|
Electride 01
An electride is an ionic compound in which an electron is the anion. Solutions of alkali metals in ammonia are electride salts. In the case of sodium, these blue Solution (chemistry), solutions consist of [Na(NH3)6]+ and solvated electrons: :Na + 6 NH3 → [Na(NH3)6]+ + e− The cation [Na(NH3)6]+ is an octahedral molecular geometry, octahedral coordination complex. Solid salts Addition of a complexant like crown ether or 2.2.2-Cryptand, [2.2.2]-cryptand to a solution of [Na(NH3)6]+e− affords [Na (crown ether)]+e− or [Na(2,2,2-crypt)]+e−. Evaporation of these solutions yields a blue-black paramagnetic solid with the formula [Na(2,2,2-crypt)]+e−. Most solid electride salts decompose above 240 K, although [Ca24Al28O64]4+(e−)4 is stable at room temperature. In these salts, the electron is delocalized between the cations. Electrides are paramagnetism, paramagnetic, and are Mott insulators. Properties of these salts have been analyzed. Thorium compounds, ThI2 and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electride 01
An electride is an ionic compound in which an electron is the anion. Solutions of alkali metals in ammonia are electride salts. In the case of sodium, these blue Solution (chemistry), solutions consist of [Na(NH3)6]+ and solvated electrons: :Na + 6 NH3 → [Na(NH3)6]+ + e− The cation [Na(NH3)6]+ is an octahedral molecular geometry, octahedral coordination complex. Solid salts Addition of a complexant like crown ether or 2.2.2-Cryptand, [2.2.2]-cryptand to a solution of [Na(NH3)6]+e− affords [Na (crown ether)]+e− or [Na(2,2,2-crypt)]+e−. Evaporation of these solutions yields a blue-black paramagnetic solid with the formula [Na(2,2,2-crypt)]+e−. Most solid electride salts decompose above 240 K, although [Ca24Al28O64]4+(e−)4 is stable at room temperature. In these salts, the electron is delocalized between the cations. Electrides are paramagnetism, paramagnetic, and are Mott insulators. Properties of these salts have been analyzed. Thorium compounds, ThI2 and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cerium
Cerium is a chemical element with the symbol Ce and atomic number 58. Cerium is a soft, ductile, and silvery-white metal that tarnishes when exposed to air. Cerium is the second element in the lanthanide series, and while it often shows the +3 oxidation state characteristic of the series, it also has a stable +4 state that does not oxidize water. It is also considered one of the rare-earth elements. Cerium has no known biological role in humans but is not particularly toxic, except with intense or continued exposure. Despite always occurring in combination with the other rare-earth elements in minerals such as those of the monazite and bastnäsite groups, cerium is easy to extract from its ores, as it can be distinguished among the lanthanides by its unique ability to be oxidized to the +4 state in aqueous solution. It is the most common of the lanthanides, followed by neodymium, lanthanum, and praseodymium. It is the 25th-most abundant element, making up 66 ppm of the Ear ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salts
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively charged sodium ions and negatively charged chloride ions. The component ions in a salt compound can be either inorganic, such as chloride (Cl−), or organic, such as acetate (). Each ion can be either monatomic, such as fluoride (F−), or polyatomic, such as sulfate (). Types of salt Salts can be classified in a variety of ways. Salts that produce hydroxide ions when dissolved in water are called ''alkali salts'' and salts that produce hydrogen ions when dissolved in water are called ''acid salts''. ''Neutral salts'' are those salts that are neither acidic nor basic. Zwitterions contain an anionic and a cationic centre in the same molecule, but are not considered salts. Examples of zwitterions are amino acids, many metabolites, peptides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
F-center
An F center or Farbe center (from the original German ''Farbzentrum'', where ''Farbe'' means ''color'' and ''zentrum'' means center) is a type of crystallographic defect in which an anionic vacancy in a crystal lattice is occupied by one or more unpaired electrons. Electrons in such a vacancy in a crystal lattice tend to absorb light in the visible spectrum such that a material that is usually Transparency (optics), transparent becomes colored. The greater the number of F centers, the more intense the color of the compound. F centers are a type of color center. This is used to identify many compounds, especially zinc oxide (yellow). History Before the discovery of point defects it was already known that some crystals can be discolored using various methods. In 1830 T.J. Pearsall discovered that fluorspar could be discolored using violet light. Thirty years later similar results were achieved by melting crystals together with a specific metal. In 1921 Wilhelm Röntgen, W. Röntgen e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Single-layer Materials
In materials science, the term single-layer materials or 2D materials refers to crystalline solids consisting of a single layer of atoms. These materials are promising for some applications but remain the focus of research. Single-layer materials derived from single elements generally carry the -ene suffix in their names, e.g. graphene. Single-layer materials that are compounds of two or more elements have -ane or -ide suffixes. 2D materials can generally be categorized as either 2D allotropes of various elements or as compounds (consisting of two or more covalently bonding elements). It is predicted that there are hundreds of stable single-layer materials. The atomic structure and calculated basic properties of these and many other potentially synthesisable single-layer materials, can be found in computational databases. 2D materials can be produced using mainly two approaches: top-down exfoliation and bottom-up synthesis. The exfoliation methods include sonication, mechanical, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Disodium Helide
Disodium helide (Na2He) is a compound of helium and sodium that is stable at high pressures above . It was first predicted using the USPEX crystal structure prediction algorithm and then synthesised in 2016. Synthesis Na2He was predicted to be thermodynamically stable over 160 GPa and dynamically stable over 100 GPa. This means it should be possible to form at the higher pressure and then decompress to 100 GPa, but below that it would decompose. Compared with other binary compounds of other elements and helium, it was predicted to be stable at the lowest pressure of any such combination. This also means, for example, that a helium-potassium compound is predicted to require much higher pressures of the order of terapascals. The material was synthesized by putting tiny plates of sodium in a diamond anvil cell along with helium at 1600 bar and then compressing to 130 GPa and heating to 1,500 K with a laser. Disodium helide is predicted to be an insulator and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scientific Reports
''Scientific Reports'' is a peer-reviewed open-access scientific mega journal published by Nature Portfolio, covering all areas of the natural sciences. The journal was established in 2011. The journal states that their aim is to assess solely the scientific validity of a submitted paper, rather than its perceived importance, significance, or impact. In September 2016, the journal became the largest in the world by number of articles, overtaking '' PLOS ONE''. Abstracting and indexing The journal is abstracted and indexed in the Chemical Abstracts Service, the Science Citation Index Expanded, and selectively in Index Medicus/MEDLINE/PubMed. According to the ''Journal Citation Reports'', the journal has a 2021 impact factor 4.996. Reviewing policy The ''Guide to Referees'' states that to be published, "a paper must be scientifically valid and technically sound in methodology and analysis", and reviewers have to ensure manuscripts "are not assessed based on their perceived impor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Physical Review Letters
''Physical Review Letters'' (''PRL''), established in 1958, is a peer-reviewed, scientific journal that is published 52 times per year by the American Physical Society. As also confirmed by various measurement standards, which include the ''Journal Citation Reports'' impact factor and the journal ''h''-index proposed by Google Scholar, many physicists and other scientists consider ''Physical Review Letters'' to be one of the most prestigious journals in the field of physics. ''According to Google Scholar, PRL is the journal with the 9th journal h-index among all scientific journals'' ''PRL'' is published as a print journal, and is in electronic format, online and CD-ROM. Its focus is rapid dissemination of significant, or notable, results of fundamental research on all topics related to all fields of physics. This is accomplished by rapid publication of short reports, called "Letters". Papers are published and available electronically one article at a time. When published in s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Localization Function
In quantum chemistry, the electron localization function (ELF) is a measure of the likelihood of finding an electron in the neighborhood space of a reference electron located at a given point and with the same spin. Physically, this measures the extent of spatial localization of the reference electron and provides a method for the mapping of electron pair probability in multielectronic systems. ELF's usefulness stems from the observation that it allows electron localization to be analyzed in a chemically intuitive way. For example, the shell structure of heavy atoms is obvious when plotting ELF against the radial distance from the nucleus; the ELF for radon has six clear maxima, whereas the electronic density decreases monotonically and the radially weighted density fails to show all shells. When applied to molecules, an analysis of the ELF shows a clear separation between the core and valence electron, and also shows covalent bonds and lone pairs, in what has been called "a fai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
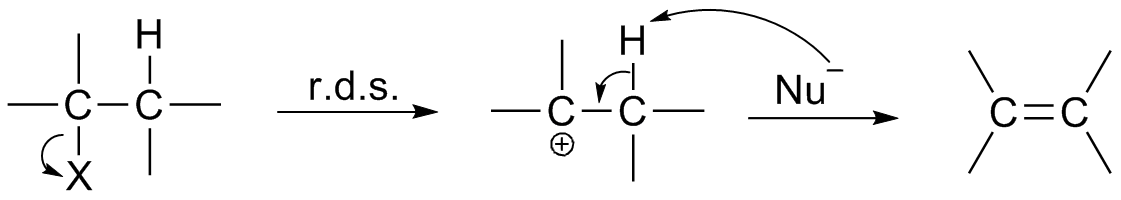
Reaction Intermediate
In chemistry, a reaction intermediate or an intermediate is a molecular entity that is formed from the reactants (or preceding intermediates) but is consumed in further reactions in stepwise chemical reactions that contain multiple elementary steps. Intermediates are the reaction product of one elementary step, but do not appear in the chemical equation for an overall chemical equation. For example, consider this hypothetical stepwise reaction: :A + B -> C + D The reaction includes two elementary steps: :A + B -> X :X -> C + D In this example, X is a reaction intermediate. IUPAC definition The IUPAC Gold Book defines an ''intermediate'' as a compound that has a lifetime greater than a molecular vibration that is formed (directly or indirectly) from the reactants and reacts further to give (either directly or indirectly) the products of a chemical reaction. The lifetime condition distinguishes true, chemically distinct intermediates from vibrational states or such transition st ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Amide
Sodium amide, commonly called sodamide (systematic name sodium azanide), is the inorganic compound with the formula . It is a salt composed of the sodium cation and the azanide anion. This solid, which is dangerously reactive toward water, is white, but commercial samples are typically gray due to the presence of small quantities of metallic iron from the manufacturing process. Such impurities do not usually affect the utility of the reagent. conducts electricity in the fused state, its conductance being similar to that of NaOH in a similar state. has been widely employed as a strong base in organic synthesis. Preparation and structure Sodium amide can be prepared by the reaction of sodium with ammonia gas, but it is usually prepared by the reaction in liquid ammonia using iron(III) nitrate as a catalyst. The reaction is fastest at the boiling point of the ammonia, c. −33 °C. An electride, , is formed as a reaction intermediate. : is a salt-like material and as such, c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Birch Reduction
The Birch reduction is an organic reaction that is used to convert arenes to cyclohexadienes. The reaction is named after the Australian chemist Arthur Birch and involves the organic reduction of aromatic rings in an amine solvent (traditionally liquid ammonia) with an alkali metal (traditionally sodium) and a proton source (traditionally an alcohol). Unlike catalytic hydrogenation, Birch reduction does not reduce the aromatic ring all the way to a cyclohexane. An example is the reduction of naphthalene in ammonia and ethanol: Reaction mechanism and regioselectivity A solution of sodium in liquid ammonia consists of the intensely blue electride salt a(NH3)xsup>+ e−. The solvated electrons add to the aromatic ring to give a radical anion, which then abstracts a proton from the alcohol. The process then repeats at either the ''ortho'' or ''para'' position (depending on substituents) to give the final diene. The residual double bonds do not stabilize further radical add ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



.png)