|
Law Of Definite Composition
In chemistry, the law of definite proportions, sometimes called Proust's law, or law of constant composition states that a given chemical compound always contains its component elements in fixed ratio (by mass) and does not depend on its source and method of preparation. For example, oxygen makes up about 8/9 of the mass of any sample of pure water, while hydrogen makes up the remaining 1/9 of the mass: the mass of two elements in a compound are always in the same ratio. Along with the law of multiple proportions, the law of definite proportions forms the basis of stoichiometry. History The law of constant proportion was given by Joseph Proust in 1797. This observation was first made by the English theologian and chemist Joseph Priestley, and Antoine Lavoisier, a French nobleman and chemist centered on the process of combustion. The law of definite proportions might seem obvious to the modern chemist, inherent in the very definition of a chemical compound. At the end of the 18 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the science, scientific study of the properties and behavior of matter. It is a natural science that covers the Chemical element, elements that make up matter to the chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during a Chemical reaction, reaction with other Chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the properties ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
William Prout
William Prout FRS (; 15 January 1785 – 9 April 1850) was an English chemist, physician, and natural theologian. He is remembered today mainly for what is called Prout's hypothesis. Biography Prout was born in Horton, Gloucestershire in 1785 and educated at 17 years of age by a clergyman, followed by the Redland Academy at Bristol and Edinburgh University, where he graduated in 1811 with an MD. His professional life was spent as a practising physician in London, but he also occupied himself with chemical research. He was an active worker in biological chemistry and carried out many analyses of the secretions of living organisms, which he believed were produced by the breakdown of bodily tissues. In 1823, he discovered that stomach juices contain hydrochloric acid, which can be separated from gastric juice by distillation. In 1827, he proposed the classification of substances in food into sugars and starches, oily bodies, and albumen, which would later become known as ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ocean
The ocean (also the sea or the world ocean) is the body of salt water that covers approximately 70.8% of the surface of Earth and contains 97% of Earth's water. An ocean can also refer to any of the large bodies of water into which the world ocean is conventionally divided."Ocean." ''Merriam-Webster.com Dictionary'', Merriam-Webster, [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atmospheric
An atmosphere () is a layer of gas or layers of gases that envelop a planet, and is held in place by the gravity of the planetary body. A planet retains an atmosphere when the gravity is great and the temperature of the atmosphere is low. A stellar atmosphere is the outer region of a star, which includes the layers above the opacity (optics), opaque photosphere; stars of low temperature might have outer atmospheres containing compound molecules. The atmosphere of Earth is composed of nitrogen (78%), oxygen (21%), argon (0.9%), Carbon dioxide in Earth's atmosphere, carbon dioxide (0.04%) and trace gases. Most organisms use oxygen for respiration (physiology), respiration; lightning and bacteria perform nitrogen fixation to produce ammonia that is used to make nucleotides and amino acids; plants, algae, and cyanobacteria use carbon dioxide for photosynthesis. The layered composition of the atmosphere minimises the harmful effects of sunlight, ultraviolet radiation, the solar win ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Astronomy
Astronomy () is a natural science that studies astronomical object, celestial objects and phenomena. It uses mathematics, physics, and chemistry in order to explain their origin and chronology of the Universe, evolution. Objects of interest include planets, natural satellite, moons, stars, nebulae, galaxy, galaxies, and comets. Relevant phenomena include supernova explosions, gamma ray bursts, quasars, blazars, pulsars, and cosmic microwave background radiation. More generally, astronomy studies everything that originates beyond atmosphere of Earth, Earth's atmosphere. Cosmology is a branch of astronomy that studies the universe as a whole. Astronomy is one of the oldest natural sciences. The early civilizations in recorded history made methodical observations of the night sky. These include the Babylonian astronomy, Babylonians, Greek astronomy, Greeks, Indian astronomy, Indians, Egyptian astronomy, Egyptians, Chinese astronomy, Chinese, Maya civilization, Maya, and many anc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radiometric Dating
Radiometric dating, radioactive dating or radioisotope dating is a technique which is used to date materials such as rocks or carbon, in which trace radioactive impurities were selectively incorporated when they were formed. The method compares the abundance of a naturally occurring radioactive isotope within the material to the abundance of its decay products, which form at a known constant rate of decay. The use of radiometric dating was first published in 1907 by Bertram Boltwood and is now the principal source of information about the absolute age of rocks and other geological features, including the age of fossilized life forms or the age of Earth itself, and can also be used to date a wide range of natural and man-made materials. Together with stratigraphic principles, radiometric dating methods are used in geochronology to establish the geologic time scale. Among the best-known techniques are radiocarbon dating, potassium–argon dating and uranium–lead dating. By al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numbers) due to different numbers of neutrons in their nuclei. While all isotopes of a given element have almost the same chemical properties, they have different atomic masses and physical properties. The term isotope is formed from the Greek roots isos ( ἴσος "equal") and topos ( τόπος "place"), meaning "the same place"; thus, the meaning behind the name is that different isotopes of a single element occupy the same position on the periodic table. It was coined by Scottish doctor and writer Margaret Todd in 1913 in a suggestion to the British chemist Frederick Soddy. The number of protons within the atom's nucleus is called its atomic number and is equal to the number of electrons in the neutral (non-ionized) atom. Each atomic numbe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
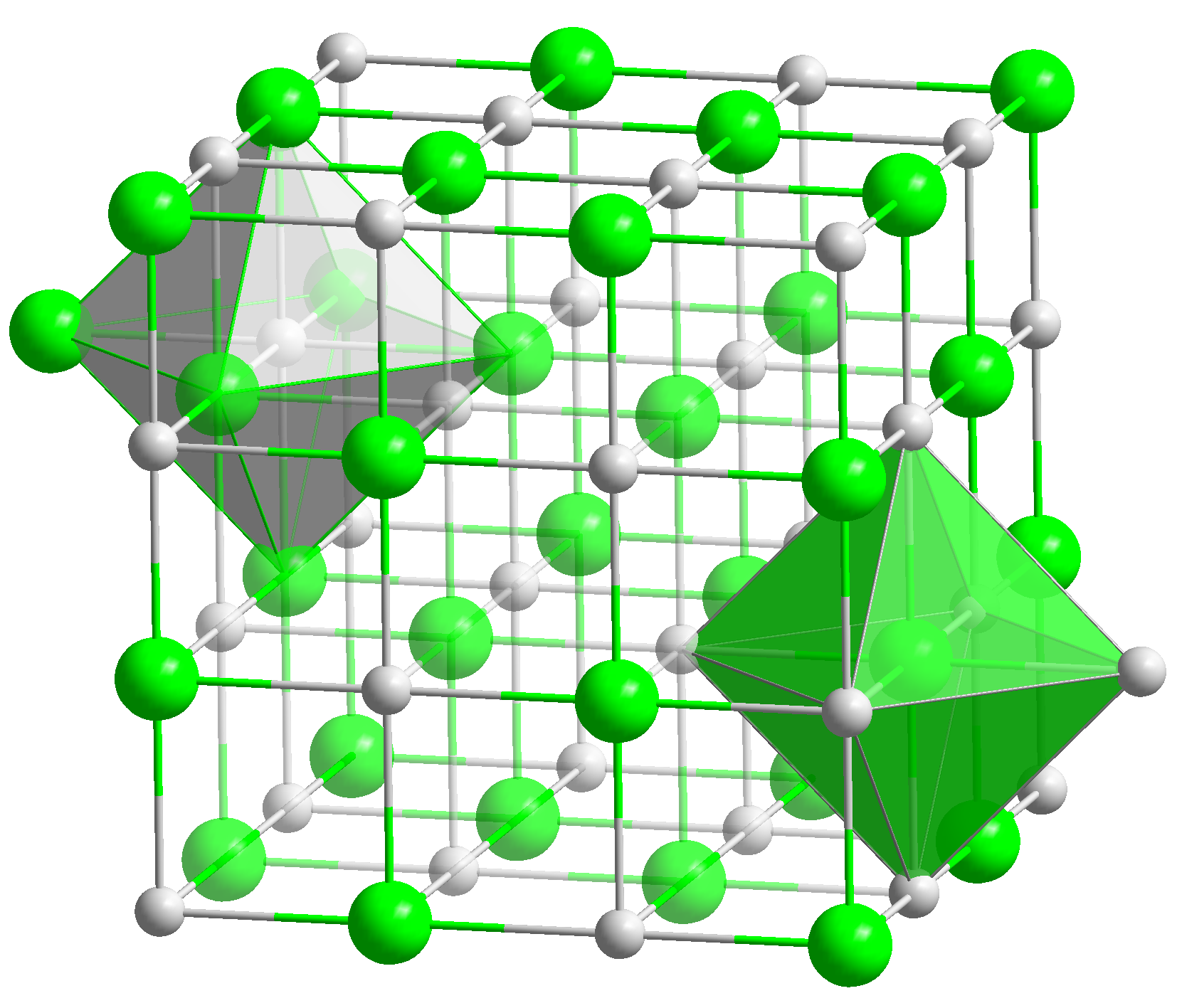
Wüstite
Wüstite ( Fe O) is a mineral form of iron(II) oxide found with meteorites and native iron. It has a grey colour with a greenish tint in reflected light. Wüstite crystallizes in the isometric-hexoctahedral crystal system in opaque to translucent metallic grains. It has a Mohs hardness of 5 to 5.5 and a specific gravity of 5.88. Wüstite is a typical example of a non-stoichiometric compound. Wüstite was named after Fritz Wüst (1860–1938), a German metallurgist and founding director of the ''Kaiser-Wilhelm-Institut für Eisenforschung'' (presently Max Planck Institute for Iron Research GmbH). In addition to its type locality in Germany, it has been reported from Disko Island, Greenland; the Jharia coalfield, Jharkhand, India; and as inclusions in diamonds in a number of kimberlite pipes. It also is reported from deep sea manganese nodules. Its presence indicates a highly reducing environment. Wüstite redox buffer Iron minerals on the earth's surface are typically r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron Oxide
Iron oxides are chemical compounds composed of iron and oxygen. Several iron oxides are recognized. All are black magnetic solids. Often they are non-stoichiometric. Oxyhydroxides are a related class of compounds, perhaps the best known of which is rust. Iron oxides and oxyhydroxides are widespread in nature and play an important role in many geological and biological processes. They are used as iron ores, pigments, catalysts, and in thermite, and occur in hemoglobin. Iron oxides are inexpensive and durable pigments in paints, coatings and colored concretes. Colors commonly available are in the "earthy" end of the yellow/orange/red/brown/black range. When used as a food coloring, it has E number E172. Stoichiometries Iron oxides feature as ferrous ( Fe(II)) or ferric ( Fe(III)) or both. They adopt octahedral or tetrahedral coordination geometry. Only a few oxides are significant at the earth's surface, particularly wüstite, magnetite, and hematite. * Oxides of FeII ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Non-stoichiometric Compound
In chemistry, non-stoichiometric compounds are chemical compounds, almost always solid inorganic compounds, having elemental composition whose proportions cannot be represented by a ratio of small natural numbers (i.e. an empirical formula); most often, in such materials, some small percentage of atoms are missing or too many atoms are packed into an otherwise perfect lattice work. Contrary to earlier definitions, modern understanding of non-stoichiometric compounds view them as homogeneous, and not mixtures of stoichiometric chemical compounds. Since the solids are overall electrically neutral, the defect is compensated by a change in the charge of other atoms in the solid, either by changing their oxidation state, or by replacing them with atoms of different elements with a different charge. Many metal oxides and sulfides have non-stoichiometric examples; for example, stoichiometric iron(II) oxide, which is rare, has the formula , whereas the more common material is nonstoichi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Binding Energies
In physics and chemistry, binding energy is the smallest amount of energy required to remove a particle from a system of particles or to disassemble a system of particles into individual parts. In the former meaning the term is predominantly used in condensed matter physics, atomic physics, and chemistry, whereas in nuclear physics the term ''separation energy'' is used. A bound system is typically at a lower energy level than its unbound constituents. According to relativity theory, a decrease in the total energy of a system is accompanied by a decrease in the total mass, where . Types of binding energy There are several types of binding energy, each operating over a different distance and energy scale. The smaller the size of a bound system, the higher its associated binding energy. Mass–energy relation A bound system is typically at a lower energy level than its unbound constituents because its mass must be less than the total mass of its unbound constituents. For sys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass Defect
Nuclear binding energy in experimental physics is the minimum energy that is required to disassemble the nucleus of an atom into its constituent protons and neutrons, known collectively as nucleons. The binding energy for stable nuclei is always a positive number, as the nucleus must gain energy for the nucleons to move apart from each other. Nucleons are attracted to each other by the strong nuclear force. In theoretical nuclear physics, the nuclear binding energy is considered a negative number. In this context it represents the energy of the nucleus relative to the energy of the constituent nucleons when they are infinitely far apart. Both the experimental and theoretical views are equivalent, with slightly different emphasis on what the binding energy means. The mass of an atomic nucleus is less than the sum of the individual masses of the free constituent protons and neutrons. The difference in mass can be calculated by the Einstein equation, , where ''E'' is the nuclear b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |