|
Orders Of Magnitude (voltage)
To help compare different orders of magnitude, the following list describes various voltage levels. SI multiple Notes External links {{Orders of magnitude Voltage Voltage, also known as electric pressure, electric tension, or (electric) potential difference, is the difference in electric potential between two points. In a static electric field, it corresponds to the work needed per unit of charge to m ... Voltage ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Order Of Magnitude
An order of magnitude is an approximation of the logarithm of a value relative to some contextually understood reference value, usually 10, interpreted as the base of the logarithm and the representative of values of magnitude one. Logarithmic distributions are common in nature and considering the order of magnitude of values sampled from such a distribution can be more intuitive. When the reference value is 10, the order of magnitude can be understood as the number of digits in the base-10 representation of the value. Similarly, if the reference value is one of some powers of 2, since computers store data in a binary format, the magnitude can be understood in terms of the amount of computer memory needed to store that value. Differences in order of magnitude can be measured on a base-10 logarithmic scale in “decades” (i.e., factors of ten). Examples of numbers of different magnitudes can be found at Orders of magnitude (numbers). Definition Generally, the order of magnitude ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
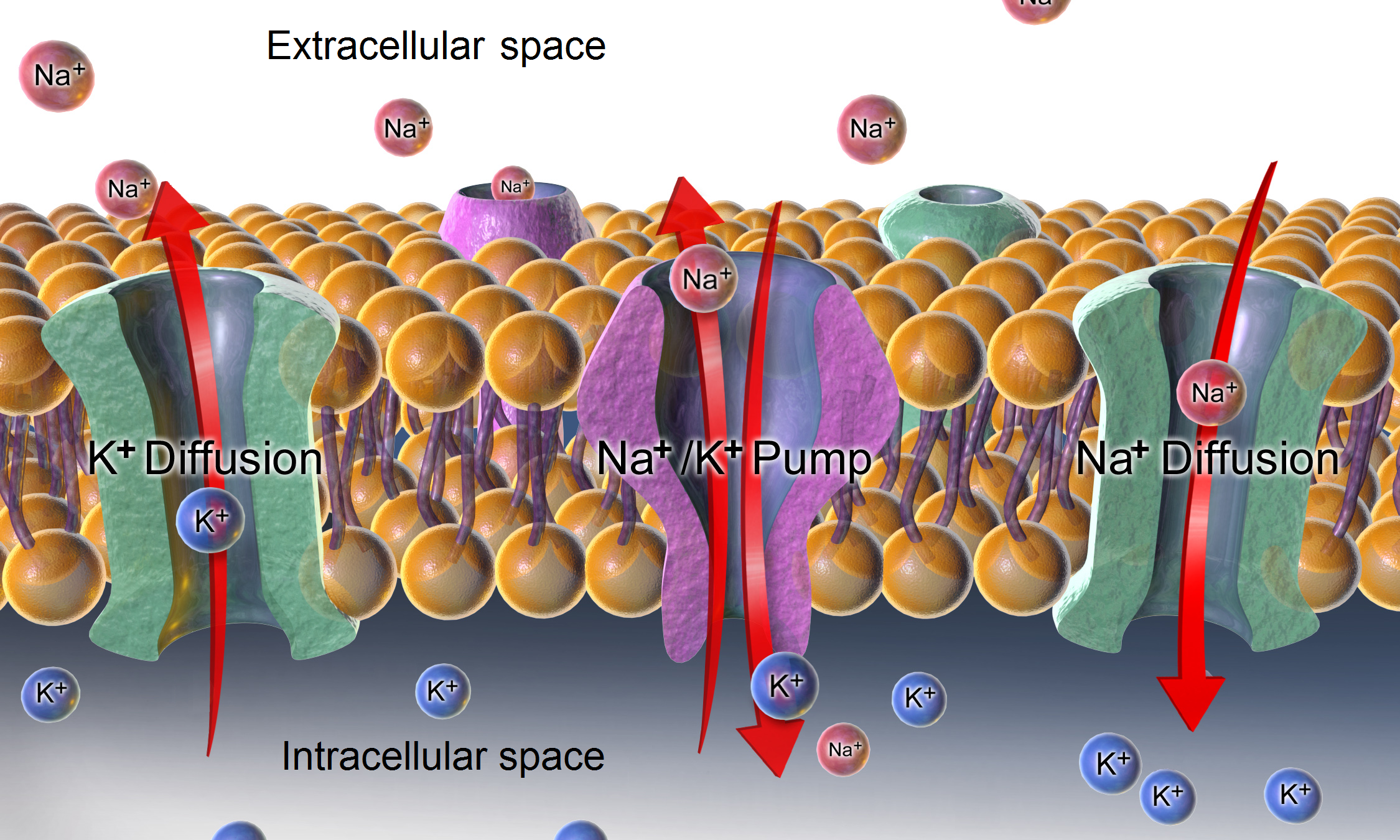
Resting Potential
A relatively static membrane potential which is usually referred to as the ground value for trans-membrane voltage. The relatively static membrane potential of quiescent cells is called the resting membrane potential (or resting voltage), as opposed to the specific dynamic electrochemical phenomena called action potential and graded membrane potential. Apart from the latter two, which occur in excitable cells (neurons, muscles, and some secretory cells in glands), membrane voltage in the majority of non-excitable cells can also undergo changes in response to environmental or intracellular stimuli. The resting potential exists due to the differences in membrane permeabilities for potassium, sodium, calcium, and chloride ions, which in turn result from functional activity of various ion channels, ion transporters, and exchangers. Conventionally, resting membrane potential can be defined as a relatively stable, ground value of transmembrane voltage in animal and plant cells. Beca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkaline Battery
An alkaline battery (IEC code: L) is a type of primary battery where the electrolyte (most commonly potassium hydroxide) has a pH value above 7. Typically these batteries derive energy from the reaction between zinc metal and manganese dioxide, nickel and cadmium, or nickel and hydrogen. Compared with zinc–carbon batteries of the Leclanché cell or zinc chloride types, alkaline batteries have a higher energy density and longer shelf life, yet provide the same voltage. The alkaline battery gets its name because it has an alkaline electrolyte of potassium hydroxide (KOH) instead of the acidic ammonium chloride (NH4Cl) or zinc chloride (ZnCl2) electrolyte of the zinc–carbon batteries. Other battery systems also use alkaline electrolytes, but they use different active materials for the electrodes. Alkaline batteries account for 80% of manufactured batteries in the US and over 10 billion individual units produced worldwide. In Japan, alkaline batteries account for 46% of all ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Standard Electrode Potential
In electrochemistry, standard electrode potential E^\ominus, or E^\ominus_, is a measure of the reducing power of any element or compound. The IUPAC "Gold Book" defines it as: ''"the value of the standard emf (electromotive force) of a cell in which molecular hydrogen under standard pressure is oxidized to solvated protons at the left-hand electrode"''. Background The basis for an electrochemical cell, such as the galvanic cell, is always a redox reaction which can be broken down into two half-reactions: oxidation at anode (loss of electron) and reduction at cathode (gain of electron). Electricity is produced due to the difference of electric potential between the individual potentials of the two metal electrodes with respect to the electrolyte. Although the overall potential of a cell can be measured, there is no simple way to accurately measure the electrode/electrolyte potentials in isolation. The electric potential also varies with temperature, concentration and pressure. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic table. In some respects, zinc is chemically similar to magnesium: both elements exhibit only one normal oxidation state (+2), and the Zn2+ and Mg2+ ions are of similar size.The elements are from different metal groups. See periodic table. Zinc is the 24th most abundant element in Earth's crust and has five stable isotopes. The most common zinc ore is sphalerite (zinc blende), a zinc sulfide mineral. The largest workable lodes are in Australia, Asia, and the United States. Zinc is refined by froth flotation of the ore, roasting, and final extraction using electricity ( electrowinning). Zinc is an essential trace element for humans, animals, plants and for microorganisms and is necessary for prenatal and postnatal development. It ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement. Copper is one of the few metals that can occur in nature in a directly usable metallic form ( native metals). This led to very early human use in several regions, from circa 8000 BC. Thousands of years later, it was the first metal to be smelted from sulfide ores, circa 5000 BC; the first metal to be cast into a shape in a mold, c. 4000 BC; and the first metal to be purposely alloyed with another metal, tin, to create ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lemon Battery
A lemon battery is a simple battery often made for the purpose of education. Typically, a piece of zinc metal (such as a galvanized nail) and a piece of copper (such as a penny) are inserted into a lemon and connected by wires. Power generated by reaction of the metals is used to power a small device such as a light-emitting diode (LED). The lemon battery is similar to the first electrical battery invented in 1800 by Alessandro Volta, who used brine (salt water) instead of lemon juice. The lemon battery illustrates the type of chemical reaction ( oxidation-reduction) that occurs in batteries. The zinc and copper are called the electrodes, and the juice inside the lemon is called the electrolyte. There are many variations of the lemon cell that use different fruits (or liquids) as electrolytes and metals other than zinc and copper as electrodes. Use in school projects There are numerous sets of instructions for making lemon batteries and for obtaining components such as light-em ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Integrated Circuit
An integrated circuit or monolithic integrated circuit (also referred to as an IC, a chip, or a microchip) is a set of electronic circuits on one small flat piece (or "chip") of semiconductor material, usually silicon. Large numbers of tiny MOSFETs (metal–oxide–semiconductor field-effect transistors) integrate into a small chip. This results in circuits that are orders of magnitude smaller, faster, and less expensive than those constructed of discrete electronic components. The IC's mass production capability, reliability, and building-block approach to integrated circuit design has ensured the rapid adoption of standardized ICs in place of designs using discrete transistors. ICs are now used in virtually all electronic equipment and have revolutionized the world of electronics. Computers, mobile phones and other home appliances are now inextricable parts of the structure of modern societies, made possible by the small size and low cost of ICs such as modern computer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CMOS
Complementary metal–oxide–semiconductor (CMOS, pronounced "sea-moss", ) is a type of metal–oxide–semiconductor field-effect transistor (MOSFET) fabrication process that uses complementary and symmetrical pairs of p-type and n-type MOSFETs for logic functions. CMOS technology is used for constructing integrated circuit (IC) chips, including microprocessors, microcontrollers, memory chips (including CMOS BIOS), and other digital logic circuits. CMOS technology is also used for analog circuits such as image sensors (CMOS sensors), data converters, RF circuits (RF CMOS), and highly integrated transceivers for many types of communication. The CMOS process was originally conceived by Frank Wanlass at Fairchild Semiconductor and presented by Wanlass and Chih-Tang Sah at the International Solid-State Circuits Conference in 1963. Wanlass later filed US patent 3,356,858 for CMOS circuitry and it was granted in 1967. commercialized the technology with the trademark "COS-MO ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IC Power Supply Pin
IC power-supply pins denote a voltage and current supply terminals in electric, electronics engineering, and in Integrated circuit design. Integrated circuits (ICs) have at least two pins that connect to the power rails of the circuit in which they are installed. These are known as the power-supply pins. However, the labeling of the pins varies by IC family and manufacturer. The double subscript notation usually corresponds to a first letter in a given IC family (transistors) notation of the terminals (e.g. VDD supply for a drain terminal in FETs etc.). The simplest labels are V+ and V−, but internal design and historical traditions have led to a variety of other labels being used. V+ and V− may also refer to the non-inverting (+) and inverting (−) voltage inputs of ICs like op amps. For power supplies, sometimes one of the supply rails is referred to as ground (abbreviated "GND") positive and negative voltages are relative to the ground. In digital electronics, negat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diode
A diode is a two-terminal electronic component that conducts current primarily in one direction (asymmetric conductance); it has low (ideally zero) resistance in one direction, and high (ideally infinite) resistance in the other. A diode vacuum tube or thermionic diode is a vacuum tube with two electrodes, a heated cathode and a plate, in which electrons can flow in only one direction, from cathode to plate. A semiconductor diode, the most commonly used type today, is a crystalline piece of semiconductor material with a p–n junction connected to two electrical terminals. Semiconductor diodes were the first semiconductor electronic devices. The discovery of asymmetric electrical conduction across the contact between a crystalline mineral and a metal was made by German physicist Ferdinand Braun in 1874. Today, most diodes are made of silicon, but other semiconducting materials such as gallium arsenide and germanium are also used. Among many uses, diodes are found in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Threshold Voltage
The threshold voltage, commonly abbreviated as Vth or VGS(th), of a field-effect transistor (FET) is the minimum gate-to-source voltage (VGS) that is needed to create a conducting path between the source and drain terminals. It is an important scaling factor to maintain power efficiency. When referring to a junction field-effect transistor (JFET), the threshold voltage is often called pinch-off voltage instead. This is somewhat confusing since ''pinch off'' applied to insulated-gate field-effect transistor (IGFET) refers to the channel pinching that leads to current saturation behaviour under high source–drain bias, even though the current is never off. Unlike ''pinch off'', the term ''threshold voltage'' is unambiguous and refers to the same concept in any field-effect transistor. Basic principles In n-channel ''enhancement-mode'' devices, a conductive channel does not exist naturally within the transistor, and a positive gate-to-source voltage is necessary to create one su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.jpg)


