|
Iodometry
Iodometry, known as iodometric titration, is a method of volumetric chemical analysis, a redox titration where the appearance or disappearance of elementary iodine indicates the end point. Note that iodometry involves indirect titration of iodine liberated by reaction with the analyte, whereas iodimetry involves direct titration using iodine as the titrant. Redox titration using sodium thiosulphate, (usually) as a reducing agent is known as iodometric titration since it is used specifically to titrate iodine. The iodometric titration is a general method to determine the concentration of an oxidising agent in solution. In an iodometric titration, a starch solution is used as an indicator since it can absorb the that is released. This absorption will cause the solution to change its colour from deep blue to light yellow when titrated with standardised thiosulfate solution. This indicates the end point of the titration. Iodometry is commonly used to analyse the concentration of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodine
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a semi-lustrous, non-metallic solid at standard conditions that melts to form a deep violet liquid at , and boils to a violet gas at . The element was discovered by the French chemist Bernard Courtois in 1811 and was named two years later by Joseph Louis Gay-Lussac, after the Ancient Greek 'violet-coloured'. Iodine occurs in many oxidation states, including iodide (I−), iodate (), and the various periodate anions. It is the least abundant of the stable halogens, being the sixty-first most abundant element. As the heaviest essential mineral nutrient, iodine is required for the synthesis of thyroid hormones. Iodine deficiency affects about two billion people and is the leading preventable cause of intellectual disabilities. The dominant producers of iodine today are Chile and Japan. Due to its high atomic number and ease of attachment to organic compound ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodine
Iodine is a chemical element with the symbol I and atomic number 53. The heaviest of the stable halogens, it exists as a semi-lustrous, non-metallic solid at standard conditions that melts to form a deep violet liquid at , and boils to a violet gas at . The element was discovered by the French chemist Bernard Courtois in 1811 and was named two years later by Joseph Louis Gay-Lussac, after the Ancient Greek 'violet-coloured'. Iodine occurs in many oxidation states, including iodide (I−), iodate (), and the various periodate anions. It is the least abundant of the stable halogens, being the sixty-first most abundant element. As the heaviest essential mineral nutrient, iodine is required for the synthesis of thyroid hormones. Iodine deficiency affects about two billion people and is the leading preventable cause of intellectual disabilities. The dominant producers of iodine today are Chile and Japan. Due to its high atomic number and ease of attachment to organic compound ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Volumetric Analysis
Titration (also known as titrimetry and volumetric analysis) is a common laboratory method of quantitative chemical analysis to determine the concentration of an identified analyte (a substance to be analyzed). A reagent, termed the ''titrant'' or ''titrator'', is prepared as a standard solution of known concentration and volume. The titrant reacts with a solution of ''analyte'' (which may also be termed the ''titrand'') to determine the analyte's concentration. The volume of titrant that reacted with the analyte is termed the ''titration volume''. History and etymology The word "titration" descends from the French word ''titrer'' (1543), meaning the proportion of gold or silver in coins or in works of gold or silver; i.e., a measure of fineness or purity. ''Tiltre'' became ''titre'', which thus came to mean the "fineness of alloyed gold", and then the "concentration of a substance in a given sample". In 1828, the French chemist Joseph Louis Gay-Lussac first used ''titre'' as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox Titration
A redox titration is a type of titration based on a redox reaction between the analyte and titrant. It may involve the use of a redox indicator and/or a potentiometer. A common example of a redox titration is treating a solution of iodine with a reducing agent to produce iodide using a starch indicator to help detect the endpoint. Iodine (I2) can be reduced to iodide (I−) by, say, thiosulfate (, and when all iodine is spent the blue colour disappears. This is called an iodometric titration. Most often of all, the reduction of iodine to iodide is the last step in a series of reactions where the initial reactions are used to convert an unknown amount of the solute (the substance being analyzed) to an equivalent amount of iodine, which may then be titrated. Sometimes other halogens (or haloalkanes) than iodine are used in the intermediate reactions because they are available in better measurable standard solutions and/or react more readily with the solute. The extra steps in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Thiosulphate
Sodium thiosulfate (sodium thiosulphate) is an inorganic compound with the formula . Typically it is available as the white or colorless pentahydrate, . The solid is an efflorescent (loses water readily) crystalline substance that dissolves well in water. Sodium thiosulfate is used in gold mining, water treatment, analytical chemistry, the development of silver-based photographic film and prints, and medicine. The medical uses of sodium thiosulfate include treatment of cyanide poisoning and pityriasis. It is on the World Health Organization's List of Essential Medicines. Uses Sodium thiosulfate is used predominantly in industry. For example, it is used to convert dyes to their soluble colorless forms, which are called leuco. It is also used to bleach "wool, cotton, silk, ...soaps, glues, clay, sand, bauxite, and... edible oils, edible fats, and gelatin." Medical uses Sodium thiosulfate is used in the treatment of cyanide poisoning. Other uses include topical treatment of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiosulfate
Thiosulfate ( IUPAC-recommended spelling; sometimes thiosulphate in British English) is an oxyanion of sulfur with the chemical formula . Thiosulfate also refers to the compounds containing this anion, which are the salts of thiosulfuric acid, e.g. sodium thiosulfate . Thiosulfate also refers to the esters of thiosulfuric acid, e.g. ''O'',''S''-dimethyl thiosulfate . The prefix thio- indicates that the thiosulfate is a sulfate with one oxygen replaced by sulfur. Thiosulfate is tetrahedral at the central S atom. Thiosulfate salts occur naturally. Thiosulfate ion has C3v symmetry, and is produced by certain biochemical processes. It rapidly dechlorinates water and is notable for its use to halt bleaching in the paper-making industry. Thiosulfate salts are mainly used in dying in textiles and the bleaching of natural substances. Sodium thiosulfate, commonly called ''hypo'' (from "hyposulfite"), was widely used in photography to fix black and white negatives and prints after the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient times and were powdered for use as medicine and cosmetics, often known by the Arabic name kohl. The earliest known description of the metal in the West was written in 1540 by Vannoccio Biringuccio. China is the largest producer of antimony and its compounds, with most production coming from the Xikuangshan Mine in Hunan. The industrial methods for refining antimony from stibnite are roasting followed by reduction with carbon, or direct reduction of stibnite with iron. The largest applications for metallic antimony are in alloys with lead and tin, which have improved properties for solders, bullets, and plain bearings. It improves the rigidity of lead-alloy plates in lead–acid batteries. Antimony trioxide is a prominent additive for halo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
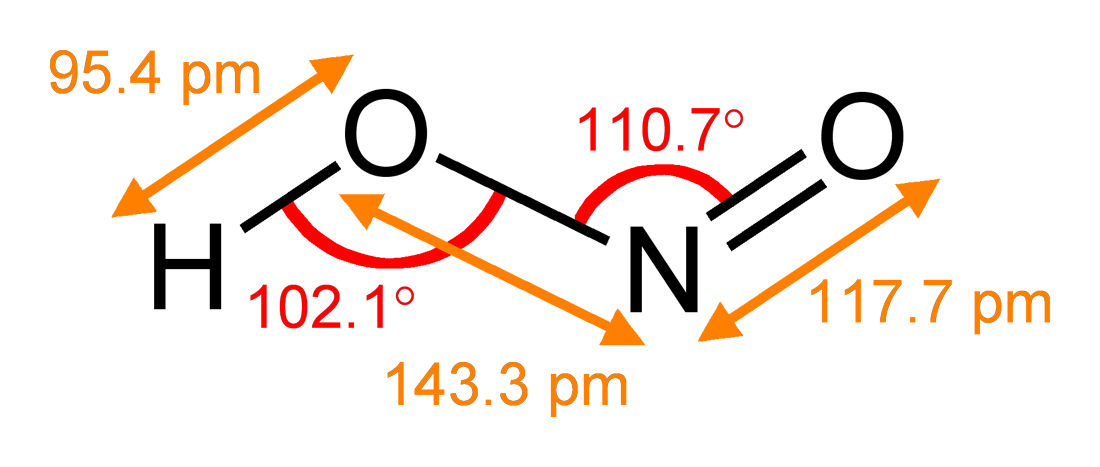
Nitrite
The nitrite polyatomic ion, ion has the chemical formula . Nitrite (mostly sodium nitrite) is widely used throughout chemical and pharmaceutical industries. The nitrite anion is a pervasive intermediate in the nitrogen cycle in nature. The name nitrite also refers to organic compounds having the –ONO group, which are esters of nitrous acid. Production Sodium nitrite is made industrially by passing a mixture of nitrogen oxides into aqueous sodium hydroxide or sodium carbonate solution: : The product is purified by recrystallization. Alkali metal nitrites are thermally stable up to and beyond their melting point (441 °C for KNO2). Ammonium nitrite can be made from dinitrogen trioxide, N2O3, which is formally the anhydride of nitrous acid: :2 NH3 + H2O + N2O3 → 2 NH4NO2 Structure The nitrite ion has a symmetrical structure (C2v molecular point group, symmetry), with both N–O bonds having equal length and a bond angle of about 115°. In valence bond theory, it is des ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Erlenmeyer Flask
An Erlenmeyer flask, also known as a conical flask (British English) or a titration flask, is a type of laboratory flask which features a flat bottom, a conical body, and a cylindrical neck. It is named after the German chemist Emil Erlenmeyer (1825–1909), who created it in 1860. Erlenmeyer flasks have wide bases, with sides that taper upward to a short vertical neck. They may be graduated, and often spots of ground glass or enamel are used where they can be labeled with a pencil. It differs from the beaker in its tapered body and narrow neck. Depending on the application, they may be constructed from glass or plastic, in a wide range of volumes. The mouth of the Erlenmeyer flask may have a beaded lip that can be stopped or covered. Alternatively, the neck may be fitted with ground glass or other connector for use with more specialized stoppers or attachment to other apparatus. A Büchner flask is a common design modification for filtration under vacuum. Uses In chemistr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary Standard
A primary standard in metrology is a standard that is sufficiently accurate such that it is not calibrated by or subordinate to other standards. Primary standards are defined via other quantities like length, mass and time. Primary standards are used to calibrate other standards referred to as working standards.Skoog, Douglas A., Donald M. West and F. James Holler. "Fundamentals of Analytical Chemistry 8th ed." Harcourt Brace College Publishers. 1995 ''Holt Science and Technology: Physical Science''. Ed. Rinehart and Winston, Inc. Holt. Holt McDougal (July 2000). . See Hierarchy of Standards. In chemistry Standards are used in analytical chemistry. Here, a primary standard is typically a reagent which can be weighed easily, and which is so pure that its weight is truly representative of the number of moles of substance contained. Features of a primary standard include: # High purity # Stability (low reactivity) # Low hygroscopicity (to minimize weight changes due to humidity) # H ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Tetrachloride
Carbon tetrachloride, also known by many other names (such as tetrachloromethane, also IUPAC nomenclature of inorganic chemistry, recognised by the IUPAC, carbon tet in the cleaning industry, Halon-104 in firefighting, and Refrigerant-10 in HVACR) is an organic compound with the chemical formula CCl4. It is a colourless liquid with a "sweet" smell that can be detected at low levels. It is practically incombustible at lower temperatures. It was formerly widely used in fire extinguishers, as a precursor to refrigerants and as a cleaning agent, but has since been phased out because of environmental and safety concerns. Exposure to high concentrations of carbon tetrachloride (including vapor) can affect the central nervous system and degenerate the liver and kidneys. Prolonged exposure can be fatal. Properties In the carbon tetrachloride molecule, four chlorine atoms are positioned symmetrically as corners in a tetrahedron, tetrahedral configuration joined to a central carbon atom by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethyl Ether
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula , sometimes abbreviated as (see Pseudoelement symbols). It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It is commonly used as a solvent in laboratories and as a starting fluid for some engines. It was formerly used as a general anesthetic, until non-flammable drugs were developed, such as halothane. It has been used as a recreational drug to cause intoxication. Production Most diethyl ether is produced as a byproduct of the vapor-phase hydration of ethylene to make ethanol. This process uses solid-supported phosphoric acid catalysts and can be adjusted to make more ether if the need arises. Vapor-phase dehydration of ethanol over some alumina catalysts can give diethyl ether yields of up to 95%. Diethyl ether can be prepared both in laboratories and on an industrial scale by the acid ether synthesis. Ethanol is mixed with a stro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |