable isotopes: gallium-69 and gallium-71. The most commercially important radioisotopes are gallium-67 and gallium-68. Gallium-67 (half-life 3.3 days) is a gamma-emitting isotope (the gamma ray emitted immediately after electron capture) used in standard nuclear medical imaging, in procedures usually referred to as gallium scans. It is usually used as the free ion, Ga3+. It is the longest-lived radioisotope of gallium. The shorter-lived gallium-68 (half-life 68 minutes) is a positron-emitting isotope generated in very small quantities from germanium-68 in gallium-68 generators or in much greater quantities by proton bombardment of 68Zn in low-energy medical cyclotrons, for use in a small minority of diagnostic PET scans. For this use, it is usually attached as a tracer to a carrier molecule (for example the somatostatin analogue DOTATOC), which gives the resulting radiopharmaceutical a different tissue-uptake specificity fro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Positron Emission Tomography
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as radiotracers to visualize and measure changes in metabolic processes, and in other physiological activities including blood flow, regional chemical composition, and absorption. Different tracers are used for various imaging purposes, depending on the target process within the body. For example, -FDG is commonly used to detect cancer, NaF is widely used for detecting bone formation, and oxygen-15 is sometimes used to measure blood flow. PET is a common imaging technique, a medical scintillography technique used in nuclear medicine. A radiopharmaceutical — a radioisotope attached to a drug — is injected into the body as a tracer. When the radiopharmaceutical undergoes beta plus decay, a positron is emitted, and when the positron collides with an ordinary electron, the two particles annihilate and gamma rays are emitted. These gamma rays are detecte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octreotide
Octreotide, sold under the brand name Sandostatin among others, is an octapeptide that mimics natural somatostatin pharmacologically, though it is a more potent inhibitor of growth hormone, glucagon, and insulin than the natural hormone. It was first synthesized in 1979 by the chemist Wilfried Bauer, and binds predominantly to the somatostatin receptors SSTR2 and SSTR5. It was approved for use in the United States in 1988. Octreotide (Mycapssa) was approved for medical use in the European Union in 2022. , octreotide (Mycapssa) is the first and only oral somatostatin analog (SSA) approved by the FDA. Medical uses Tumors Octreotide is used for the treatment of growth hormone producing tumors (acromegaly and gigantism), when surgery is contraindicated, pituitary tumors that secrete thyroid-stimulating hormone (thyrotropinomata), diarrhea and flushing episodes associated with carcinoid syndrome, and diarrhea in people with vasoactive intestinal peptide-secreting tumors ( VIPomas). ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |