|
Methiopropamine Metabolism Colored
Methiopropamine (MPA) is a thiophene ring-based structural analog of methamphetamine originally reported in 1942. Chemically it is not a phenethylamine or amphetamine and is not their functional analog either. It originally appeared for public sale in the UK in December 2010 as a "research chemical" or "legal high", recently branded as Blow. It has limited popularity as a recreational stimulant. Pharmacology Methiopropamine functions as a selective norepinephrine-dopamine reuptake inhibitor that is approximately 1.85 times more selective for norepinephrine than dopamine. It is approximately one third as potent as dextroamphetamine as a norepinephrine reuptake inhibitor and one fifth as much as a dopamine reuptake inhibitor. It displays negligible activity as a serotonin reuptake inhibitor. Methiopropamine has the potential for significant acute toxicity with cardiovascular, gastrointestinal, and psychotic symptoms. Metabolism For ''N''-alkyl amphetamines, deamination an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
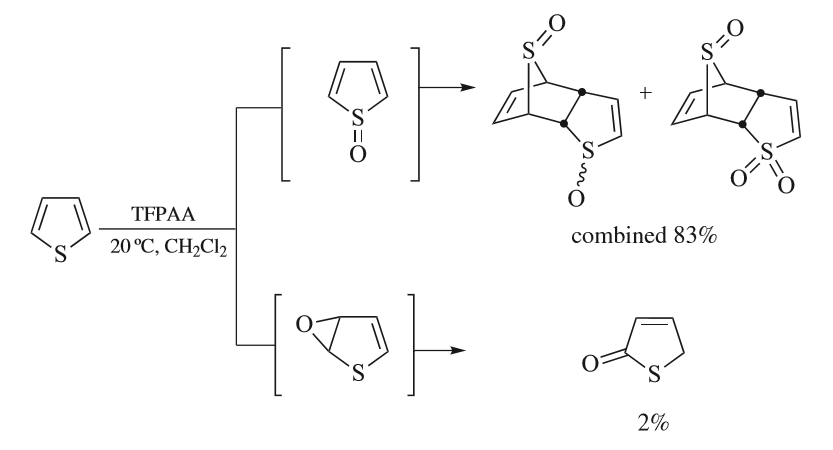
Thiophene
Thiophene is a heterocyclic compound with the formula C4H4S. Consisting of a planar five-membered ring, it is aromatic as indicated by its extensive substitution reactions. It is a colorless liquid with a benzene-like odor. In most of its reactions, it resembles benzene. Compounds analogous to thiophene include furan (C4H4O), selenophene (C4H4Se) and pyrrole (C4H4NH), which each vary by the heteroatom in the ring. Isolation and occurrence Thiophene was discovered as a contaminant in benzene. It was observed that isatin (an indole) forms a blue dye if it is mixed with sulfuric acid and crude benzene. The formation of the blue indophenin had long been believed to be a reaction of benzene itself. Viktor Meyer was able to isolate thiophene as the actual substance responsible for this reaction. Thiophene and especially its derivatives occur in petroleum, sometimes in concentrations up to 1–3%. The thiophenic content of oil and coal is removed via the hydrodesulfurization (HDS) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Demethylation
Demethylation is the chemical process resulting in the removal of a methyl group (CH3) from a molecule. A common way of demethylation is the replacement of a methyl group by a hydrogen atom, resulting in a net loss of one carbon and two hydrogen atoms. The counterpart of demethylation is methylation. In biochemistry In biochemical systems, the process of demethylation is catalyzed by demethylases. These enzymes oxidize N-methyl groups, which occur in histones and some forms of DNA: :R2N-CH3 + O → R2N-H + CH2O One such oxidative enzyme family is the cytochrome P450. Alpha-ketoglutarate-dependent hydroxylases are active for demethylation of DNA, operating by a similar pathway. These reactions exploit the weak C-H bond adjacent to amines. In particular, 5-methylcytosines in DNA can be demethylated by TET enzymes as illustrated in the figure. TET enzymes are dioxygenases in the family of alpha-ketoglutarate-dependent hydroxylases. A TET enzyme is an alpha-ketoglut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethylphenidate
Ethylphenidate (EPH), also known as Baxtercaine in the United Kingdom is a psychostimulant and a close analog of methylphenidate. Ethylphenidate acts as both a dopamine reuptake inhibitor and norepinephrine reuptake inhibitor, meaning it effectively boosts the levels of the norepinephrine and dopamine neurotransmitters in the brain, by binding to, and partially blocking the transporter proteins that normally remove those monoamines from the synaptic cleft. However, considering the close similarities between ethylphenidate and methylphenidate and the fact that methylphenidate, like cocaine, actually does not primarily act as a "classical" reuptake inhibitor, but rather as an "inverse agonist at the DAT" (also called a "negative allosteric modulator at the DAT"), it is at least very likely that ethylphenidate also primarily acts as an inverse DAT agonist instead of (or at least only secondarily) as a classical reuptake inhibitor (which could be called a "competitive antagonist at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methiopropamine Synthesis
Methiopropamine (MPA) is a thiophene ring-based structural analog of methamphetamine originally reported in 1942. Chemically it is not a phenethylamine or amphetamine and is not their functional analog either. It originally appeared for public sale in the UK in December 2010 as a "research chemical" or "legal high", recently branded as Blow. It has limited popularity as a recreational stimulant. Pharmacology Methiopropamine functions as a selective norepinephrine-dopamine reuptake inhibitor that is approximately 1.85 times more selective for norepinephrine than dopamine. It is approximately one third as potent as dextroamphetamine as a norepinephrine reuptake inhibitor and one fifth as much as a dopamine reuptake inhibitor. It displays negligible activity as a serotonin reuptake inhibitor. Methiopropamine has the potential for significant acute toxicity with cardiovascular, gastrointestinal, and psychotic symptoms. Metabolism For ''N''-alkyl amphetamines, deamination an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylamine
Methylamine is an organic compound with a formula of . This colorless gas is a derivative of ammonia, but with one hydrogen atom being replaced by a methyl group. It is the simplest primary amine. Methylamine is sold as a solution in methanol, ethanol, tetrahydrofuran, or water, or as the anhydrous gas in pressurized metal containers. Industrially, methylamine is transported in its anhydrous form in pressurized railcars and tank trailers. It has a strong odor similar to rotten fish. Methylamine is used as a building block for the synthesis of numerous other commercially available compounds. Industrial production Methylamine is prepared commercially by the reaction of ammonia with methanol in the presence of an aluminosilicate catalyst. Dimethylamine and trimethylamine are co-produced; the reaction kinetics and reactant ratios determine the ratio of the three products. The product most favored by the reaction kinetics is trimethylamine. : In this way, an estimated 115,000 tons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Tribromide
Phosphorus tribromide is a colourless liquid with the formula P Br3. The liquid fumes in moist air due to hydrolysis and has a penetrating odour. It is used in the laboratory for the conversion of alcohols to alkyl bromides. Preparation PBr3 is prepared by treating red phosphorus with bromine. An excess of phosphorus is used in order to prevent formation of PBr5: :2 P + 3 Br2 → 2 PBr3 Because the reaction is highly exothermic, it is often conducted in the presence of a diluent such as PBr3. Reactions Phosphorus tribromide, like PCl3 and PF3, has both properties of a Lewis base and a Lewis acid. For example, with a Lewis acid such as boron tribromide it forms stable 1 :1 adducts such as Br3B · PBr3. At the same time PBr3 can react as an electrophile or Lewis acid in many of its reactions, for example with amines. The most important reaction of PBr3 is with alcohols, where it replaces an OH group with a bromine atom to produce an alkyl bromide. All three bromides ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propylene Oxide
Propylene oxide is an acutely toxic and carcinogenic organic compound with the molecular formula CH3CHCH2O. This colourless volatile liquid with an odour similar to ether, is produced on a large scale industrially. Its major application is its use for the production of polyether polyols for use in making polyurethane plastics. It is a chiral epoxide, although it is commonly used as a racemic mixture. This compound is sometimes called 1,2-propylene oxide to distinguish it from its isomer 1,3-propylene oxide, better known as oxetane. Production Industrial production of propylene oxide starts from propylene. Two general approaches are employed, one involving hydrochlorination and the other involving oxidation. In 2005, about half of the world production was through chlorohydrin technology and one half via oxidation routes. The latter approach is growing in importance. Hydrochlorination route The traditional route proceeds via the conversion of propene to propylene chlorohydrin a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiopropamine
Thiopropamine is a stimulant drug which is an analogue of amphetamine where the phenyl ring has been replaced by thiophene. It has similar stimulant effects to amphetamine but with around one third the potency. The N-methyl and thiophen-3-yl analogues are also known and are somewhat more potent, though still generally weaker than the corresponding amphetamines. Pharmacology Like amphetamine and most of its analogues, thiopropamine most likely is a norepinephrine–dopamine reuptake inhibitor and/or releasing agent. Thiopropamine is likely to be metabolized into active 4-hydroxymethiopropamine and thiophene S- oxides. These are further deaminated by CYP2C in liver transforming them into inactive 1-(Thiophen-2-yl)-2-propan-2-one which is a phenylacetone derivative. Propan-2-amines are not metabolized by monoamine oxidases and most actually behave as competitive Competition is a rivalry where two or more parties strive for a common goal which cannot be shared: where o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrophilic
A hydrophile is a molecule or other molecular entity that is attracted to water molecules and tends to be dissolved by water.Liddell, H.G. & Scott, R. (1940). ''A Greek-English Lexicon'' Oxford: Clarendon Press. In contrast, hydrophobes are not attracted to water and may seem to be repelled by it. Hygroscopics ''are'' attracted to water, but are not dissolved by water. Molecules A hydrophilic molecule or portion of a molecule is one whose interactions with water and other polar substances are more thermodynamically favorable than their interactions with oil or other hydrophobic solvents. They are typically charge-polarized and capable of hydrogen bonding. This makes these molecules soluble not only in water but also in other polar solvents. Hydrophilic molecules (and portions of molecules) can be contrasted with hydrophobic molecules (and portions of molecules). In some cases, both hydrophilic and hydrophobic properties occur in a single molecule. An example of these amphiph ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiophene-2-carboxylic Acid
Thiophene-2-carboxylic acid is an organic compound with the formula SC4H3CO2H. It is one of two monocarboxylic acids of thiophene, the other being thiophene-3-carboxylic acid. Copper(I) thiophene-2-carboxylate is a catalyst for Ullmann coupling reactions. Synthesis It can be prepared by the oxidation of thiophene-2-carboxaldehyde or, more practically, 2-acetylthiophene.. Applications and reactions left, Suprofen, which is produced from thiophene-2-carboxylic acid, is the active ingredient in some eye drops. Upon treatment with lithium diisopropylamide, LDA, thiophene-2-carboxylic acid undergoes double deprotonation to give the 5-lithio derivative, a precursor to many 5-substituted derivatives. Thiophene-2-carboxylic acid has been widely studied as a substrate in coupling reaction A coupling reaction in organic chemistry is a general term for a variety of reactions where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a mai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenylacetone
Phenylacetone is an organic compound with the chemical formula C6H5CH2COCH3. It is a colorless oil that is soluble in organic solvents. This substance is used in the manufacture of methamphetamine and amphetamine, where it is commonly known as P2P. Due to the illicit uses in clandestine chemistry, it was declared a schedule II controlled substance in the United States in 1980. In humans, phenylacetone occurs as a metabolite of amphetamine and methamphetamine via FMO3-mediated oxidative deamination. Synthesis One method of creating phenylacetone is via: : phenylacetic acid (C8H8O2) + acetic anhydride (C4H6O3) + pyridine catalyst → phenylacetone (C9H10O) + carbon dioxide (CO2) + water (H2O) Sodium acetate has been used instead of pyridine. In the 20th century chloroacetone, benzene, and anhydrous aluminum chloride were used to synthesize phenylacetone.https://pubs.acs.org/doi/10.1021/ja01863a506 62, 6, 1622. https://doi.org/10.1021/ja01863a506 The reaction included Friedel� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Liver
The liver is a major Organ (anatomy), organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the Protein biosynthesis, synthesis of proteins and biochemicals necessary for digestion and growth. In humans, it is located in the quadrant (anatomy), right upper quadrant of the abdomen, below the thoracic diaphragm, diaphragm. Its other roles in metabolism include the regulation of Glycogen, glycogen storage, decomposition of red blood cells, and the production of hormones. The liver is an accessory digestive organ that produces bile, an alkaline fluid containing cholesterol and bile acids, which helps the fatty acid degradation, breakdown of fat. The gallbladder, a small pouch that sits just under the liver, stores bile produced by the liver which is later moved to the small intestine to complete digestion. The liver's highly specialized biological tissue, tissue, consisting mostly of hepatocytes, regulates a w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |