|
Oestrone
Estrone (E1), also spelled oestrone, is a steroid, a weak estrogen, and a minor female sex hormone. It is one of three major endogenous estrogens, the others being estradiol and estriol. Estrone, as well as the other estrogens, are synthesized from cholesterol and secreted mainly from the gonads, though they can also be formed from adrenal androgens in adipose tissue. Relative to estradiol, both estrone and estriol have far weaker activity as estrogens. Estrone can be converted into estradiol, and serves mainly as a precursor or metabolic intermediate of estradiol. It is both a precursor and metabolite of estradiol. In addition to its role as a natural hormone, estrone has been used as a medication, for instance in menopausal hormone therapy; for information on estrone as a medication, see the estrone (medication) article. Biological activity Estrone is an estrogen, specifically an agonist of the estrogen receptors ERα and ERβ. It is a far less potent estrogen than is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Estrone (medication)
Estrone (E1), sold under the brand names Estragyn, Kestrin, and Theelin among many others, is an estrogen medication and naturally occurring steroid hormone which has been used in menopausal hormone therapy and for other indications. It has been provided as an aqueous suspension or oil solution given by injection into muscle and as a vaginal cream applied inside of the vagina. It can also be taken by mouth as estradiol/estrone/estriol (brand name Hormonin) and in the form of prodrugs like estropipate (estrone sulfate; brand name Ogen) and conjugated estrogens (mostly estrone sulfate; brand name Premarin). Side effects of estrogens like estrone include breast tenderness, breast enlargement, headache, nausea, fluid retention, and edema, among others. Estrone is a naturally occurring and bioidentical estrogen, or an agonist of the estrogen receptor, the biological target of estrogens like endogenous estradiol. It is a relatively weak estrogen, with much lower activity than es ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Steroid
A steroid is a biologically active organic compound with four rings arranged in a specific molecular configuration. Steroids have two principal biological functions: as important components of cell membranes that alter membrane fluidity; and as signaling molecules. Hundreds of steroids are found in plants, animals and fungi. All steroids are manufactured in cells from the sterols lanosterol (opisthokonts) or cycloartenol (plants). Lanosterol and cycloartenol are derived from the cyclization of the triterpene squalene. The steroid core structure is typically composed of seventeen carbon atoms, bonded in four " fused" rings: three six-member cyclohexane rings (rings A, B and C in the first illustration) and one five-member cyclopentane ring (the D ring). Steroids vary by the functional groups attached to this four-ring core and by the oxidation state of the rings. Sterols are forms of steroids with a hydroxy group at position three and a skeleton derived from cholestane. ''A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Menopausal Hormone Therapy
Hormone replacement therapy (HRT), also known as menopausal hormone therapy or postmenopausal hormone therapy, is a form of hormone therapy used to treat symptoms associated with female menopause. These symptoms can include hot flashes, vaginal atrophy, accelerated skin aging, vaginal dryness, decreased muscle mass, sexual dysfunction, and bone loss or osteoporosis. They are in large part related to the diminished levels of sex hormones that occur during menopause. Estrogens and progestogens are the main hormone drugs used in HRT. Progesterone is the main female sex hormone that occurs naturally and is also manufactured into a drug that is used in menopausal hormone therapy. Although both classes of hormones can have symptomatic benefit, progestogen is specifically added to estrogen regimens, unless the uterus has been removed, to avoid the increased risk of endometrial cancer. Unopposed estrogen therapy promotes endometrial hyperplasia and increases the risk of cancer, while ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand (biochemistry)
In biochemistry and pharmacology, a ligand is a substance that forms a complex with a biomolecule to serve a biological purpose. The etymology stems from ''ligare'', which means 'to bind'. In protein-ligand binding, the ligand is usually a molecule which produces a signal by binding to a site on a target protein. The binding typically results in a change of conformational isomerism (conformation) of the target protein. In DNA-ligand binding studies, the ligand can be a small molecule, ion, or protein which binds to the DNA double helix. The relationship between ligand and binding partner is a function of charge, hydrophobicity, and molecular structure. Binding occurs by intermolecular forces, such as ionic bonds, hydrogen bonds and Van der Waals forces. The association or docking is actually reversible through dissociation. Measurably irreversible covalent bonding between a ligand and target molecule is atypical in biological systems. In contrast to the definition of lig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Prohormone
A prohormone is a committed Precursor (chemistry), precursor of a hormone consisting of peptide hormones synthesized together that has a minimal hormonal effect by itself because of its expression-suppressing structure, often created by protein folding and binding additional peptide chains to certain ends, that makes hormone receptor binding sites located on its peptide hormone chain segments inaccessible. Prohormones can travel the blood stream as a hormone in an inactivated form, ready to be activated later in the cell by post-translational modification. The body naturally produces prohormones as a way to regulate hormone expression, making them an optimal storage and transportation unit for inactive hormones. Once prohormones are needed to be expressed, prohormone convertase, a protein, cleaves the prohormones and separates them into one or more active hormones. Often in nature, this cleaving process happens immediately, and a prohormone is quickly converted to a set of one or mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
In Vivo
Studies that are ''in vivo'' (Latin for "within the living"; often not italicized in English) are those in which the effects of various biological entities are tested on whole, living organisms or cells, usually animals, including humans, and plants, as opposed to a tissue extract or dead organism. This is not to be confused with experiments done ''in vitro'' ("within the glass"), i.e., in a laboratory environment using test tubes, Petri dishes, etc. Examples of investigations ''in vivo'' include: the pathogenesis of disease by comparing the effects of bacterial infection with the effects of purified bacterial toxins; the development of non-antibiotics, antiviral drugs, and new drugs generally; and new surgical procedures. Consequently, animal testing and clinical trials are major elements of ''in vivo'' research. ''In vivo'' testing is often employed over ''in vitro'' because it is better suited for observing the overall effects of an experiment on a living subject. In dr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biotransformation
Biotransformation is the biochemical modification of one chemical compound or a mixture of chemical compounds. Biotransformations can be conducted with whole cells, their lysates, or purified enzymes. Increasingly, biotransformations are effected with purified enzymes. Major industries and life-saving technologies depend on biotransformations. Advantages and disadvantages Compared to the conventional production of chemicals, biotransformations are often attractive because their selectivities can be high, limiting the coproduction of undesirable coproducts. Generally operating under mild temperatures and pressures in aqueous solutions, many biotransformations are "green". The catalysts, i.e. the enzymes, are amenable to improvement by genetic manipulation. Biotechnology usually is restrained by substrate scope. Petrochemicals for example are often not amenable to biotransformations, especially on the scale required for some applications, e.g. fuels. Biotransformations can be s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
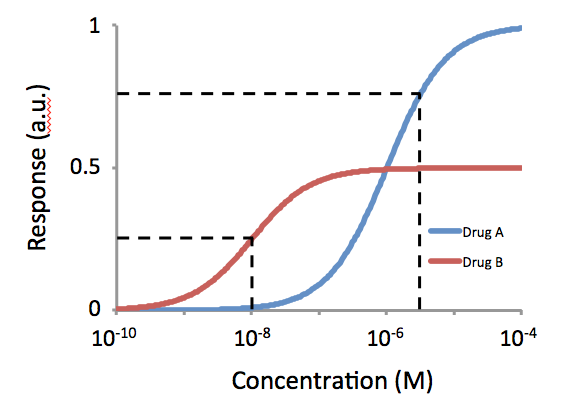
Relative Transactivational Capacities
Intrinsic activity (IA) and efficacy refer to the relative ability of a drug-receptor complex to produce a maximum functional response. This must be distinguished from the affinity, which is a measure of the ability of the drug to bind to its molecular target, and the EC50, which is a measure of the potency of the drug and which is proportional to both efficacy and affinity. This use of the word "efficacy" was introduced by Stephenson (1956) to describe the way in which agonists vary in the response they produce, even when they occupy the same number of receptors. High efficacy agonists can produce the maximal response of the receptor system while occupying a relatively low proportion of the receptors in that system. There is a distinction between efficacy and intrinsic activity. Mechanism of efficacy Agonists of lower efficacy are not as efficient at producing a response from the drug-bound receptor, by stabilizing the active form of the drug-bound receptor. Therefore, they may ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Relative Binding Affinities
In biochemistry and pharmacology, a ligand is a substance that forms a complex with a biomolecule to serve a biological purpose. The etymology stems from ''ligare'', which means 'to bind'. In protein-ligand binding, the ligand is usually a molecule which produces a signal by binding to a site on a target protein. The binding typically results in a change of conformational isomerism (conformation) of the target protein. In DNA-ligand binding studies, the ligand can be a small molecule, ion, or protein which binds to the DNA double helix. The relationship between ligand and binding partner is a function of charge, hydrophobicity, and molecular structure. Binding occurs by intermolecular forces, such as ionic bonds, hydrogen bonds and Van der Waals forces. The association or docking is actually reversible through dissociation. Measurably irreversible covalent bonding between a ligand and target molecule is atypical in biological systems. In contrast to the definition of ligand ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Subcutaneous Injection
Subcutaneous administration is the insertion of medications beneath the skin either by injection or infusion. A subcutaneous injection is administered as a bolus into the subcutis, the layer of skin directly below the dermis and epidermis, collectively referred to as the cutis. The instruments are usually a hypodermic needle and a syringe. Subcutaneous injections are highly effective in administering medications such as insulin, morphine, diacetylmorphine and goserelin. Subcutaneous administration may be abbreviated as SC, SQ, subcu, sub-Q, SubQ, or subcut. Subcut is the preferred abbreviation to reduce the risk of misunderstanding and potential errors. Subcutaneous tissue has few blood vessels and so drugs injected here are for slow, sustained rates of absorption, often with some amount of depot effect. Compared with other routes of administration, it is slower than intramuscular injections but still faster than intradermal injections. Subcutaneous infusion (as opposed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potency (pharmacology)
In the field of pharmacology, potency is a measure of drug activity expressed in terms of the amount required to produce an effect of given intensity. A highly potent drug (e.g., fentanyl, alprazolam, risperidone, bumetanide, bisoprolol) evokes a given response at low concentrations, while a drug of lower potency (meperidine, diazepam, ziprasidone, furosemide, metoprolol) evokes the same response only at higher concentrations. Higher potency does not necessarily mean greater effectiveness or more side effects. The IUPHAR The International Union of Basic and Clinical Pharmacology (IUPHAR) is a voluntary, non-profit association representing the interests of scientists in pharmacology-related fields to facilitate ''Better Medicines through Global Education and Resear ... has stated that 'potency' is ''"an imprecise term that should always be further defined"'', for instance as EC_, IC_, ED_, LD_ and so on. See also * Reaction inhibitor § Potency References Further readin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ERβ
Estrogen receptor beta (ERβ) also known as NR3A2 (nuclear receptor subfamily 3, group A, member 2) is one of two main types of estrogen receptor—a nuclear receptor which is activated by the sex hormone estrogen. In humans ERβ is encoded by the ''ESR2'' gene. Function ERβ is a member of the family of estrogen receptors and the superfamily of nuclear receptor transcription factors. The gene product contains an N-terminal DNA binding domain and C-terminal ligand binding domain and is localized to the nucleus, cytoplasm, and mitochondria. Upon binding to 17-β-estradiol, estriol or related ligands, the encoded protein forms homo-dimers or hetero-dimers with estrogen receptor α that interact with specific DNA sequences to activate transcription. Some isoforms dominantly inhibit the activity of other estrogen receptor family members. Several alternatively spliced transcript variants of this gene have been described, but the full-length nature of some of these variants h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |