|
Polydispersity
In chemistry, the dispersity is a measure of the heterogeneity of sizes of molecules or particles in a mixture. A collection of objects is called uniform if the objects have the same size, shape, or mass. A sample of objects that have an inconsistent size, shape and mass distribution is called non-uniform. The objects can be in any form of chemical dispersion, such as particles in a colloid, droplets in a cloud, crystals in a rock, or polymer macromolecules in a solution or a solid polymer mass. Polymers can be described by molecular mass distribution; a population of particles can be described by size, surface area, and/or mass distribution; and thin films can be described by film thickness distribution. IUPAC has deprecated the use of the term ''polydispersity index'', having replaced it with the term ''dispersity'', represented by the symbol Đ (pronounced D-strokeStepto, R. F. T.; Gilbert, R. G.; Hess, M.; Jenkins, A. D.; Jones, R. G.; Kratochvíl P. (2009).Dispersity in Po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Living Polymerization
In polymer chemistry, living polymerization is a form of chain growth polymerization where the ability of a growing polymer chain to terminate has been removed. This can be accomplished in a variety of ways. Chain termination and chain transfer reactions are absent and the rate of chain initiation is also much larger than the rate of chain propagation. The result is that the polymer chains grow at a more constant rate than seen in traditional chain polymerization and their lengths remain very similar (i.e. they have a very low polydispersity index). Living polymerization is a popular method for synthesizing block copolymers since the polymer can be synthesized in stages, each stage containing a different monomer. Additional advantages are predetermined molar mass and control over end-groups. Living polymerization is desirable because it offers precision and control in macromolecular synthesis. This is important since many of the novel/useful properties of polymers res ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Number Average Molecular Weight
In polymer chemistry, the molar mass distribution (or molecular weight distribution) describes the relationship between the number of moles of each polymer species () and the molar mass () of that species. In linear polymers, the individual polymer chains rarely have exactly the same degree of polymerization and molar mass, and there is always a distribution around an average value. The molar mass distribution of a polymer may be modified by polymer fractionation. Definitions of molar mass average Different average values can be defined, depending on the statistical method applied. In practice, four averages are used, representing the weighted mean taken with the mole fraction, the weight fraction, and two other functions which can be related to measured quantities: *''Number average molar mass'' (), also loosely referred to as ''number average molecular weight'' (NAMW). *''Mass average molar mass'' (), where stands for weight; also commonly referred to as ''weight av ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carothers' Equation
In step-growth polymerization, the Carothers equation (or Carothers' equation) gives the degree of polymerization, , for a given fractional monomer conversion, . There are several versions of this equation, proposed by Wallace Carothers, who invented nylon in 1935. Linear polymers: two monomers in equimolar quantities The simplest case refers to the formation of a strictly linear polymer by the reaction (usually by condensation) of two monomers in equimolar quantities. An example is the synthesis of nylon-6,6 whose formula is from one mole of hexamethylenediamine, , and one mole of adipic acid, . For this case :\bar_n=\frac In this equation * is the number-average value of the degree of polymerization, equal to the average number of monomer units in a polymer molecule. For the example of nylon-6,6 \bar_n = 2n ( diamine units and diacid units). *p=\tfrac is the extent of reaction (or conversion to polymer), defined by ** is the number of molecules present initially as monome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer () is a chemical substance, substance or material that consists of very large molecules, or macromolecules, that are constituted by many repeat unit, repeating subunits derived from one or more species of monomers. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compound (chemistry), compounds, produces unique physical property, physical properties including toughness, high rubber elasticity, elasticity, viscoelasticity, and a tendency to form Amorphous solid, amorphous and crystallization of polymers, semicrystalline structures rath ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fulvic Acid
Humic substances (HS) are colored relatively recalcitrant organic compounds naturally formed during long-term decomposition and transformation of biomass residues. The color of humic substances varies from bright yellow to light or dark brown leading to black. The term comes from humus, which in turn comes from the Latin word ''humus'', meaning "soil, earth". Humic substances represent the major part of organic matter in soil, peat, coal, and sediments, and are important components of dissolved natural organic matter (NOM) in lakes (especially dystrophic lakes), rivers, and sea water. Humic substances account for 50 – 90% of cation exchange capacity in soils. "Humic substances" is an umbrella term covering humic acid, fulvic acid and humin, which differ in solubility. By definition, humic acid (HA) is soluble in water at neutral and alkaline pH, but insoluble at acidic pH < 2. Fulvic acid (FA) is soluble in water at any pH. Humin is not soluble in water at any pH. This definiti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Damköhler Numbers
The Damköhler numbers (Da) are dimensionless numbers used in chemical engineering to relate the chemical reaction timescale ( reaction rate) to the transport phenomena rate occurring in a system. It is named after German chemist Gerhard Damköhler, who worked in chemical engineering, thermodynamics, and fluid dynamics. The Karlovitz number (Ka) is related to the Damköhler number by Da = 1/Ka. In its most commonly used form, the first Damköhler number (DaI) relates particles' characteristic residence time scale in a fluid region to the reaction timescale. The residence time scale can take the form of a convection time scale, such as volumetric flow rate through the reactor for continuous ( plug flow or stirred tank) or semibatch chemical processes: : \mathrm = \frac In reacting systems that include interphase mass transport, the first Damköhler number can be written as the ratio of the chemical reaction rate to the mass transfer rate : \mathrm_ = \frac It is also defin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Geometric Distribution
In probability theory and statistics, the geometric distribution is either one of two discrete probability distributions: * The probability distribution of the number X of Bernoulli trials needed to get one success, supported on \mathbb = \; * The probability distribution of the number Y=X-1 of failures before the first success, supported on \mathbb_0 = \ . These two different geometric distributions should not be confused with each other. Often, the name ''shifted'' geometric distribution is adopted for the former one (distribution of X); however, to avoid ambiguity, it is considered wise to indicate which is intended, by mentioning the support explicitly. The geometric distribution gives the probability that the first occurrence of success requires k independent trials, each with success probability p. If the probability of success on each trial is p, then the probability that the k-th trial is the first success is :\Pr(X = k) = (1-p)^p for k=1,2,3,4,\dots The above form of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
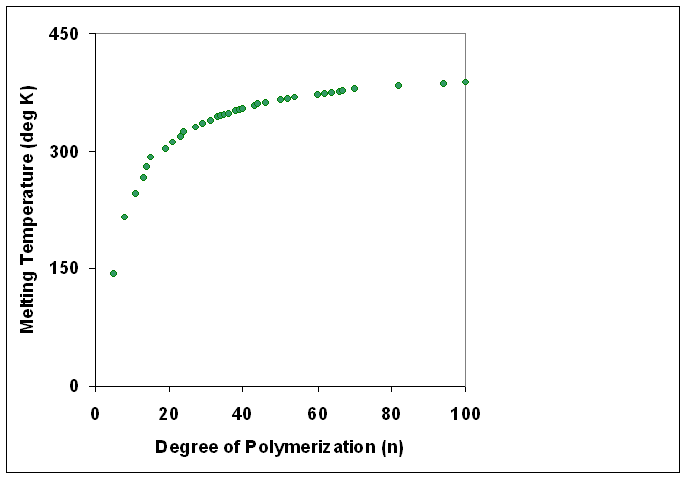
Degree Of Polymerization
The degree of polymerization, or DP, is the number of structural unit, monomeric units in a macromolecule or polymer or oligomer molecule. For a homopolymer, there is only one type of monomeric unit and the ''number-average'' degree of polymerization is given by \overline_n\equiv\overline_n=\frac, where \overline_n is the Molar mass distribution#Number average molar mass, number-average molecular weight and M_0 is the molecular weight of the monomer unit. The overlines indicate arithmetic mean values. For most industrial purposes, degrees of polymerization in the thousands or tens of thousands are desired. This number does not reflect the variation in molecule size of the polymer that typically occurs, it only represents the mean number of monomeric units. Some authors, however, define DP as the number of repeat units, where for copolymers the repeat unit may not be identical to the monomeric unit.Fried J.R. "Polymer Science and Technology" (Pearson Prentice-Hall, 2nd edn 2003), p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Continuous Stirred-tank Reactor
The continuous stirred-tank reactor (CSTR), also known as vat- or backmix reactor, mixed flow reactor (MFR), or a continuous-flow stirred-tank reactor (CFSTR), is a common model for a chemical reactor in chemical engineering and environmental engineering. A CSTR often refers to a model used to estimate the key unit operation variables when using a continuous agitated-tank reactor to reach a specified output. The mathematical model works for all fluids: liquids, gases, and slurries. The behavior of a CSTR is often approximated or modeled by that of an ideal CSTR, which assumes perfect mixing. In a perfectly mixed reactor, reagent is instantaneously and uniformly mixed throughout the reactor upon entry. Consequently, the output composition is identical to composition of the material inside the reactor, which is a function of residence time and reaction rate. The CSTR is the ideal limit of complete mixing in reactor design, which is the complete opposite of a plug flow reactor (P ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |