|
Ribofuranose
Ribose is a simple sugar and carbohydrate with molecular formula C5H10O5 and the linear-form composition H−(C=O)−(CHOH)4−H. The naturally-occurring form, , is a component of the ribonucleotides from which RNA is built, and so this compound is necessary for coding, decoding, regulation and expression of genes. It has a structural analog, deoxyribose, which is a similarly essential component of DNA. is an unnatural sugar that was first prepared by Emil Fischer and Oscar Piloty in 1891. It was not until 1909 that Phoebus Levene and Walter Jacobs recognised that was a natural product, the enantiomer of Fischer and Piloty's product, and an essential component of nucleic acids. Fischer chose the name "ribose" as it is a partial rearrangement of the name of another sugar, arabinose, of which ribose is an epimer at the 2' carbon; both names also relate to gum arabic, from which arabinose was first isolated and from which they prepared . Like most sugars, ribose exists as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deoxyribose
Deoxyribose, or more precisely 2-deoxyribose, is a monosaccharide with idealized formula H−(C=O)−(CH2)−(CHOH)3−H. Its name indicates that it is a deoxy sugar, meaning that it is derived from the sugar ribose by loss of a hydroxy group. Discovered in 1929 by Phoebus Levene, deoxyribose is most notable for its presence in DNA. Since the pentose sugars arabinose and ribose only differ by the stereochemistry at C2′, 2-deoxyribose and 2-deoxyarabinose are equivalent, although the latter term is rarely used because ribose, not arabinose, is the precursor to deoxyribose. Structure Several isomers exist with the formula H−(C=O)−(CH2)−(CHOH)3−H, but in deoxyribose all the hydroxyl groups are on the same side in the Fischer projection. The term "2-deoxyribose" may refer to either of two enantiomers: the biologically important -2-deoxyribose and to the rarely encountered mirror image -2-deoxyribose.C Bernelot-Moens and B Demple (1989), ''Multiple DNA repair activities f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deoxyribose
Deoxyribose, or more precisely 2-deoxyribose, is a monosaccharide with idealized formula H−(C=O)−(CH2)−(CHOH)3−H. Its name indicates that it is a deoxy sugar, meaning that it is derived from the sugar ribose by loss of a hydroxy group. Discovered in 1929 by Phoebus Levene, deoxyribose is most notable for its presence in DNA. Since the pentose sugars arabinose and ribose only differ by the stereochemistry at C2′, 2-deoxyribose and 2-deoxyarabinose are equivalent, although the latter term is rarely used because ribose, not arabinose, is the precursor to deoxyribose. Structure Several isomers exist with the formula H−(C=O)−(CH2)−(CHOH)3−H, but in deoxyribose all the hydroxyl groups are on the same side in the Fischer projection. The term "2-deoxyribose" may refer to either of two enantiomers: the biologically important -2-deoxyribose and to the rarely encountered mirror image -2-deoxyribose.C Bernelot-Moens and B Demple (1989), ''Multiple DNA repair activities f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Emil Fischer
Hermann Emil Louis Fischer (; 9 October 1852 – 15 July 1919) was a German chemist and 1902 recipient of the Nobel Prize in Chemistry. He discovered the Fischer esterification. He also developed the Fischer projection, a symbolic way of drawing asymmetric carbon atoms. He also hypothesized lock and key mechanism of enzyme action. He never used his first given name, and was known throughout his life simply as Emil Fischer. Early years and career Fischer was born in Euskirchen, near Cologne, the son of Laurenz Fischer, a businessman, and his wife Julie Poensgen. After graduating he wished to study natural sciences, but his father compelled him to work in the family business until determining that his son was unsuitable. Fischer then attended the University of Bonn in 1871, but switched to the University of Strasbourg in 1872. He earned his doctorate in 1874 under Adolf von Baeyer with his study of phthaleins, and was appointed to a position at the university. After eight ye ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Haworth Projection
In chemistry, a Haworth projection is a common way of writing a structural formula to represent the cyclic structure of monosaccharides with a simple three-dimensional perspective. Haworth projection approximate the shapes of the actual molecules better for furanoses -which are in reality nearly planar- than for pyranoses which exist in solution in the chair conformation. Organic chemistry and especially biochemistry are the areas of chemistry that use the Haworth projection the most. The Haworth projection was named after the British chemist Sir Norman Haworth. A Haworth projection has the following characteristics: * Carbon is the implicit type of atom. In the example on the right, the atoms numbered from 1 to 6 are all carbon atoms. Carbon 1 is known as the anomeric carbon. * Hydrogen atoms on carbon are implicit. In the example, atoms 1 to 6 have extra hydrogen atoms not depicted. * A thicker line indicates atoms that are closer to the observer. In the example on the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Science Education (journal)
''Science Education'' is a bimonthly peer-reviewed scientific journal covering science education. It was established in 1916 as ''General Science Quarterly'', obtaining its current name in 1929. The editors-in-chief are Sherry A. Southerland (Florida State University) and John Settlage (University of Connecticut). According to the ''Journal Citation Reports'', the journal has a 2020 impact factor The impact factor (IF) or journal impact factor (JIF) of an academic journal is a scientometric index calculated by Clarivate that reflects the yearly mean number of citations of articles published in the last two years in a given journal, as i ... of 4.593, ranking it 31st out of 265 journals in the category "Education & Educational Research)". References External links * Publications established in 1916 Bimonthly journals Wiley (publisher) academic journals Science education journals English-language journals {{edu-journal-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gum Arabic
Gum arabic, also known as gum sudani, acacia gum, Arabic gum, gum acacia, acacia, Senegal gum, Indian gum, and by other names, is a natural gum originally consisting of the hardened sap of two species of the '' Acacia'' tree, ''Senegalia senegal'' and ''Vachellia seyal.'' The term "gum arabic" does not legally indicate a particular botanical source, however. The gum is harvested commercially from wild trees, mostly in Sudan (80%) and throughout the Sahel, from Senegal to Somalia. The name "gum Arabic" (''al-samgh al-'arabi'') was used in the Middle East at least as early as the 9th century. Gum arabic first found its way to Europe via Arabic ports, so retained its name. Gum arabic is a complex mixture of glycoproteins and polysaccharides, predominantly polymers of arabinose and galactose. It is soluble in water, edible, and used primarily in the food industry and soft-drink industry as a stabilizer, with E number E414 (I414 in the US). Gum arabic is a key ingredient ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epimer
In stereochemistry, an epimer is one of a pair of diastereomers. The two epimers have opposite configuration at only one stereogenic center out of at least two. All other stereogenic centers in the molecules are the same in each. Epimerization is the interconversion of one epimer to the other epimer. Doxorubicin and epirubicin are two epimers that are used as drugs. Examples The stereoisomers β-D- glucopyranose and β-D- mannopyranose are epimers because they differ only in the stereochemistry at the C-2 position. The hydroxy group in β-D-glucopyranose is equatorial (in the "plane" of the ring), while in β-D-mannopyranose the C-2 hydroxy group is axial (up from the "plane" of the ring). These two molecules are epimers but, because they are not mirror images of each other, are not enantiomers. (Enantiomers have the same name, but differ in D and L classification.) They are also not sugar anomers, since it is not the anomeric carbon involved in the stereochemistry. Simila ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arabinose
Arabinose is an aldopentose – a monosaccharide containing five carbon atoms, and including an aldehyde (CHO) functional group. For biosynthetic reasons, most saccharides are almost always more abundant in nature as the "D"-form, or structurally analogous to D-glyceraldehyde.The D/L nomenclature does not refer to the molecule's optical rotation properties but to its structural analogy to glyceraldehyde. However, L-arabinose is in fact more common than D-arabinose in nature and is found in nature as a component of biopolymers such as hemicellulose and pectin. The L-arabinose operon, also known as the araBAD operon, has been the subject of much biomolecular research. The operon directs the catabolism of arabinose in ''E. coli'', and it is dynamically activated in the presence of arabinose and the absence of glucose. A classic method for the organic synthesis of arabinose from glucose is the Wohl degradation. : Etymology Arabinose gets its name from gum arabic, from which i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Academic Press
Academic Press (AP) is an academic book publisher founded in 1941. It was acquired by Harcourt, Brace & World in 1969. Reed Elsevier bought Harcourt in 2000, and Academic Press is now an imprint of Elsevier. Academic Press publishes reference books, serials and online products in the subject areas of: * Communications engineering * Economics * Environmental science * Finance * Food science and nutrition * Geophysics * Life sciences * Mathematics and statistics * Neuroscience * Physical sciences * Psychology Well-known products include the ''Methods in Enzymology'' series and encyclopedias such as ''The International Encyclopedia of Public Health'' and the ''Encyclopedia of Neuroscience''. See also * Akademische Verlagsgesellschaft (AVG) — the German predecessor, founded in 1906 by Leo Jolowicz (1868–1940), the father of Walter Jolowicz Walter may refer to: People * Walter (name), both a surname and a given name * Little Walter, American blues harmonica player Marion Wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
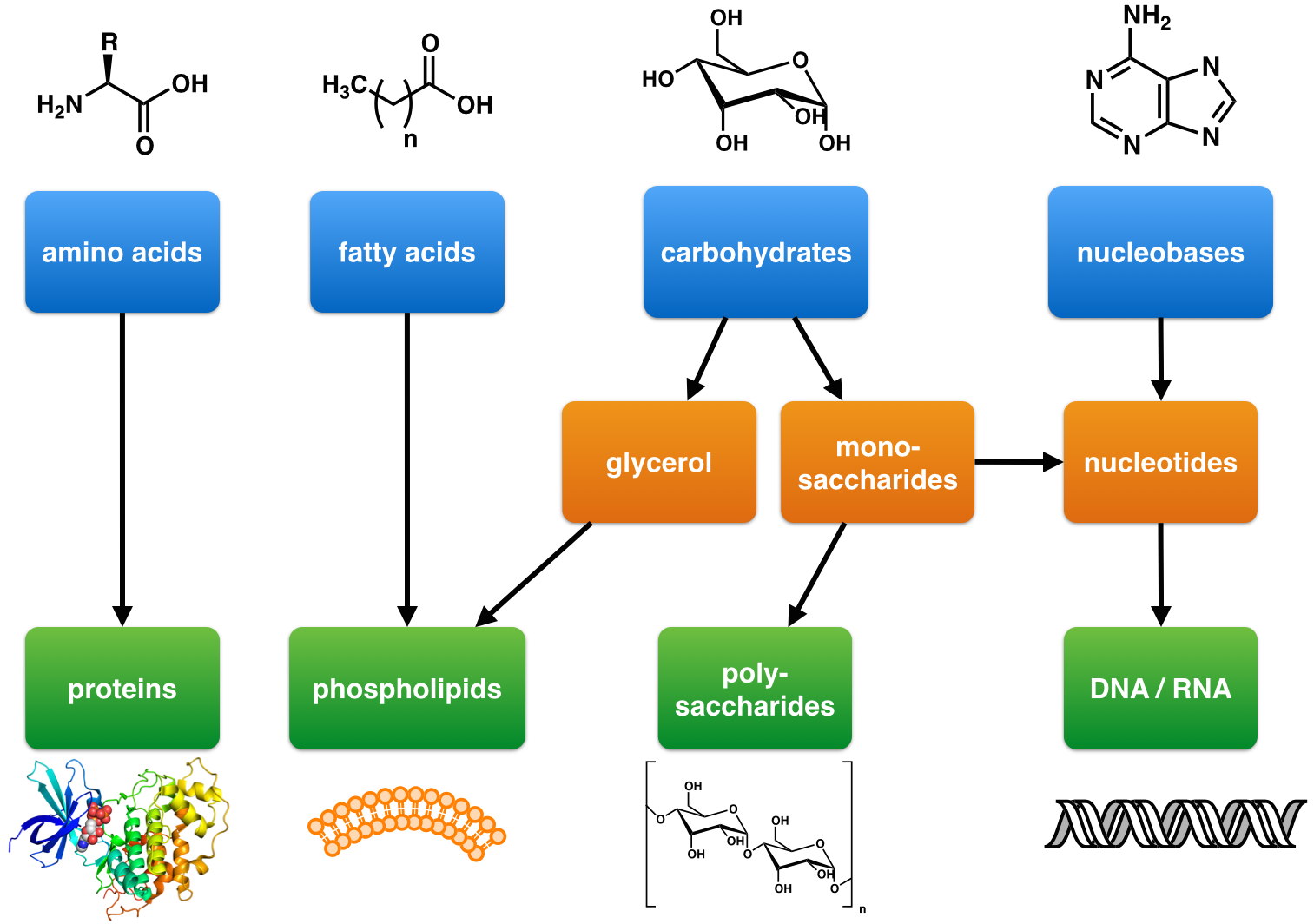
Nucleic Acid
Nucleic acids are biopolymers, macromolecules, essential to all known forms of life. They are composed of nucleotides, which are the monomers made of three components: a 5-carbon sugar, a phosphate group and a nitrogenous base. The two main classes of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). If the sugar is ribose, the polymer is RNA; if the sugar is the ribose derivative deoxyribose, the polymer is DNA. Nucleic acids are naturally occurring chemical compounds that serve as the primary information-carrying molecules in cells and make up the genetic material. Nucleic acids are found in abundance in all living things, where they create, encode, and then store information of every living cell of every life-form on Earth. In turn, they function to transmit and express that information inside and outside the cell nucleus to the interior operations of the cell and ultimately to the next generation of each living organism. The encoded information is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enantiomer
In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical antipode – is one of two stereoisomers that are non-superposable onto their own mirror image. Enantiomers are much like one's right and left hands, when looking at the same face, they cannot be superposed onto each other. No amount of reorientation will allow the four unique groups on the chiral carbon (see Chirality (chemistry)) to line up exactly. The number of stereoisomers a molecule has can be determined by the number of chiral carbons it has. Stereoisomers include both enantiomers and diastereomers. Diastereomers, like enantiomers, share the same molecular formula and are non-superposable onto each other however, they are not mirror images of each other. A molecule with chirality rotates plane-polarized light. A mixture of equals a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Natural Product
A natural product is a natural compound or substance produced by a living organism—that is, found in nature. In the broadest sense, natural products include any substance produced by life. Natural products can also be prepared by chemical synthesis (both semisynthesis and total synthesis) and have played a central role in the development of the field of organic chemistry by providing challenging synthetic targets. The term natural product has also been extended for commercial purposes to refer to cosmetics, dietary supplements, and foods produced from natural sources without added artificial ingredients. Within the field of organic chemistry, the definition of natural products is usually restricted to organic compounds isolated from natural sources that are produced by the pathways of primary or secondary metabolism. Within the field of medicinal chemistry, the definition is often further restricted to secondary metabolites. Secondary metabolites (or specialized metabolites ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |