|
Dihydrolipoamide Dehydrogenase
Dihydrolipoamide dehydrogenase (DLD), also known as dihydrolipoyl dehydrogenase, mitochondrial, is an enzyme that in humans is encoded by the ''DLD'' gene. DLD is a flavoprotein enzyme that oxidizes dihydrolipoamide to lipoamide. Dihydrolipoamide dehydrogenase (DLD) is a mitochondrial enzyme that plays a vital role in energy metabolism in eukaryotes. This enzyme is required for the complete reaction of at least five different multi-enzyme complexes. Additionally, DLD is a flavoenzyme oxidoreductase that contains a reactive disulfide bridge and a FAD cofactor that are directly involved in catalysis. The enzyme associates into tightly bound homodimers required for its enzymatic activity. File:Lipoamide-2D-skeletal.png, Lipoamide File:Dihydrolipoamide.svg, Dihydrolipoamide Structure The protein encoded by the DLD gene comes together with another protein to form a dimer in the central metabolic pathway. Several amino acids within the catalytic pocket have been identified as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the reaction ra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Active Site
In biology and biochemistry, the active site is the region of an enzyme where substrate molecules bind and undergo a chemical reaction. The active site consists of amino acid residues that form temporary bonds with the substrate (binding site) and residues that catalyse a reaction of that substrate (catalytic site). Although the active site occupies only ~10–20% of the volume of an enzyme, it is the most important part as it directly catalyzes the chemical reaction. It usually consists of three to four amino acids, while other amino acids within the protein are required to maintain the tertiary structure of the enzymes. Each active site is evolved to be optimised to bind a particular substrate and catalyse a particular reaction, resulting in high specificity. This specificity is determined by the arrangement of amino acids within the active site and the structure of the substrates. Sometimes enzymes also need to bind with some cofactors to fulfil their function. The active si ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Failure To Thrive
Failure to thrive (FTT), also known as weight faltering or faltering growth, indicates insufficient weight gain or absence of appropriate physical growth in children. FTT is usually defined in terms of weight, and can be evaluated either by a low weight for the child's age, or by a low rate of increase in the weight. The term "failure to thrive" has been used in different ways, as there is no objective standard or universally accepted definition for when to diagnose FTT. One definition describes FTT as a fall in one or more weight centile spaces on a World Health Organization (WHO) growth chart depending on birth weight or when weight is below the 2nd percentile of weight for age irrespective of birth weight. Another definition of FTT is a weight for age that is consistently below the 5th percentile or weight for age that falls by at least two major percentile lines on a growth chart. While weight loss after birth is normal and most babies return to their birth weight by three wee ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reperfusion Injury
Reperfusion injury, sometimes called ischemia-reperfusion injury (IRI) or reoxygenation injury, is the tissue damage caused when blood supply returns to tissue ('' re-'' + '' perfusion'') after a period of ischemia or lack of oxygen (anoxia or hypoxia). The absence of oxygen and nutrients from blood during the ischemic period creates a condition in which the restoration of circulation results in inflammation and oxidative damage through the induction of oxidative stress rather than (or along with) restoration of normal function. Reperfusion injury is distinct from cerebral hyperperfusion syndrome (sometimes called "Reperfusion syndrome"), a state of abnormal cerebral vasodilation. Mechanisms Reperfusion of ischemic tissues is often associated with microvascular injury, particularly due to increased permeability of capillaries and arterioles that lead to an increase of diffusion and fluid filtration across the tissues. Activated endothelial cells produce more reactive oxygen sp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein Moonlighting
Protein moonlighting (or gene sharing) is a phenomenon by which a protein can perform more than one function. Ancestral moonlighting proteins originally possessed a single function but through evolution, acquired additional functions. Many proteins that moonlight are enzymes; others are receptors, ion channels or chaperones. The most common primary function of moonlighting proteins is enzymatic catalysis, but these enzymes have acquired secondary non-enzymatic roles. Some examples of functions of moonlighting proteins secondary to catalysis include signal transduction, transcriptional regulation, apoptosis, motility, and structural. Protein moonlighting may occur widely in nature. Protein moonlighting through gene sharing differs from the use of a single gene to generate different proteins by alternative RNA splicing, DNA rearrangement, or post-translational processing. It is also different from multifunctionality of the protein, in which the protein has multiple domains, eac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
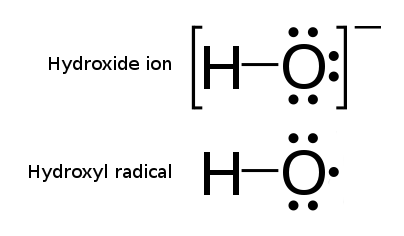
Hydroxyl Radical
The hydroxyl radical is the diatomic molecule . The hydroxyl radical is very stable as a dilute gas, but it decays very rapidly in the condensed phase. It is pervasive in some situations. Most notably the hydroxyl radicals are produced from the decomposition of hydroperoxides (ROOH) or, in atmospheric chemistry, by the reaction of excited atomic oxygen with water. It is also important in the field of radiation chemistry, since it leads to the formation of hydrogen peroxide and oxygen, which can enhance corrosion and SCC in coolant systems subjected to radioactive environments. In organic synthesis, hydroxyl radicals are most commonly generated by photolysis of 1-hydroxy-2(1''H'')-pyridinethione. Notation The unpaired electron of the hydroxyl radical is officially represented by a middle dot, •, beside the O. Biology Hydroxyl radicals can occasionally be produced as a byproduct of immune action. Macrophages and microglia most frequently generate this compound when exp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferrous
In chemistry, the adjective Ferrous indicates a compound that contains iron(II), meaning iron in its +2 oxidation state, possibly as the divalent cation Fe2+. It is opposed to "ferric" or iron(III), meaning iron in its +3 oxidation state, such as the trivalent cation Fe3+.ferrous entry in the online dictionary. Accessed on 2008-04-19. This usage has been largely replaced by the nomenclature, which calls for the oxidation state being indicated by Roman numerals in parentheses, such as |
Superoxide
In chemistry, a superoxide is a compound that contains the superoxide ion, which has the chemical formula . The systematic name of the anion is dioxide(1−). The reactive oxygen ion superoxide is particularly important as the product of the one-electron reduction of dioxygen , which occurs widely in nature. Molecular oxygen (dioxygen) is a diradical containing two unpaired electrons, and superoxide results from the addition of an electron which fills one of the two degenerate molecular orbitals, leaving a charged ionic species with a single unpaired electron and a net negative charge of −1. Both dioxygen and the superoxide anion are free radicals that exhibit paramagnetism. Superoxide was historically also known as "hyperoxide". Salts Superoxide forms salts with alkali metals and alkaline earth metals. The salts caesium superoxide (), rubidium superoxide (), potassium superoxide (), and sodium superoxide () are prepared by the reaction of with the respective alkali me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ubiquinone
Coenzyme Q, also known as ubiquinone and marketed as CoQ10, is a coenzyme family that is ubiquitous in animals and most bacteria (hence the name ubiquinone). In humans, the most common form is coenzyme Q10 or ubiquinone-10. It is a 1,4-benzoquinone, where Q refers to the quinone chemical group and 10 refers to the number of isoprenyl chemical subunits in its tail. In natural ubiquinones, the number can be anywhere from 6 to 10. This family of fat-soluble substances, which resemble vitamins, is present in all respiring eukaryotic cells, primarily in the mitochondria. It is a component of the electron transport chain and participates in aerobic cellular respiration, which generates energy in the form of ATP. Ninety-five percent of the human body's energy is generated this way. Organs with the highest energy requirements—such as the heart, liver, and kidney—have the highest CoQ10 concentrations. There are three redox states of CoQ: fully oxidized (ubiquinone), semiquinone ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitric Oxide
Nitric oxide (nitrogen oxide or nitrogen monoxide) is a colorless gas with the formula . It is one of the principal oxides of nitrogen. Nitric oxide is a free radical: it has an unpaired electron, which is sometimes denoted by a dot in its chemical formula In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, ... (•N=O or •NO). Nitric oxide is also a heteronuclear diatomic molecule, a class of molecules whose study spawned early modern molecular orbital theory, theories of chemical bonding. An important Reaction intermediate, intermediate in chemical industry, industrial chemistry, nitric oxide forms in combustion systems and can be generated by lightning in thunderstorms. In mammals, including humans, nitric oxide is a signaling molecule in many physiological and pathological pro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferric
In chemistry, iron(III) refers to the element iron in its +3 oxidation state. In ionic compounds (salts), such an atom may occur as a separate cation (positive ion) denoted by Fe3+. The adjective ferric or the prefix ferri- is often used to specify such compounds — as in "ferric chloride" for iron(III) chloride, . The adjective "ferrous" is used instead for iron(II) salts, containing the cation Fe2+. The word ferric is derived from the Latin word ''ferrum'' for iron. Iron(III) metal centres also occur in coordination complexes, such as in the anion ferrioxalate, , where three bidentate oxalate ions surrounding the metal centre; or, in organometallic compounds, such as the ferrocenium cation , where two cyclopentadienyl anions are bound to the FeIII centre. Iron is almost always encountered in the oxidation states 0 (as in the metal), +2, or +3. Iron(III) is usually the most stable form in air, as illustrated by the pervasiveness of rust, an insoluble iron(III)-containing ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diaphorase
Diaphorase may refer to: * Cytochrome b5 reductase, an enzyme * NADH dehydrogenase, an enzyme * NADPH dehydrogenase In enzymology, a NADPH dehydrogenase () is an enzyme that catalysis, catalyzes the chemical reaction :NADPH + H+ + acceptor \rightleftharpoons NADP+ + reduced acceptor The 3 substrate (biochemistry), substrates of this enzyme are nicotinamide ade ..., an enzyme {{Short pages monitor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


_oxide.jpg)
-oxide-sample.jpg)