|
Organoselenium
Organoselenium compounds (or seleno-organic) are chemical compounds containing carbon-to-selenium chemical bonds. Organoselenium chemistry is the corresponding science exploring their properties and reactivity. Selenium belongs with oxygen and sulfur to the group 16 elements or chalcogens, and similarities in chemistry are to be expected. Organoselenium compounds are found at trace levels in ambient waters, soils and sediments. Selenium can exist with oxidation state −2, +2, +4, +6. Se(II) is the dominant form in organoselenium chemistry. Down the group 16 column, the bond strength becomes increasingly weaker (234 kilojoule, kJ/mole (unit), mol for the C−Se bond and 272 kJ/mol for the C−S bond) and the bond lengths longer (C−Se 198 pm, C−S 181 pm and C−O 141 pm). Selenium compounds are more nucleophilic than the corresponding sulfur compounds and also more acidic. The pKa, p''K''a values of XH2 are 16 for oxygen, 7 for sulfur and 3.8 for selenium. In contrast to sulfoxi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organoselenium Compounds
Organoselenium compounds (or seleno-organic) are chemical compounds containing carbon-to-selenium chemical bonds. Organoselenium chemistry is the corresponding science exploring their properties and reactivity. Selenium belongs with oxygen and sulfur to the group 16 elements or chalcogens, and similarities in chemistry are to be expected. Organoselenium compounds are found at trace levels in ambient waters, soils and sediments. Selenium can exist with oxidation state −2, +2, +4, +6. Se(II) is the dominant form in organoselenium chemistry. Down the group 16 column, the bond strength becomes increasingly weaker (234 kilojoule, kJ/mole (unit), mol for the C−Se bond and 272 kJ/mol for the C−S bond) and the bond lengths longer (C−Se 198 pm, C−S 181 pm and C−O 141 pm). Selenium compounds are more nucleophilic than the corresponding sulfur compounds and also more acidic. The pKa, p''K''a values of XH2 are 16 for oxygen, 7 for sulfur and 3.8 for selenium. In contrast to sulfoxi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenium
Selenium is a chemical element with the symbol Se and atomic number 34. It is a nonmetal (more rarely considered a metalloid) with properties that are intermediate between the elements above and below in the periodic table, sulfur and tellurium, and also has similarities to arsenic. It seldom occurs in its elemental state or as pure ore compounds in the Earth's crust. Selenium – from Greek ( 'Moon') – was discovered in 1817 by , who noted the similarity of the new element to the previously discovered tellurium (named for the Earth). Selenium is found in metal sulfide ores, where it partially replaces the sulfur. Commercially, selenium is produced as a byproduct in the refining of these ores, most often during production. Minerals that are pure selenide or selenate compounds are known but rare. The chief commercial uses for selenium today are glassmaking and pigments. Selenium is a semiconductor and is used in photocells. Applications in electronics, once important, have been ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
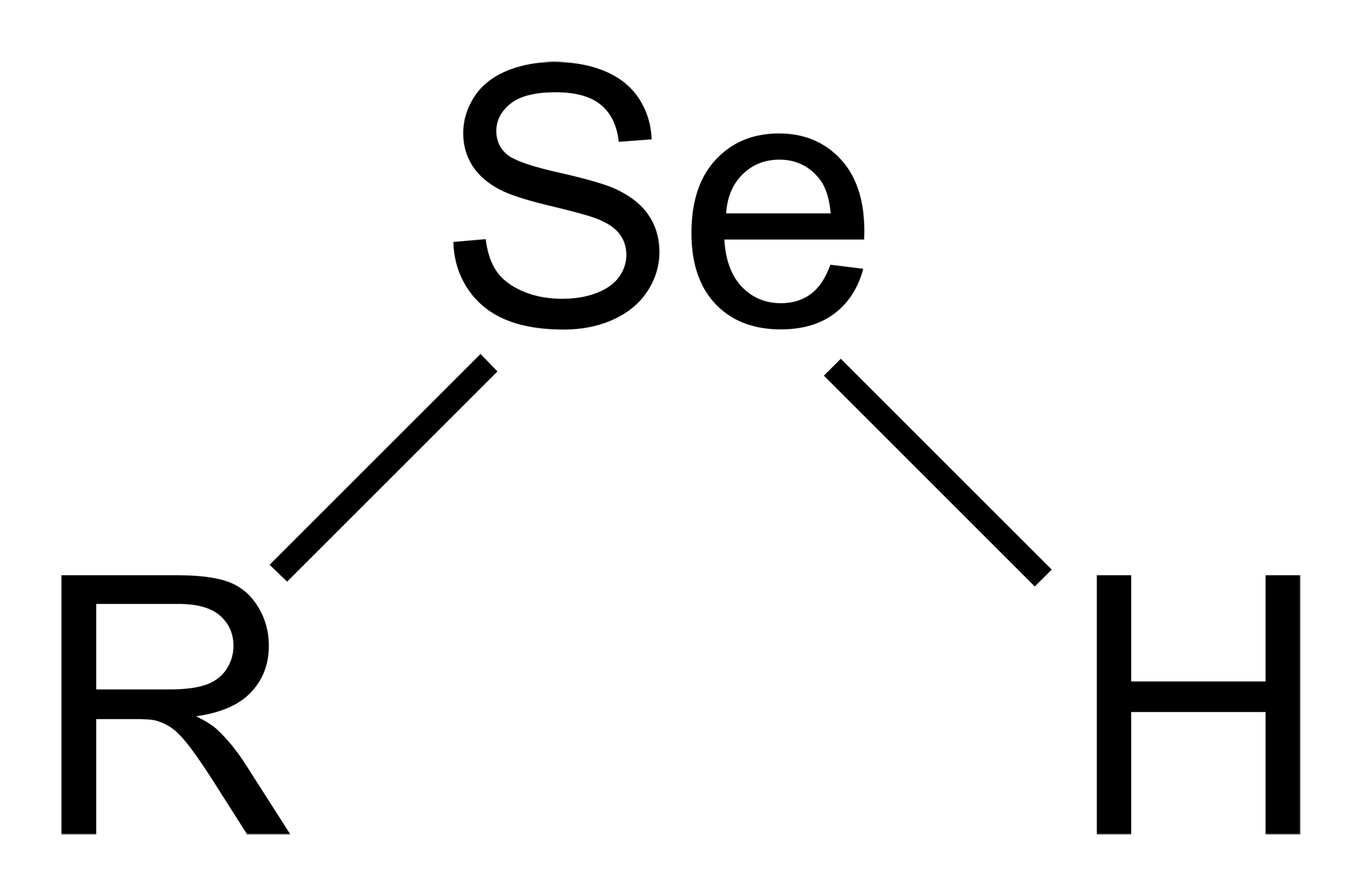
Selenol
Selenols are organic compounds that contain the functional group with the connectivity C–Selenium, Se–H. Selenols are sometimes also called selenomercaptans and selenothiols. Selenols are one of the principal classes of organoselenium compounds. The best known member of the group is the amino acid selenocysteine. Structure, bonding, properties Selenols are structurally similar to thiols, but the C-Se bond is about 8% longer at 196 pm. The C–Se–H angle approaches 90°. The bonding involves almost pure p-orbitals on Se, hence the near 90 angles. The Se–H bond energy is weaker than the S–H bond, consequently selenols are easily oxidized and serve as H-atom donors. The Se-H bond is much weaker than the S-H bond as reflected in their respective bond dissociation energy (BDE). For C6H5Se-H, the BDE is 326 kJ/mol, while for C6H5S-H, the BDE is 368 kJ/mol. Selenol acids are about 1000 times stronger than thiols: the p''K''a of CH3SeH is 5.2 vs 8.3 for CH3SH. Deprotonation affo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethyl Selenide
Diethyl selenide is an organoselenium compound with the formula . First reported in 1836, it was the first organoselenium compound to be discovered. It is the selenium analogue of diethyl ether. It has a strong and unpleasant smell. Occurrence Diethyl selenide has been detected in biofuel produced from plantain peel. It is also a minor air pollutant in some areas. Preparation It may be prepared by a substitution reaction similar to the Williamson ether synthesis: reaction of a metal selenide, such as sodium selenide, with two equivalents of ethyl iodide or similar reagent to supply the ethyl group In organic chemistry, an ethyl group (abbr. Et) is an alkyl substituent with the formula , derived from ethane (). ''Ethyl'' is used in the International Union of Pure and Applied Chemistry's nomenclature of organic chemistry for a saturated ...s: References {{reflist Organoselenium compounds Foul-smelling chemicals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzeneselenol
Benzeneselenol, also known as selenophenol, is the organoselenium compound with the formula C6H5SeH, often abbreviated PhSeH. It is the selenium analog of phenol. This colourless, malodorous compound is a reagent in organic synthesis. Synthesis Benzeneselenol is prepared by the reaction of phenylmagnesium bromide and selenium: :PhMgBr + Se → PhSeMgBr :PhSeMgBr + HCl → PhSeH + MgBrCl Since benzeneselenol does not have a long shelflife, it is often generated in situ. A common method is by reduction of diphenyldiselenide. A further reason for this conversion is that often, it is the anion that is sought. Reactions More so than thiophenol, benzeneselenol is easily oxidized by air. The facility of this reaction reflects the weakness of the Se-H bond, bond dissociation energy of which is estimated to be between 67 and 74 kcal/mol. In contrast, the S-H BDE for thiophenol is near 80 kcal/mol. The product is diphenyl diselenide as shown in this idealized equation: : ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylselenide
Dimethyl selenide is the organoselenium compound with the formula (CH3)2Se. This colorless, malodorous, liquid is the simplest selenoether. It occurs in trace amounts in anaerobic environments. Dimethyl selenide is prepared by treating Se2- sources with electrophilic methylating agents such as methyl iodide Iodomethane, also called methyl iodide, and commonly abbreviated "MeI", is the chemical compound with the formula CH3I. It is a dense, colorless, volatile liquid. In terms of chemical structure, it is related to methane by replacement of one h ...: :Na2Se + 2 CH3I → (CH3)2Se + 2 NaI References {{DEFAULTSORT:Organoselenium Chemistry Organoselenium compounds Selenium(−II) compounds Selenoethers Foul-smelling chemicals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diphenyldiselenide
Diphenyl diselenide is the chemical compound with the formula (C6H5)2Se2, abbreviated Ph2Se2. This orange-coloured solid is the oxidized derivative of benzeneselenol. It is used as a source of the PhSe unit in organic synthesis. Preparation and properties Ph2Se2 is prepared by the oxidation of benzeneselenoate, the conjugate base of benzeneselenol which is generated via the Grignard reagent: : PhMgBr + Se → PhSeMgBr :2 PhSeMgBr + Br2 → Ph2Se2 + 2 MgBr2 The molecule has idealized C2-symmetry, like hydrogen peroxide and related molecules. The Se-Se bond length of 2.29 Å the C-Se-Se-C dihedral angle is 82° and the C-Se-Se angles are near 110°. Antioxidant action "Dietary supplementation with Ph2Se2 prevented CH3HgCl-induced locomotor impairment. This effect appeared to be mediated by antioxidant action. Ph2Se2 may be a viable approach to prevention or reduction CH3HgCl-mediated neurotoxic effects." The protective effects of diphenyl diselenide against met ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Group 16 Element
The chalcogens (ore forming) ( ) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. Group 16 consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive elements polonium (Po) and livermorium (Lv). Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. The word "chalcogen" is derived from a combination of the Greek word () principally meaning copper (the term was also used for bronze/brass, any metal in the poetic sense, ore or coin), and the Latinized Greek word , meaning ''born'' or ''produced''. Sulfur has been known since antiquity, and oxygen was recognized as an element in the 18th century. Selenium, tellurium and polonium were discovered in the 19th century, and livermorium in 2000. All of the chalcogens ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Selenide
Sodium selenide is an inorganic compound of sodium and selenium with the chemical formula Na2Se. Preparation This colourless solid is prepared by the reaction of selenium with a solution of sodium in liquid ammonia at −40 °C.Brauer, G. ed. (1963) ''Handbook of Preparative Inorganic Chemistry'', 2nd Ed., Academic Press, NY., Vol. 1. p. 421. Alternatively, sodium selenide can be prepared by the reaction of gaseous hydrogen selenide with metallic sodium at 100 °C. Reactions Like other alkali metal chalcogenides, this material is highly sensitive to water, easily undergoing hydrolysis to give mixtures of sodium biselenide (NaSeH) and hydroxide. This hydrolysis occurs because of the extreme basicity of the Se2− ion. :Na2Se + H2O → NaHSe + NaOH Similarly, sodium selenide is readily oxidized to polyselenides, a conversion signaled by off-white samples. Sodium selenide reacts with acids to produce toxic hydrogen selenide gas. :Na2Se + 2 HCl → H2Se + 2 NaCl The compound r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases. ''Nucleophilic'' describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. History The terms ''nucleophile'' and ''electrophile'' were introduced by Christopher Kelk Ingold in 1933, replacing the terms ''anionoid'' and ''cationoid' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be classified into two varieties: if the alkyl or aryl groups are the same on both sides of the oxygen atom, then it is a simple or symmetrical ether, whereas if they are different, the ethers are called mixed or unsymmetrical ethers. A typical example of the first group is the solvent and anaesthetic diethyl ether, commonly referred to simply as "ether" (). Ethers are common in organic chemistry and even more prevalent in biochemistry, as they are common linkages in carbohydrates and lignin. Structure and bonding Ethers feature bent C–O–C linkages. In dimethyl ether, the bond angle is 111° and C–O distances are 141 pm. The barrier to rotation about the C–O bonds is low. The bonding of oxygen in ethers, alcohols, and water is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylmagnesium Bromide
Phenylmagnesium bromide, with the simplified formula , is a magnesium-containing organometallic compound. It is commercially available as a solution in diethyl ether or tetrahydrofuran (THF). Phenylmagnesium bromide is a Grignard reagent. It is often used as a synthetic equivalent for the phenyl "Ph−" synthon. Preparation Phenylmagnesium bromide is commercially available as solutions of diethyl ether or THF. Laboratory preparation involves treating bromobenzene with magnesium metal, usually in the form of turnings. A small amount of iodine may be used to activate the magnesium to initiate the reaction. Coordinating solvents such as ether or THF, are required to solvate (complex) the magnesium(II) center. The solvent must be aprotic since alcohols and water contain an acidic proton and thus react with phenylmagnesium bromide to give benzene. Carbonyl-containing solvents, such as acetone and ethyl acetate, are also incompatible with the reagent. Structure Although phenylmagn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


.png)

