Wüstite on:
[Wikipedia]
[Google]
[Amazon]
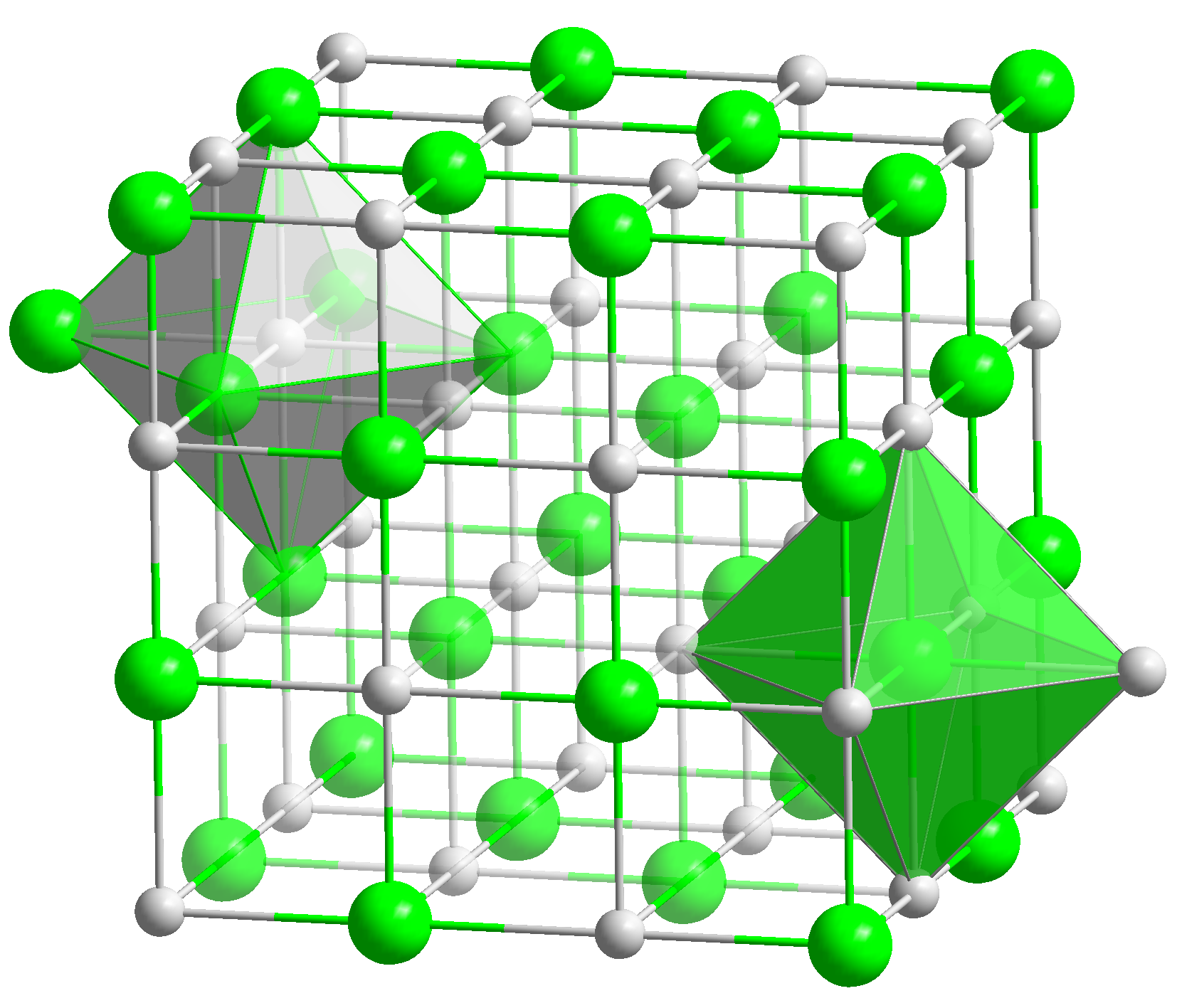 Wüstite ( Fe O) is a mineral form of
Wüstite ( Fe O) is a mineral form of
\underset + \underset -> + \underset
The formula for magnetite is more accurately written as FeO·Fe2O3 than as Fe3O4. Magnetite is one part FeO and one part Fe2O3, rather than a
Mineral Data Pub. PDF file
Accessed 3/5/2006
Accessed 3/5/2006
Accessed 3/5/2006
Webmineral data
Accessed 3/5/2006 {{DEFAULTSORT:Wustite Cubic minerals Iron(II) minerals Minerals in space group 225 Non-stoichiometric compounds Oxide minerals
 Wüstite ( Fe O) is a mineral form of
Wüstite ( Fe O) is a mineral form of iron(II) oxide
Iron(II) oxide or ferrous oxide is the inorganic compound with the formula FeO. Its mineral form is known as wüstite. One of several iron oxides, it is a black-colored powder that is sometimes confused with rust, the latter of which consists ...
found with meteorite
A meteorite is a solid piece of debris from an object, such as a comet, asteroid, or meteoroid, that originates in outer space and survives its passage through the atmosphere to reach the surface of a planet or Natural satellite, moon. When the ...
s and native iron
Telluric iron, also called native iron, is iron that originated on Earth, and is found in a metallic form rather than as an ore. Telluric iron is extremely rare, with only one known major deposit in the world, located in Greenland.
Introduction
Wi ...
. It has a grey colour with a greenish tint in reflected light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 tera ...
. Wüstite crystallizes in the isometric-hexoctahedral crystal system in opaque to translucent metallic grains. It has a Mohs hardness
The Mohs scale of mineral hardness () is a qualitative ordinal scale, from 1 to 10, characterizing scratch resistance of various minerals through the ability of harder material to scratch softer material.
The scale was introduced in 1812 by th ...
of 5 to 5.5 and a specific gravity
Relative density, or specific gravity, is the ratio of the density (mass of a unit volume) of a substance to the density of a given reference material. Specific gravity for liquids is nearly always measured with respect to water (molecule), wa ...
of 5.88. Wüstite is a typical example of a non-stoichiometric compound
In chemistry, non-stoichiometric compounds are chemical compounds, almost always solid inorganic compounds, having elemental composition whose proportions cannot be represented by a ratio of small natural numbers (i.e. an empirical formula); m ...
.
Wüstite was named after Fritz Wüst
Fritz originated as a German nickname for Friedrich, or Frederick (''Der Alte Fritz'', and ''Stary Fryc'' were common nicknames for King Frederick II of Prussia and Frederick III, German Emperor) as well as for similar names including Fridolin an ...
(1860–1938), a German metallurgist
Metallurgy is a domain of materials science and engineering that studies the physical and chemical behavior of metallic elements, their inter-metallic compounds, and their mixtures, which are known as alloys.
Metallurgy encompasses both the sc ...
and founding director of the ''Kaiser-Wilhelm-Institut für Eisenforschung'' (presently Max Planck Institute for Iron Research GmbH).
In addition to its type locality in Germany, it has been reported from Disko Island
Disko Island ( kl, Qeqertarsuaq, da, Diskoøen) is a large island in Baffin Bay, off the west coast of Greenland. It has an area of ,Jharia
Jharia is a neighbourhood in Dhanbad city in Dhanbad Sadar subdivision of Dhanbad district in Jharkhand state, India. Jharia's economy is heavily dependent on the local coal fields, used to make coke. However, fires in the coal fields have made ...
coalfield, Jharkhand
Jharkhand (; ; ) is a state in eastern India. The state shares its border with the states of West Bengal to the east, Chhattisgarh to the west, Uttar Pradesh to the northwest, Bihar to the north and Odisha to the south. It has an area of . It ...
, India; and as inclusions in diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
s in a number of kimberlite
Kimberlite is an igneous rock and a rare variant of peridotite. It is most commonly known to be the main host matrix for diamonds. It is named after the town of Kimberley in South Africa, where the discovery of an diamond called the Star of ...
pipes. It also is reported from deep sea manganese nodule
Polymetallic nodules, also called manganese nodules, are mineral concretions on the sea bottom formed of concentric layers of iron and manganese hydroxides around a core. As nodules can be found in vast quantities, and contain valuable metals, de ...
s.
Its presence indicates a highly reducing environment
A reducing atmosphere is an atmospheric condition in which oxidation is prevented by removal of oxygen and other oxidizing gases or vapours, and which may contain actively reducing gases such as hydrogen, carbon monoxide, and gases such as hydro ...
.
Wüstite redox buffer
Iron minerals on the earth's surface are typically richly oxidized, forminghematite
Hematite (), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of . ...
, with Fe3+ state, or in somewhat less oxidizing environments, magnetite
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the ...
, with a mixture of Fe3+ and Fe2+. Wüstite, in geochemistry, defines a ''redox buffer
In geology, a redox buffer is an assemblage of minerals or compounds that constrains oxygen fugacity as a function of temperature. Knowledge of the redox conditions (or equivalently, oxygen fugacities) at which a rock forms and evolves can be im ...
'' of oxidation within rocks at which point the rock is so reduced that Fe3+, and thus hematite, is absent.
As the redox state of a rock is further reduced, magnetite is converted to wüstite. This occurs by conversion of the Fe3+ ions in magnetite to Fe2+ ions. An example reaction is presented below:
:solid solution
A solid solution, a term popularly used for metals, is a homogenous mixture of two different kinds of atoms in solid state and have a single crystal structure. Many examples can be found in metallurgy, geology, and solid-state chemistry. The word ...
of wüstite and hematite
Hematite (), also spelled as haematite, is a common iron oxide compound with the formula, Fe2O3 and is widely found in rocks and soils. Hematite crystals belong to the rhombohedral lattice system which is designated the alpha polymorph of . ...
. The magnetite is termed a ''redox buffer'' because until all Fe3+ magnetite is converted to Fe2+ the oxide mineral assemblage of iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in f ...
remains wüstite-magnetite, and furthermore the redox state of the rock remains at the same level of oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
fugacity
In chemical thermodynamics, the fugacity of a real gas is an effective partial pressure which replaces the mechanical partial pressure in an accurate computation of the chemical equilibrium constant. It is equal to the pressure of an ideal gas whic ...
. This is similar to buffering in the H+/OH− acid–base system of water.
Once the Fe3+ is consumed, then oxygen must be stripped from the system to further reduce it and wüstite is converted to native iron. The oxide mineral equilibrium assemblage of the rock becomes wüstite–magnetite–iron.
In nature, the only natural systems which are chemically reduced enough to even attain a wüstite–magnetite composition are rare, including carbonate-rich skarn
Skarns or tactites are hard, coarse-grained metamorphic rocks that form by a process called metasomatism. Skarns tend to be rich in calcium-magnesium-iron-manganese-aluminium silicate minerals, which are also referred to as calc-silicate minerals ...
s, meteorites, fulgurite
Fulgurites (), commonly known as "fossilized lightning", are natural tubes, clumps, or masses of sintered, vitrified, and/or fused soil, sand, rock, organic debris and other sediments that sometimes form when lightning discharges into ground. ...
s and lightning-affected rock, and perhaps the mantle where reduced carbon is present, exemplified by the presence of diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
or graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
.
Effects upon silicate minerals
The ratio of Fe2+ to Fe3+ within a rock determines, in part, the silicate mineral assemblage of the rock. Within a rock of a given chemical composition, iron enters minerals based on the bulk chemical composition and the mineral phases which are stable at that temperature and pressure. Iron may only enter minerals such aspyroxene
The pyroxenes (commonly abbreviated to ''Px'') are a group of important rock-forming inosilicate minerals found in many igneous and metamorphic rocks. Pyroxenes have the general formula , where X represents calcium (Ca), sodium (Na), iron (Fe II) ...
and olivine
The mineral olivine () is a magnesium iron silicate with the chemical formula . It is a type of nesosilicate or orthosilicate. The primary component of the Earth's upper mantle, it is a common mineral in Earth's subsurface, but weathers quickl ...
if it is present as Fe2+; Fe3+ cannot enter the lattice of fayalite
Fayalite (, commonly abbreviated to Fa) is the iron-rich end-member of the olivine solid-solution series. In common with all minerals in the olivine group, fayalite crystallizes in the orthorhombic system (space group ''Pbnm'') with cell para ...
olivine and thus for every two Fe3+ ions, one Fe2+ is used and one molecule of magnetite is created.
In chemically reduced rocks, magnetite may be absent due to the propensity of iron to enter olivine, and wüstite may only be present if there is an excess of iron above what can be used by silica. Thus, wüstite may only be found in silica-undersaturated compositions which are also heavily chemically reduced, satisfying both the need to remove all Fe3+ and to maintain iron outside of silicate minerals.
In nature, carbonate rocks, potentially carbonatite
Carbonatite () is a type of intrusive rock, intrusive or extrusive rock, extrusive igneous rock defined by mineralogic composition consisting of greater than 50% carbonate minerals. Carbonatites may be confused with marble and may require geoche ...
, kimberlite
Kimberlite is an igneous rock and a rare variant of peridotite. It is most commonly known to be the main host matrix for diamonds. It is named after the town of Kimberley in South Africa, where the discovery of an diamond called the Star of ...
s, carbonate-bearing melilitic rocks, and other rare alkaline rocks may satisfy these criteria. However, wüstite is not reported in most of these rocks in nature, potentially because the redox state necessary to drive magnetite to wüstite is so rare.
Role in nitrogen fixation
Approximately 2-3% of the world's energy budget is allocated to theHaber process
The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German chemists Fritz Haber and C ...
for ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous was ...
() production, which relies on wüstite-derived catalysts. The industrial catalyst is derived from finely ground iron powder, which is usually obtained by reduction of high-purity magnetite
Magnetite is a mineral and one of the main iron ores, with the chemical formula Fe2+Fe3+2O4. It is one of the oxides of iron, and is ferrimagnetic; it is attracted to a magnet and can be magnetized to become a permanent magnet itself. With the ...
(Fe3O4). The pulverized iron metal is burnt (oxidized) to give magnetite or wüstite of a defined particle size. The magnetite (or wüstite) particles are then partially reduced, removing some of the oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as wel ...
in the process. The resulting catalyst particles consist of a core of magnetite, encased in a shell of wüstite, which in turn is surrounded by an outer shell of iron metal. The catalyst maintains most of its bulk volume during the reduction, resulting in a highly porous high-surface-area material, which enhances its effectiveness as a catalyst.
Historical use
According to Vagn Fabritius Buchwald, wüstite was an important component during theIron Age
The Iron Age is the final epoch of the three-age division of the prehistory and protohistory of humanity. It was preceded by the Stone Age (Paleolithic, Mesolithic, Neolithic) and the Bronze Age (Chalcolithic). The concept has been mostly appl ...
to facilitate the process of forge welding
Forge welding (FOW), also called fire welding, is a solid-state welding process that joins two pieces of metal by heating them to a high temperature and then hammering them together. It may also consist of heating and forcing the metals together ...
. In ancient times, when blacksmithing was performed using a charcoal forge
A forge is a type of hearth used for heating metals, or the workplace (smithy) where such a hearth is located. The forge is used by the smith to heat a piece of metal to a temperature at which it becomes easier to shape by forging, or to th ...
, the deep charcoal
Charcoal is a lightweight black carbon residue produced by strongly heating wood (or other animal and plant materials) in minimal oxygen to remove all water and volatile constituents. In the traditional version of this pyrolysis process, cal ...
pit in which the steel or iron was placed provided a highly reducing, virtually oxygen-free environment, producing a thin wüstite layer on the metal. At the welding temperature, the iron becomes highly reactive with oxygen, and will spark and form thick layers of slag
Slag is a by-product of smelting (pyrometallurgical) ores and used metals. Broadly, it can be classified as ferrous (by-products of processing iron and steel), ferroalloy (by-product of ferroalloy production) or non-ferrous/base metals (by-prod ...
when exposed to the air, which makes welding the iron or steel nearly impossible. To solve this problem, ancient blacksmiths would toss small amounts of sand onto the white-hot metal. The silica in the sand reacts with the wüstite to form fayalite
Fayalite (, commonly abbreviated to Fa) is the iron-rich end-member of the olivine solid-solution series. In common with all minerals in the olivine group, fayalite crystallizes in the orthorhombic system (space group ''Pbnm'') with cell para ...
, which melts just below the welding temperature. This produced an effective flux
Flux describes any effect that appears to pass or travel (whether it actually moves or not) through a surface or substance. Flux is a concept in applied mathematics and vector calculus which has many applications to physics. For transport ph ...
that shielded the metal from oxygen and helped extract oxides and impurities, leaving a pure surface that can weld readily. Although the ancients had no knowledge of how this worked, the ability to weld iron contributed to the movement out of the Bronze Age
The Bronze Age is a historic period, lasting approximately from 3300 BC to 1200 BC, characterized by the use of bronze, the presence of writing in some areas, and other early features of urban civilization. The Bronze Age is the second pri ...
and into the modern.
Related minerals
Wüstite forms a solid solution withpericlase
Periclase is a magnesium mineral that occurs naturally in contact metamorphic rocks and is a major component of most basic refractory bricks. It is a cubic form of magnesium oxide ( Mg O). In nature it usually forms a solid solution with wüstit ...
( MgO), and iron substitutes for magnesium. Periclase, when hydrated, forms brucite
Brucite is the mineral form of magnesium hydroxide, with the chemical formula Mg( OH)2. It is a common alteration product of periclase in marble; a low-temperature hydrothermal vein mineral in metamorphosed limestones and chlorite schists; and ...
(Mg(O H)2), a common product of serpentinite
Serpentinite is a rock composed predominantly of one or more serpentine group minerals, the name originating from the similarity of the texture of the rock to that of the skin of a snake. Serpentinite has been called ''serpentine'' or ''ser ...
metamorphic reaction
A metamorphic reaction is a chemical reaction that takes place during the geological process of metamorphism wherein one assemblage of minerals is transformed into a second assemblage which is stable under the new temperature/pressure conditions r ...
s.
Oxidation and hydration of wüstite forms goethite
Goethite (, ) is a mineral of the diaspore group, consisting of iron(III) oxide-hydroxide, specifically the "α" polymorph. It is found in soil and other low-temperature environments such as sediment. Goethite has been well known since ancient t ...
and limonite
Limonite () is an iron ore consisting of a mixture of hydrated iron(III) oxide-hydroxides in varying composition. The generic formula is frequently written as FeO(OH)·H2O, although this is not entirely accurate as the ratio of oxide to hydroxid ...
.
Zinc, aluminium, and other metals may substitute for iron in wüstite.
Wüstite in dolomite Dolomite may refer to:
*Dolomite (mineral), a carbonate mineral
*Dolomite (rock), also known as dolostone, a sedimentary carbonate rock
*Dolomite, Alabama, United States, an unincorporated community
*Dolomite, California, United States, an unincor ...
skarn
Skarns or tactites are hard, coarse-grained metamorphic rocks that form by a process called metasomatism. Skarns tend to be rich in calcium-magnesium-iron-manganese-aluminium silicate minerals, which are also referred to as calc-silicate minerals ...
s may be related to siderite
Siderite is a mineral composed of iron(II) carbonate (FeCO3). It takes its name from the Greek word σίδηρος ''sideros,'' "iron". It is a valuable iron mineral, since it is 48% iron and contains no sulfur or phosphorus. Zinc, magnesium and ...
(iron(II) carbonate), wollastonite
Wollastonite is a calcium inosilicate mineral ( Ca Si O3) that may contain small amounts of iron, magnesium, and manganese substituting for calcium. It is usually white. It forms when impure limestone or dolomite is subjected to high temperature ...
, enstatite
Enstatite is a mineral; the magnesium endmember of the pyroxene silicate mineral series enstatite (MgSiO3) – ferrosilite (FeSiO3). The magnesium rich members of the solid solution series are common rock-forming minerals found in igneous and m ...
, diopside
Diopside is a monoclinic pyroxene mineral with composition . It forms complete solid solution series with hedenbergite () and augite, and partial solid solutions with orthopyroxene and pigeonite. It forms variably colored, but typically dull ...
, and magnesite
Magnesite is a mineral with the chemical formula (magnesium carbonate). Iron, manganese, cobalt, and nickel may occur as admixtures, but only in small amounts.
Occurrence
Magnesite occurs as veins in and an alteration product of ultramafic ro ...
.
See also
*Normative mineralogy
Normative mineralogy is a calculation of the composition of a rock sample that estimates the ''idealised mineralogy'' of a rock based on a quantitative chemical analysis according to the principles of geochemistry.
Normative mineral calculations ...
* Skarn
Skarns or tactites are hard, coarse-grained metamorphic rocks that form by a process called metasomatism. Skarns tend to be rich in calcium-magnesium-iron-manganese-aluminium silicate minerals, which are also referred to as calc-silicate minerals ...
References
Mineral Data Pub. PDF file
Accessed 3/5/2006
Accessed 3/5/2006
Accessed 3/5/2006
Webmineral data
Accessed 3/5/2006 {{DEFAULTSORT:Wustite Cubic minerals Iron(II) minerals Minerals in space group 225 Non-stoichiometric compounds Oxide minerals