|
Ytterbium Compounds
Ytterbium compounds are chemical compounds that contain the element ytterbium (Yb). The chemical behavior of ytterbium is similar to that of the rest of the lanthanides. Most ytterbium compounds are found in the +3 oxidation state, and its salts in this oxidation state are nearly colorless. Like europium, samarium, and thulium, the trihalides of ytterbium can be reduced to the dihalides by hydrogen, zinc dust, or by the addition of metallic ytterbium. The +2 oxidation state occurs only in solid compounds and reacts in some ways similarly to the alkaline earth metal compounds; for example, ytterbium(II) oxide (YbO) shows the same structure as calcium oxide (CaO). Halides Ytterbium forms both dihalides and trihalides with the halogens fluorine, chlorine, bromine, and iodine. The dihalides are susceptible to oxidation to the trihalides at room temperature and disproportionate to the trihalides and metallic ytterbium at high temperature: :3 YbX2 → 2 YbX3 + Yb (X = F, Cl, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium
Ytterbium is a chemical element with the symbol Yb and atomic number 70. It is a metal, the fourteenth and penultimate element in the lanthanide series, which is the basis of the relative stability of its +2 oxidation state. However, like the other lanthanides, its most common oxidation state is +3, as in its oxide, halides, and other compounds. In aqueous solution, like compounds of other late lanthanides, soluble ytterbium compounds form complexes with nine water molecules. Because of its closed-shell electron configuration, its density and melting and boiling points differ significantly from those of most other lanthanides. In 1878, the Swiss chemist Jean Charles Galissard de Marignac separated from the rare earth "erbia" another independent component, which he called " ytterbia", for Ytterby, the village in Sweden near where he found the new component of erbium. He suspected that ytterbia was a compound of a new element that he called "ytterbium" (in total, four elements were ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium(III) Chloride
Ytterbium(III) chloride (Ytterbium, YbChlorine, Cl3) is an inorganic chemical compound. It reacts with NiCl2 to form a very effective Catalysis, catalyst for the Reductive dechlorination, reductive dehalogenation of aryl halides. It is poisonous if injected, and mildly toxic by ingestion. It is an experimental Teratology, teratogen, known to Irritation, irritate the skin and eyes. When heated to Chemical decomposition, decomposition it emits Toxicity, toxic fumes of Chloride, Cl−. History The synthesis of YbCl3 was first reported by Jan Hoogschagen in 1946. It is now a commercially available source of Yb3+ ions and therefore of significant chemical interest. Chemical properties The valence electron configuration of Yb+3 (from YbCl3) is 4''f''135''s''25''p''6, which has crucial implications for the chemical behaviour of Yb+3. Also, the size of Yb+3 governs its catalytic behaviour and biological applications. For example, while both Ce+3 and Yb+3 have a single unpaired ''f'' elec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium(III) Oxide
Ytterbium(III) oxide is the chemical compound with the formula Yb2O3. It is one of the more commonly encountered compounds of ytterbium. It has the "rare-earth C-type sesquioxide" structure which is related to the fluorite structure with one quarter of the anions removed, leading to ytterbium atoms in two different six coordinate (non-octahedral) environments. Wells A.F. (1984) ''Structural Inorganic Chemistry'' 5th edition Oxford Science Publications Uses * Colorant for glasses and enamels * Dopant for garnet crystals in lasers * Optical fibers See also * Active laser medium The active laser medium (also called gain medium or lasing medium) is the source of optical gain within a laser. The gain results from the stimulated emission of photons through electronic or molecular transitions to a lower energy state from a h ... References External links Information at WebElements Ytterbium compounds Sesquioxides {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
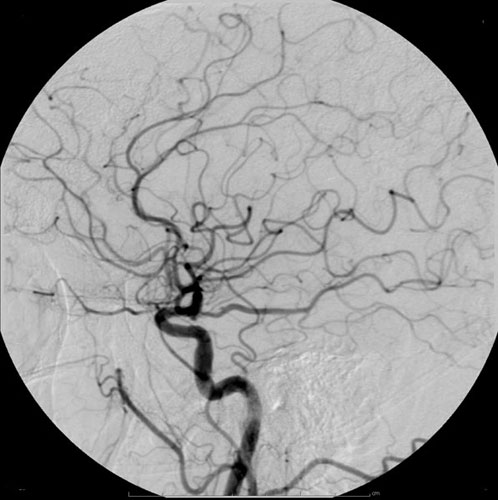
X-ray Contrast Agent
Radiocontrast agents are substances used to enhance the visibility of internal structures in X-ray-based imaging techniques such as computed tomography (contrast CT), projectional radiography, and fluoroscopy. Radiocontrast agents are typically iodine, or more rarely barium sulfate. The contrast agents absorb external X-rays, resulting in decreased exposure on the X-ray detector. This is different from radiopharmaceuticals used in nuclear medicine which emit radiation. Magnetic resonance imaging (MRI) functions through different principles and thus MRI contrast agents have a different mode of action. These compounds work by altering the magnetic properties of nearby hydrogen nuclei. Types and uses Radiocontrast agents used in X-ray examinations can be grouped in positive (iodinated agents, barium sulfate), and negative agents (air, carbon dioxide, methylcellulose). Iodine (circulatory system) Iodinated contrast contains iodine. It is the main type of radiocontrast used for i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluoride
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typically have distinctive bitter tastes, and are odorless. Its salts and minerals are important chemical reagents and industrial chemicals, mainly used in the production of hydrogen fluoride for fluorocarbons. Fluoride is classified as a weak base since it only partially associates in solution, but concentrated fluoride is corrosive and can attack the skin. Fluoride is the simplest fluorine anion. In terms of charge and size, the fluoride ion resembles the hydroxide ion. Fluoride ions occur on Earth in several minerals, particularly fluorite, but are present only in trace quantities in bodies of water in nature. Nomenclature Fluorides include compounds that contain ionic fluoride and those in which fluoride does not dissociate. The nom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tooth Filling
Dental restoration, dental fillings, or simply fillings are treatments used to restore the function, integrity, and morphology of missing tooth structure resulting from caries or external trauma as well as to the replacement of such structure supported by dental implants. They are of two broad types—''direct'' and ''indirect''—and are further classified by location and size. A root canal filling, for example, is a restorative technique used to fill the space where the dental pulp normally resides. Tooth preparation Restoring a tooth to good form and function requires two steps: # preparing the tooth for placement of restorative material or materials, and # placement of these materials. The process of preparation usually involves cutting the tooth with a rotary dental handpiece and dental burrs, a dental laser, or though air abrasion to make space for the planned restorative materials and to remove any dental decay or portions of the tooth that are structurally unsound. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium(III) Fluoride
Ytterbium(III) fluoride () is an inorganic chemical compound A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ... that is insoluble in water. Like other Ytterbium compounds, it is a rather unremarkable white substance. Ytterbium fluoride has found a niche usage as a radio-opaque agent in the dental industry to aid in the identification of fillings under X-ray examination. References Fluorides Lanthanide halides Ytterbium compounds {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coupling Reactions
A coupling reaction in organic chemistry is a general term for a variety of reactions where two fragments are joined together with the aid of a metal catalyst. In one important reaction type, a main group organometallic compound of the type R-M (R = organic fragment, M = main group center) reacts with an organic halide of the type R'-X with formation of a new carbon-carbon bond in the product R-R'. The most common type of coupling reaction is the cross coupling reaction. Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki were awarded the 2010 Nobel Prize in Chemistry for developing palladium-catalyzed cross coupling reactions. Broadly speaking, two types of coupling reactions are recognized: *Heterocouplings combine two different partners, such as in the Heck reaction of an alkene (RC=CH) and an alkyl halide (R'-X) to give a substituted alkene, or the Corey–House synthesis of an alkane by the reaction of a lithium diorganylcuprate (R2CuLi) with an organyl (pseudo)halide ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reducing Agent
In chemistry, a reducing agent (also known as a reductant, reducer, or electron donor) is a chemical species that "donates" an electron to an (called the , , , or ). Examples of substances that are commonly reducing agents include the Earth metals, formic acid, oxalic acid, and sulfite compounds. In their pre-reaction states, reducers have extra electrons (that is, they are by themselves reduced) and oxidizers lack electrons (that is, they are by themselves oxidized). This is commonly expressed in terms of their oxidation states. An agent's oxidation state describes its degree of loss of electrons, where the higher the oxidation state then the fewer electrons it has. So initially, prior to the reaction, a reducing agent is typically in one of its lower possible oxidation states; its oxidation state increases during the reaction while that of the oxidizer decreases. Thus in a redox reaction, the agent whose oxidation state increases, that "loses/Electron donor, donates electrons ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
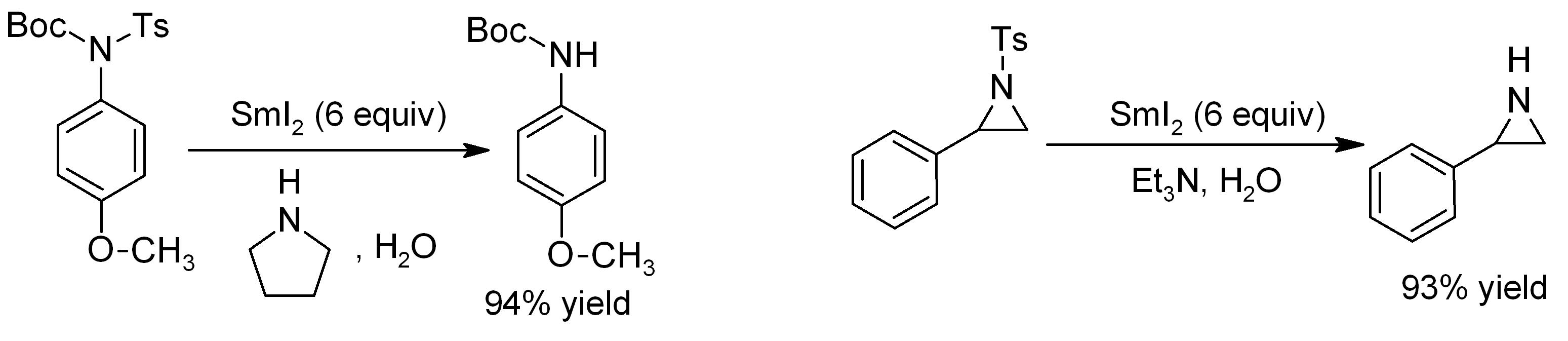
Samarium(II) Iodide
Samarium(II) iodide is an inorganic compound with the formula SmI2. When employed as a solution for organic synthesis, it is known as Kagan's reagent. SmI2 is a green solid and solutions are green as well. It is a strong one-electron reducing agent that is used in organic synthesis. Structure In samarium(II) iodide, the metal centers are seven-coordinate with a face-capped octahedral geometry. In its ether adducts, samarium remains heptacoordinate with five ether and two terminal iodide ligands. Preparation Samarium iodide is easily prepared in nearly quantitative yields from samarium metal and either diiodomethane or 1,2-diiodoethane. When prepared in this way, its solutions is most often used without purification of the inorganic reagent. Solid, solvent-free SmI2 forms by high temperature decomposition of samarium(III) iodide (SmI3).G. Jantsch, N. Skalla: "Zur Kenntnis der Halogenide der seltenen Erden. IV. – Über Samarium(II)jodid und den thermischen Abbau des Samari ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ytterbium(II) Iodide
Ytterbium(II) iodide is an iodide of ytterbium, with the chemical formula of YbI2. It is a yellow solid. Preparation Ytterbium(II) iodide can be prepared by heating ytterbium(III) iodide: :\mathrm It can also be prepared by reacting metallic ytterbium with 1,2-diiodoethane in tetrahydrofuran: :\mathrm Although the reaction takes place at room temperature, due to the sensitivity of the reagents it is necessary to work anhydrous and under inert gas. Otherwise, if oxygen is present, rapid oxidation to ytterbium(III) takes place. This can be visually recognized by the color change from green to yellow solution. Properties and uses Ytterbium(II) iodide is a yellow solid that is very sensitive to air and moisture and is rapidly oxidized to ytterbium(III). It reacts with water to produce hydrogen gas and basic iodides, and reacts violently with acids. Ytterbium(II) iodide sinters at 0.01 Torr from about 780 °C and gives a viscous melt at about 920 °C. It begin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diels–Alder Reaction
In organic chemistry, the Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. It is the prototypical example of a pericyclic reaction with a concerted mechanism. More specifically, it is classified as a thermally-allowed +2cycloaddition with Woodward–Hoffmann symbol π4s_+_π2s.html" ;"title="sub>π4s + π2s">sub>π4s + π2s It was first described by Otto Diels and Kurt Alder in 1928. For the discovery of this reaction, they were awarded the Nobel Prize in Chemistry in 1950. Through the simultaneous construction of two new carbon–carbon bonds, the Diels–Alder reaction provides a reliable way to form six-membered rings with good control over the regio- and stereochemical outcomes. Consequently, it has served as a powerful and widely applied tool for the introduction of chemical complexity in the synthesis of natural products and new materials. The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
_chloride.jpg)