|
FAT1
Protocadherin FAT1 is a protein that in humans is encoded by the ''FAT1'' gene. Function This gene is an ortholog of the ''Drosophila'' fat gene, which encodes a tumor suppressor essential for controlling cell proliferation during Drosophila development. The gene product is a member of the cadherin superfamily, a group of integral membrane proteins characterized by the presence of cadherin-type repeats. This gene is expressed at high levels in a number of fetal epithelia. Transcript variants derived from alternative splicing and/or alternative promoter usage exist, but they have not been fully described. The murine Fat1 knockout mouse is not embryonically lethal but pups die within 48-hours due to the abnormal fusion of foot processes of the podocytes within the kidney. These Fat1 knockout mice also showed partially penetrant but often severe midline defects including holoprosencephaly, microphthalmia-anophthalmia and in rare cases cyclopia. It has been shown that the EVH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hippo Signaling Pathway
The Hippo signaling pathway, also known as the Salvador-Warts-Hippo (SWH) pathway, is a signaling pathway that controls organ size in animals through the regulation of cell proliferation and apoptosis. The pathway takes its name from one of its key signaling components—the protein kinase Hippo (Hpo). Mutations in this gene lead to tissue overgrowth, or a "hippopotamus"-like phenotype. A fundamental question in developmental biology is how an organ knows to stop growing after reaching a particular size. Organ growth relies on several processes occurring at the cellular level, including cell division and programmed cell death (or apoptosis). The Hippo signaling pathway is involved in restraining cell proliferation and promoting apoptosis. As many cancers are marked by unchecked cell division, this signaling pathway has become increasingly significant in the study of human cancer. The Hippo pathway also has a critical role in stem cell and tissue specific progenitor cell self-r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Podocyte
Podocytes are cells in Bowman's capsule in the kidneys that wrap around capillaries of the glomerulus. Podocytes make up the epithelial lining of Bowman's capsule, the third layer through which filtration of blood takes place. Bowman's capsule filters the blood, retaining large molecules such as proteins while smaller molecules such as water, salts, and sugars are filtered as the first step in the formation of urine. Although various viscera have epithelial layers, the name visceral epithelial cells usually refers specifically to podocytes, which are specialized epithelial cells that reside in the visceral layer of the capsule. One type of specialized epithelial cell is podocalyxin. The podocytes have long foot processes called ''pedicels'', for which the cells are named ('' podo-'' + '' -cyte''). The pedicels wrap around the capillaries and leave slits between them. Blood is filtered through these slits, each known as a filtration slit, slit diaphragm, or slit pore. Several ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glypican 3
Glypican-3 is a protein that, in humans, is encoded by the ''GPC3'' gene. The ''GPC3'' gene is located on human X chromosome (Xq26) where the most common gene (Isoform 2, GenBank Accession No.: NP_004475) encodes a 70-kDa core protein with 580 amino acids. Three variants have been detected that encode alternatively spliced forms termed Isoforms 1 (NP_001158089), Isoform 3 (NP_001158090) and Isoform 4 (NP_001158091). Structure and function The protein core of GPC3 consists of two subunits, where the N-terminal subunit has a size of ~40 kDa and the C-terminal subunit is ~30 kDa. Six glypicans (GPC1-6) have been identified in mammals. Cell surface heparan sulfate proteoglycans are composed of a membrane-associated protein core substituted with a variable number of heparan sulfate chains. Members of the glypican-related integral membrane proteoglycan family (GRIPS) contain a core protein anchored to the cytoplasmic membrane via a glycosyl phosphatidylinositol linkage. These proteins m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
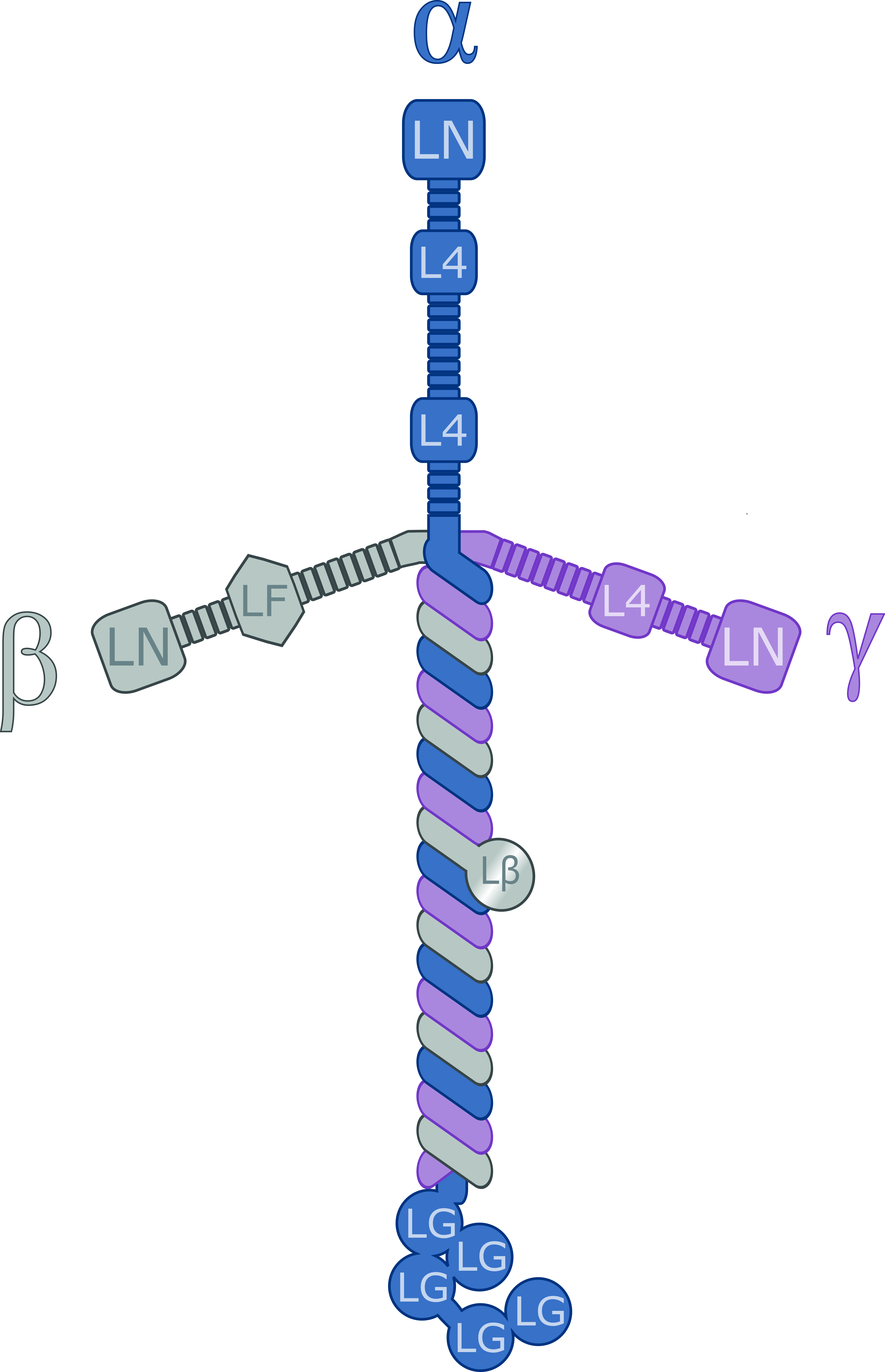
Laminin
Laminins are a family of glycoproteins of the extracellular matrix of all animals. They are major components of the basal lamina (one of the layers of the basement membrane), the protein network foundation for most cells and organs. The laminins are an important and biologically active part of the basal lamina, influencing cell differentiation, migration, and adhesion. Laminins are heterotrimeric proteins with a high molecular mass (~400 to ~900 kDa). They contain three different chains (α, β and γ) encoded by five, four, and three paralogous genes in humans, respectively. The laminin molecules are named according to their chain composition. Thus, laminin-511 contains α5, β1, and γ1 chains. Fourteen other chain combinations have been identified ''in vivo''. The trimeric proteins intersect to form a cross-like structure that can bind to other cell membrane and extracellular matrix molecules. The three shorter arms are particularly good at binding to other laminin molecule ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid resid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
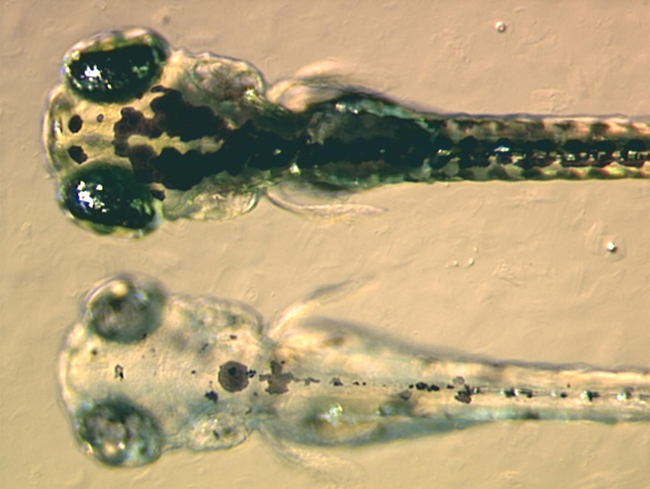
Zebrafish
The zebrafish (''Danio rerio'') is a freshwater fish belonging to the minnow family (Cyprinidae) of the order Cypriniformes. Native to South Asia, it is a popular aquarium fish, frequently sold under the trade name zebra danio (and thus often called a " tropical fish" although both tropical and subtropical). It is also found in private ponds. The zebrafish is an important and widely used vertebrate model organism in scientific research, for example in drug development, in particular pre-clinical development. It is also notable for its regenerative abilities, and has been modified by researchers to produce many transgenic strains. Taxonomy The zebrafish is a derived member of the genus ''Brachydanio'', of the family Cyprinidae. It has a sister-group relationship with '' Danio aesculapii''. Zebrafish are also closely related to the genus '' Devario'', as demonstrated by a phylogenetic tree of close species. Distribution Range The zebrafish is native to fresh wat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
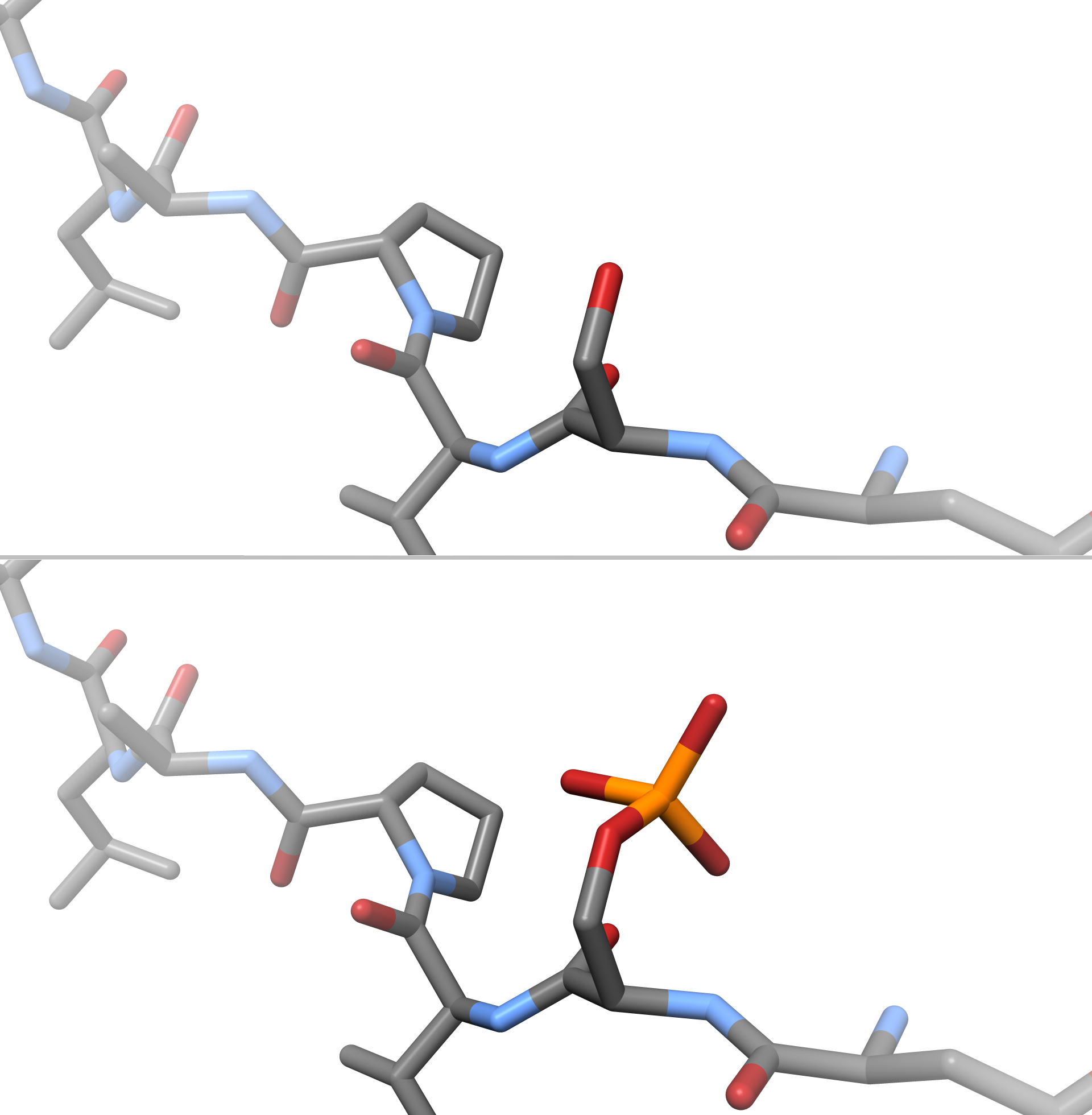
Phosphorylated
In chemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology and could be driven by natural selection. Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License. Protein phosphorylation often activates (or deactivates) many enzymes. Glucose Phosphorylation of sugars is often the first stage in their catabolism. Phosphorylation allows cells to accumulate sugars because the phosphate group prevents the molecules from diffusing back across their transporter. Phosphorylation of glucose is a key reaction in sugar metabolism. The chemical equation for the conversion of D-glucose to D-glucose-6-phosphate in the first step of glycolysis is given by :D-glucose + ATP → D-glucose-6-phosphate + ADP :ΔG° = −16.7 kJ/mol (° indicates measurement at standard condition) Hepatic cells are freely permeable to glucose, and the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterodimer
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word ''dimer'' has roots meaning "two parts", ''di-'' + '' -mer''. A protein dimer is a type of protein quaternary structure. A protein homodimer is formed by two identical proteins. A protein heterodimer is formed by two different proteins. Most protein dimers in biochemistry are not connected by covalent bonds. An example of a non-covalent heterodimer is the enzyme reverse transcriptase, which is composed of two different amino acid chains. An exception is dimers that are linked by disulfide bridges such as the homodimeric protein NEMO. Some proteins contain specialized domains to ensure dimerization (dimerization domains) and specificity. The G protein-coupled cannabinoid receptors have the ability to form both homo- and heterodimers with sever ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Furin
Furin is a protease, a proteolytic enzyme that in humans and other animals is encoded by the ''FURIN'' gene. Some proteins are inactive when they are first synthesized, and must have sections removed in order to become active. Furin cleaves these sections and activates the proteins. It was named furin because it was in the upstream region of an oncogene known as FES. The gene was known as FUR (FES Upstream Region) and therefore the protein was named furin. Furin is also known as PACE (Paired basic Amino acid Cleaving Enzyme). A member of family S8, furin is a subtilisin-like peptidase. Function The protein encoded by this gene is an enzyme that belongs to the subtilisin-like proprotein convertase family. The members of this family are proprotein convertases that process latent precursor proteins into their biologically active products. This encoded protein is a calcium-dependent serine endoprotease that can efficiently cleave precursor proteins at their paired basic amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
EGF Domain
The EGF-like domain is an evolutionary conserved protein domain, which derives its name from the epidermal growth factor where it was first described. It comprises about 30 to 40 amino-acid residues and has been found in a large number of mostly animal proteins. Most occurrences of the EGF-like domain are found in the extracellular domain of membrane-bound proteins or in proteins known to be secreted. An exception to this is the prostaglandin-endoperoxide synthase. The EGF-like domain includes 6 cysteine residues which in the epidermal growth factor have been shown to form 3 disulfide bonds. The structures of 4-disulfide EGF-domains have been solved from the laminin and integrin proteins. The main structure of EGF-like domains is a two-stranded β-sheet followed by a loop to a short C-terminal, two-stranded β-sheet. These two β-sheets are usually denoted as the major (N-terminal) and minor (C-terminal) sheets. EGF-like domains frequently occur in numerous tandem copies in prot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wnt Signaling Pathway
The Wnt signaling pathways are a group of signal transduction pathways which begin with proteins that pass signals into a cell through cell surface receptors. The name Wnt is a portmanteau created from the names Wingless and Int-1. Wnt signaling pathways use either nearby cell-cell communication (paracrine) or same-cell communication (autocrine). They are highly evolutionarily conserved in animals, which means they are similar across animal species from fruit flies to humans. Three Wnt signaling pathways have been characterized: the canonical Wnt pathway, the noncanonical planar cell polarity pathway, and the noncanonical Wnt/calcium pathway. All three pathways are activated by the binding of a Wnt-protein ligand to a Frizzled family receptor, which passes the biological signal to the Dishevelled protein inside the cell. The canonical Wnt pathway leads to regulation of gene transcription, and is thought to be negatively regulated in part by the SPATS1 gene. The noncanonical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beta-catenin
Catenin beta-1, also known as beta-catenin (β-catenin), is a protein that in humans is encoded by the ''CTNNB1'' gene. Beta-catenin is a dual function protein, involved in regulation and coordination of cell–cell adhesion and gene transcription. In humans, the CTNNB1 protein is encoded by the ''CTNNB1'' gene. In '' Drosophila'', the homologous protein is called ''armadillo''. β-catenin is a subunit of the cadherin protein complex and acts as an intracellular signal transducer in the Wnt signaling pathway. It is a member of the catenin protein family and homologous to γ-catenin, also known as plakoglobin. Beta-catenin is widely expressed in many tissues. In cardiac muscle, beta-catenin localizes to adherens junctions in intercalated disc structures, which are critical for electrical and mechanical coupling between adjacent cardiomyocytes. Mutations and overexpression of β-catenin are associated with many cancers, including hepatocellular carcinoma, colorectal ca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |