|
Coomassie Brilliant Blue
Coomassie brilliant blue is the name of two similar triphenylmethane dyes that were developed for use in the textile industry but are now commonly used for staining proteins in analytical biochemistry. Coomassie brilliant blue G-250 differs from Coomassie brilliant blue R-250 by the addition of two methyl groups. The name "Coomassie" is a registered trademark of Imperial Chemical Industries. Name and discovery The name Coomassie was adopted at the end of the 19th century as a trade name by the Blackley-based dye manufacturer Levinstein Ltd, in marketing a range of acid wool dyes. In 1896 during the Fourth Anglo–Ashanti War, British forces had occupied the town of Coomassie (modern-day Kumasi in Ghana). In 1918 Levinstein Ltd became part of British Dyestuffs, which in 1926 became part of Imperial Chemical Industries. Although ICI still owns the Coomassie trademark, the company no longer manufactures the dyes. The blue disulfonated triphenylmethane dyes were first produced in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colour Index International
Colour Index International is a reference database jointly maintained by the Society of Dyers and Colourists and the American Association of Textile Chemists and Colorists. It currently contains over 27,000 individual products listed under 13,000 Colour Index Generic Names. It was first printed in 1925 but is now published solely on the World Wide Web. The index serves as a common reference database of manufactured colour products and is used by manufacturers and consumers, such as artists and decorators. Colorants (both dyes and pigments) are listed using a dual classification which use the Colour Index Generic Name (the prime identifier) and Colour Index Constitution Numbers. These numbers are prefixed with C.I. or CI, for example, C.I. 15510. (This abbreviation is sometimes mistakenly thought to be CL, due to the font used to display it.) A detailed record of products available on the market is presented under each Colour Index reference. For each product name, Colour Index ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Dissociation Constant
In chemistry, an acid dissociation constant (also known as acidity constant, or acid-ionization constant; denoted ) is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction :HA A^- + H^+ known as dissociation in the context of acid–base reactions. The chemical species HA is an acid that dissociates into , the conjugate base of the acid and a hydrogen ion, . The system is said to be in equilibrium when the concentrations of its components will not change over time, because both forward and backward reactions are occurring at the same rate. The dissociation constant is defined by :K_\text = \mathrm, or :\mathrmK_\ce = - \log_ K_\text = \log_\frac where quantities in square brackets represent the concentrations of the species at equilibrium. Theoretical background The acid dissociation constant for an acid is a direct consequence of the underlying thermodynamics of the dissociation reaction; the p''K''a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Colloid
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others extend the definition to include substances like aerosols and gels. The term colloidal suspension refers unambiguously to the overall mixture (although a narrower sense of the word '' suspension'' is distinguished from colloids by larger particle size). A colloid has a dispersed phase (the suspended particles) and a continuous phase (the medium of suspension). The dispersed phase particles have a diameter of approximately 1 nanometre to 1 micrometre. Some colloids are translucent because of the Tyndall effect, which is the scattering of light by particles in the colloid. Other colloids may be opaque or have a slight color. Colloidal suspensions are the subject of interface and colloid science. This field of study was introduced in 1845 by Ital ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetic Acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water and other trace elements. Acetic acid is the second simplest carboxylic acid (after formic acid). It is an important chemical reagent and industrial chemical, used primarily in the production of cellulose acetate for photographic film, polyvinyl acetate for wood glue, and synthetic fibres and fabrics. In households, diluted acetic acid is often used in descaling agents. In the food industry, acetic acid is controlled by the food additive code E260 as an acidity regulator and as a condiment. In biochemistry, the acetyl group, derived from acetic acid, is fundamental to all forms of life. When bound to coenzyme A, it is central to the metabolism of carbohydrates and fats. The global demand for acetic aci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical and the simplest aliphatic alcohol, with the formula C H3 O H (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a light, volatile, colourless, flammable liquid with a distinctive alcoholic odour similar to that of ethanol (potable alcohol). A polar solvent, methanol acquired the name wood alcohol because it was once produced chiefly by the destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide. Methanol consists of a methyl group linked to a polar hydroxyl group. With more than 20 million tons produced annually, it is used as a precursor to other commodity chemicals, including formaldehyde, acetic acid, methyl tert-butyl ether, methyl benzoate, anisole, peroxyacids, as well as a host of more specialised chemicals. Occurrence Small amounts of methanol are present in normal, healthy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
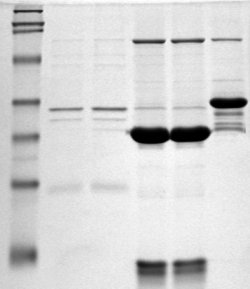
Polyacrylamide Gel
Polyacrylamide gel electrophoresis (PAGE) is a technique widely used in biochemistry, forensic chemistry, genetics, molecular biology and biotechnology to separate biological macromolecules, usually proteins or nucleic acids, according to their Electrophoresis, electrophoretic mobility. Electrophoretic mobility is a function of the length, conformation, and charge of the molecule. Polyacrylamide gel electrophoresis is a powerful tool used to analyze RNA samples. When polyacrylamide gel is denatured after electrophoresis, it provides information on the sample composition of the RNA species. Hydration reaction, Hydration of acrylonitrile results in formation of acrylamide molecules () by nitrile hydratase. Acrylamide monomer is in a powder state before addition of water. Acrylamide is toxic to the human nervous system, therefore all safety measures must be followed when working with it. Acrylamide is soluble in water and upon addition of free-radical initiators it polymerizes resul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfosalicylic Acid
Sulfosalicylic acid is used in urine tests to determine urine protein content. The chemical causes the precipitation of dissolved proteins, which is measured from the degree of turbidity. , Family Practice Notebook website It is also used for integral colour . With water it is used as a shuttle solution for the CAS assay to test for siderophore. See also *Salicylic acid
Salicylic acid is an organic compound with the formula HOC6H4CO2H. A colorless, bitter-tasting solid, it is a precursor to and a metabolite of aspirin (acet ...
[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cellulose Acetate
In biochemistry, cellulose acetate refers to any acetate ester of cellulose, usually cellulose diacetate. It was first prepared in 1865. A bioplastic, cellulose acetate is used as a film base in photography, as a component in some coatings, and as a frame material for eyeglasses; it is also used as a synthetic fiber in the manufacture of cigarette filters and playing cards. In cellulose acetate film, photographic film, cellulose acetate film replaced nitrate film in the 1950s, being far less flammable and cheaper to produce. History In 1865, French chemist Paul Schützenberger discovered that cellulose reacts with acetic anhydride to form cellulose acetate. The German chemists Arthur Eichengrün and Theodore Becker invented the first soluble forms of cellulose acetate in 1903. In 1904, Camille Dreyfus (chemist), Camille Dreyfus and his younger brother Henri Dreyfus, Henri performed chemical research and development on cellulose acetate in a shed in their father's garden in Basel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Lauryl Sulfate
Sodium dodecyl sulfate (SDS) or sodium lauryl sulfate (SLS), sometimes written sodium laurilsulfate, is an organic compound with the formula . It is an anionic surfactant used in many cleaning and hygiene products. This compound is the sodium Salt (chemistry), salt of the 12-carbon an organosulfate. Its hydrocarbon tail combined with a Chemical polarity, polar "Detergent#Chemical classifications of detergents, headgroup" give the compound amphiphilic properties that make it useful as a detergent. SDS is also component of mixtures produced from inexpensive coconut oil, coconut and palm oils. SDS is a common component of many domestic cleaning, personal hygiene and cosmetic, pharmaceutical, and food products, as well as of industrial and commercial cleaning and product formulations. Physicochemical properties The critical micelle concentration (CMC) in water at 25 °C is 8.2 mM, and the aggregation number at this concentration is usually considered to be about 62. The micelle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bradford Assay
The Bradford protein assay was developed by Marion M. Bradford in 1976. It is a quick and accurate spectroscopic analytical procedure used to measure the concentration of protein in a solution. The reaction is dependent on the amino acid composition of the measured proteins. Principle The Bradford assay, a colorimetric protein assay, is based on an absorbance shift of the dye Coomassie brilliant blue G-250. The Coomassie brilliant blue G-250 dye exists in three forms: anionic (blue), neutral (green), and cationic (red). Under acidic conditions, the red form of the dye is converted into its blue form, binding to the protein being assayed. If there's no protein to bind, then the solution will remain brown. The dye forms a strong, noncovalent complex with the protein's carboxyl group by van der Waals force and amino group through electrostatic interactions. During the formation of this complex, the red form of Coomassie dye first donates its free electron to the ionizable groups ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Keratin
Keratin () is one of a family of structural fibrous proteins also known as ''scleroproteins''. Alpha-keratin (α-keratin) is a type of keratin found in vertebrates. It is the key structural material making up scales, hair, nails, feathers, horns, claws, hooves, and the outer layer of skin among vertebrates. Keratin also protects epithelial cells from damage or stress. Keratin is extremely insoluble in water and organic solvents. Keratin monomers assemble into bundles to form intermediate filaments, which are tough and form strong unmineralized epidermal appendages found in reptiles, birds, amphibians, and mammals. Excessive keratinization participate in fortification of certain tissues such as in horns of cattle and rhinos, and armadillos' osteoderm. The only other biological matter known to approximate the toughness of keratinized tissue is chitin. Keratin comes in two types, the primitive, softer forms found in all vertebrates and harder, derived forms found only among saur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |