|
Perfluoromethyldiethylamine
Perfluoromethyldiethylamine is a tertiary perfluorinated amine with the chemical formula C5F13N. The compound consists of two pentafluoroethyl and one trifluoromethyl groups connected to nitrogen. The compound is produced for the electronics industry, along with other perfluoroalkylamines. Unlike ordinary amines, perfluoroamines are nonbasic. Preparation and use Perfluoroalkylamines are prepared by electrofluorination of the parent amine using hydrogen fluoride as solvent and source of fluorine. The compound has two commercial uses, although details are often proprietary. It is a component in Fluosol, artificial blood. This application exploits the high solubility of oxygen and carbon dioxide in the solvent, as well as the low viscosity and toxicity. It is also a component of Fluorinert Fluorinert is the trademarked brand name for the line of electronics coolant liquids sold commercially by 3M. As perfluorinated compounds (PFCs), all Fluorinert variants have an extremely h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Perfluorinated Compound
A perfluorinated compound (PFC) or perfluoro compound is an organofluorine compound containing only carbon-fluorines and C−C bonds, as well as potentially heteroatoms. Perfluorinated compounds have properties that result from the presence of fluorocarbons (containing only C−F and C−C bonds) and any functional group. Common functional groups in PFCs are OH, CO2H, chlorine, O, and SO3H. Electrofluorination is the predominant method of production. Some of these compounds known as perfluoroalkanes can remain in our atmosphere for a long time. They bioaccumulate due to their chemical stability. Because of their potential contribution to climate change, they were regulated under the Kyoto Protocol. Some fluorosurfactants have proven toxic in animal testing while widespread industrial applications continue. Applications Perfluorinated compounds are used ubiquitously: For example, fluorosurfactants are widely used in the production of teflon (PTFE) and related fluorinated pol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Formula
In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as parentheses, dashes, brackets, commas and ''plus'' (+) and ''minus'' (−) signs. These are limited to a single typographic line of symbols, which may include Subscript and superscript, subscripts and superscripts. A chemical formula is not a chemical nomenclature, chemical name, and it contains no words. Although a chemical formula may imply certain simple chemical structures, it is not the same as a full chemical structural formula. Chemical formulae can fully specify the structure of only the simplest of molecules and chemical substances, and are generally more limited in power than chemical names and structural formulae. The simplest types of chemical formulae are called ''empirical formulae'', which use letters and numbers ind ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
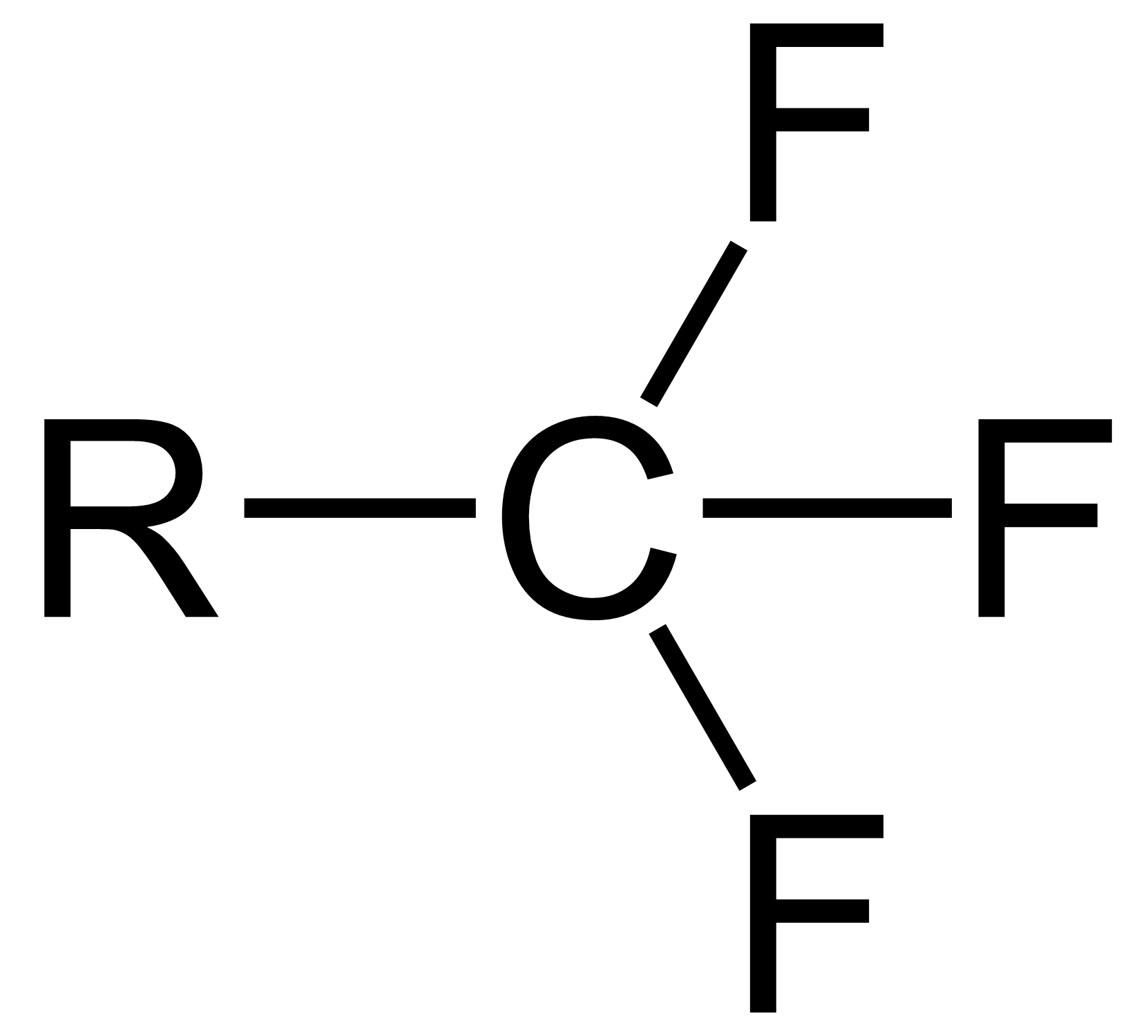
Trifluoromethyl
The trifluoromethyl group is a functional group that has the formula -CF3. The naming of is group is derived from the methyl group (which has the formula -CH3), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane H–, 1,1,1-trifluoroethane –, and hexafluoroacetone –CO–. Compounds with this group are a subclass of the organofluorines. Properties The trifluoromethyl group has a significant electronegativity that is often described as being intermediate between the electronegativities of fluorine and chlorine. For this reason, trifluoromethyl-substituted compounds are often strong acids, such as trifluoromethanesulfonic acid and trifluoroacetic acid Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a structural analogue of acetic acid with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms and is a colorless liquid with a .... Conversely, the trifluoromethyl group ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Fluoride
Hydrogen fluoride (fluorane) is an inorganic compound with the chemical formula . This colorless gas or liquid is the principal industrial source of fluorine, often as an aqueous solution called hydrofluoric acid. It is an important feedstock in the preparation of many important compounds including pharmaceuticals and polymers, e.g. polytetrafluoroethylene (PTFE). HF is widely used in the petrochemical industry as a component of superacids. Hydrogen fluoride boils at near room temperature, much higher than other hydrogen halides. Hydrogen fluoride is an extremely dangerous gas, forming corrosive and penetrating hydrofluoric acid upon contact with moisture. The gas can also cause blindness by rapid destruction of the corneas. History In 1771 Carl Wilhelm Scheele prepared the aqueous solution, hydrofluoric acid in large quantities, although hydrofluoric acid had been known in the glass industry before then. French chemist Edmond Frémy (1814–1894) is credited with discoveri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluosol
A blood substitute (also called artificial blood or blood surrogate) is a substance used to mimic and fulfill some functions of biological blood. It aims to provide an alternative to blood transfusion, which is transferring blood or blood-based products from one person into another. Thus far, there are no well-accepted ''oxygen-carrying'' blood substitutes, which is the typical objective of a red blood cell transfusion; however, there are widely available non-blood volume expanders for cases where only volume restoration is required. These are helping doctors and surgeons avoid the risks of disease transmission and immune suppression, address the chronic blood donor shortage, and address the concerns of Jehovah's Witnesses and others who have religious objections to receiving transfused blood. The main categories of "oxygen-carrying" blood substitutes being pursued are hemoglobin-based oxygen carriers (HBOC) and perfluorocarbon emulsions. Oxygen therapeutics are in clinical tri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorinert
Fluorinert is the trademarked brand name for the line of electronics coolant liquids sold commercially by 3M. As perfluorinated compounds (PFCs), all Fluorinert variants have an extremely high Global Warming Potential (GWP), so should be used with caution (see below). It is an electrically insulating, stable fluorocarbon-based fluid, which is used in various cooling applications. It is mainly used for cooling electronics. Different molecular formulations are available with a variety of boiling points, allowing it to be used in "single-phase" applications, where it remains a liquid, or for "two-phase" applications, where the liquid boils to remove additional heat by evaporative cooling. An example of one of the compounds 3M uses is FC-72 ( perfluorohexane, C6F14). Perfluorohexane is used for low-temperature heat-transfer applications due to its boiling point. Another example is FC-75, perfluoro(2-butyl-tetrahydrofurane). There are 3M fluids that can handle up to , such as FC-70 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Perfluorinated Compounds
A perfluorinated compound (PFC) or perfluoro compound is an organofluorine compound containing only carbon-fluorines and C−C bonds, as well as potentially heteroatoms. Perfluorinated compounds have properties that result from the presence of fluorocarbons (containing only C−F and C−C bonds) and any functional group. Common functional groups in PFCs are OH, CO2H, chlorine, O, and SO3H. Electrofluorination is the predominant method of production. Some of these compounds known as perfluoroalkanes can remain in our atmosphere for a long time. They bioaccumulate due to their chemical stability. Because of their potential contribution to climate change, they were regulated under the Kyoto Protocol. Some fluorosurfactants have proven toxic in animal testing while widespread industrial applications continue. Applications Perfluorinated compounds are used ubiquitously: For example, fluorosurfactants are widely used in the production of teflon (PTFE) and related fluorinated po ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amines
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group (these may respectively be called alkylamines and arylamines; amines in which both types of substituent are attached to one nitrogen atom may be called alkylarylamines). Important amines include amino acids, biogenic amines, trimethylamine, and aniline; Inorganic derivatives of ammonia are also called amines, such as monochloramine (). The substituent is called an amino group. Compounds with a nitrogen atom attached to a carbonyl group, thus having the structure , are called amides and have different chemical properties from amines. Classification of amines Amines can be classified according to the nature and number of substituents on nitrogen. Aliphatic amines contain only H and alkyl substituents. Aromatic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |