|
Natrolite
Natrolite is a tectosilicate mineral species belonging to the zeolite group. It is a hydrated sodium and aluminium silicate with the formula . The type locality is Hohentwiel, Hegau, Germany. It was named natrolite by Martin Heinrich Klaproth in 1803. The name is derived from ''natron'' (), the Greek word for soda, in reference to the sodium content, and ''lithos'' (), meaning stone. Needle stone or needle-zeolite are other informal names, alluding to the common acicular habit of the crystals, which are often very slender and are aggregated in divergent tufts. The crystals are frequently epitaxial overgrowths of natrolite, mesolite, and gonnardite in various orders. Properties Larger crystals most commonly have the form of a square prism terminated by a low pyramid, the prism angle being nearly a right angle. The crystals are tetragonal in appearance, though actually orthorhombic. There are perfect cleavages parallel to the faces of the prism. The mineral also ofte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gonnardite
Gonnardite is a comparatively rare, fibrous zeolite, natrolite subgroup. Older papers claim that a complete solid solution exists between tetranatrolite and gonnardite, but tetranatrolite was discredited as a separate species in 1999.American Mineralogist (1999) 84: 1445-1450 A series, based on the disorder of the silicon-aluminum in the framework, appears to exist between Na-rich gonnardite and natrolite, Na2(Si3Al2)O10·2H2O.Tschernich, Zeolites of the World (1992) Geoscience Press, pages 215 to 225 Gonnardite was named in 1896 after Ferdinand Pierre Joseph Gonnard (1833–1923), who was Professor of Mining Engineering at the University of Lyon, France. Crystallography Orthorhombic-bipyramidal class 2/m 2/m 2/m and tetragonal-scalenoidal class 2m (orthorhombic with a very close to b, or tetragonal with a equal to b). Unit Cell Parameters: a = b = 13.21 Å, c = 6.622 Å, Z = 2Mineralogical Magazine (1998) 62: 548Space Group: I2d Crystal habit Crystals are prismatic, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zeolite
Zeolites are microporous, crystalline aluminosilicate materials commonly used as commercial adsorbents and catalysts. They mainly consist of silicon, aluminium, oxygen, and have the general formula ・y where is either a metal ion or H+. These positive ions can be exchanged for others in a contacting electrolyte solution. exchanged zeolites are particularly useful as solid acid catalysts. The term ''zeolite'' was originally coined in 1756 by Swedish mineralogist Axel Fredrik Cronstedt, who observed that rapidly heating a material, believed to have been stilbite, produced large amounts of steam from water that had been adsorbed by the material. Based on this, he called the material ''zeolite'', from the Greek , meaning "to boil" and , meaning "stone". Zeolites occur naturally but are also produced industrially on a large scale. , 253 unique zeolite frameworks have been identified, and over 40 naturally occurring zeolite frameworks are known. Every new zeolite structure th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mineral
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid chemical compound with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Minerals'; p. 1. In the series ''Geology: Landforms, Minerals, and Rocks''. Rosen Publishing Group. The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or are organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks. The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral, or may be an aggregate of two or more different types of minerals, spacially segregated into disti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesolite
Mesolite is a tectosilicate mineral with formula Na2 Ca2( Al2 Si3 O10)3·8 H2O. It is a member of the zeolite group and is closely related to natrolite which it also resembles in appearance. Mesolite crystallizes in the orthorhombic system and typically forms fibrous, acicular prismatic crystals or masses. Radiating sprays of needlelike crystals are not uncommon. It is vitreous in luster and clear to white in color. It has a Mohs hardness of 5 to 5.5 and a low specific gravity of 2.2 to 2.4. The refractive indices are nα=1.505 nβ=1.505 nγ=1.506. Occurrence It was first described in 1816 for an occurrence in the Cyclopean Islands near Catania, Sicily. From the Greek ''mesos'', "middle", as its composition lies between natrolite and scolecite. Like other zeolites, mesolite occurs as void fillings in amygdaloidal basalt also in andesites and hydrothermal veins. Images File:Mesolite (6911826414).jpg, Mesolite File:Mesolite-Quartz-238750.jpg, "Puff ball" of mesolite in a bas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acicular (crystal Habit)
__NOTOC__ Acicular, in mineralogy, refers to a crystal habit composed of slender, needle-like crystals. Crystals with this habit tend to be fragile. Complete, undamaged acicular specimens are uncommon. The term "acicular" derives from the Late Latin "acicula" meaning "little needle". Strictly speaking, the word refers to a growth habit that is slender and tapering to a point. Prismatic crystals are not acicular; however, colloquial usage has altered the commonly understood meaning of the word. When writing for mineralogical publications, authors should restrict their usage of "acicular" to crystals with the tapering growth habit. To add to the confusion, some minerals are described with various morphological terms. For example, natrolite is often described as slender prismatic and millerite is often described as filiform or capillary. Examples Minerals with an acicular habit include mesolite, natrolite, malachite, gypsum, rutile, brochantite and bultfonteinite. Crystals of d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. History In the early tenth century, the Persian physician and alchemist Abu Bakr al-Razi ( 865–925, Latin: Rhazes) conducted experiments with sal ammoniac ( ammonium chloride) and vitriol (hydrated sulfates of various metals), which he distilled together, thus producing the gas hydrogen chloride. In doing so, al-Razi may have stumbled upon a primitive method for producing hydrochloric acid, as perhaps manifested in the following recipe from his ("The Book of Secrets"): However, it appears that in most of his experiments al-Razi disregarded the gaseous products, concentrating instead on the color c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lustre (mineralogy)
Lustre (British English) or luster (American English; see spelling differences) is the way light interacts with the surface of a crystal, rock, or mineral. The word traces its origins back to the Latin ''lux'', meaning "light", and generally implies radiance, gloss, or brilliance. A range of terms are used to describe lustre, such as ''earthy'', ''metallic'', ''greasy'', and ''silky''. Similarly, the term ''vitreous'' (derived from the Latin for glass, ''vitrum'') refers to a glassy lustre. A list of these terms is given below. Lustre varies over a wide continuum, and so there are no rigid boundaries between the different types of lustre. (For this reason, different sources can often describe the same mineral differently. This ambiguity is further complicated by lustre's ability to vary widely within a particular mineral species). The terms are frequently combined to describe intermediate types of lustre (for example, a "vitreous greasy" lustre). Some minerals exhibit unu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Specific Gravity
Relative density, or specific gravity, is the ratio of the density (mass of a unit volume) of a substance to the density of a given reference material. Specific gravity for liquids is nearly always measured with respect to water (molecule), water at its densest (at ); for gases, the reference is air at room temperature (). The term "relative density" (often abbreviated r.d. or RD) is often preferred in scientific usage, whereas the term "specific gravity" is deprecation, deprecated. If a substance's relative density is less than 1 then it is less dense than the reference; if greater than 1 then it is denser than the reference. If the relative density is exactly 1 then the densities are equal; that is, equal volumes of the two substances have the same mass. If the reference material is water, then a substance with a relative density (or specific gravity) less than 1 will float in water. For example, an ice cube, with a relative density of about 0.91, will float. A substance wi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hardness
In materials science, hardness (antonym: softness) is a measure of the resistance to localized plastic deformation induced by either mechanical indentation or abrasion (mechanical), abrasion. In general, different materials differ in their hardness; for example hard metals such as titanium and beryllium are harder than soft metals such as sodium and metallic tin, or wood and common plastics. Macroscopic hardness is generally characterized by strong intermolecular bonds, but the behavior of solid materials under force is complex; therefore, hardness can be measured in different ways, such as scratch hardness, indentation hardness, and rebound hardness. Hardness is dependent on ductility, elasticity (physics), elastic stiffness, plasticity (physics), plasticity, deformation (mechanics), strain, strength of materials, strength, toughness, viscoelasticity, and viscosity. Common examples of hard matter are ceramics, concrete, certain metals, and superhard materials, which can be cont ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Igneous Rock
Igneous rock (derived from the Latin word ''ignis'' meaning fire), or magmatic rock, is one of the three main rock types, the others being sedimentary and metamorphic. Igneous rock is formed through the cooling and solidification of magma or lava. The magma can be derived from partial melts of existing rocks in either a planet's mantle or crust. Typically, the melting is caused by one or more of three processes: an increase in temperature, a decrease in pressure, or a change in composition. Solidification into rock occurs either below the surface as intrusive rocks or on the surface as extrusive rocks. Igneous rock may form with crystallization to form granular, crystalline rocks, or without crystallization to form natural glasses. Igneous rocks occur in a wide range of geological settings: shields, platforms, orogens, basins, large igneous provinces, extended crust and oceanic crust. Geological significance Igneous and metamorphic rocks make up 90–95% of the top ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silica
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , most commonly found in nature as quartz and in various living organisms. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and most abundant families of materials, existing as a compound of several minerals and as a synthetic product. Notable examples include fused quartz, fumed silica, silica gel, opal and aerogels. It is used in structural materials, microelectronics (as an electrical insulator), and as components in the food and pharmaceutical industries. Structure In the majority of silicates, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central Si atomsee 3-D Unit Cell. Thus, SiO2 forms 3-dimensional network solids in which each silicon atom is covalently bonded in a tetrahedral manner to 4 oxygen atoms. In contrast, CO2 is a linear molecule. The starkly different structures of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
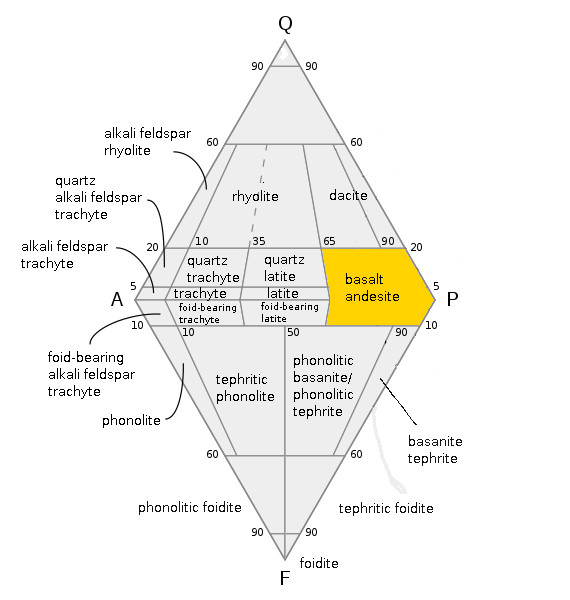
Basalt
Basalt (; ) is an aphanitic (fine-grained) extrusive igneous rock formed from the rapid cooling of low-viscosity lava rich in magnesium and iron (mafic lava) exposed at or very near the surface of a rocky planet or moon. More than 90% of all volcanic rock on Earth is basalt. Rapid-cooling, fine-grained basalt is chemically equivalent to slow-cooling, coarse-grained gabbro. The eruption of basalt lava is observed by geologists at about 20 volcanoes per year. Basalt is also an important rock type on other planetary bodies in the Solar System. For example, the bulk of the plains of Venus, which cover ~80% of the surface, are basaltic; the lunar maria are plains of flood-basaltic lava flows; and basalt is a common rock on the surface of Mars. Molten basalt lava has a low viscosity due to its relatively low silica content (between 45% and 52%), resulting in rapidly moving lava flows that can spread over great areas before cooling and solidifying. Flood basalts are thick sequence ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |