|
Glyoxal
Glyoxal is an organic compound with the chemical formula OCHCHO. It is the smallest dialdehyde (a compound with two aldehyde groups). It is a crystalline solid, white at low temperatures and yellow near the melting point (15 °C). The liquid is yellow, and the vapor is green.O'Neil, M.J. (2001): ''The Merck Index'', 13th Edition, page 803. Pure glyoxal is not commonly encountered because glyoxal is usually handled as a 40% aqueous solution (density near 1.24 g/mL). It forms a series of hydrates, including oligomers. For many purposes, these hydrated oligomers behave equivalently to glyoxal. Glyoxal is produced industrially as a precursor to many products. Production Glyoxal was first prepared and named by the German-British chemist Heinrich Debus (1824–1915) by reacting ethanol with nitric acid. Commercial glyoxal is prepared either by the gas-phase oxidation of ethylene glycol in the presence of a silver or copper catalyst (the Laporte process) or by the liquid-phase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methylglyoxal
Methylglyoxal (MGO) is the organic compound with the formula CH3C(O)CHO. It is a reduced derivative of pyruvic acid. It is a reactive compound that is implicated in the biology of diabetes. Methylglyoxal is produced industrially by degradation of carbohydrates using overexpressed methylglyoxal synthase. Chemical structure Gaseous methylglyoxal has two carbonyl groups, an aldehyde and a ketone. In the presence of water, it exists as hydrates and oligomers. The formation of these hydrates is indicative of the high reactivity of MGO, which is relevant to its biological behavior. Biochemistry Biosynthesis and biodegradation In organisms, methylglyoxal is formed as a side-product of several metabolic pathways. Methylglyoxal mainly arises as side products of glycolysis involving glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. It is also thought to arise via the degradation of acetone and threonine. Illustrative of the myriad pathways to MGO, aristolochic acid caused 12 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
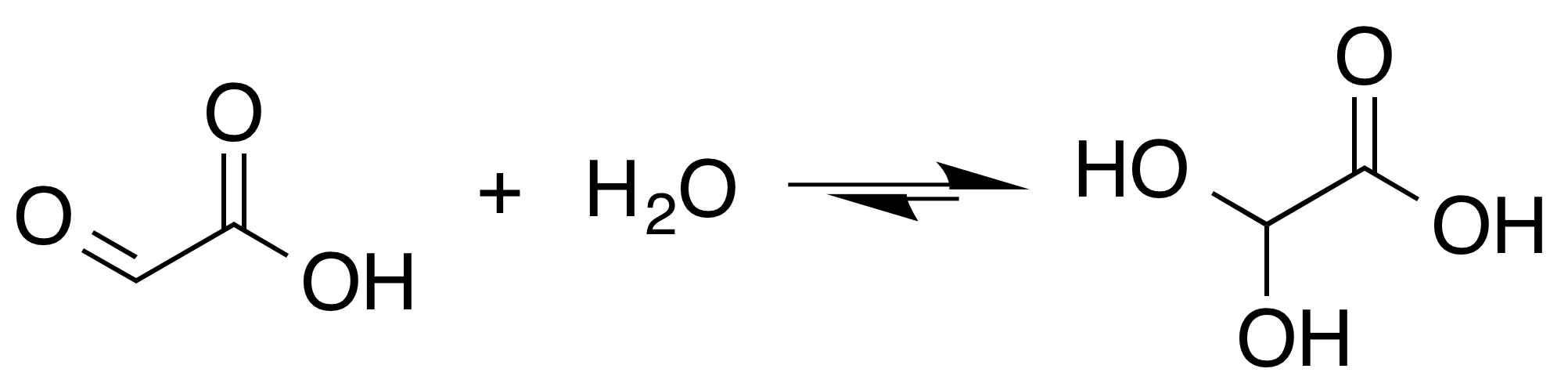
Glyoxylic Acid
Glyoxylic acid or oxoacetic acid is an organic compound. Together with acetic acid, glycolic acid, and oxalic acid, glyoxylic acid is one of the C2 carboxylic acids. It is a colourless solid that occurs naturally and is useful industrially. Structure and nomenclature Although the structure of glyoxylic acid is described as having an aldehyde functional group, the aldehyde is only a minor component of the form most prevalent in some situations. Instead, it often exists as a hydrate or a cyclic dimer. For example, in the presence of water, the carbonyl rapidly converts to a geminal diol (described as the "monohydrate"). The equilibrium constant (''K'') is 300 for the formation of dihydroxyacetic acid at room temperature: : In solution, the monohydrate exists in equilibrium with a hemi acylal dimer form:Georges Mattioda and Yani Christidis “Glyoxylic Acid” Ullmann's Encyclopedia of Industrial Chemistry, 2002, Wiley-VCH, Weinheim. : In isolation, the aldehyde structure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetaldehyde
Acetaldehyde (IUPAC systematic name ethanal) is an organic chemical compound with the formula CH3 CHO, sometimes abbreviated by chemists as MeCHO (Me = methyl). It is a colorless liquid or gas, boiling near room temperature. It is one of the most important aldehydes, occurring widely in nature and being produced on a large scale in industry. Acetaldehyde occurs naturally in coffee, bread, and ripe fruit, and is produced by plants. It is also produced by the partial oxidation of ethanol by the liver enzyme alcohol dehydrogenase and is a contributing cause of hangover after alcohol consumption. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Consumption of disulfiram inhibits acetaldehyde dehydrogenase, the enzyme responsible for the metabolism of acetaldehyde, thereby causing it to build up in the body. The International Agency for Research on Cancer (IARC) has listed acetaldehyde as a Group 1 carcinogen. Acetaldehyde is "one of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenious Acid
Selenous acid (or selenious acid) is the chemical compound with the formula . Structurally, it is more accurately described by . It is the principal oxoacid of selenium; the other being selenic acid. Formation and properties Selenous acid is analogous to sulfurous acid, but it is more readily isolated. Selenous acid is easily formed upon the addition of selenium dioxide to water. As a crystalline solid, the compound can be seen as pyramidal molecules that are interconnected with hydrogen bonds. In solution it is a diprotic acid: : (p''K''a = 2.62) : (p''K''a = 8.32) It is moderately oxidizing in nature, but kinetically slow. In 1 M : : (''E''o = +0.74 V) In 1 M : : (''E''o = −0.37 V) Selenous acid is hygroscopic. Uses The major use is in protecting and changing the color of steel, especially steel parts on firearms. The so-called cold-bluing process uses selenous acid, copper(II) nitrate, and nitric acid to change the color of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heinrich Debus (chemist)
Heinrich Debus (13 July 1824 – 9 December 1915) was a German chemist. Education and career In 1838, he attended a trade school in Kassel, where he was taught by Robert Wilhelm Bunsen. He studied chemistry from 1845 to 1848 in Marburg, and served as Bunsen's assistant from 1847. In 1848, he earned his doctorate by investigating a red madder dye. He completed his habilitation in 1851 after Bunsen left for Breslau. At the suggestion of Frederick Augustus Genth, Debus was named Bunsen's successor at Marburg. Later in 1851 he left for England to be a chemistry teacher at Queenwood College followed from 1868 to 1870 by the position as Science Master at Clifton College, Bristol. He then taught at Guy's Hospital, London from 1870 until appointed foundation Professor of Chemistry in 1873 at the new Royal Naval College, Greenwich, where he remained until his retirement and return to Germany. In 1858, Debus first synthesized imidazole from glyoxal, ammonia, and formaldehyde. The Debus ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Propanedial
Malondialdehyde (MDA) is the organic compound with the nominal formula CH2(CHO)2. A colorless liquid, malondialdehyde is a highly reactive compound that occurs as the enol. It occurs naturally and is a marker for oxidative stress. Structure and synthesis Malondialdehyde mainly exists as the enol:V. Nair, C. L. O'Neil, P. G. Wang "Malondialdehyde", ''Encyclopedia of Reagents for Organic Synthesis'', 2008, John Wiley & Sons, New York. Article Online Posting Date: March 14, 2008 :CH2(CHO)2 → HOC(H)=CH-CHO In organic solvents, the ''cis''-isomer is favored, whereas in water the ''trans''-isomer predominates. The equilibrium is rapid and is inconsequential for many purposes. In the laboratory it can be generated in situ by hydrolysis of its acetal 1,1,3,3-tetramethoxypropane, which is commercially available and shelf-stable, unlike malondialdehyde. Malondialdehyde is easily deprotonated to give the sodium salt of the enolate (m.p. 245 °C). Biosynthesis and reactivity ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. The metal is found in the Earth's crust in the pure, free elemental form ("native silver"), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc Refining (metallurgy), refining. Silver has long been valued as a precious metal. Silver metal is used in many bullion coins, sometimes bimetallism, alongside gold: while it is more abundant than gold, it is much less abundant as a native metal. Its purity is typically measured on a per-mille basis; a 94%-pure alloy is described as "0.940 fine". As one of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyruvic Acid
Pyruvic acid (CH3COCOOH) is the simplest of the alpha-keto acids, with a carboxylic acid and a ketone functional group. Pyruvate, the conjugate base, CH3COCOO−, is an intermediate in several metabolic pathways throughout the cell. Pyruvic acid can be made from glucose through glycolysis, converted back to carbohydrates (such as glucose) via gluconeogenesis, or to fatty acids through a reaction with acetyl-CoA. It can also be used to construct the amino acid alanine and can be converted into ethanol or lactic acid via fermentation. Pyruvic acid supplies energy to cells through the citric acid cycle (also known as the Krebs cycle) when oxygen is present ( aerobic respiration), and alternatively ferments to produce lactate when oxygen is lacking. Chemistry In 1834, Théophile-Jules Pelouze distilled tartaric acid and isolated glutaric acid and another unknown organic acid. Jöns Jacob Berzelius characterized this other acid the following year and named pyruvic acid bec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetylacetone
Acetylacetone is an organic compound with the chemical formula . It is a colorless liquid, classified as a 1,3- diketone. It exists in equilibrium with a tautomer . These tautomers interconvert so rapidly under most conditions that they are treated as a single compound in most applications. It is a colorless liquid that is a precursor to acetylacetonate anion (commonly abbreviated acac−), a bidentate ligand. It is also a building block for the synthesis of heterocyclic compounds. Properties Tautomerism The keto and enol tautomers of acetylacetone coexist in solution. The enol form has C2v symmetry, meaning the hydrogen atom is shared equally between the two oxygen atoms. In the gas phase, the equilibrium constant, ''K''keto→enol, is 11.7, favoring the enol form. The two tautomeric forms can be distinguished by NMR spectroscopy, IR spectroscopy and other methods. The equilibrium constant tends to be high in nonpolar solvents; when k = >1, the enol form is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usually gaseous or liquid) as the reactant, or heterogeneous, whose components are not in the same phase. Enzymes and other biocatalysts are often considered as a third category. Catalysis is ubiquitous in chemical industry of all kinds. Estimates are that 90% of all commercially produced chemical products involve catalysts at some sta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement. Copper is one of the few metals that can occur in nature in a directly usable metallic form (native metals). This led to very early human use in several regions, from circa 8000 BC. Thousands of years later, it was the first metal to be smelted from sulfide ores, circa 5000 BC; the first metal to be cast into a shape in a mold, c. 4000 BC; and the first metal to be purposely alloyed with another metal, tin, to create br ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trosly-Breuil
Trosly-Breuil () is a commune in the Oise department in northern France. In 1964, Canadian Jean Vanier invited two men, Raphael Simi and Philippe Seux, to leave the institutions where they lived and live with him in Trosly-Breuil. Their time together led to the establishment of L'Arche at Trosly-Breuil, a community for people with disabilities to live with those who cared for them. Since that time L'Arche communities have been established in fifty countries around the world. See also * Communes of the Oise department The following is a list of the 679 communes of the Oise department of France. The communes cooperate in the following intercommunalities (as of 2020):Communes of Oise {{Oise-geo-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |