|
Levopropoxyphene
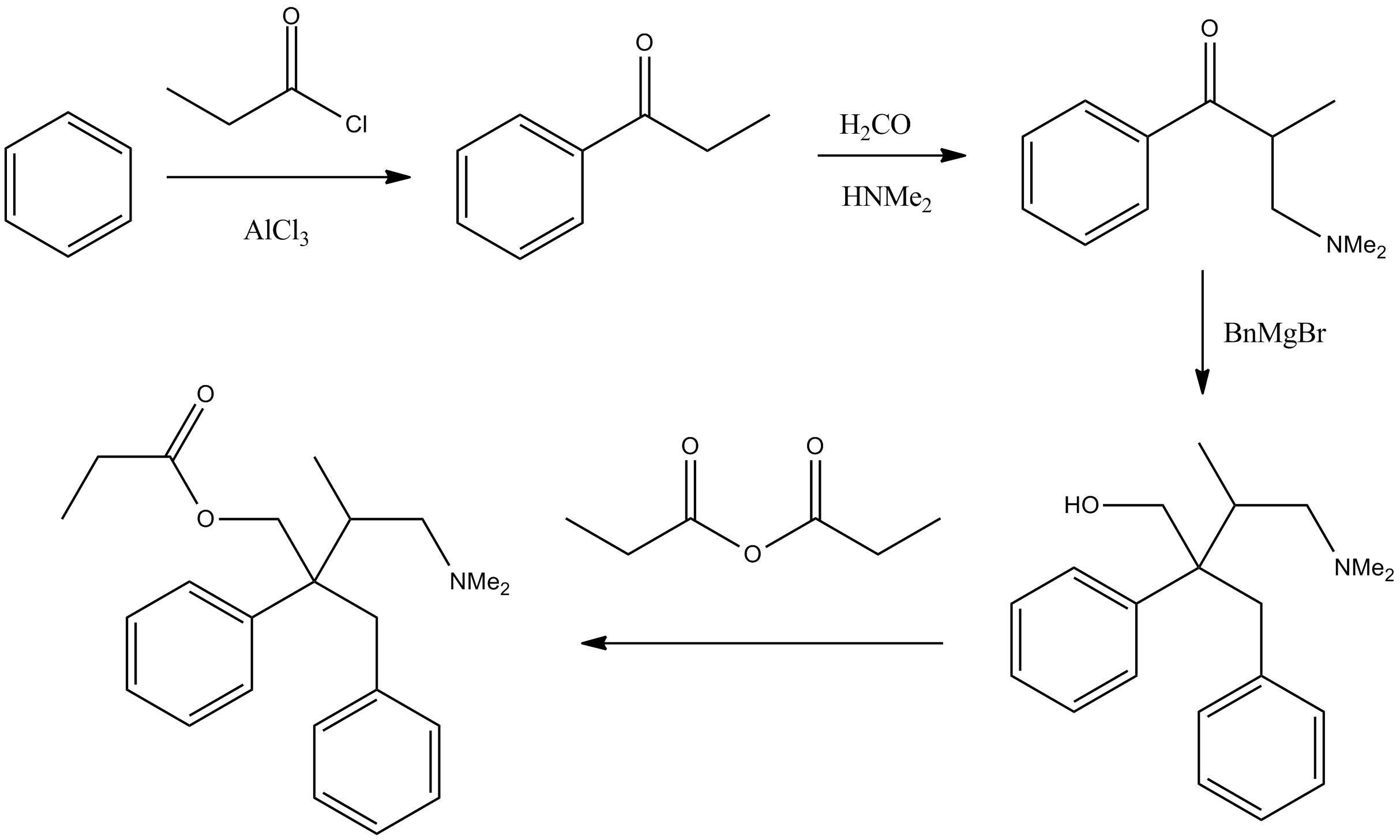
Levopropoxyphene is an antitussive. It is an optical isomer of dextropropoxyphene. The racemic mixture is called propoxyphene. Only the dextro-isomer (dextropropoxyphene) has an analgesic effect; the levo-isomer appears to exert only an antitussive effect. It was formerly marketed in the U.S. by Eli Lilly under the tradename Novrad (a reversal of Darvon) as an antitussive. Unlike many antitussives, it binds poorly to the sigma-1 receptor. Synthesis #Mannich reaction of propiophenone with formaldehyde and dimethylamine affords the corresponding aminoketone. #Reaction of the ketone with benzylmagnesium bromide gives the amino alcohol. It is of note that this intermediate fails to show analgesic activity in animal assays. #Esterification of the alcohol by means of propionic anhydride affords the propionate. Chirality The presence of two chiral centers in this molecule means that the compound can exist as any of four isomers. The biologic activity has been found to be associated wi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dextropropoxyphene
Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive (cough suppressant) and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia (pain relief) is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration. Dextropropoxyphene is sometimes combined with acetaminophen. Trade names include Darvocet-N, Di-Gesic, and Darvon with APAP (for dextropropoxyphene and paracetamol). The British approved name (i.e. the generic name of the active ingredient) of the paracetamol/dextropropoxyphene preparation is co-proxamol (sold under a vari ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propoxyphene
Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive (cough suppressant) and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia (pain relief) is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration. Dextropropoxyphene is sometimes combined with acetaminophen. Trade names include Darvocet-N, Di-Gesic, and Darvon with APAP (for dextropropoxyphene and paracetamol). The British approved name (i.e. the generic name of the active ingredient) of the paracetamol/dextropropoxyphene preparation is co-proxamol (sold under a var ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Darvon
Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive (cough suppressant) and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia (pain relief) is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration. Dextropropoxyphene is sometimes combined with acetaminophen. Trade names include Darvocet-N, Di-Gesic, and Darvon with APAP (for dextropropoxyphene and paracetamol). The British approved name (i.e. the generic name of the active ingredient) of the paracetamol/dextropropoxyphene preparation is co-proxamol (sold under a var ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propoxyphene Synthesis
Dextropropoxyphene is an analgesic in the opioid category, patented in 1955 and manufactured by Eli Lilly and Company. It is an optical isomer of levopropoxyphene. It is intended to treat mild pain and also has antitussive (cough suppressant) and local anaesthetic effects. The drug has been taken off the market in Europe and the US due to concerns of fatal overdoses and heart arrhythmias. It is still available in Australia, albeit with restrictions after an application by its manufacturer to review its proposed banning. Its onset of analgesia (pain relief) is said to be 20–30 minutes and peak effects are seen about 1.5–2.0 hours after oral administration. Dextropropoxyphene is sometimes combined with acetaminophen. Trade names include Darvocet-N, Di-Gesic, and Darvon with APAP (for dextropropoxyphene and paracetamol). The British approved name (i.e. the generic name of the active ingredient) of the paracetamol/dextropropoxyphene preparation is co-proxamol (sold under a v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antitussive
Cold medicines are a group of medications taken individually or in combination as a treatment for the symptoms of the common cold and similar conditions of the upper respiratory tract. The term encompasses a broad array of drugs, including analgesics, antihistamines and decongestants, among many others. It also includes drugs which are marketed as cough suppressants or antitussives, but their effectiveness in reducing cough symptoms is unclear or minimal. While they have been used by 10% of American children in any given week, they are not recommended in Canada or the United States in children six years or younger because of lack of evidence showing effect and concerns of harm. In 2020, one version containing codeine and guaifenesin was the 377th most commonly prescribed medication in the United States with more than 400thousand prescriptions. Types There are a number of different cough and cold medications, which may be used for various coughing symptoms. The commercially a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Optical Isomer
In chemistry, a molecule or ion is called chiral () if it cannot be superposed on its mirror image by any combination of rotations, translations, and some conformational changes. This geometric property is called chirality (). The terms are derived from Ancient Greek χείρ (''cheir'') 'hand'; which is the canonical example of an object with this property. A chiral molecule or ion exists in two stereoisomers that are mirror images of each other, called enantiomers; they are often distinguished as either "right-handed" or "left-handed" by their absolute configuration or some other criterion. The two enantiomers have the same chemical properties, except when reacting with other chiral compounds. They also have the same physical properties, except that they often have opposite optical activities. A homogeneous mixture of the two enantiomers in equal parts is said to be racemic, and it usually differs chemically and physically from the pure enantiomers. Chiral molecules will u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Racemate
In chemistry, a racemic mixture, or racemate (), is one that has equal amounts of left- and right-handed enantiomers of a chiral molecule or salt. Racemic mixtures are rare in nature, but many compounds are produced industrially as racemates. History The first known racemic mixture was racemic acid, which Louis Pasteur found to be a mixture of the two enantiomeric isomers of tartaric acid. He manually separated the crystals of a mixture by hand, starting from an aqueous solution of the sodium ammonium salt of racemate tartaric acid. Pasteur benefited from the fact that ammonium tartrate salt that gives enantiomeric crystals with distinct crystal forms (at 77 °F). Reasoning from the macroscopic scale down to the molecular, he reckoned that the molecules had to have non-superimposable mirror images. A sample with only a single enantiomer is an ''enantiomerically pure'' or ''enantiopure'' compound. Etymology From racemic acid found in grapes; from Latin ''racemus'', meani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sigma-1 Receptor
The sigma-1 receptor (σ1R), one of two sigma receptor subtypes, is a chaperone protein at the endoplasmic reticulum (ER) that modulates calcium signaling through the IP3 receptor. In humans, the σ1 receptor is encoded by the ''SIGMAR1'' gene. The σ1 receptor is a transmembrane protein expressed in many different tissue types. It is particularly concentrated in certain regions of the central nervous system. It has been implicated in several phenomena, including cardiovascular function, schizophrenia, clinical depression, the effects of cocaine abuse, and cancer. Much is known about the binding affinity of hundreds of synthetic compounds to the σ1 receptor. An endogenous ligand for the σ1 receptor has yet to be conclusively identified, but tryptaminergic trace amines and neuroactive steroids have been found to activate the receptor. Especially progesterone, but also testosterone, pregnenolone sulfate, and dehydroepiandrosterone sulfate (DHEA-S) bind to the σ1 receptor. C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mannich Reaction
In organic chemistry, the Mannich reaction is a three-component organic reaction that involves the amino alkylation of an acidic proton next to a carbonyl () functional group by formaldehyde () and a primary or secondary amine () or ammonia (). The final product is a β-amino-carbonyl compound also known as a Mannich base. Reactions between aldimines and α-methylene carbonyls are also considered Mannich reactions because these imines form between amines and aldehydes. The reaction is named after Carl Mannich. center, 500px, Scheme 1 - Ammonia or an amine reacts with formaldehyde and an alpha acidic proton of a carbonyl compound to a beta amino carbonyl compound. The Mannich reaction starts with the nucleophilic addition of an amine to a carbonyl group followed by dehydration to the Schiff base. The Schiff base is an electrophile which reacts in a second step in an electrophilic addition with an enol formed from a carbonyl compound containing an acidic alpha-proton. The Mann ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propiophenone
Propiophenone (shorthand: benzoylethane or BzEt) is an aryl ketone. It is a colorless, sweet-smelling liquid that is insoluble in water, but miscible with organic solvents. It is used in the preparation of other compounds. Production Propiophenone can be prepared by Friedel–Crafts reaction of propanoyl chloride and benzene. It is also prepared commercially by ketonization of benzoic acid and propionic acid over calcium acetate Calcium acetate is a chemical compound which is a calcium salt of acetic acid. It has the formula Ca(C2H3O2)2. Its standard name is calcium acetate, while calcium ethanoate is the systematic name. An older name is acetate of lime. The anhydrous ... and alumina at 450–550 °C: :C6H5CO2H + CH3CH2CO2H → C6H5C(O)CH2CH3 + CO2 + H2O Ludwig Claisen discovered that α-methoxystyrene forms this compound when heated for an hour at 300 °C (65% yield). Uses 120px, left, Phenmetrazine, derived from propiophenone, is an appetite suppressant."> ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formaldehyde
Formaldehyde ( , ) (systematic name methanal) is a naturally occurring organic compound with the formula and structure . The pure compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde (refer to section Forms below), hence it is stored as an aqueous solution (formalin), which is also used to store animal specimens. It is the simplest of the aldehydes (). The common name of this substance comes from its similarity and relation to formic acid. Formaldehyde is an important precursor to many other materials and chemical compounds. In 1996, the installed capacity for the production of formaldehyde was estimated at 8.7 million tons per year. It is mainly used in the production of industrial resins, e.g., for particle board and coatings. Forms Formaldehyde is more complicated than many simple carbon compounds in that it adopts several diverse forms. These compounds can often be used interchangeably and can be interconverted. *Molecular formald ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylamine
Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around 40%. An estimated 270,000 tons were produced in 2005. Structure and synthesis The molecule consists of a nitrogen atom with two methyl substituents and one proton. Dimethylamine is a weak base and the pKa of the ammonium CH3--CH3 is 10.73, a value above methylamine (10.64) and trimethylamine (9.79). Dimethylamine reacts with acids to form salts, such as dimethylamine hydrochloride, an odorless white solid with a melting point of 171.5 °C. Dimethylamine is produced by catalytic reaction of methanol and ammonia at elevated temperatures and high pressure: :2 CH3OH + NH3 → (CH3)2NH + 2 H2O Natural occurrence Dimethylamine is found quite widely distributed in animals and plants, and is present in many foods at the level of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |