|
Dimethylamidophosphoric Dicyanide
Dimethylamidophosphoric dicyanide is an important chemical for the final process of synthesizing Tabun, a nerve agent used as a chemical weapon. Preparation Dimethylamidophosphoric dicyanide could be prepared by reacting Dimethylamidophosphoric dichloride with sodium cyanide. : Safety This chemical is very flammable, highly toxic, and reactive. If ingested or absorbed through skin, it will cause mild nerve agent symptoms directly as well as blood agent symptoms due to release of HCN. If mixed with water, it gives off poisonous hydrogen cyanide fumes and dimethylamidophosphoric acid. See also * Organophosphate poisoning * Hydrogen cyanide Hydrogen cyanide, sometimes called prussic acid, is a chemical compound with the formula HCN and structure . It is a colorless, extremely poisonous, and flammable liquid that boils slightly above room temperature, at . HCN is produced on an ... References {{Chemical agents Organophosphates Nerve agent precursors Cyano compoun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tabun (nerve Agent)
Tabun or GA is an extremely toxic synthetic organophosphorus compound. It is a clear, colorless, and tasteless liquid with a faint fruity odor.Facts About Tabun National Terror Alert Response System It is classified as a because it fatally interferes with normal functioning of the mammalian nervous system. Its production is strictly controlled and stockpiling outlawed by the Chemical Weapons Convention of 1993. Tabun is the first of the ''G-series'' nerve agents along with GB ( [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylamidophosphoric Dichloride
Dimethylamidophosphoric dichloride is an important chemical for few industrial purposes. It is an important chemical for synthesizing phosphoramidates as well as Nerve agent GA which is used as a chemical weapon. Safety This chemical is also corrosive, flammable and will cause mild nerve agent symptoms if ingested or absorbed through skin due to its nature. It will react with water giving off hydrogen chloride vapors and dimethylamidophosphoric acid. History First synthesis of the substance dates back to turn of the 19th century, when a student of German chemistry professor August Michaelis Ernst Ratzlaff made dimethylamidophosphoric dichloride as well as its diethyl analog for experiments of another PhD student of Michaelis Adolph Schall. He obtained a PhD at University of Rostock with a thesis titled ''Über die Einwirkung primärer und sekundärer Amine auf Phosphoroxychlorid und Äthoxylphosphoroxychlorid'' later in 1901. The high toxicity of the substance (as well as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Cyanide
Sodium cyanide is a poisonous compound with the formula Na C N. It is a white, water-soluble solid. Cyanide has a high affinity for metals, which leads to the high toxicity of this salt. Its main application, in gold mining, also exploits its high reactivity toward metals. It is a moderately strong base. Production and chemical properties Sodium cyanide is produced by treating hydrogen cyanide with sodium hydroxide: :HCN + NaOH → NaCN + H2O Worldwide production was estimated at 500,000 tons in the year 2006. Formerly it was prepared by the Castner process involving the reaction of sodium amide with carbon at elevated temperatures. : NaNH2 + C → NaCN + H2 The structure of solid NaCN is related to that of sodium chloride. The anions and cations are each six-coordinate. Potassium cyanide (KCN) adopts a similar structure. When treated with acid, it forms the toxic gas hydrogen cyanide: : NaCN + H+ → HCN + Na+ Because the salt is derived from a weak aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthesis Of Dimethylamidophosphoric Dicyanide-2D-by-AHRLS
Synthesis or synthesize may refer to: Science Chemistry and biochemistry *Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors **Organic synthesis, the chemical synthesis of organic compounds ***Total synthesis, the complete organic synthesis of complex organic compounds, usually without the aid of biological processes ***Convergent synthesis or linear synthesis, a strategy to improve the efficiency of multi-step chemical syntheses **Dehydration synthesis, a chemical synthesis resulting in the loss of a water molecule *Biosynthesis, the creation of an organic compound in a living organism, usually aided by enzymes **Photosynthesis, a biochemical reaction using a carbon molecule to produce an organic molecule, using sunlight as a catalyst **Chemosynthesis, the synthesis of biological compounds into organic waste, using methane or an oxidized molecule as a catalyst **Amino acid synthesis, the synthesis of an amino acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Cyanide
Hydrogen cyanide, sometimes called prussic acid, is a chemical compound with the formula HCN and structure . It is a colorless, extremely poisonous, and flammable liquid that boils slightly above room temperature, at . HCN is produced on an industrial scale and is a highly valued precursor to many chemical compounds ranging from polymers to pharmaceuticals. Large-scale applications are for the production of potassium cyanide and adiponitrile, used in mining and plastics, respectively. It is more toxic than solid cyanide compounds due to its volatile nature. Structure and general properties Hydrogen cyanide is a linear molecule, with a triple bond between carbon and nitrogen. The tautomer of HCN is HNC, hydrogen isocyanide. Hydrogen cyanide is weakly acidic with a p''K''a of 9.2. It partially ionizes in water solution to give the cyanide anion, CN−. A solution of hydrogen cyanide in water, represented as HCN, is called ''hydrocyanic acid''. The salts of the cyanide ani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Poisonous
Poison is a chemical substance that has a detrimental effect to life. The term is used in a wide range of scientific fields and industries, where it is often specifically defined. It may also be applied colloquially or figuratively, with a broad sense. Whether something is considered a poison may change depending on the amount, the circumstances, and what living things are present. Poisoning could be accidental or deliberate, and if the cause can be identified there may be ways to neutralise the effects or minimise the symptoms. In biology, a poison is a chemical substance causing death, injury or harm to organisms or their parts. In medicine, poisons are a kind of toxin that are delivered passively, not actively. In industry the term may be negative, something to be removed to make a thing safe, or positive, an agent to limit unwanted pests. In ecological terms, poisons introduced into the environment can later cause unwanted effects elsewhere, or in other parts of the food ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organophosphate Poisoning
Organophosphate poisoning is poisoning due to organophosphates (OPs). Organophosphates are used as insecticides, medications, and nerve agents. Symptoms include increased saliva and tear production, diarrhea, vomiting, small pupils, sweating, muscle tremors, and confusion. While onset of symptoms is often within minutes to hours, some symptoms can take weeks to appear. Symptoms can last for days to weeks. Organophosphate poisoning occurs most commonly as a suicide attempt in farming areas of the developing world and less commonly by accident. Exposure can be from drinking, breathing in the vapors, or skin exposure. The underlying mechanism involves the inhibition of acetylcholinesterase (AChE), leading to the buildup of acetylcholine (ACh) in the body. Diagnosis is typically based on the symptoms and can be confirmed by measuring butyrylcholinesterase activity in the blood. Carbamate poisoning can present similarly. Prevention efforts include banning very toxic types of organo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Cyanide
Hydrogen cyanide, sometimes called prussic acid, is a chemical compound with the formula HCN and structure . It is a colorless, extremely poisonous, and flammable liquid that boils slightly above room temperature, at . HCN is produced on an industrial scale and is a highly valued precursor to many chemical compounds ranging from polymers to pharmaceuticals. Large-scale applications are for the production of potassium cyanide and adiponitrile, used in mining and plastics, respectively. It is more toxic than solid cyanide compounds due to its volatile nature. Structure and general properties Hydrogen cyanide is a linear molecule, with a triple bond between carbon and nitrogen. The tautomer of HCN is HNC, hydrogen isocyanide. Hydrogen cyanide is weakly acidic with a p''K''a of 9.2. It partially ionizes in water solution to give the cyanide anion, CN−. A solution of hydrogen cyanide in water, represented as HCN, is called ''hydrocyanic acid''. The salts of the cyanide ani ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organophosphates
In organic chemistry, organophosphates (also known as phosphate esters, or OPEs) are a class of organophosphorus compounds with the general structure , a central phosphate molecule with alkyl or aromatic substituents. They can be considered as esters of phosphoric acid. Like most functional groups, organophosphates occur in a diverse range of forms, with important examples including key biomolecules such as DNA, RNA and ATP, as well as many insecticides, herbicides, nerve agents and flame retardants. OPEs have been widely used in various products as flame retardants, plasticizers, and performance additives to engine oil. The popularity of OPEs as flame retardants came as a substitution for the highly regulated brominated flame retardants. The low cost of production and compatibility to diverse polymers made OPEs to be widely used in industry including textile, furniture, electronics as plasticizers and flame retardants. These compounds are added to the final product physi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
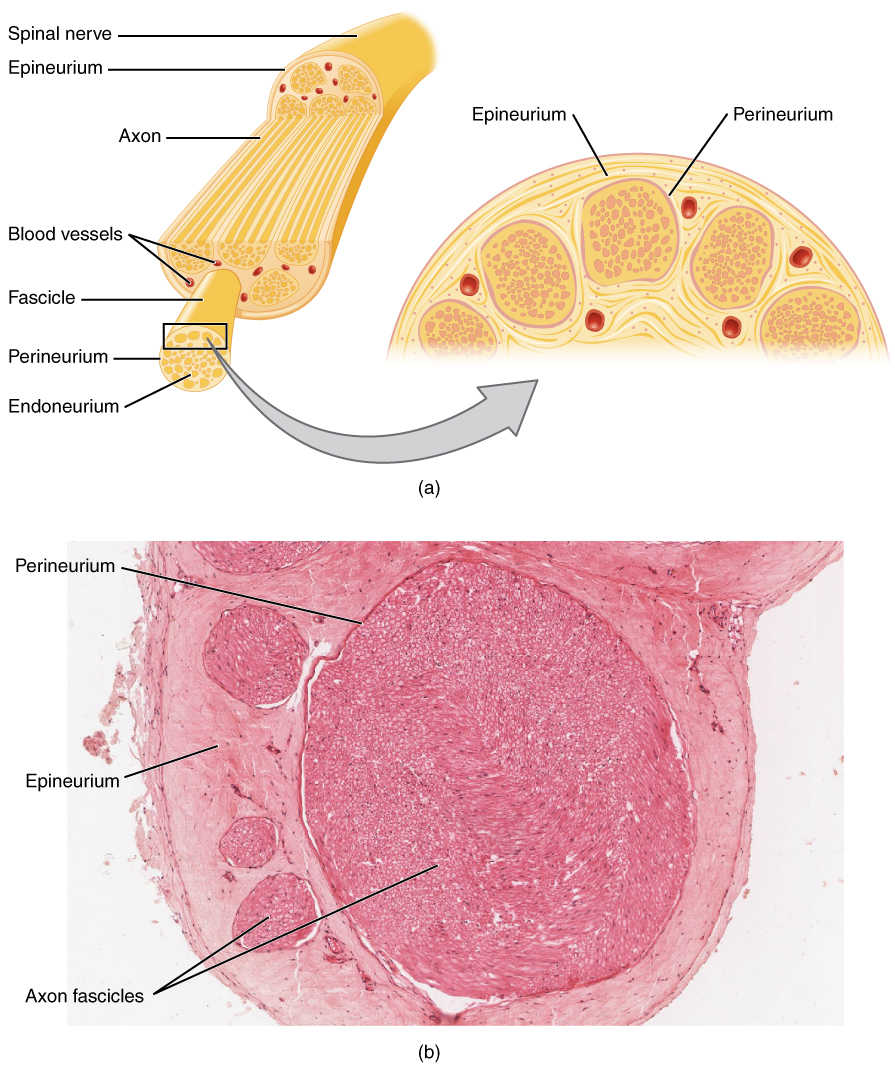
Nerve Agent Precursors
A nerve is an enclosed, cable-like bundle of nerve fibers (called axons) in the peripheral nervous system. A nerve transmits electrical impulses. It is the basic unit of the peripheral nervous system. A nerve provides a common pathway for the electrochemical nerve impulses called action potentials that are transmitted along each of the axons to peripheral organs or, in the case of sensory nerves, from the periphery back to the central nervous system. Each axon, within the nerve, is an extension of an individual neuron, along with other supportive cells such as some Schwann cells that coat the axons in myelin. Within a nerve, each axon is surrounded by a layer of connective tissue called the endoneurium. The axons are bundled together into groups called fascicles, and each fascicle is wrapped in a layer of connective tissue called the perineurium. Finally, the entire nerve is wrapped in a layer of connective tissue called the epineurium. Nerve cells (often called neurons) are f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |