|
Klopman-Salem Equation
In the theory of chemical reactivity, the Klopman-Salem equation describes the energetic change that occurs when two species approach each other in the course of a reaction and begin to interact, as their associated molecular orbitals begin to overlap with each other and atoms bearing partial charges begin to experience attractive or repulsive electrostatic forces. First described independently by Gilles Klopman and Lionel Salem in 1968, this relationship provides a mathematical basis for the key assumptions of frontier molecular orbital theory In chemistry, frontier molecular orbital theory is an application of MO theory describing HOMO/LUMO interactions. History In 1952, Kenichi Fukui published a paper in the ''Journal of Chemical Physics'' titled "A molecular theory of reactivity i ... (i.e., theory of HOMO-LUMO interactions) and hard soft acid base (HSAB) theory. Conceptually, it highlights the importance of considering both electrostatic interactions and orbital interact ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reactivity (chemistry)
In chemistry, reactivity is the impulse for which a chemical substance undergoes a chemical reaction, either by itself or with other materials, with an overall release of energy. ''Reactivity'' refers to: * the chemical reactions of a single substance, * the chemical reactions of two or more substances that interact with each other, * the systematic study of sets of reactions of these two kinds, * methodology that applies to the study of reactivity of chemicals of all kinds, * experimental methods that are used to observe these processes * theories to predict and to account for these processes. The chemical reactivity of a single substance (reactant) covers its behavior in which it: * Decomposes * Forms new substances by addition of atoms from another reactant or reactants * Interacts with two or more other reactants to form two or more products The chemical reactivity of a substance can refer to the variety of circumstances (conditions that include temperature, pressure, prese ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Species
A chemical species is a chemical substance or ensemble composed of chemically identical molecular entity, molecular entities that can explore the same set of molecular energy levels on a characteristic or delineated time scale. These energy levels determine the way the chemical species will interact with others (engaging in chemical bonds, etc.). The species can be an atom, molecule, ion, or radical, and it has a specific chemical name and chemical formula. The term is also applied to a set of chemically identical atomic or molecular structural units in a solid array. In supramolecular chemistry, chemical species are those supramolecular structures whose interactions and associations are brought about via intermolecular bonding and debonding actions, and function to form the basis of this branch of chemistry. For instance: * The chemical species argon is an atom, atomic species of formula Ar; * dioxygen and ozone are different molecule, molecular species, of respective formulas O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
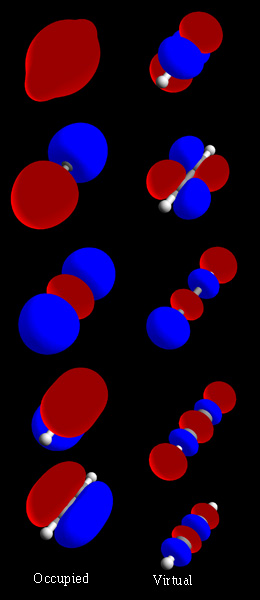
Molecular Orbital
In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of finding an electron in any specific region. The terms ''atomic orbital'' and ''molecular orbital'' were introduced by Robert S. Mulliken in 1932 to mean ''one-electron orbital wave functions''. At an elementary level, they are used to describe the ''region'' of space in which a function has a significant amplitude. In an isolated atom, the orbital electrons' location is determined by functions called atomic orbitals. When multiple atoms combine chemically into a molecule, the electrons' locations are determined by the molecule as a whole, so the atomic orbitals combine to form molecular orbitals. The electrons from the constituent atoms occupy the molecular orbitals. Mathematically, molecular orbitals are an approximate solution to the Schrödin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Partial Charge
A partial charge is a non-integer charge value when measured in elementary charge units. Partial charge is more commonly called net atomic charge. It is represented by the Greek lowercase letter 𝛿, namely 𝛿− or 𝛿+. Partial charges are created due to the asymmetric distribution of electrons in chemical bonds. For example, in a polar covalent bond like HCl, the shared electron oscillates between the bonded atoms. The resulting partial charges are a property only of zones within the distribution, and not the assemblage as a whole. For example, chemists often choose to look at a small space surrounding the nucleus of an atom: When an electrically neutral atom bonds chemically to another neutral atom that is more electronegative, its electrons are partially drawn away. This leaves the region about that atom's nucleus with a partial positive charge, and it creates a partial negative charge on the atom to which it is bonded. In such a situation, the distributed charges taken ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coulomb's Law
Coulomb's inverse-square law, or simply Coulomb's law, is an experimental law of physics that quantifies the amount of force between two stationary, electrically charged particles. The electric force between charged bodies at rest is conventionally called ''electrostatic force'' or Coulomb force. Although the law was known earlier, it was first published in 1785 by French physicist Charles-Augustin de Coulomb, hence the name. Coulomb's law was essential to the development of the theory of electromagnetism, maybe even its starting point, as it made it possible to discuss the quantity of electric charge in a meaningful way. The law states that the magnitude of the electrostatic force of attraction or repulsion between two point charges is directly proportional to the product of the magnitudes of charges and inversely proportional to the square of the distance between them. Coulomb studied the repulsive force between bodies having electrical charges of the same sign: Coulomb also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gilles Klopman
Gilles Klopman (February 24, 1933 – January 10, 2015) was the Charles F. Mabery Professor of Research in Chemistry, Oncology and Environmental Health Sciences Director of the Laboratory for Decision Support Methodologies at Case Western Reserve University in Cleveland, Ohio, and Adjunct Professor of Environmental and Occupational Health, (University of Pittsburgh) Dr. Klopman was educated in Belgium and the United States in theoretical chemistry, physical organic chemistry (L. es Sc., University of Brussels (Belgium), 1956, Dr. es Sc., University of Brussels, 1960, Postdoctoral Fellow, University of Texas, 1965–66) Structure-Activity Studies of Biologically Active Molecules Professor Klopman’s work has involved the evaluation of chemical reactivity and includes experimental determination of reactivity indices and substituent constants to the development of reactivity theories . He has contributed significantly to the concept of charge and orbital controlled reactions where ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lionel Salem
Lionel Salem (born 5 March 1937, Paris) is a French theoretical chemist, former research director at the French National Centre for Scientific Research (CNRS), retired since 1999. He is a member of the International Academy of Quantum Molecular Science which named him its annual award winner in 1975 ''for his work on photochemical processes and on chemical reaction mechanisms.'' . He has contributed to the theories of forces between molecules, of conjugated molecules, of organic reaction mechanisms and of heterogeneous catalysis. He developed the electronic theory of diradicals, as well as the concepts of diradical and zwitterionic states. In 1968 he described the energy change for the approach of two molecules as a function of the properties of the orbitals of the two molécules; this approach, pursued independently by Gilles Klopman, led to the Klopman-Salem equation In the theory of chemical reactivity, the Klopman-Salem equation describes the energetic change that occurs w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Frontier Molecular Orbital Theory
In chemistry, frontier molecular orbital theory is an application of MO theory describing HOMO/LUMO interactions. History In 1952, Kenichi Fukui published a paper in the ''Journal of Chemical Physics'' titled "A molecular theory of reactivity in aromatic hydrocarbons." Though widely criticized at the time, he later shared the Nobel Prize in Chemistry with Roald Hoffmann for his work on reaction mechanisms. Hoffman's work focused on creating a set of four pericyclic reactions in organic chemistry, based on orbital symmetry, which he coauthored with Robert Burns Woodward, entitled "The Conservation of Orbital Symmetry." Fukui's own work looked at the frontier orbitals, and in particular the effects of the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO) on reaction mechanisms, which led to it being called Frontier Molecular Orbital Theory (FMO Theory). He used these interactions to better understand the conclusions of the Woodward–Hoffma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hard And Soft Acids And Bases
HSAB concept is a jargon for "hard and soft (Lewis) acids and bases". HSAB is widely used in chemistry for explaining stability of compounds, reaction mechanisms and pathways. It assigns the terms 'hard' or 'soft', and 'acid' or 'base' to chemical species. 'Hard' applies to species which are small, have high charge states (the charge criterion applies mainly to acids, to a lesser extent to bases), and are weakly polarizable. 'Soft' applies to species which are big, have low charge states and are strongly polarizable. The theory is used in contexts where a qualitative, rather than quantitative, description would help in understanding the predominant factors which drive chemical properties and reactions. This is especially so in transition metal chemistry, where numerous experiments have been done to determine the relative ordering of ligands and transition metal ions in terms of their hardness and softness. HSAB theory is also useful in predicting the products of metathesis rea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quantum Chemistry
Quantum chemistry, also called molecular quantum mechanics, is a branch of physical chemistry focused on the application of quantum mechanics to chemical systems, particularly towards the quantum-mechanical calculation of electronic contributions to physical and chemical properties of Molecule, molecules, Material, materials, and solutions at the atomic level. These calculations include systematically applied approximations intended to make calculations computationally feasible while still capturing as much information about important contributions to the computed Wave function, wave functions as well as to observable properties such as structures, spectra, and thermodynamic properties. Quantum chemistry is also concerned with the computation of quantum effects on molecular dynamics and chemical kinetics. Chemists rely heavily on spectroscopy through which information regarding the Quantization (physics), quantization of energy on a molecular scale can be obtained. Common metho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |